Abstract
Gastric cancer (GC) remains poor prognosis and survival issues due to the resistance of chemotherapies, such as cisplatin. The long non-coding RNA small nucleolar RNA host gene 7 (lncRNA-SNHG7) is known as an oncogenic molecule in diverse cancers. Here, we demonstrate that SNHG7 was significantly upregulated in gastric cancer and positively correlated with cisplatin resistance of gastric cancer cells that SNHG7 was significantly upregulated in cisplatin resistant cells. Silencing SNHG7 dramatically sensitized cisplatin resistant cells. In contrast, a negative correlation between lncRNA-SNHG7 and miR-34a was found that miR-34a was downregulated in gastric cancer patient tissues and significantly sensitized cisplatin resistant gastric cancer cells. Intriguingly, bioinformatical analysis indicated miR-34a has putative biding site for SNHG7 and such negative association between SNHG7 and miR-34a was verified in gastric cancer tissues. The cisplatin resistant cells displayed increased glycolysis rate and SNHG7 promoted cellular glycolysis rate of gastric cancer cells. Luciferase assay illustrated LDHA, a glycolysis enzyme, was the direct target of miR-34a. Importantly, inhibiting SNHG7 successfully suppressed LDHA expressions and sensitized cisplatin resistant cells and such inhibitory effects could be recovered by further anti-miR-34a. These findings suggest an important regulator mechanism for the SNHG7-mediated cisplatin resistance via miR-34a/LDHA-glycolysis axis.
Introduction
Gastric cancer (GC) is one of the most common cancers worldwide and represents a high incidence of cancer-related death [1]. Currently, GC is the fifth most frequently diagnosed cancer and the third leading cause of cancer death [1, 2]. The primary treatment approach is a surgical resection with combination of chemotherapy such as cisplatin, a platinum-based compound [3]. Despite the recent discoveries in surgical techniques and chemotherapeutic agents, the mortality and average 5-year survival rate did not effectively improved [4]. Seriously, although impressive clinical responses were observed from those chemotherapeutic agents initially, development of acquired chemoresistant cancer phenotypes limited the clinical applications of them [5]. Thus, investigation of underlining molecular mechanisms for chemoresistance is an urgent task to effectively improve treatment results.
Long non-coding RNAs (lncRNAs) are a group of RNAs with relative long (
In cancer cells, the rate of glucose metabolism is dramatically higher than untransformed cells, even with enough oxygen, a phenomenon known as the “Warburg effect” [15]. Moreover, the Warburg effect is apparently associated with chemosensitivity of cancer cells [16]. A recent study revealed that 5-Fu resistant cancer cells displayed upregulated glycolysis rate, and such chemoresistance could be effectively overcome by inhibiting glycolysis enzyme, PKM2 [17], suggesting glycolysis inhibition of cancer cells could enhance the chemotherapeutic effects of anti-cancer agents.
In this study, we aimed to explore the functions of SNHG7 in chemoresistant gastric cancer cells. The potential mechanisms of SNHG7/miR-34a-mediated cisplatin resistance will be investigated. In addition, the specific roles of SNHG7 in regulating the cellular glucose metabolism of gastric cancer cells will be examined. Our study will contribute to develop novel and promising targets against the cisplatin resistant gastric cancer.
Materials and methods
Patient samples
This study was approved by the Medical Ethics Committee of the Fourth Medical Center of PLA General Hospital (number 2020KY012-KS001). Human gastric cancer tissue samples used for this study came from 30 gastric cancer and 30 normal gastric tissues. Samples were collected from the Department of General Surgery, the Fourth Medical Center of PLA General Hospital from January 2017 to June 2018. All patients received no radiotherapy and/or chemotherapy before surgery. Upon dissection, tissues were stored immediately at liquid nitrogen and then transferred to
Cell culture and reagents
The human gastric epithelial cell line and gastric cancer cell lines were obtained from the American Type Culture Collection (ATCC). Cells were cultured in RPMI 1640 medium (Thermo Fisher Scientific, Inc., Carlsbad, CA, USA) with 10% fetal bovine serum (FBS), 100 units/ml penicillin, and 100
Prediction of lncRNA-miRNA and miRNA-mRNA interactions
The interaction between lnRNA-SNHG7 and miR-34a was predicted by starBase of ENCORI (
Plasmid, shRNA and miRNAs transfection
The gastric cancer cells were cultured to about 80% confluence in 24 or 96-well plates then cells using were transfected with the indicated shRNA, miRNAs, or plasmid DNA using Lipofectamine 2000 (Thermo Fisher Scientific, Inc., Carlsbad, CA, USA) according to the manufacturer’s instructions. The shRNA-SNHG7 or negative control was conducted by RiboBio (Guangzhou, China). The miR-34a precursor, miR-34a inhibitors or negative control were purchased from GenePharma (Shanghai, China) and transfected at 50 nM. The overexpression plasmid of LDHA was purchased from Origen.com and transfected at 2 ug. After 48 hours transfection, the cells were harvested for downstream experiments.
RNA isolation and qRT-PCR
The total RNA was extracted from the gastric tumor tissues or adjacent normal gastric tissues by the Trizol method according to the manufacturer’s instructions. First-strand cDNA was synthesized by reversely transcribing RNA into complementary DNA from 500 ng total RNA using a SuperScript First-Standard Synthesis System (Invitrogen, Carlsbad, CA, USA) following the manufacturer’s protocol. qRT-PCR experiments were performed using the SYBR Green qPCR Master Mix (ThermoFisher Scientific, Shanghai, China). The fold changes were analyzed using the 2
Luciferase reporter assays
The luciferase assay was performed according to previous descriptions [20]. 5
Measurements of glucose metabolism rate
The glucose uptake and lactate product measurements were performed using the glucose test kit (Applygen Technologies, Beijing, China) and the L-lactate assay kit (BioVision, Milpitas, CA, USA) according to the manufacturer’s instruction. Results were normalized according to the cell number by dividing the readings from treated cells to that of control cells. Experiments were performed in triplicate and repeated three times.
Cell viability assay
The cell viability assay was performed by MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) method (Sigma-Aldrich, Shanghai, China) according to the manufacture’s instruction. Briefly, cells (5
Western blot assay
Total proteins were extracted from gastric cancer tissues or cells using the RIPA buffer with protease inhibitor cocktail (Bio-Rad, Hercules, CA, USA) according to the instructions of the manufacturer. After 15 min incubation with lysis buffer on ice, samples were centrifuged at 10,000 g for 10 min at 4
Statistical analysis
Results were analyzed by the Prism 6.0 software package (GraphPad Software, Inc.). All experiments were performed three times. Paired student
SNHG7 is upregulated in gastric tumor and contributes to cisplatin resistance. (A) The expressions of SNHG7 in 30 pairs of gastric cancer tissues and adjacent normal tissues were detected by qRT-PCR. (B) The expressions of SNHG7 in normal gastric epithelial cells and five gastric cancer cell lines were detected by qRT-PCR. 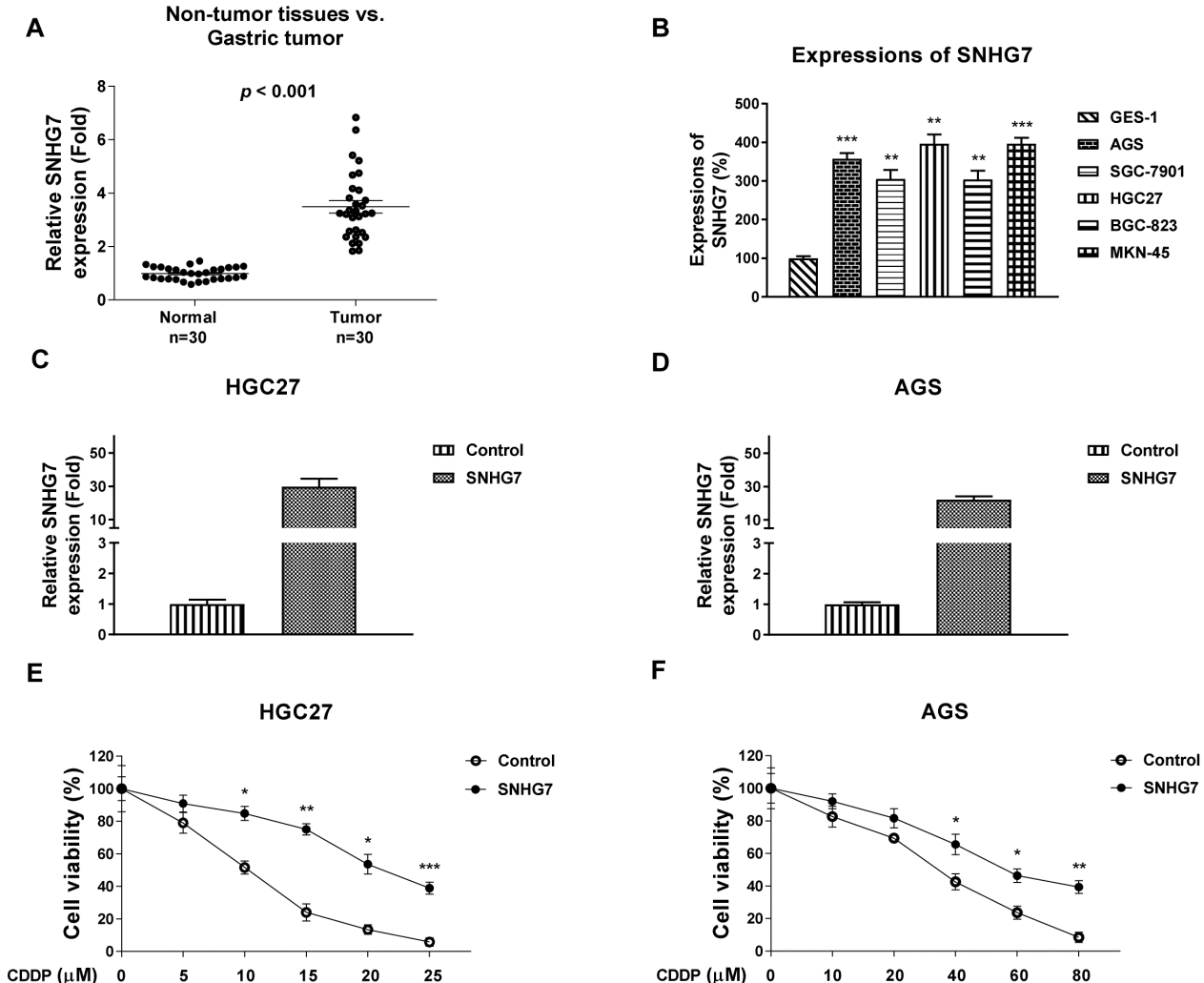
SNHG7 is positively correlated with cisplatin resistance of gastric cancer cells. (A) Verifying the cisplatin resistance from HGC27 cells. HGC27 parental and cisplatin resistant cells were treated with cisplatin at 0, 5, 10, 15, 20 or 25 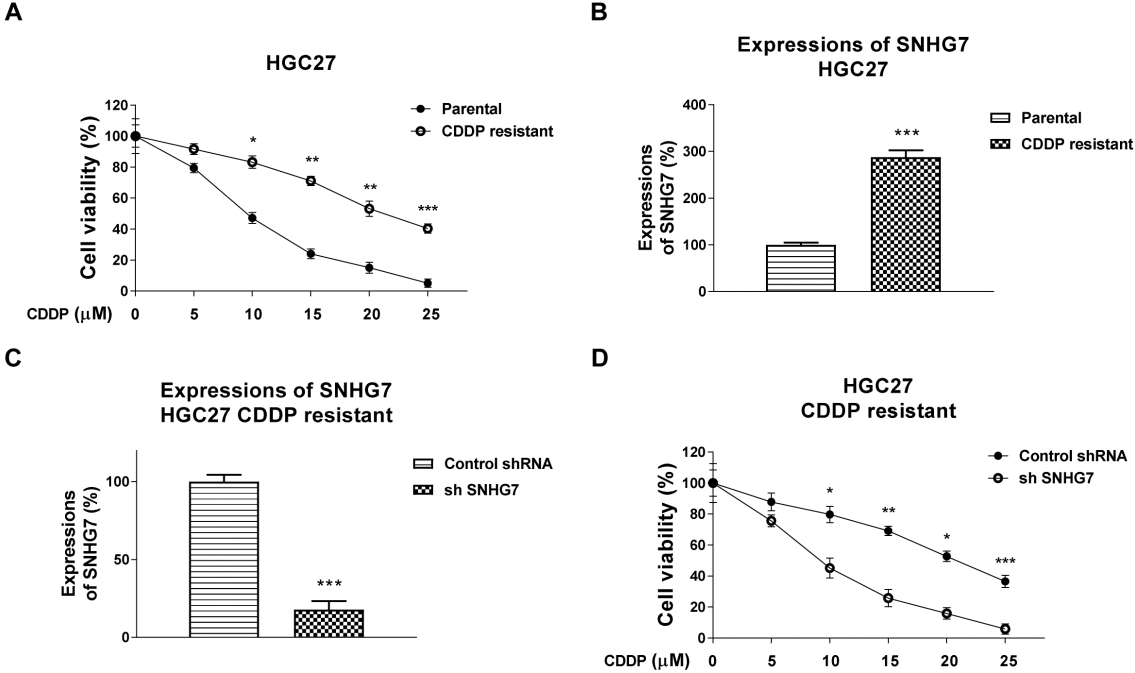
miR-34a is downregulated in gastric cancers and negatively correlates with cisplatin resistance. (A) The expressions of miR-34a in 30 pairs of gastric cancer tissues and adjacent normal tissues were detected by qRT-PCR. (B) The expressions of miR-34a in normal gastric epithelial cells and five gastric cancer cell lines were detected by qRT-PCR. Human U6 was an internal control. (C) Expressions of miR-34a from HGC27 parental and cisplatin resistant cells were detected by qRT-PCR. (D) HGC27 cisplatin resistant without or with miR-34a overexpression were treated with cisplatin at the indicated concentrations for 48 hours, cell viabilities were measured by MTT assay. The error bars in graphs represented SD, and each experiment was repeated three times. 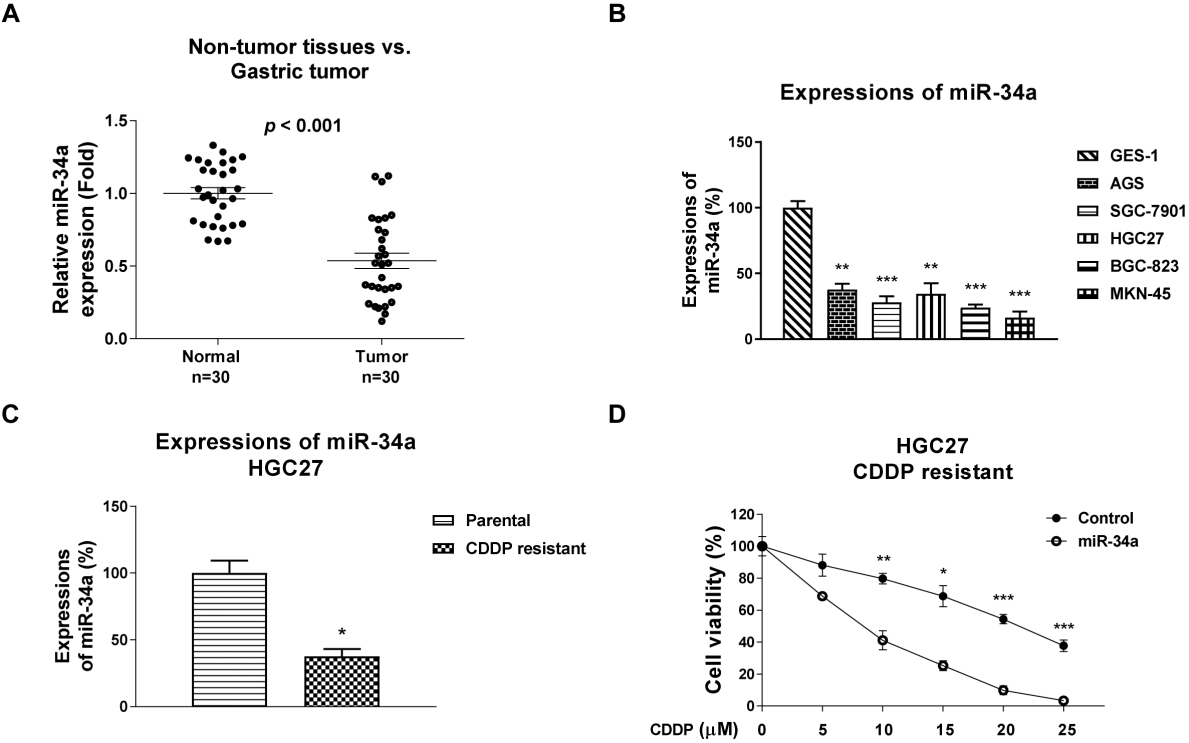
SNHG7 inhibits miR-34a expression through interaction with miR-34a. (A) Prediction of binding sequence of miR-34a on SNHG7 from starBase. (B) Negative correlation between miR-34a and SNHG7 expression in gastric cancer tissues. (C) HGC27 and AGS cells were transfected with control or SNHG7 overexpression plasmid for 48 hours, expressions of miR-34a were determined by qRT-PCR. (D) HGC27 and AGS cells were transfected with control shRNA or SNHG7 shRNA for 48 hours, expressions of miR-34a were determined by qRT-PCR. The error bars in graphs represented SD, and each experiment was repeated three times. 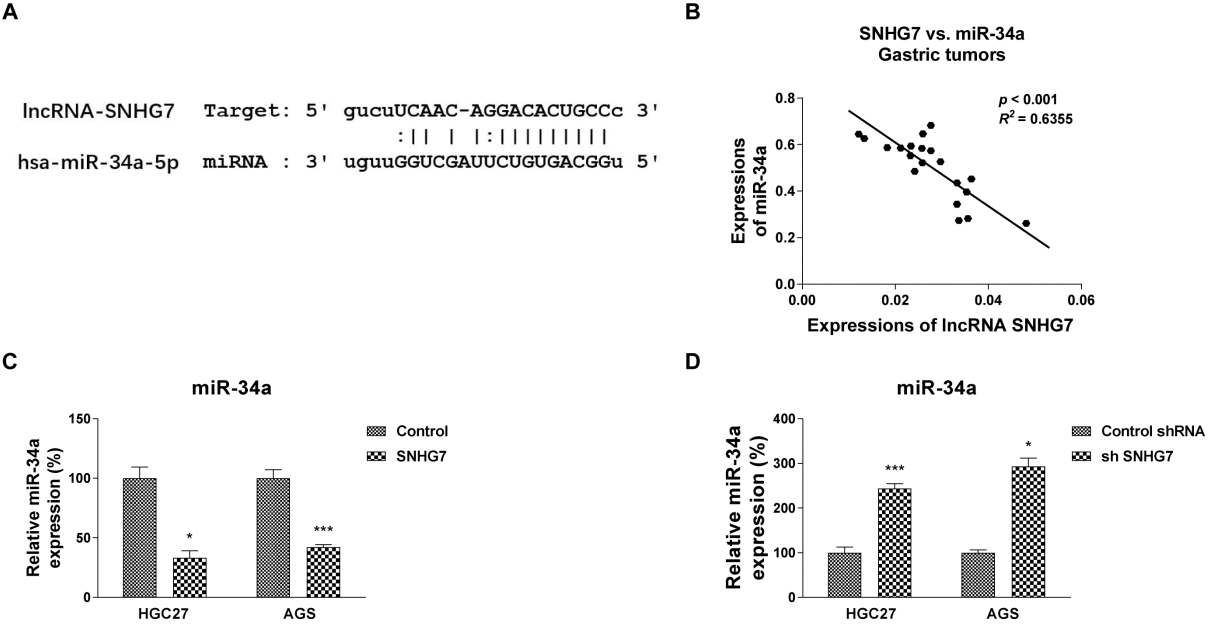
SNHG7 promotes glycolysis rates of gastric cancer cells. (A) The glucose uptake and lactate product were measured from the HGC27 parental and cisplatin resistant cells. (B) Protein expressions of glycolysis enzymes from HGC27 parental and cisplatin resistant cells were determined by Western blot. 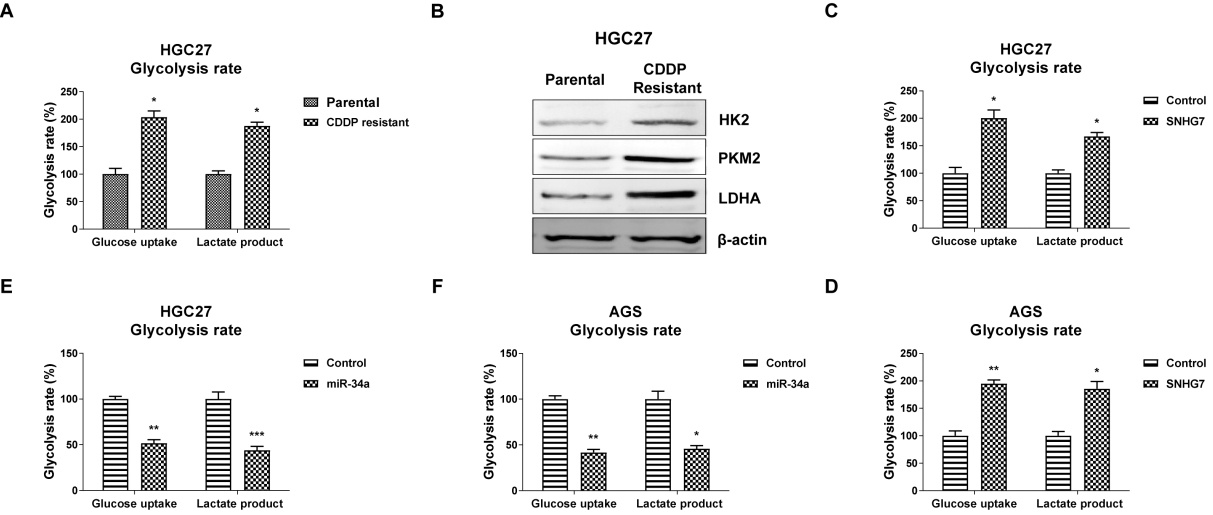
miR-34a directly targets 3’UTR of LDHA mRNA. (A) Bioinformatic analysis predicted that 3’UTR of LDHA mRNA harbored miR-34a binding sites. (B) HGC27 and AGS cells were transfected with control miRNAs or miR-34a. The protein expressions of LDHA were determined by Western blot. 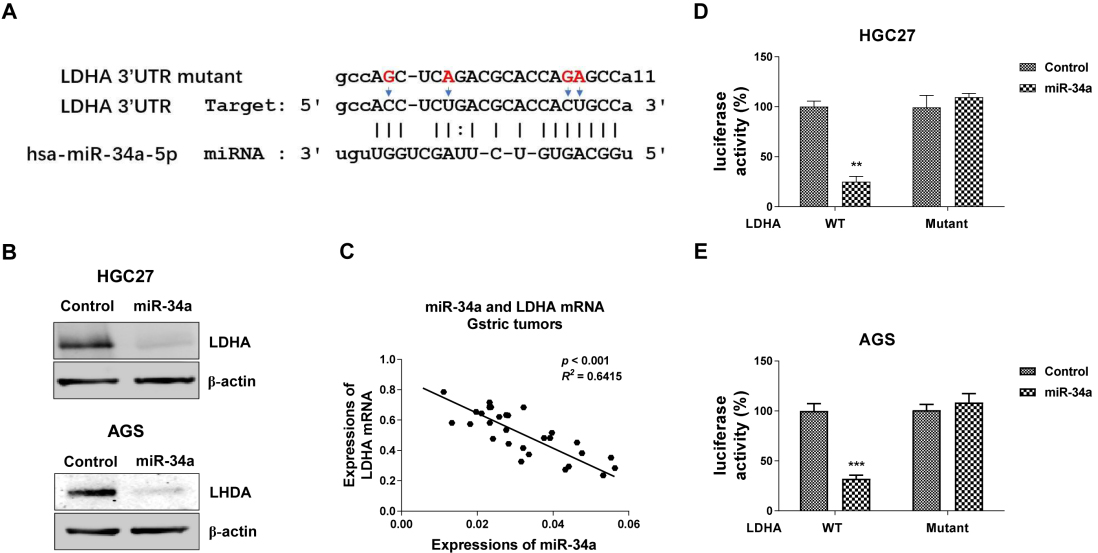
Restoration of LDHA recovers the glycolysis and cisplatin resistance of gastric cancer cells. (A) HGC27 cisplatin resistant cells were transfected with control, miR-34a alone or miR-34a plus LDHA overexpression plasmid for 48 hours, the expressions of LDHA were measured by Western blot. 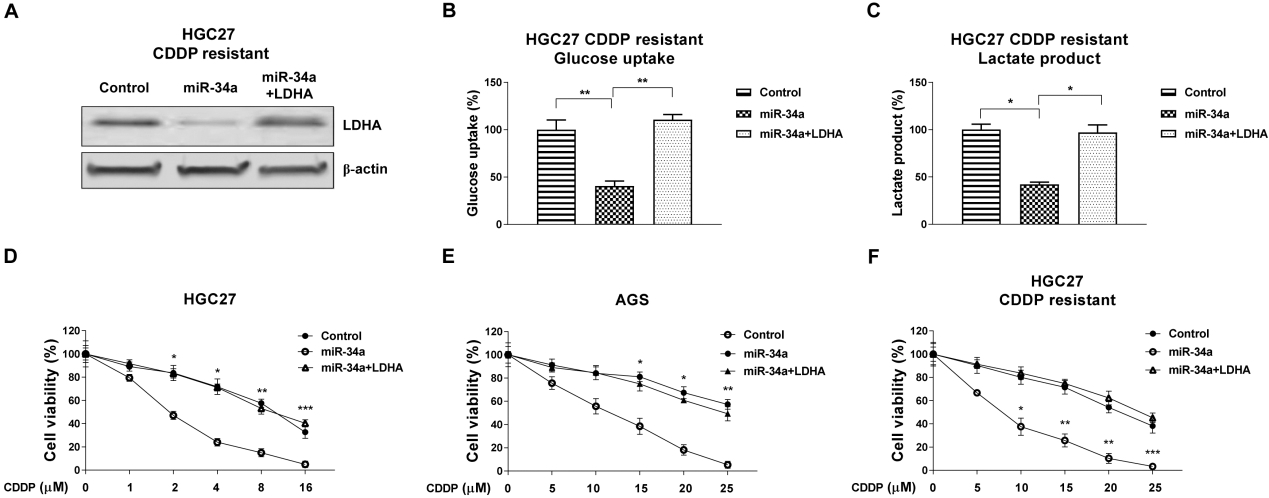
The SNHG7-promoted cisplatin resistance is through miR-34a/LDHA axis. (A) HGC27 cells were transfected with control, SNHG7 shRNA alone or SNHG7 shRNA plus miR-34a inhibitor for 48 hours, protein expressions of LDHA were detected by Western blot. 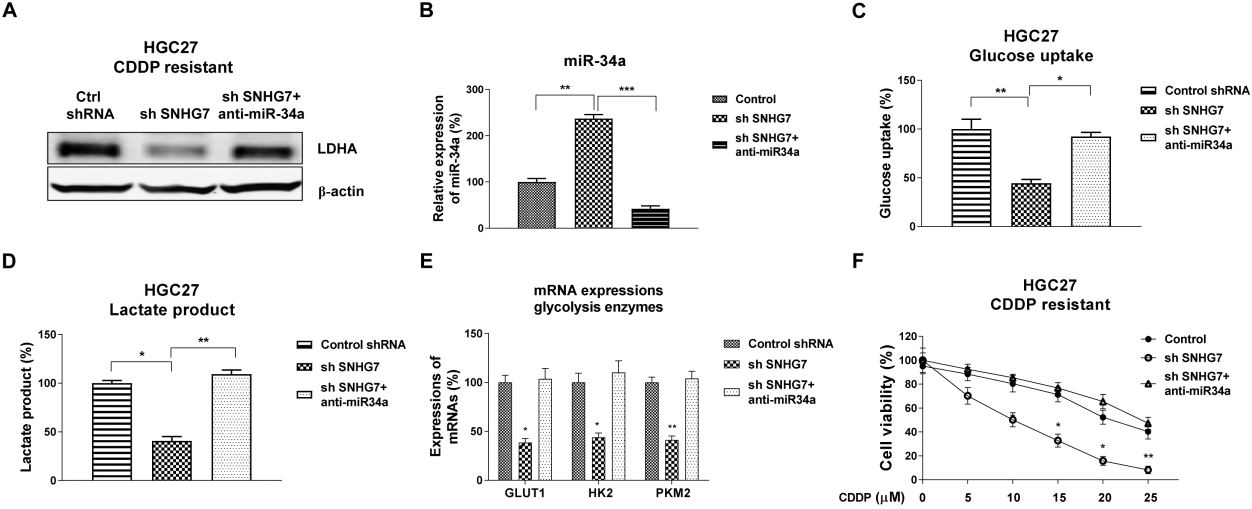
LncRNA-SNHG7 is upregulated in gastric cancer and positively correlates with cisplatin resistance
To investigate the clinical relevance of SNHG7 during the progression of gastric cancer, we collected the gastric tumor tissues and their corresponding adjacent normal tissues (
miR-34a is downregulated in gastric cancer and negatively correlate with cisplatin resistance
We next to evaluate the molecular mechanisms for the SNHG7-mediated cisplatin resistance. Previous studies have demonstrated lncRNAs can act as miRNA sponges to suppress their regulatory effects on targets mRNAs [19]. Moreover, miR-34a has been reported to be negatively correlated with cisplatin resistance in osteosarcoma [21], suggesting miR-34a might be a downstream effector of SNHG7, which involves in cisplatin resistance. Expectedly, the miR-34a expressions were significantly downregulated in tumor tissues compared with gastric normal tissues (Fig. 3A). Similar trend was observed that miR-34a was suppressed in gastric cancer cells compared with normal gastric epithelial cells (Fig. 3B). Consistently, miR-34a was found downregulated in cisplatin resistant HGC27 cells compared with parental cells (Fig. 3C). Thus, we hypothesized miR-34a could sensitizes gastric cancer cells to chemotherapy. Results in Fig. 3D illustrated overexpression of miR-34a significantly increased the sensitivity of cisplatin resistant cells (Fig. 3D). The IC50 of miR-34a overexpressing cells was 7.62
SNHG7 suppresses miR-34a expression via sponging
Accumulating evidence indicated that lncRNAs contain motifs which are complementary to miRNAs sequences to negatively regulate miRNAs. A putative complementary sequence was predicted for miR-34a in SNHG7 Starbase (
Cisplatin resistant gastric cancer cells showed elevated glycolysis rate
Accumulating evidence suggests that malignant cancer cells demand increased glucose metabolism via a higher anaerobic glycolysis rate, a phenomenon was known as “the Warburg effect” [15]. Moreover, the acquired chemoresistance of cancer cells were frequently associated with higher glycolysis rate [16]. To investigate the molecular mechanism for the SNHG7/miR-34a-meidated cisplatin resistance, we compared the glycolysis rates of HGC27 parental and cisplatin resistant cells. Interestingly, the glucose uptake and lactate product, two glycolysis speed limit steps [15], as well as glycolysis key enzymes were apparently increased in cisplatin resistant cells (Fig. 5A and B), indicating the dysregulated cellular glycolysis rate was tightly associated with cisplatin resistance. Consistent results from Fig. 5C and D demonstrated overexpression of SNHG7 facilitated glucose uptake and lactate product of gastric cancer cells. On the contrary, overexpression of miR-34a significantly suppressed glycolysis rates of HGC27 and AGS cells (Fig. 5E and F). These results suggested targeting the SNHG7-promoted cellular glycolysis rate might contribute to anti-chemoresistance of gastric tumor.
miR-34a directly targets glycolysis enzyme LDHA to increase the cisplatin sensitivity of gastric cancer cells
To identify the potential target of miR-34a, we analyzed the miR-34a binding sites on mRNAs through the TargetScan databases. Among candidates, we found the 3’-UTR of human LDHA, a glycolysis key enzyme which catalyzes the convert of pyruvate to lactate, contains a putative miR-34a binding region (Fig. 6A). Consistent results from Spearman’s correlation analysis demonstrated overexpression of miR-34a significantly decreased the protein levels of LDHA (Fig. 6B). Importantly, the reverse correlation between miR-34a and LDHA mRNA expressions was found in gastric tumor specimen (Fig. 6C). To verify whether miR-34a could directly bind to the 3-UTR region of LDHA mRNA, a luciferase reporter assay was performed by co-transfecting control miRNA, miR-34a precursor and original-LDHA 3’UTR or nucleotides mutant LDHA 3’UTR (Fig. 6A) into HGC27 and AGS cells. Luciferase assay showed that miR-34a significantly decreased the luciferase activity of plasmid containing the wild type LDHA 3’-UTR but not the binding site mutant LDHA 3’-UTR. Taken together, the above results strongly demonstrated miR-34a could directly target 3’UTR of LDHA mRNA in gastric cancer cells and tumor. To test whether the miR-34a/LDHA-mediated glycolysis inhibition led to cisplatin sensitization, we transfected HGC27 cisplatin resistant cells with control miRNAs, or miR-34a precursor or miR-34a plus LDHA overexpression plasmid. Western blot results demonstrated co-transfection of miR-34a and LDHA successfully rescued the LDHA protein expressions (Fig. 7A). Furthermore, the glucose uptake (Fig. 7B) and lactate product (Fig. 7C) were significantly recovered in miR-34a and LDHA co-transfected cells. We next asked whether the miR-34a/LDHA axis was response for the miR-34a-mediated cisplatin sensitization by rescue experiments. As we expected, restoration of LDHA expression in miR-34a overexpressed cells contradicted the miR-34a-mediated cisplatin sensitization in gastric cancer cells (Fig. 7D and E). Importantly, recovering LDHA successfully restored the cisplatin resistance of HGC27 cisplatin resistant cells (Fig. 7F).
The acquired cisplatin resistance is through the SNHG7-miR-34a-glycolysis axis
Our results revealed SNHG7 negatively regulated miR-34a, suggesting that the miR-34a/LDHA binding was associated with the SNHG7-mediated cisplatin resistance. To test this hypothesis, lncRNA-SNHG7 was knocked down in HGC27 cells, or knock down SNHG7 plus anti-miR-34a treatment. We detected inhibition of endogenous SNHG7 significantly induced miR-34a expressions and downregulated LDHA expressions (Fig. 8A and B). Moreover, knockdown SNHG7 plus anti-miR-34a recovered the LDHA protein levels in HGC27 cells (Fig. 8A and B), suggesting SNHG7 could upregulate LDHA through suppressing miR-34a in gastric cancer cells. Furthermore, inhibiting the SNHG7 downregulation-induced miR-34a apparently recovered glucose uptake and lactate product (Fig. 8C and D). Consistent results in Fig. 8E showed GLUT1, HK2 and PKM2 which are glycolysis key enzymes were significantly downregulated by SNHG7 knocking down, however, such downregulation was rescued by further miR-34a inhibition (Fig. 8E). Finally, we transfected HGC27 cisplatin resistant cells with control, sh SNHG7 or sh SNHG7 plus anti-miR-34a, followed by treatments with increased concentrations of cisplatin. Expectedly, SHHG7 knockdown cells showed increased cisplatin sensitivity, however, such sensitization effect was further recovered by miR-34a inhibition, suggesting the SNHG7-mediated cisplatin resistance was through the miR-34a-LDHA axis.
Discussion
Gastric cancer, one of the most common types of cancer, represents a high incidence of cancer-related death. Although impressive clinical responses were observed from primary therapeutic approaches such as surgical resection with combination of chemotherapy, a large group of patients developed acquired chemoresistant cancer phenotypes, which limit the clinical applications of them [3, 4]. Thus, investigation of effective therapeutic treatments against chemoresistance is an urgent task. Previous studies revealed multiple signaling molecules which were known important regulators for gastric cancer cisplatin sensitivity, such as RAP2A which led to increase cisplatin resistance [22]. In addition, the GPR30-mediated epithelial-mesenchymal transition (EMT) also contributed to cisplatin resistance [23]. Accumulated evidence demonstrated that lncRNAs have critical regulators during cancer development by functioning as oncogenes or tumor suppressors [7]. This study revealed the oncogenic roles of lncRNA SNHG7 in gastric cancer. We demonstrated that SNHG7 was significantly upregulated in gastric cancer tissues and cells and positively associated with cisplatin resistance. SNHG7 was apparently upregulated cisplatin resistant gastric cancer cell line, indicating targeting SNHG7 might effectively sensitize gastric cancer cells to cisplatin. Although previous studies have described oncogenic roles of SHHG7 in multiple cancers, our results firstly demonstrated the functions of SNHG7 in cisplatin resistance of gastric cancer.
It is widely recognized that miRNAs play an essential role in regulating cancer cell malignant progresses [13]. Among them, miR-34a was known to be downregulated and act as a tumor suppressive miRNA in multiple cancers [24]. In addition, miR-129 and miR-424 have been reported to regulate cisplatin resistance of gastric cancer through direct targeting SMURF1 and MAPK3, respectively [25, 26]. Recent studies demonstrated lncRNAs exert its function to negatively regulate gene expressions through directly interaction with microRNAs as competing endogenous RNAs to further upregulate the targets of miRNAs. SNHG7 has been revealed to directly inhibit miR-5095, which targeted CTNNB1 mRNA to subsequently suppress the Wnt/
Cancer cells display adaption to tumor micro-environment by switching oxidative phosphorylation (OXPHOS) to less efficient glycolysis for not only producing energy but suppling building blocks for their high proliferation rate, a phenomenon called Warburg effect. An increasing number of recent studies have demonstrated that the Warburg effect favors tumor chemoresistance by diverse mechanisms [15]. Consequently, inhibition of glycolysis effectively decreased the cell proliferation rate and overcame drug resistance [16]. In this study, we consistently showed cisplatin resistant cells have upregulated glycolysis rate and lncRNA-NEAT1 expressions, compared with parental cells. Restoration of LDHA in miR-34a overexpressing gastric cancer cells recovers cisplatin resistance. Importantly, our date demonstrated that inhibition of NEAT1 significantly sensitized cisplatin resistant cells and such sensitization was further blocked by miR-34a inhibition, suggesting the NEAT1-promoted cisplatin resistance was through miR-34a/LDHA axis.
In summary, we report lncRNA-NEAT1 contributed to cisplatin resistance of gastric cancer cells. NEAT1 suppressed miR-34a expression, leading to a de-repression of LDHA. These findings emphasized an ceRNA role of SNHG7 against miR-34a via regulation of glycolysis during chemoresistance, presenting lncRNA-NEAT1 as a promising therapeutic target for overcoming cisplatin resistance of gastric cancer.
Footnotes
Conflict of interest
The authors in this study declare no conflicts of interest.
