Abstract
BACKGROUND:
The majority of ovarian cancer cases are diagnosed at an advanced stage with poor prognosis. This study evaluates autoantibodies against tumor antigens to identify candidate biomarkers for early detection of ovarian cancer in women at increased risk.
OBJECTIVE:
To assess the immunoreactivity of paraneoplastic antigens and tumor associated antigens with high-grade serous ovarian cancer (HGSOC) samples.
METHODS:
Five paraneoplastic antigens along with three tumor-associated antigens were evaluated with HGSOC patient serum samples. Validation screening was performed with
RESULTS:
TRIM21 achieved the highest sensitivity in the first validation screening of 33% with 100% specificity. Combining TRIM21 with NY-ESO-1, TP53, and PAX8 provided 67% sensitivity with 94% specificity, and 56% sensitivity at 98% specificity. These four markers resulted in 46% sensitivity with 98% specificity in the second validation cohort; TRIM21 achieved the highest individual sensitivity of 36%.
CONCLUSIONS:
Autoantibodies to TRIM21, NY-ESO-1, and TP53 may complement CA125 in screening of women at genetic risk for ovarian cancer.
Introduction
Early detection of ovarian cancer
Ovarian cancer is the fifth leading cause of cancer-related deaths in women. Stage I ovarian cancer is defined by localized cancer in the ovaries or fallopian tubes, with only 15% of cases diagnosed at this stage [1]. The majority of cases are diagnosed at an advanced stage, defined as stage II when the tumor has spread to organs within the pelvis, stage III which involves the peritoneal surface of the pelvis or abdomen and surrounding lymph nodes, and stage IV with metastasis beyond the abdominal cavity [1]. Worldwide, the 5-year age-standardized net survival for early stage ovarian cancer is 80%, which decreases to 30% for advanced disease [2].
Commonly, detection consists of a two-step test that involves measurement of circulating antigen CA125 either at a value
Screening in increased-risk populations has the potential to be beneficial given a higher incidence relative to the general population [3, 6, 7]. Those with a family history of ovarian cancer are at an increased-risk, including hereditary breast and ovarian cancer patients with BRCA1 and BRCA2 mutations which are frequently found in the germline of Type II high-grade serous ovarian cancers (HGSOC), and Lynch syndrome with mismatch repair gene mutations [8]. Seventy percent of all epithelial ovarian cancer cases are HGSOC; the present study evaluates a series of autoantibodies for use as diagnostic biomarkers in serum samples from patients with HGSOC [1].
Paraneoplastic autoantibodies associated with ovarian cancer
Paraneoplastic autoantibodies are associated with autoimmune syndromes that develop from the unre- gulated immune response initiated by tumor antigens. These antigens are also expressed by normal cells in the muscle or nervous system, resulting in a paraneoplastic syndrome indicated by neurological symptoms or muscle weakness. These syndromes present months to years prior to tumor diagnosis in an estimated 70% of cases; interestingly the autoimmune symptoms can resolve upon removal of the tumor, and return of symptoms can indicate tumor recurrence [9]. Cancers associated with paraneoplastic syndromes include thymoma, lung, breast, and ovarian adenocarcinomas, and groups of autoantibodies associated with various paraneoplastic syndromes are more specific for the tumor type than the syndrome [10]. Although paraneoplastic syndromes are rare, autoantibodies associated with paraneoplastic syndromes can be detected in patients with cancer who do not present with paraneoplastic syndromes [11]. For example, two paraneoplastic antigens associated with lung cancer, SOX2 and Hu-D, are included in an FDA-approved early detection autoantibody test for lung cancer [12]. It was previously reported that 36% of SCLC patients without paraneoplastic syndrome had reactivity to at least one SOX family member, and 16–25% of SCLC patients without a paraneoplastic syndrome harbored anti-Hu antibodies [11]. The aim of this study was to identify antigens to detect autoantibodies that may have individually low frequencies, but as a panel provide enhanced sensitivity to detect ovarian cancer. To that end, we evaluated a panel of five paraneoplastic antigens along with three tumor-associated antigens that were previously reported to have high immuno-reactivity or enhanced expression in ovarian cancer.
Two paraneoplastic syndromes that have been reported prior to diagnosis of ovarian cancer are polymyositis and paraneoplastic cerebellar degeneration [10]. Polymyositis is defined by muscle inflammation, and dermatomyositis by an accompanying skin rash. These myopathies are associated with the autoantibodies HARS, SRP-19, TRIM21, and Mi-2 [13]. Cerebellar degeneration results from damage to Purkinje cells and is associated with anti-neuronal antibodies (ANA), in particular CDR2 and CDR2L antigens [14]. CDR2 antibodies have previously been reported to have a low frequency, 2.3%, in ovarian cancer sera [15].
Serum sample patient population (
164), analyzed on western blot and ELISA
Serum sample patient population (
Patient population of serum samples analyzed on ELISA and western blot.
Of the eight antigens examined in a large-scale validation using western blot and ELISA, the antigens TRIM21, NY-ESO-1, TP53 and PAX8 combined to yield the highest sensitivity of 67% with 94% specificity, and 56% sensitivity with 98% specificity. A second validation study of those four antigens on western blot with an independent sample set maintained 98% specificity with 46% sensitivity.
Collection of patient samples
Samples were obtained from patients at Karmanos Cancer Institute, St. John Hospital and Oakwood Hospital in Detroit, MI, and at the Mayo Clinic, Rochester, MN. Additional specimens were provided by the Cooperative Human Tissue Network (CHTN) and Gynecologic Oncology Group specimen banks. All samples were collected prior to surgery or therapy. Healthy control sera were collected as part of a large-scale community outreach project. Blood was collected via venipuncture, centrifuged at 2,500 rpm at 4
For the Validation I and Validation II studies, the early and late stage HGSOC group is comprised of females age 18 and over diagnosed with epithelial ovarian cancer. In these studies we used serum collected from 19 early stage and 95 late stage serous ovarian cancer patients prior to treatment or surgery. The benign ovarian cyst group included 100 samples. One hundred healthy controls were self-reported to be free of cancer and potentially confounding benign conditions such as ovarian cysts, uterine fibroids, or endometriosis (Table 1). The HARS antigen was not processed with the Validation II sample set, with the exception of
Line blots
To determine which myositis-associated antigens were most useful as biomarkers for ovarian cancer, we evaluated line blots from Euroimmun (EUROIMMUN, Leubeck, Germany) and for onconeuronal antigens associated with paraneoplastic syndromes we evaluated line blots from Ravo Diagnostika (Ravo Diagnostika, Freiburg, Germany). Line blots were processed per manufacturer protocol, and incubated with a serum sample dilution of 1:100. The antigens included on the Euroimmun myositis profile line blots are: TRIM21, OJ, EJ, PL-12, PL-7, SRP, HARS, PM-SCL75, PM-SCL100, KU, and MI-2. The antigens included on Ravo Diagnostika paraneoplastic antigen line blots are: Hu-D, CDR2, RI, CRMP5, AMPHIPHYSIN, MA1, MA2, SOX1, and GAD65. Line blot reactivity was scored from 0–5 for each antigen by two independent observers blinded to the patient sample status.
Cloning strategy for recombinant expression vectors for protein expression
The tumor antigens were first PCR amplified using forward primers (containing a 6X Histidine tag and T7 tag) and reverse primers using cDNA template prepared from the ovarian cancer cell lines. The PCR products were column purified, restriction digested and ligated to pET-21b bacterial expression vector (EMD Millipore Corporation, San Diego, CA, USA). The ligated DNA was then transformed into BL21-DE3 strain and the positive colonies were sequenced. These expression vectors were employed for in vivo production of recombinant His-tagged proteins in BL21-DE3 bacterial strain. The SRP-19 expression plasmid was kindly gifted by Dr. Howard M. Fried, University of North Carolina at Chapel Hill [16]. The human TP53 (1–393) expression plasmid was a gift from Cheryl Arrowsmith (Addgene plasmid #24859; RRID:Addgene_24859; http://n2t.net/addgene: 24859) [17]. All cDNA expression plasmids used in this study were fully DNA sequenced.
Production and purification of recombinant His-tagged proteins
The BL21-DE3 bacterial cells bearing clones were grown overnight in 5 mL LB with 50
Western blot
Western blots were performed with 0.3–0.5
Western blot of healthy control serum and early stage HGSOC serum diluted at 1:300 with 7 antigens in Validation I study. A. Secondary antibody anti-HIS tag IgG loading control. B. Secondary antibody anti-human IgG. Scans quantified on Odyssey software; background-corrected integrated intensity of anti-human IgG antibody (IRDye800) normalized as ratio to anti-His tag antibody (IRDye700) per antigen.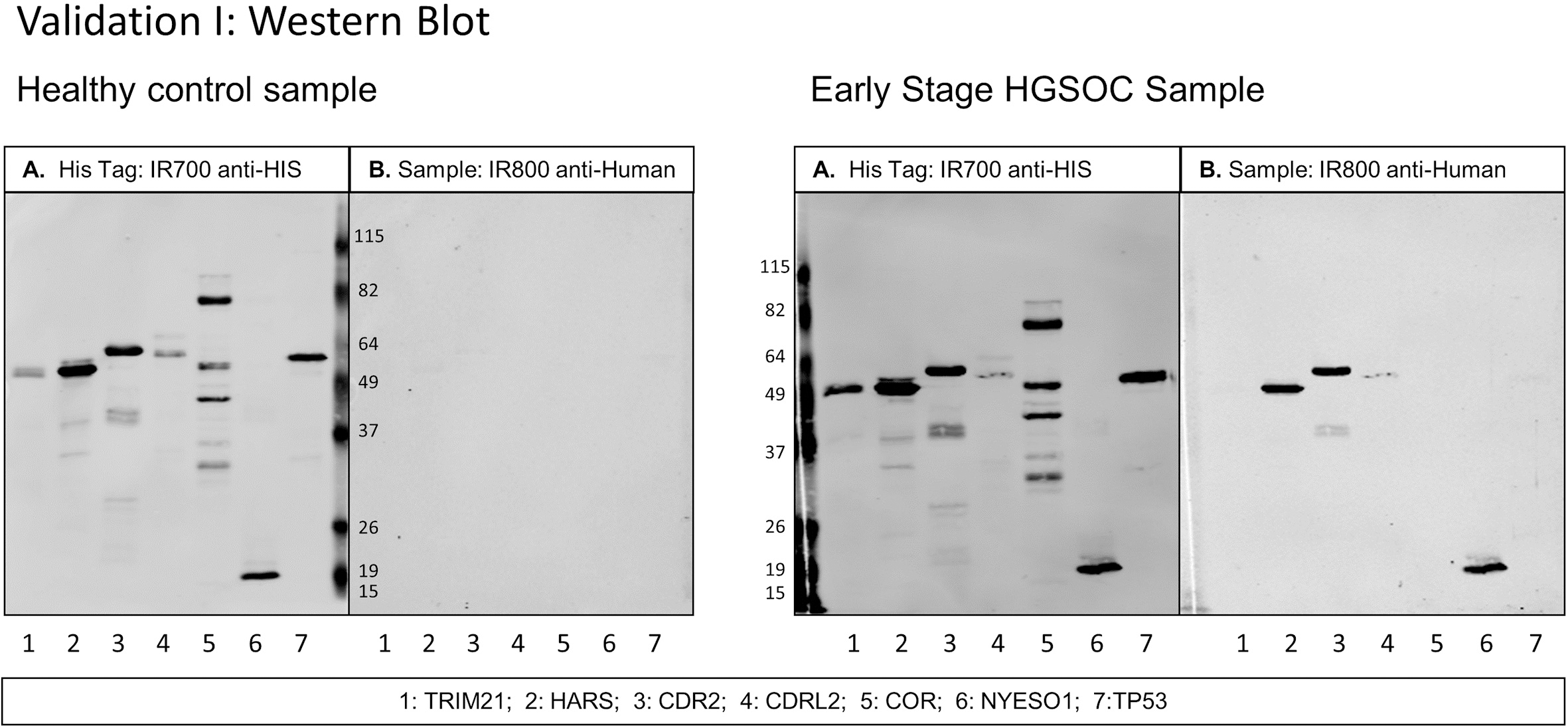
The serum set described in Table 1 was processed on ELISA as follows: purified antigens were coated in duplicate wells at concentrations from 0.3–1.5
A standard curve using serum with known reactivity to TRIM21 (The Binding Site, San Diego, CA, USA) at five dilutions ranging from 1:75 to 1:1200 was included on each plate to account for plate-to-plate and day-to-day variation. In addition, a pair of non-coated wells were blocked with donkey serum and incubated with each patient serum. These patient serum specific background values were subtracted from the antigen values for each patient sample. The serum set described in Table 1 was processed on ELISA. Each measurement was run in duplicate per plate, with four plates processed per day over 11 days. For 88 replicate measurements of the 1:75 dilution of the positive control standard curve, the coefficients of variation (CV) for the variance components in the ELISA assay are as follows: Intra-assay CV (within plate) 0.0281; Inter-assay CV (plate to plate) 0.0749; and CV day-to-day 0.0898.
Statistical methods
For the ELISA data analysis, to adjust for day-to-day variability among the 44 ELISA plates measured across 11 days, we utilized the positive control measurements to construct the standard curve, which was measured on all plates. The linear curve consisted of 6 dilutions of a positive control serum sample (The Binding Site, San Diego, CA, USA) measured against TRIM21 in duplicate. A linear mixed model with log optical density (OD) as the response; dilution (treated as a factor), protein (either TRIM21 or BKG) and their interaction as the fixed effects; plate nested within day as a random effect; an estimated correlation structure between duplicate observations; and unequal variance within each protein/dilution combination. From this model, we extracted the random effects terms to adjust the observed log (OD) values. After averaging the duplicates and exponenting resultant value, the appropriate control adjusted OD was subtracted to produce the normalized OD measurement. This normalized OD measurement was then used to compare to the values from the western blot analysis.
Description of paraneoplastic antigens evaluated against HGSOC sera
Description of paraneoplastic antigens evaluated against HGSOC sera
The 8 antigens selected for the large-scale screening on western blot and ELISA are: HARS, TRIM21, CORTACTIN, CDR2, CDR2L, TP53, NY-ESO-1, and PAX8.
Quantification of autoantibody binding on western blot for each sample was measured over 15 days for Validation I for each of the 8 antigens, and measured over 16 days for Validation II for 4 antigens, utilizing multiple membranes per day. Samples were randomized per category of HGSOC, benign, and healthy with each category evenly distributed per day, and labeled so that the experimenters were blinded to the sample category. The quantification values for both the IRDye700 and IRDye800 channels were log transformed after the addition of a small constant (0.01) to ensure all values were positive. The difference between the log transformed IRDye700 and IRDye800 values for each antigen for each sample is the pre-adjustment analysis metric. We employed a mixed model to develop adjustment factors to account for the between-day variability. We utilized the estimated day-specific random effects (from a model including the log difference as the response, antigen as the fixed effect and day as the random effect) to account for day-to-day variability. The log difference minus the day-specific random effect was used as the final analysis metric. Subsequently, for each antigen, the mean and standard deviation of the analysis metric was computed using the healthy samples. For each antigen, the mean and standard deviation was used to create standardized values [(Obs-Mean)/StdDev)]. This standardized value was used in the figures and tables presented in this manuscript.
Study design
Selection of 8 antigens from evaluation of full-length recombinant proteins and homologous epitopes on line blot, western blot, and ELISA
We previously identified ovarian cancer-related epitopes by screening unbiased random-peptide phage display cDNA libraries using ovarian cancer sera and found that many shared amino acid sequence homology with paraneoplastic antigens. Two phage-borne epitopes, 4B7 and 3A9, displayed 100% homology to the myositis associated antigens HARS and SRP-19, respectively [18]. Additionally, two ovarian cancer epitopes, 4F10 and 4E8, showed partial homology to the paraneoplastic antigens TRIM21 and Hu-D. An initial survey of autoantibodies to 20 paraneoplastic antigens in HGSOC sera [10, 19] was performed, including the four antigens to which ovarian cancer epitopes showed homology; HARS, SRP-19, TRIM21, and Hu-D. This study utilized two commercially available line blots in which recombinant antigens were spotted onto a membrane, with one line blot test consisting of myositis-associated antigens, and a second line blot test consisting of onconeuronal antigens associated with paraneoplastic neurological syndrome (Supplemental Fig. 1).
Sensitivity/specificity for TRIM21, NY-ESO-1, TP53, and PAX8, Validation I
Sensitivity/specificity for TRIM21, NY-ESO-1, TP53, and PAX8, Validation I
Sensitivity and specificity for TRIM21, NY-ESO-1, TP53, PAX8, HARS, CDR2L, CDR2, and CORTACTIN. Threshold (2)
A–D. Individual antigen plots for 4 antigens, TRIM21, NY-ESO-1, TP53, and PAX8 in Validation I. E. Maximum value of TRIM21, NY-ESO-1, TP53, or PAX8 in Validation I. The immunoreactivity as defined by the ratio of 800/700 is standardized for each antigen to provide the threshold of Mean 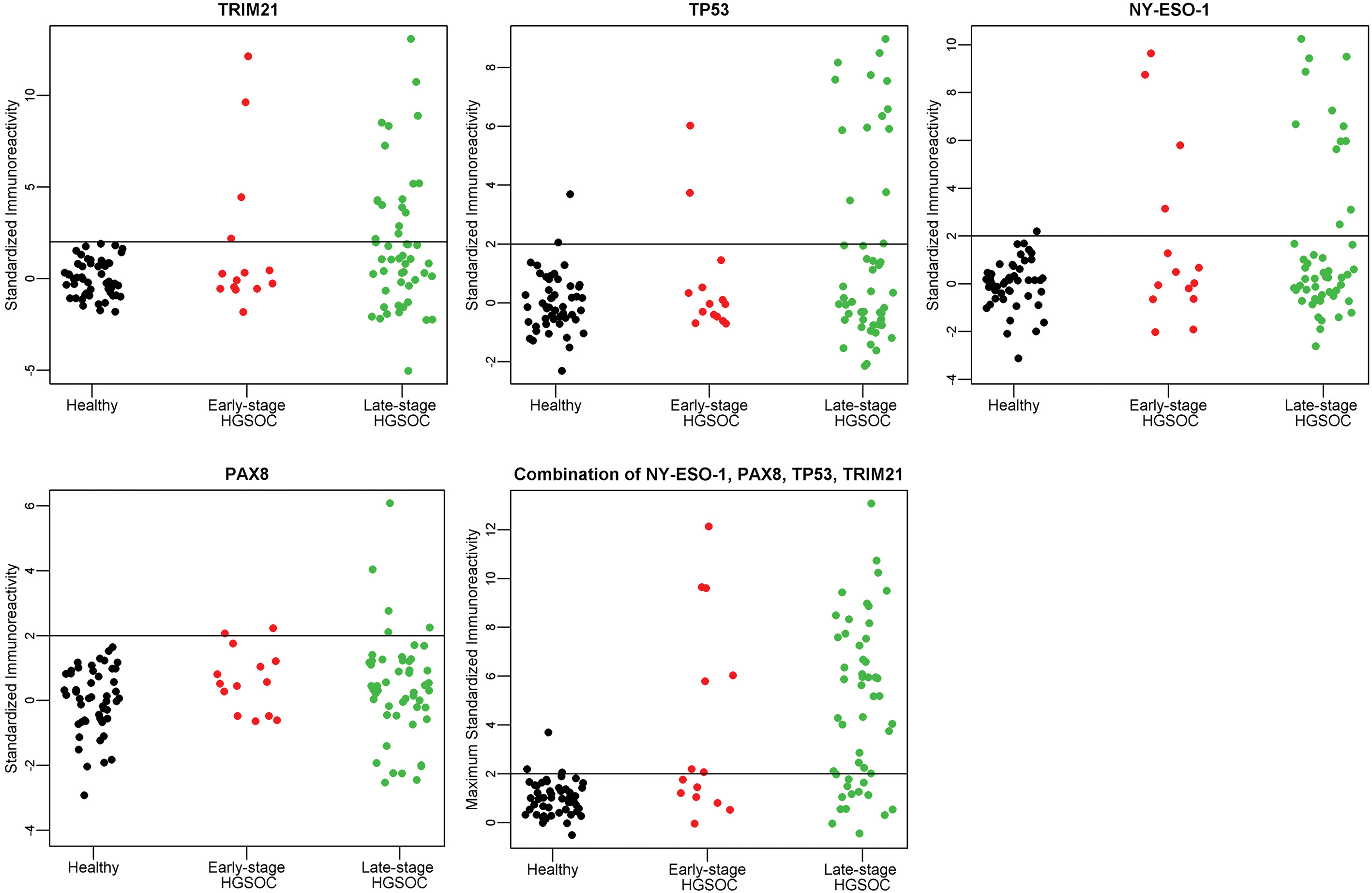
In addition, selected antigens were purified for further analysis on western blot and ELISA. The following antigens were either expressed in E. coli and purified in house, or obtained commercially for immunoreaction in western blot and ELISA assays: SRP-19, HARS, AARS, CDR2, HuD, TRIM21, TRIM33, CDR2L, CORTACTIN, CKB, NY-ESO-1, PAX8 and TP53. In addition, the four phage display epitopes with homology to paraneoplastic antigens were sub-cloned, expressed in E. coli and purified [20]. Results from western blot and ELISA screening with a serum set of
To avoid experimental bias, an independent sample set of 164 samples that had not been used to identify the biomarkers initially was used for validation of the panel of antigens listed above. The sample population described in Table 1 consists of 50 healthy control samples, 50 benign ovarian cyst samples, 50 late stage HGSOC samples, and 14 early stage HGSOC samples.
ELISA and western blot correlation
A threshold based on the 50 healthy control values for each antigen defined positive results. The assay cutoff for both the ELISA assay and the western blot screening is defined as: Mean
TRIM21 provides highest sensitivity as an individual marker in HGSOC samples
Sensitivities and specificities as calculated by the standardized thresholds based on mean
Serum sample patient population (
150), analyzed on western blot
Serum sample patient population (
Patient population of serum samples analyzed on western blot.
Western blot of healthy control serum and early stage HGSOC serum diluted at 1:300 with 4 antigens in Validation II study. A. Secondary antibody anti-HIS tag IgG loading control. B. Secondary antibody anti-human IgG. Scans quantified on Odyssey software; background-corrected integrated intensity of anti-human IgG antibody (IRDye800) normalized as ratio to anti-His tag antibody (IRDye700) per antigen.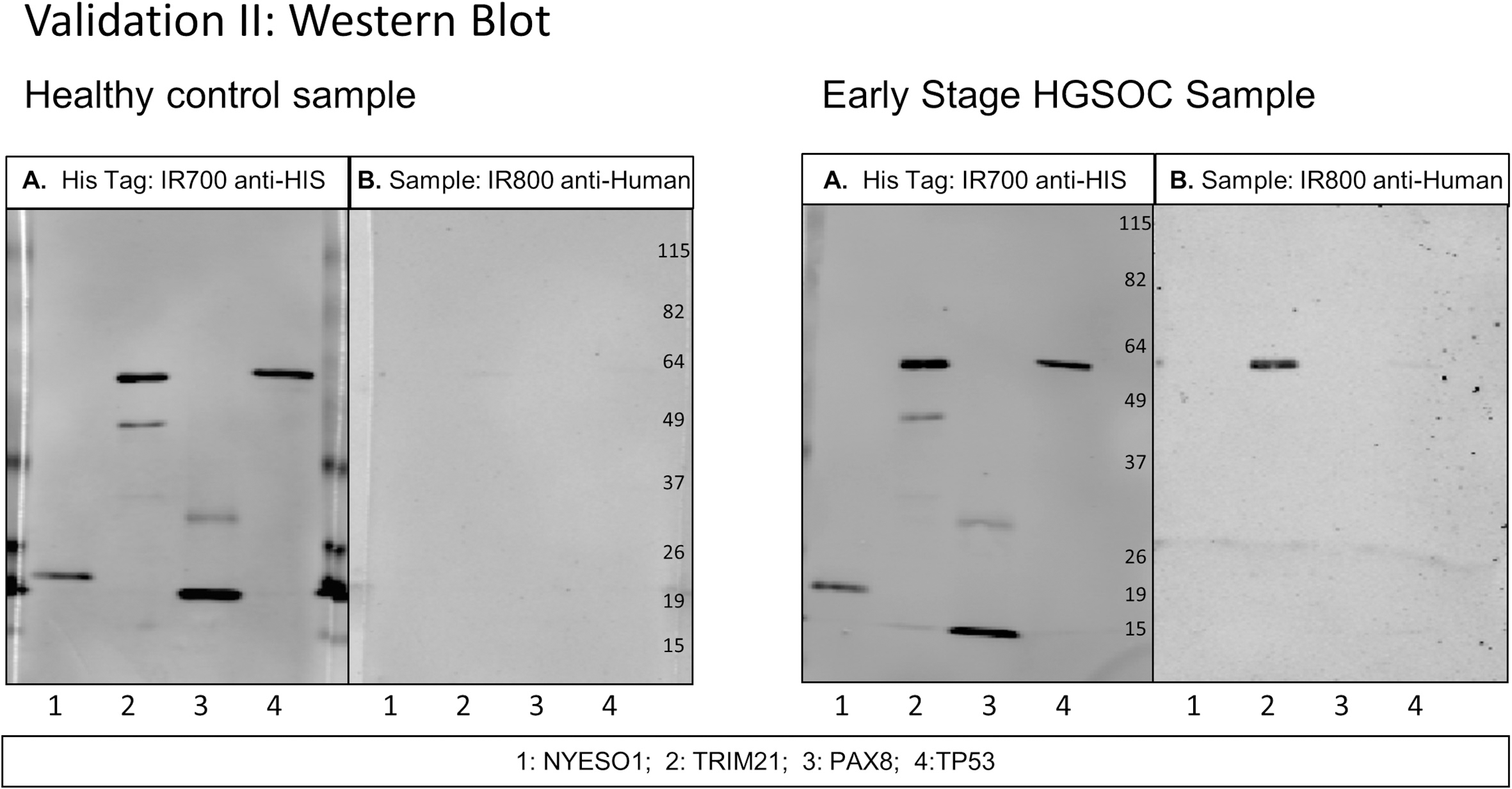
Sensitivity/specificity for TRIM21, NY-ESO-1, TP53, and PAX8, Validation II
Sensitivity and specificity for TRIM21, NY-ESO-1, TP53, and PAX8. Threshold (2)
A–D. Individual antigen plots for 4 antigens, TRIM21, NY-ESO-1, TP53, and PAX8 in Validation II. E. Maximum value of TRIM21, NY-ESO-1, TP53, or PAX8 in Validation II. The immunoreactivity as defined by the ratio of 800/700 is standardized for each antigen to provide the threshold of Mean 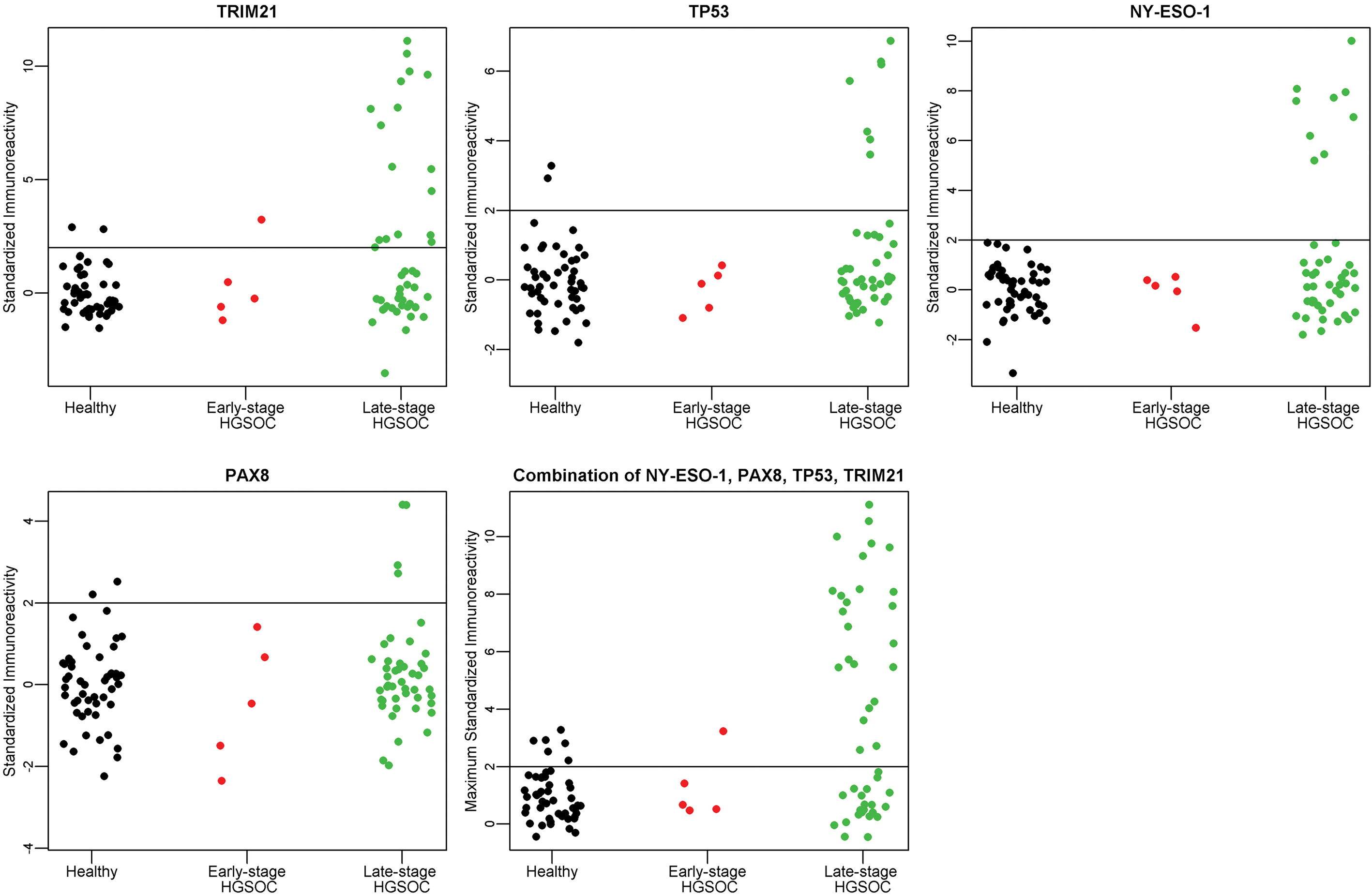
ROC curve analysis for the combination of the 4 markers: TRIM21, NY-ESO-1, TP53, and PAX8 in A. Validation I and B. Validation II.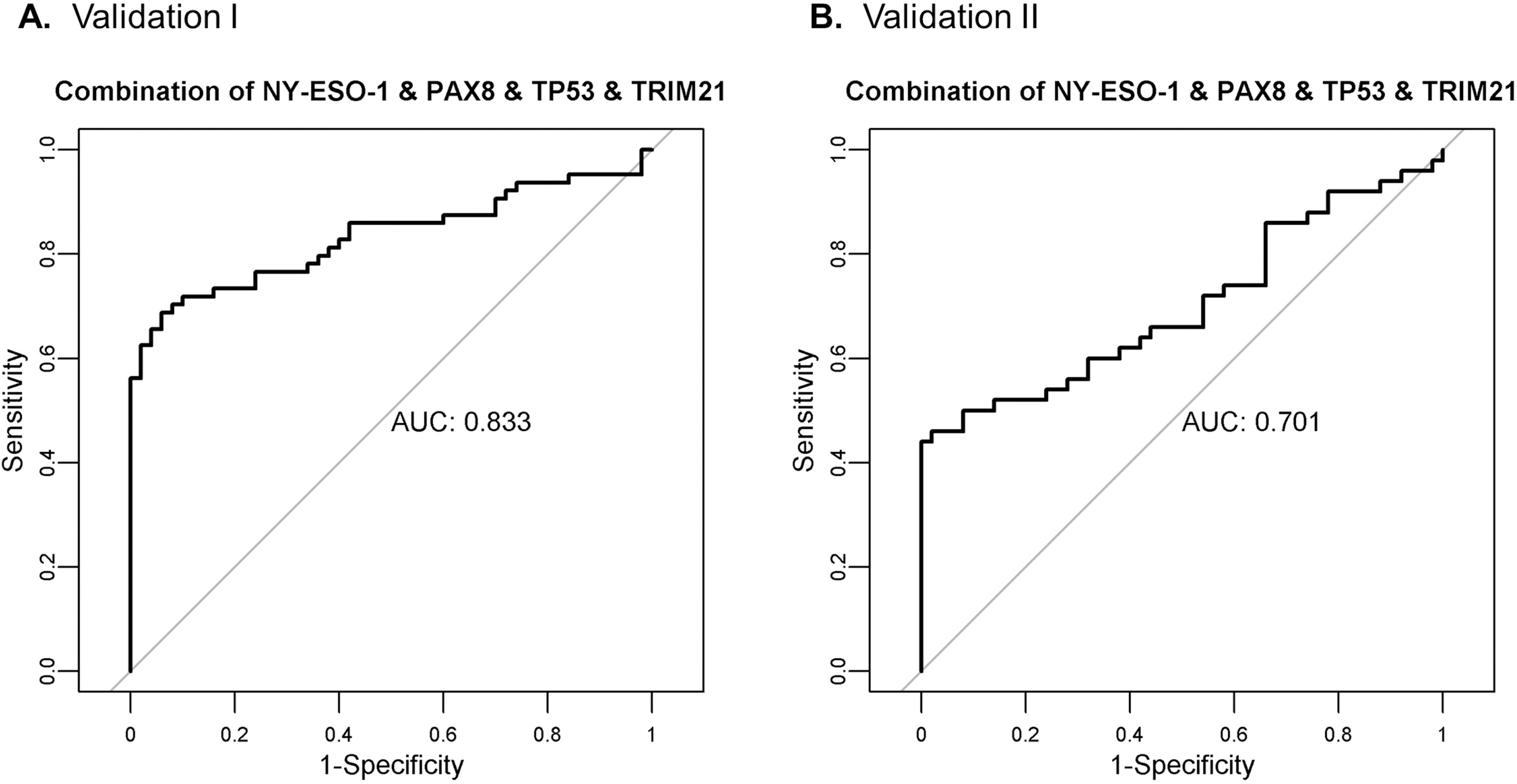
The sample set included 14 early stage samples. Individually, NY-ESO-1 and HARS detected 4 early-stage HGSOC samples (29%), TRIM21 detected 3 early-stage HGSOC samples (21%), and TP53 and PAX8 each detected 2 early-stage HGSOC samples (14%). The combination of the 5 markers yielded 50% sensitivity for detecting early stage HGSOC with 94% specificity to discriminate healthy controls. Respective sensitivities and specificities within each sample group are shown in Table 3. The combination of markers PAX8, HARS, NY-ESO-1, TP53 and TRIM21 detected 7/14 early-stage HGSOC samples. Among these five antigens, all 7/7 of the positive early stage HGSOC samples were reactive with 2 or more antigens, compared with 12/37 positive late stage HGSOC samples reacting with 2 or more antigens.
Panel of 4 markers achieves highest performance for HGSOC autoantibody detection: TRIM21, NY-ESO-1, TP53, and PAX8
The combination of the four markers TRIM21, NY-ESO-1, TP53, and PAX8 detected 42/64 HGSOC samples. With 94% specificity for the healthy control population, we can achieve 67% sensitivity (threshold
Validation II: Panel of 4 markers, TRIM21, NY-ESO-1, TP53, and PAX8, validated on independent serum set,
150
A separate serum set consisting of 50 healthy, 50 benign ovarian cyst, and 50 high-grade serous ovarian cancer samples was used for an independent validation of the 4 markers: TRIM21, NY-ESO-1, TP53, and PAX8. The patient population is described in Table 4. Patient samples were evaluated on western blot, shown in Fig. 3. In this sample set, PAX8 did not complement the 3 markers TRIM21, NY-ESO-1, and TP53. Standardized patient reactivity for each antigen is shown in Fig. 4. This validation screening of the 3 markers TRIM21, NY-ESO-1, and TP53 maintained a specificity of 98% with a sensitivity of 46% as described in Table 5. The sensitivity of the combination of the 4 markers: TRIM21, NY-ESO-1, TP53, and PAX8 for all HGSOC cases was lower in the second validation study, 50% vs. 67% with positive threshold
Although the HARS antigen was not evaluated on this sample set, the 5 early stage HGSOC samples were processed with the HARS protein resulting in 1/5 positive samples. TRIM21 was the only other antigen positive in the 5 early stage HGSOC samples; combining TRIM21 and HARS resulted in 2/5 early stage HGSOC samples positive in this set.
Discussion
Early detection of ovarian cancer has the potential to improve patient outcome. In the UK Familial Ovarian Cancer Screening Study phase II trial, 4, 348 women at high-risk for ovarian cancer were screened for CA125 every 4 months with yearly transvaginal ultrasound, which resulted in a significant stage shift in diagnosis. Of the 19 total cases detected during the 5 years of screening, 10 were stage I–II [7]. Two prospective trials from the Cancer Genetics Network and the Gynecologic Oncology Group together screened 3, 962 women at high-risk for ovarian cancer with CA125 screening every 3 months, followed by transvaginal ultrasound upon increases in CA125 above the patient’s baseline. In these trials, 3 of the 6 incident cases were stage I–II [3]. Autoantibodies to tumor antigens, produced at small tumor volumes, can be combined with serum screening of CA125 to improve sensitivity in early detection.
We previously used phage-display screening to identify autoantibody biomarkers for both early detection and recurrence of ovarian cancer [18, 20]. Two of our identified markers were epitopes for myositis-associated antigens, HARS and SRP-19. This study evaluates of a set of myositis associated and onconeuronal autoantigens for detection of autoantibodies in serum from ovarian cancer patients without a known paraneoplastic syndrome. We note that in our previous work, the antigens CDR2, TRIM21, and HARS were evaluated for reactivity to antibodies in sera from patients experiencing a recurrence of their HGSOC; however, in that study the levels of autoantibody were considered relative to a negative control antigen for each individual patient at three time points, with the goal of monitoring disease recurrence [19]. With the goal of early detection in the current study, autoantibodies were considered relative to healthy and benign control serum samples; therefore, this study reflects differences in the frequency of paraneoplastic autoantibodies in HGSOC sera relative to sera from healthy controls and women with benign ovarian cysts. As low-grade serous ovarian cancer (LGSOC) can develop step-wise from ovarian serous cystadenoma, we evaluated 22 samples from patients with early stage LGSOC with the antigens TRIM21, NY-ESO-1, TP53, PAX8, and HARS [21, 22]. We found that 1/22 early stage LGSOC samples were positive for TP53 autoantibodies, and 1/22 samples were positive for TRIM21 autoantibodies (data not shown).
Cortactin (COR), a novel biomarker for myositis, did not show HGSOC specificity [23]. We found that CDR2 and CDR2L had reactivity with healthy and benign samples. Antibodies to CDR2 have previously been reported to have low frequency in ovarian cancer, we observed 5/50 late stage and 2/14 early stage samples positive for CDR2 autoantibodies at the threshold (2)
TRIM21 was identified as a novel biomarker for ovarian cancer, with the highest individual sensitivity of 33% for all HGSOC samples at 100% specificity compared to healthy controls in Validation I, and 36% sensitivity at 96% specificity in Validation II. TRIM21 has previously been reported as a biomarker for esophageal squamous cell carcinoma and basal-like breast cancer [27, 28]. In a study of HGSOC serum samples that utilized high-density programmable protein microarrays containing 10,247 antigens, TRIM21 was identified as one of the top 39 candidate tumor antigens, which passed three rounds of serum screening in independent sample sets [4]. In the present study, the combination of TRIM21 with TP53, NY-ESO-1, and PAX8 provided a sensitivity of 67% in Validation I sample set and 50% sensitivity in the Validation II sample set. Somatic mutations in TP53 are found in in 96% of HGSOC cases [29]; autoantibodies to TP53 in HGSOC can be detected against the wild type protein as a polyclonal response [30]. NY-ESO-1 is an immunotherapy target for ovarian cancer with numerous trials evaluating vaccines targeting NY-ESO-1 as well as adoptive transfer of NY-ESO-1 specific T-cells [31]. PAX8 is expressed in the majority of HGSOCs [32]. The present study is the first to detect anti-PAX8 autoantibodies. The full-length PAX8 protein is 450 aa; the recombinant PAX8 protein used in this study consists of the amino acids 1–287, which are present on isoforms C-E.
In three recent studies within the population of women with an increased risk of ovarian cancer that evaluated CA125 using the ROCA algorithm at intervals of 3 or 4 months, an increase in detection sensitivity for early stage tumors was observed [3, 7]. The goal of identifying autoantibody biomarkers is both to complement CA125, and to provide lead-time to CA125 detection. The serum set used in this study had limited samples with data for CA125 values. Autoantibodies to TP53 have been shown by Yang et al. to be elevated in pre-diagnostic patient samples up to 11 months before detection of CA125, and in samples taken 23 months before diagnosis for CA125-negative cases [33]. In our cohort anti-TP53 antibodies were present in 2/19 early stage samples. Additional markers such as TRIM21, HARS, NY-ESO-1, and PAX8, which detected in combination 9/19 early stage samples, may show improved lead-time, thus addressing the ultimate goal of a diagnosis at an earlier stage. Determining whether TRIM21, HARS, NY-ESO-1, and PAX8 autoantibodies are also detectable in addition to TP53 in pre-diagnostic sera will be a critical step in evaluating these biomarkers for clinical use. Early detection of HGSOC has potential to provide a mortality reduction [34].
Conclusions
A panel of autoantibody biomarkers can be useful in complementing current screening methods for the early detection of ovarian cancer in women with an increased genetic risk of ovarian cancer. The presence of paraneoplastic antibodies in HGSOC sera was evaluated. In two independent sample sets, Validation I with
Footnotes
Acknowledgments
This project was supported by The Barbara and Fred Erb Chair in Cancer Genetics. Laura Hurley was supported by the Ruth L. Kirschstein National Research Service Award T32-CA009531, the DeRoy Testamentary Foundation Predoctoral Fellowship in Cancer Research, and Wayne State University Rumble Graduate Research Assistantship. We would like to acknowledge Alyssa Moskala for her assistance with preparation of figures. A special thanks to the patients and healthy volunteers who donated serum and tissue for the study. This work could not be done without their willingness to participate in research.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-190988.
