Abstract
In this study, enzyme-linked immunosorbent assay has been used to examine the frequencies of serum autoantibodies against two candidate tumor-associated antigens intensively selected from the Human Protein Atlas database, in combination with 13 tumor-associated antigens available from our lab in sera from 44 OC patients and 50 normal healthy controls. Conventional evaluation (mean + 3SD as the cutoff value to determine a positive reactivity), receiver operating characteristic curve analyses, and classification tree analysis were further used to evaluate the diagnostic performance of autoantibodies against these tumor-associated antigens (anti-tumor-associated antigens) in ovarian cancer. For single anti-tumor-associated antigen, when the cutoff values were set as mean + 3SD of normal healthy controls, NPM1, MDM2, PLAT, p53, and c-Myc could achieve sensitivity higher than 20% at 98% specificity. Combinational utilization of autoantibodies against MDM2, PLAT, NPM1, 14-3-3 Zeta, p53, and RalA achieved the optimal diagnostic performance with 72.7% sensitivity at 96% specificity. Receiver operating characteristic curve analysis showed that the area under the receiver operating characteristic curves of autoantibodies against c-Myc, NPM1, MDM2, p16, p53, and 14-3-3 Zeta were greater than 0.80. This indicated that these tumor-associated antigens held high potential to serve as diagnostic biomarkers in ovarian cancer detection. Decision tree analysis indicated that anti-c-Myc held high potential in the detection of ovarian cancer. Further studies are warranted to validate the diagnostic performance of these anti-tumor-associated antigens with high area under the receiver operating characteristic curve, including autoantibodies against c-Myc, MDM2, PLAT, NPM1, 14-3-3 Zeta, p53, and RalA.
Introduction
Although the rank of estimated new cases and the mortality rate have slightly decreased, ovarian cancer (OC) remains fifth in the cause of all cancer deaths and it is the most malignant gynecologic cancer among women.1,2 Most OC patients (61%) are diagnosed at distant stage when treatment are less curable. Patients with localized OC have a favorable 5-year survival rate compared with patients with distant metastasis.2,3 Many studies have demonstrated that a unique group of autologous cellular antigens, so-called tumor-associated antigens (TAAs), could trigger the immune system to generate autoantibodies in cancer patients.4,5 However, some of the TAAs may rapidly degrade or be cleared and only last for a limited time in the circulating system.4,6 The signals of TAAs can be amplified by their corresponding autoantibodies, and these autoantibodies are usually more stable and can last for a long time in patients’ sera compared with TAAs; thus, they have attracted more attention than other biomarkers including TAAs themselves.4,6 In our previous study, we have systematically reviewed the development in using serum TAAs or anti-TAAs as biomarkers in the diagnosis of OC. 7 CA125, HE4, OVA1 test, and Risk of Ovarian Malignancy Algorithm (ROMA) are currently used in clinical practice to evaluate the risk of OC in women with pelvic mass. Although the risk of ovarian cancer algorithm (ROCA) has been reported being able to detect more OC patients than one-time serum CA125 using the single-threshold rule in the UK Collaborative Trial of Ovarian Cancer Screening (UKCTOCS) cohort study, the positive predictive value (PPV) of the ROCA test is still limited to make it a screening tool for average risk women. 8 So, the search of OC biomarkers needs to be continued.
The Human Protein Atlas (HPA) offers protein expression database in normal human tissues as well as in cancer tissues, and it is a good resource for systematic biomarker discovery. 9 Our previous studies have demonstrated that a higher level of two proteins, MDM2 and c-Myc, which were aberrantly overexpressed in lung cancer tissues, was positively associated with their corresponding serum autoantibodies in lung cancer patients. 10 Thus, we hypothesize that aberrantly overexpressed proteins in human cancer tissues might be candidate TAAs, and their corresponding autoantibodies might be detectable in sera from cancer patients. In this study, we have selected two proteins as candidate TAAs, mesothelin (MSLN) and plasminogen activator, tissue type (PLAT or tPA), which are overexpressed in more than 75% OC tissues but have low or no expression in normal ovary tissues and other types of normal tissues according to the HPA database (version 13). Autoantibodies against these two candidate TAAs might be of value in the detection of OC.
It is reported that CA125 can bind to MSLN in a specific manner, and this binding can mediate cell–cell adhesion in OC cell line (OVCAR-3), suggesting that MSLN may play a role in OC metastasis by binding to MUC16/CA125. 11 MSLN can be shed from cancer cells to human sera. 12 MSLN was explored as a potential biomarker for mesothelioma diagnosis, prognosis, and monitoring.13–15 As reviewed in our previous study, many researchers have also explored the potential diagnostic performance of MSLN in OC detection.16–18 PLAT is involved in cell migration and tissue remodeling.19–21 There is a close association between neoplasia and increased fibrinolytic activity. As early as in the 1980s, PLAT was reported to be found in the sera of several malignancies, and it was found in all types of OCs.22,23 Since then, several studies have tried to explore the diagnostic performance of PLAT in several human malignancies including OC.24,25 Since autoantibodies are more stable and last longer than TAAs themselves, it is meaningful to explore the diagnostic performance of autoantibodies against MSLN and PLAT in OC detection.
Previous studies have shown that the lack of sensitivity for single anti-TAA could be overcome by using a panel of intensively selected TAAs or anti-TAAs.4,5,26–29 Another 13 anti-TAAs, including autoantibodies against p53, p16/INK4A, HCC1.4/RBM39, NPM1, Survivin/BIRC5, RalA, MDM2, Cyclin B1/CCNB1, c-Myc, 14-3-3 Zeta/YWHAZ, IMP1/IGF2BP1, p62/IGF2BP2, and Koc/IGF2BP3, which have been extensively studied in our lab, were also included in this study. In brief, P16/INK4A participates in cell cycle regulation, 30 P53 can regulate gene expression and prevent mutation of the genome, 31 and HCC1.4/RBM39 is a type of RNA-binding protein and can interact with estrogen receptor alpha. 32 NPM1 can promote tumor growth by inactivation of tumor suppressor P53/ARF pathway. 33 Survivin/BIRC5 has negative regulation of apoptosis or programmed cell death by inhibiting caspase activation. 34 RalA promotes anchorage-independent growth in cancer cells and functions as molecular switch to activate cell division and transport.35,36 MDM2 is a negative regulator of the P53. 37 Cyclin B1 is a type of regulatory protein that involved in mitosis. 38 C-Myc is a mutated version of Myc that involved in cell proliferation and results in the formation of tumor. 39 14-3-3 Zeta/YWHAZ is a central hub protein for many signal transduction pathways. 40 IMP1/IGF2BP1, p62/IGF2BP2, and Koc/IGF2BP3 belong to RNA-binding protein, and they are known to be involved in RNA synthesis and metabolism. 41 In this study, we try to evaluate and compare the diagnostic value of autoantibodies against two candidate TAAs (PLAT and MSLN) and another 13 anti-TAAs from our previous studies.
Materials and methods
Patients and sera
Sera from 44 OC patients and 50 healthy individuals were obtained from the sera bank in the Cancer Autoimmunity Research Laboratory from the University of Texas at El Paso (UTEP). These sera were originally provided by our clinical collaborators. All OC sera were collected from the patients prior to their treatment. Normal human sera were collected during annual health examinations from adults with no obvious evidence of malignancy. Patients’ name and identification number were blinded to the investigator, and clinical information for the sera used in this study was not available. This study was approved by the institutional review board of the UTEP.
Expression and purification of recombinant TAAs
PLAT and MSLN full-length proteins were commercially purchased from Abcam Inc. (Cambridge, MA) and BioLegend (San Diego, CA), respectively. Another 13 recombinant proteins used in this study were purified as described in our previous studies.5,26,42 These purified proteins were coated on enzyme-linked immunosorbent assay (ELISA) and plated to detect their corresponding serum autoantibodies.
ELISA
Serum autoantibodies were detected by ELISA as previously described.5,26,42 In brief, the 96-well ELISA plates were coated with 100 µL recombinant proteins overnight in the cold room (4°C). The final concentrations of coating proteins were 0.5 µg/mL (PLAT and MSLN), 1.0 µg/mL (p53, p16, Koc, RalA, and Cyclin B1), 1.5 µg/mL (MDM2 and c-Myc), and 2.0 µg/mL (HCC1.4, IMP1, NPM1, Survivin, 14-3-3 Zeta, and p62) diluted in phosphate-buffered saline (PBS). The plates were subsequently blocked with gelatin post-coating solution for 2 h at room temperature and reacted with human sera diluted to 1:100 at 37°C for 1 h. Then, the plates were incubated with horseradish peroxidase (HRP)-conjugated goat anti-human immunoglobulin G (Caltag Laboratories, Burlingame, CA) diluted to 1:4000 for 1 h as a secondary antibody, followed by washing with PBS containing 0.05% Tween-20 (PBST) and PBS. The substrate 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt (Sigma-Aldrich, St. Louis, MO) was used as the detecting agent. The optical density (OD) value of each well was read at 405 nm, and the cutoff value for determining a positive reaction was designated as the mean absorbance of the 50 normal sera + 3 standard deviations (mean + 3SD).
The HPA
The HPA portal is a publicly available database with protein expression data in 44 different normal human tissues and 20 different cancer types, as well as 46 different human cell lines. 9 According to the HPA database (version 13), we have selected two proteins, PLAT and MSLN, which were overexpressed in more than 75% OC tissues but have a low expression rate in normal tissue (less than 10%), to test whether serum autoantibodies against these two candidate TAAs can be used as OC biomarkers.
Statistical considerations
To determine whether the frequencies of autoantibodies against 15 TAAs in sera from patients with OC were significantly higher than in sera from normal individuals, data were analyzed using the chi-square test. Methods for calculating the sensitivity/specificity and positive or negative likelihood ratios (+LR/−LR) were based on the methodology provided in the book. All p values were two-sided, and those less than 0.05 were considered statistically significant. All statistical analysis was carried out using the IBM SPSS Statistics 21.0 (IBM Corp., Armonk, NY). Area under the receiver operating characteristic curves (AUCs) were generated by using GraphPad Prism 5.01 (GraphPad Software, San Diego, CA). The program RPART, freely available on the Internet, implemented in R, was used to generate the decision tree. Pruning the tree (to correct for overtraining) was undertaken using the 1-SE rule described by Breiman et al. 43
Results
Frequency of serum autoantibodies against 15 TAAs in OC and NHC
Serum autoantibodies against two candidate TAAs and 13 TAAs were detected by ELISA as previously described. In Table 1, it is shown that autoantibodies against certain TAAs can discriminate OC patients from normal healthy controls (NHCs) even when the cutoff value was set at mean + 3SD of OD values of NHCs. For single anti-TAA, autoantibodies against NPM1, MDM2, PLAT, p53, and c-Myc have achieved more than 20% sensitivity at very high specificity when used alone, while some anti-TAAs have very low sensitivity.
Frequency of autoantibodies against 15 anti-TAAs in OC and NHCs by ELISA.

TAA: tumor-associated antigen; ELISA: enzyme-linked immunosorbent assay; OC: ovarian cancer; NHC: normal healthy control; Se.: sensitivity; Sp.: specificity.
Cutoff value = mean + 3SD of NHCs.
Sensitivity and specificity are highly influenced by the selection of cutoff value. In contrast to this, the AUC is independent of cutoff value and more precise for screening purposes. This greatly facilitates comparison of the discriminating power of different biomarkers. So, we performed receiver operating characteristic (ROC) curve analysis for all these biomarkers in this study. The results are shown in Table 2 and Figure 1. ROC curve analysis revealed that the AUCs of autoantibodies against c-Myc, NPM1, MDM2, p16, p53, and 14-3-3 Zeta were all greater than 0.80. When the cutoff values are set according to the ROC curve analysis, it is shown that the sensitivity has increased without losing too much specificity. This indicates that these anti-TAAs hold high potential to serve as diagnostic biomarkers in OC detection. It is notable that anti-c-Myc is probably the most promising biomarker for OC detection with an AUC of 0.867, and 65.9% sensitivity at 92.0% specificity. As shown in Table 2, autoantibodies against Koc and p62/IMP2 seemed to have no diagnostic value in discriminating OC from NHCs since the p values of these two biomarkers were both greater than 0.05. These two biomarkers were excluded for further analysis.
Diagnostic value of autoantibodies against single anti-TAA in ovarian cancer.
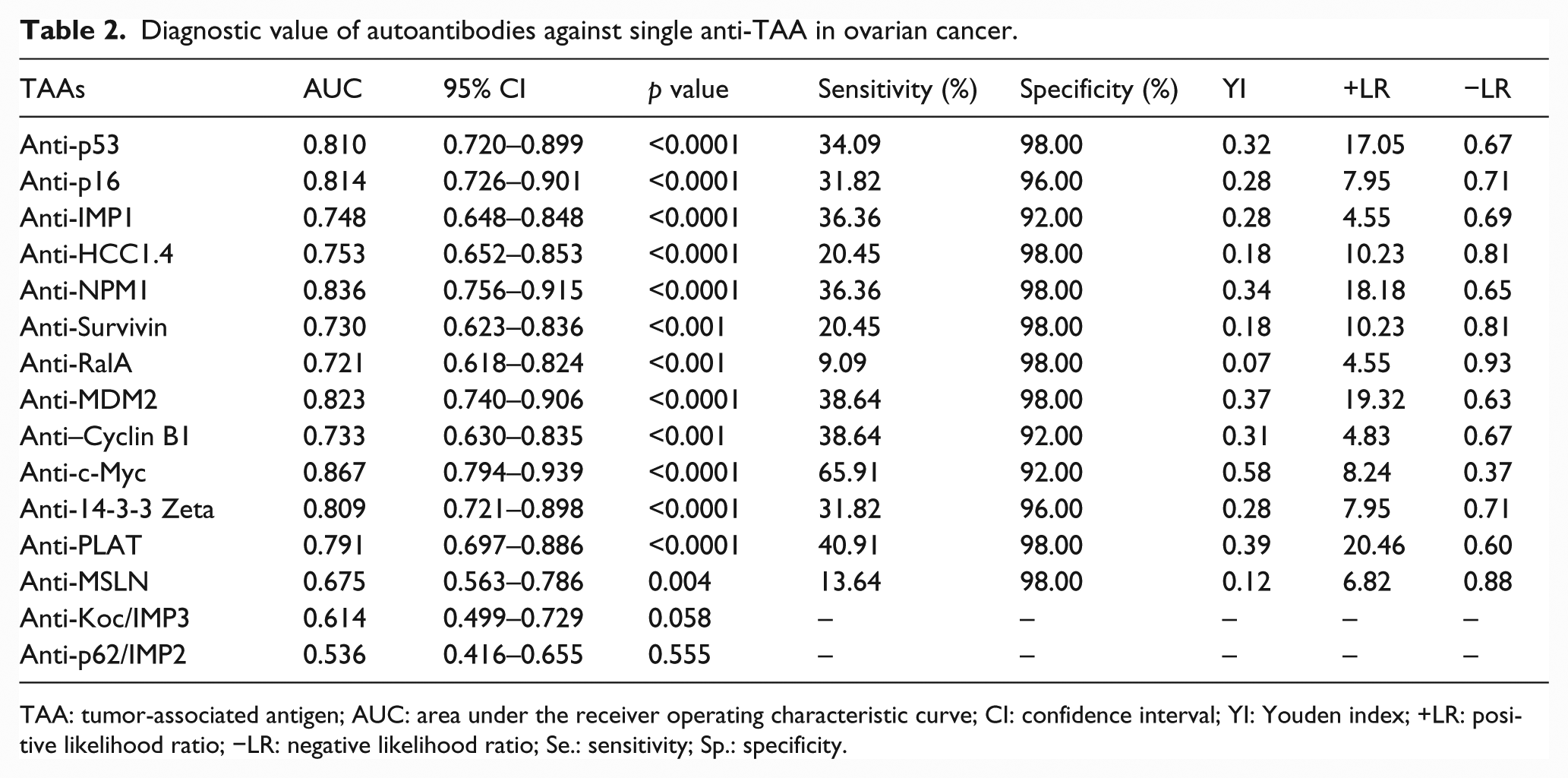
TAA: tumor-associated antigen; AUC: area under the receiver operating characteristic curve; CI: confidence interval; YI: Youden index; +LR: positive likelihood ratio; −LR: negative likelihood ratio; Se.: sensitivity; Sp.: specificity.

Comparison of ROC curves of different anti-TAAs. Autoantibodies against c-Myc, NPM1, MDM2, p16, p53, and 14-3-3 Zeta show AUC values greater than 0.800. Autoantibodies against PLAT and MSLN show AUC values of 0.791 and 0.675, respectively.
Evaluation of the diagnostic values of a panel of 15 anti-TAAs in the immunodiagnosis of OC
As described above, sensitivity for single anti-TAA varies from 0% to 27.27% when the cutoff value is set as mean + 3SD of NHCs. Our previous studies as well as other groups have reported that higher sensitivity might be obtained without losing too much specificity by using a customized mini-array or combination of multiple anti-TAAs in the diagnosis of cancers. As shown in Table 3, with successive addition of anti-TAAs to a total of six anti-TAAs (autoantibodies against MDM2, PLAT, NPM1, 14-3-3 Zeta, p53, and RalA), the optimal sensitivity increased to 72.73% at 96% specificity. When more anti-TAAs were added to the panel, the sensitivity stopped to increase and the specificity began to drop. It indicates that the panel of these six anti-TAAs can distinguish OC from NHCs.
Combinational utilization of anti-TAAs in the detection of OC.

TAA: tumor-associated antigen; OC: ovarian cancer; NHC: normal healthy control; Se.: sensitivity; Sp.: specificity.
Cutoff value = mean + 3SD.
Classification tree for discriminating OC from NHCs
In order to check whether optimal subsets of the 13 anti-TAAs for discriminating OC from NHCs exist, we used classification tree analysis as described in the previous study. 44 Classification tree for discriminating OC from NHCs is generated by using the OD values of each of the 13 anti-TAAs for each individual and further pruned according to the 1-SE rule described by Breiman et al. 43 (Supplementary Figure 1). The result of the pruned tree is shown in Figure 2. As shown in Figure 2, anti-c-Myc is the initial node and the only determinant for OC tree. This result is consistent with the result of ROC curve analysis that anti-c-Myc has the highest AUC value in OC detection. Further ROC curve analysis shows that the AUC of the pruned tree is 0.815 (Figure 3).
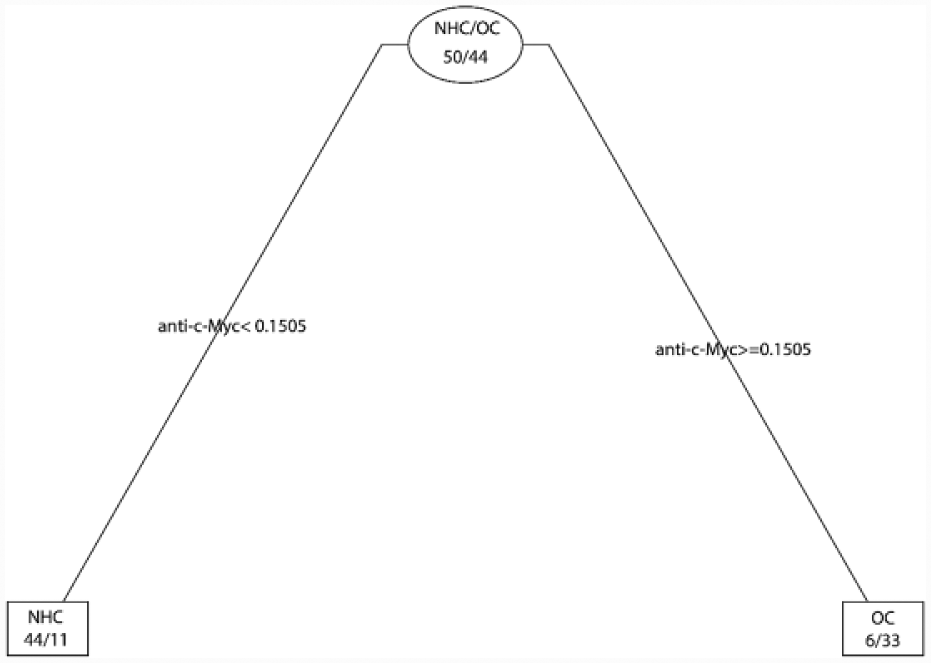
Best tree for OC detection. Anti-c-Myc is the only determinant for OC classification.

ROC curve of best tree for ovarian cancer detection. Since anti-c-Myc is the only determinant for ovarian cancer classification, the AUC value of the decision tree is close to the AUC value of anti-c-Myc.
Discussion
OC remains one of the most malignant cancers in women, and early diagnosis is essential for cancer treatment in clinical practice. We have systematically reviewed the development in using TAAs or anti-TAAs as biomarkers in the detection of OC in our previous study. 7 For single TAA or anti-TAA, the diagnostic value was insufficient to make it an ideal screening biomarker. Numerous studies have demonstrated that combinational utilization of multiple TAAs or anti-TAAs can increase sensitivity without dramatically compromising specificity. 26
In this study, we have evaluated and compared the diagnostic performance of autoantibodies against two candidate TAAs and 13 anti-TAAs in OC detection, using the conventional cutoff value as well as cutoff value determined by the ROC curve analysis, and we further performed decision tree analysis to check which anti-TAAs are critical in OC detection. For single anti-TAA, the sensitivity was very low when the cutoff value was set as mean + 3SD. Combinational utilization (“parallel connection”) of six anti-TAAs (autoantibodies against MDM2, PLAT, NPM1, 14-3-3 Zeta, p53, and RalA) has achieved the optimal diagnostic performance with 72.7% sensitivity at 96.0% specificity in OC detection. This is consistent with studies from other researchers that a panel of anti-TAAs can achieve better diagnostic performance.27–29 However, the optimal diagnostic panel might be quite different among different research groups (for more detail, please refer to Anderson and LaBaer 6 ) because the selection of TAAs or anti-TAAs is quite different between different research groups. When the cutoff value was set by using ROC curve analysis, the performance of single TAA or anti-TAA had improved. ROC curve analysis revealed that c-Myc held high potential to serve as biomarkers for OC detection with an AUC of 0.867. Several other anti-TAAs including autoantibodies against NPM1, MDM2, p16, p53, and 14-3-3 Zeta also achieved AUC greater than 0.80, indicating that they might be used as diagnostic biomarkers in OC detection. Furthermore, classification tree analysis revealed that anti-c-Myc is the only determinant for OC tree from 13 TAAs included in this analysis. However, c-Myc might not be a specific biomarker for OC detection. Myc is a multifunctional protein that participates in cell cycle progression, apoptosis, and cellular transformation. 39 C-Myc is a mutated version of Myc that can cause Myc to be constitutively expressed. This may result in the formation of cancer due to the unregulated expression of many genes caused by abnormal Myc expression. 45 A previous study has shown that tissue c-Myc expression level is positively associated with its corresponding serum autoantibodies in lung cancer. 10 Taken together, we propose that autoantibodies against c-Myc might be a good biomarker for the detection of OC, but it is not specific to OC because our previous studies have shown that anti-c-Myc autoantibodies could also distinguish breast cancer, lung cancer, gastric cancer, hepatocellular carcinoma, and esophageal squamous cell carcinoma from normal controls.26,44,46
Like c-Myc, p53 is also reported to be of value in the detection of other types of cancers besides OC, such as esophageal cancer and head and neck cancer.47,48 p53 mutation is a key event in the oncogenesis of many types of cancers, and mutated p53 can be shed into the circulating system; thus, anti-p53 autoantibodies are commonly produced accordingly. 48 But the frequency of anti-p53 autoantibody in different types of cancers varies because the rate of p53 mutation varies. The clinical utility of anti-p53 autoantibody has been reviewed by Suppiah et al. 48
In general, conventional TAAs or anti-TAAs have been studied for a long time by many research groups. However, the selection of these biomarkers is mainly based on results from previous studies. With the development of gene sequencing technology and the extensively utilization of bioinformatics in cancer research, systematic discovery of biomarkers has been made possible. The results this study confirmed that two candidate TAAs (PLAT and MSLN) intensively selected from the HPA database could be used as biomarkers for OC. However, when used alone, like many other TAAs, the sensitivities were limited when the cutoff value was set as mean + 3SD of NHCs. PLAT is a protein that has overexpression in cancer tissues of almost 20 different types of cancers included in the HPA database (version 13) but has low or no expression in normal tissues. MSLN has overexpressed in 75% (9/12) of OC tissues but has low or no expression in other types of cancers and normal tissues. Thus, we hypothesize that autoantibodies might be generated accordingly due to their abnormal overexpression in cancer tissues, and anti-PLAT might be a universal biomarker for cancers, while anti-MSLN might be an OC-specific biomarker. In our study, we have found that anti-PLAT autoantibodies have the potential to serve as biomarkers in several human malignancies, including OC, breast cancer, gastric cancer, and hepatocellular cancer (data not shown here). However, anti-MSLN autoantibodies are not specific to OC (data not shown here) because it can also be detected in hepatocellular cancer. The result does not agree with the hypothesis and therefore needs to be further validated in researches with larger sample sizes. This is mainly because the underlying mechanism of how autoantibodies are generated is still not fully understood. Our study indicates that the HPA database is a good resource for biomarker discovery. However, candidate biomarkers selected based on the HPA database need to be validated in human sera from cancer patients and normal controls.
In conclusion, combinational utilization of autoantibodies against six TAAs (MDM2, PLAT, NPM1, 14-3-3 Zeta, P53, and RalA) achieved the optimal diagnostic performance for OC when the cutoff value was set as mean + 3SD of NHCs. ROC curve analysis and classification tree analysis revealed that anti-c-Myc alone have good diagnostic performance in OC detection. However, due to the limitations of small sample size in this study, the results need to be validated in larger sample groups, and whether these TAAs or the combinations of certain TAAs can discriminate OC from other types of cancers needs to be further explored.
Footnotes
Acknowledgements
The authors thank Dr Xiaogang Su from the Department of Mathematical Sciences at the University of Texas at El Paso (UTEP) for his valuable discussion on how to plant and prune discussion trees. They also thank the Border Biomedical Research Center (BBRC) Core facilities at the UTEP for their help, which were funded by the National Institutes of Health (NIH) grant (5G12MD007592). H.S. and J.X.S. contributed equally to this work. J.X.S. was supported by the China Scholarship Council for his research in the United States.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the General Program of National Natural Science Foundation of China (No. 81172086 and No. 81372371) and the Major Project of Science and Technology in Henan Province (No. 161100311400) and Henan Key Technologies Research & Development Program of China (No. 162102410044).
