Abstract
BACKGROUNDS:
Previous studies have showed that long non-coding RNAs (lncRNAs) are critical regulators in many cancers. The aim of this study is to investigate the clinical role and functional effects of long non-coding RNA SNHG14 in pancreatic ductal adenocarcinoma (PDAC).
METHODS:
The expression of SNHG14 in 58 pairs of pancreatic cancer tissues and adjacent normal tissues was detected by quantitative real-time PCR (qRT-PCR) analysis. The correlations between SNHG14 expression and PDAC patients’ clinicopathological characteristics and prognosis were statistically assessed. Cell counting kit-8 (CCK8) and transwell cell invasion assays were employed to detect the capacities of cell proliferation and cell invasion. The western blot analysis was used to detected the expression of E-cadherin and Vimentin.
RESULTS:
In the study, we found that SNHG14 expression was higher in PDAC tissue compared to adjacent normal tissues by qRT-PCR analysis. Higher SNHG14 expression was significantly associated with advanced TNM stage and positive lymph node metastasis in PDAC patients. Furthermore, we demonstrated that higher SNHG14 expression acted as a poor predictor in PDAC patients compared with lower SNHG14 expression. Moreover, we showed that higher SNHG14 expression promoted cell proliferation, cell colony formation and cell invasion ability in PDAC. Upregulation of SNHG14 expression promoted cell invasion by affecting E-cadherin expression via interacting with EZH2.
CONCLUSIONS:
Thus, these results indicated that SNHG14 expression acts as a prognostic maker for PDAC and potential target of PDAC treatment.
Introduction
Pancreatic ductal adenocarcinoma (PDAC) is one of the most lethal malignancies worldwide. At early stage, patients with PDAC lack specific symptoms, and only approximately 20% patients have the opportunity to receive radical resection [1]. PDAC patients who have not undergone radical resection are with a median survival time
Long noncoding RNAs (lncRNAs) are defined as endogenous RNAs and more than 200 nucleotides in length [3]. Recently, some studies have indicated that lncRNAs are related to cancer initiation and progression through transcriptional and posttranscriptional regulations. Upregulation or downregulation of lncRNAs affects PDAC cell proliferation, migration, invasion and metastasis [4]. Such as, upregulated long noncoding RNA LINC01296 indicates a dismal prognosis for pancreatic ductal adenocarcinoma and promotes cell metastatic properties by affecting EMT [5]. LncRNA ZEB2-AS1 promotes pancreatic cancer cell growth and invasion through regulating the miR-204/HMGB1 axis [6]. LncRNA DLX6-AS1 promoted cancer cell proliferation and invasion by attenuating the endogenous function of miR-181b in pancreatic cancer [7]. LncRNA ADPGK-AS1 promotes pancreatic cancer progression through activating ZEB1-mediated epithelial-mesenchymal transition [8].
To this end, the involvement and regulatory functions of lncRNA SNHG14 in human tumors have been investigated. Long non-coding RNA SNHG14 induces trastuzumab resistance of breast cancer by regulating PABPC1 expression through H3K27 acetylation [9]. Long non-coding RNA SNHG14 exerts oncogenic functions in non-small cell lung cancer through acting as a miR-340 sponge [10]. The long non-coding RNA SNHG14 inhibits cell proliferation and invasion and promotes apoptosis by sponging miR-92a-3p in glioma [11]. SP1-induced up-regulation of lncRNA SNHG14 as a ceRNA promotes migration and invasion of clear cell renal cell carcinoma by regulating N-WASP [12]. However, the role of SNHG14 in PDAC remains large unknown.
In the study, we found that long non-coding RNA SNHG14 was higher expression in PDAC. Higher SNHG14 expression was significantly higher in patients with an advanced TNM stage and positive lymph node metastasis and predicted a poor prognosis in PDAC patients. Moreover, we showed that higher SNHG14 expression promoted cell proliferation, cell colony formation and cell invasion abilities in PDAC. Upregulation of SNHG14 expression promoted cell invasion by affecting E-cadherin expression via interacting with EZH2. Thus, these results indicated that SNHG14 expression acts as prognostic maker for PC and potential target of PC.
Patients and methods
Patient tissue samples
A total of 58 pairs of pancreatic cancer tissues and adjacent normal tissues were collected from patients undergoing surgical resection between March 2008 and October 2015 (mean age 55.7
Association between SNHG14 expression and clinical feathers was assessed
Association between SNHG14 expression and clinical feathers was assessed
The four human PDAC cell lines Panc1, Panc28, AsPC1 and BxPC3 and a human pancreatic ductal epithelial cell line HPDE were purchased from the American Type Culture Collection (ATCC, Manassas, VA, USA). Cells were cultured in Roswell Park Memorial Institute-1640 medium (Hyclone; Thermo Fisher Scientific, Waltham, MA, USA) and were supplemented with 10% fetal bovine serum (FBS) (Hyclone; Thermo Fisher Scientific, Waltham, MA, USA), 100 U/mL penicillin and 100 mg/mL streptomycin. All of cells lines were cultured at 37
RNA transfection
The two siRNAs for lncRNA-SNHG14, named siRNA-1 (Sense: 5’-GCAAAUGAAAGCUACCAAU-3’, Anti-sense: 5’-AUUGGUAGCUUUCAUUUGC- 3’) and siRNA-2 (Sense: 5’-GCACAAUAUCUUUGA ACUA-3’, Anti-sense: 5’-UAGUUCAAAGAUAUUG UGC-3’) were constructed. Cell transfection was performed using the Lipofectamine 2000 kit (Invitrogen; Thermo Fisher Scientific, Inc., Waltham, MA, USA) according to the manufacturer’s protocol. The knockdown efficiency was detected by using reverse transcription-quantitative polymerase chain reaction (qRT-PCR) at 48 h after cell transfection.
RNA extraction and RT-qPCR
Total RNA from tumor tissues and cells was isolated using TRIzol reagent (Invitrogen; Thermo Fisher Scientific, Inc., Waltham, MA, USA) according to the manufacturer’s protocol. RNA was reverse transcribed into cDNA using Prime Script RT Reagent kit (Invitrogen; Thermo Fisher Scientific, Inc., Waltham, MA, USA) according to the manufacturer’s protocol. QRT-PCR was performed using SYBR Premix Ex Taq (Takara Biotechnology Co., Ltd., Dalian, China), according to the manufacturer’s protocol. The experiments were performed using the ABI PRISM 7500 PCR System (Applied Biosystems; Thermo Fisher Scientific, Inc., Waltham, MA, USA), according to the manufacturer’s protocol. The primer sequences were as follows: SNHG14 (Forward) 5’-GGGTGTTTACGTA GACCAGAACC-3’, (Reverse) 5’-CTTCCAAAAGCC TTCTGCCTTAG-3’; GAPDH (Forward) 5’-GAAGGT GAAGGTCGGAGTC-3’, (Reverse) 5’-GAAGATGGT GATGGGATTTC-3’. The thermocycling conditions were as follows: 95
Cell proliferation assay
The cell proliferation was performed by using the CCK8 Kit (Dojindo, Rockville, MD, USA). Briefly, A total of 2,000 transfected cells were plated into 96-well plates. After, cells were treated with the CCK8 reagent and further cultured for 2 hours. The optical density at 450 nm was measured with a spectrophotometer (Thermo Electron Corporation, MA, USA).
Cells colony formation assay
For colony formation assay, 500 transfected cells per plate were seeded in 6-well plates and incubated for 7 days. Then, cell colonies were fixed with 4% paraformaldehyde at room temperature for 20 min and stained with 1% crystal violet. Cells were counted under a light microscope (Magnification 200
Cell invasion assay
Cellular invasion was detected using Transwell chambers with a pore size of 8
Western blot analysis
The transfected cells were lysed in radioimmunoprecipitation assay (RIPA) buffer (Beyotime Institute of Biotechnology, Haimen, China). Total protein was quantified using a Bradford protein assay (BCA, Bio-Rad Laboratories, Inc., Hercules, CA, USA). An amount of total protein (40
SNHG14 expression is significantly upregulated in PDAC. (A) The relative SNHG14 expression was higher in PDAC tissues than in corresponding normal tissues (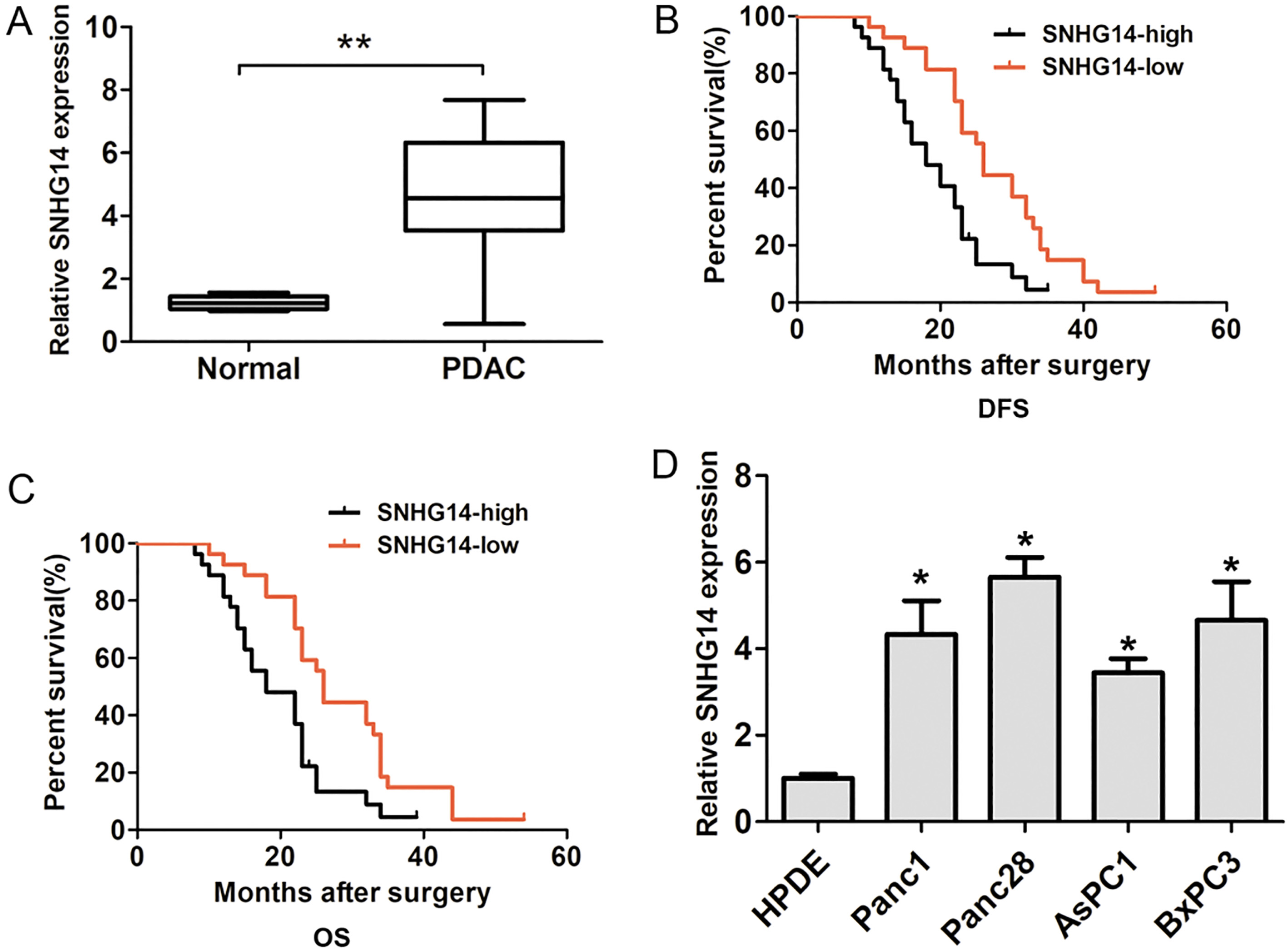
RIP experiments were performed using the EZ-Magna RIP kit (Millipore, China) following the manufacturer’s guidance. Human anti-EZH2 antibody (Santa Cruz Biotechnology, Inc., Dallas, TX, USA) or control IgG (Santa Cruz Biotechnology, Inc., Dallas, TX, USA) was used. The immunoprecipitated RNA was isolated and was further subjected to qRT-PCR analysis.
Statistical analysis
All experimental data were performed using SPSS version 19.0 (SPSS Inc., Chicago, IL, USA). One-way analysis of variance (ANOVA) was used to analyze for differences between multiple groups. The chi-square test or Student’s
Results
SNHG14 expression is significantly upregulated in PDAC
In the study, lncRNA SNHG14 expression in PDAC tissues and adjacent normal tissues was detected by using qRT-PCR analysis. The results demonstrated that lncRNA SNHG14 expression was significantly upregulated in PDAC tissues compared with adjacent normal tissues (Fig. 1A). Furthermore, the association between lncRNA SNHG14 expression and clinicopathological factors was analyzed by using Chi-square test. Our results showed that lncRNA SNHG14 expression associated with positive lymph node metastasis in patients (
The effects of SNHG14 knockdown on PDAC cell proliferation. (A) and (B) SNHG14 was knocked down by transfection of Panc1 and Panc28 cells with si-SNHG14-1, si-SNHG14-2 and si-NC. (C) and (D) Cell Counting Kit-8 assay results in si-SNHG14-1 or si-SNHG14-2-transfected and control siRNA-transfected Panc1 and Panc28 cells, 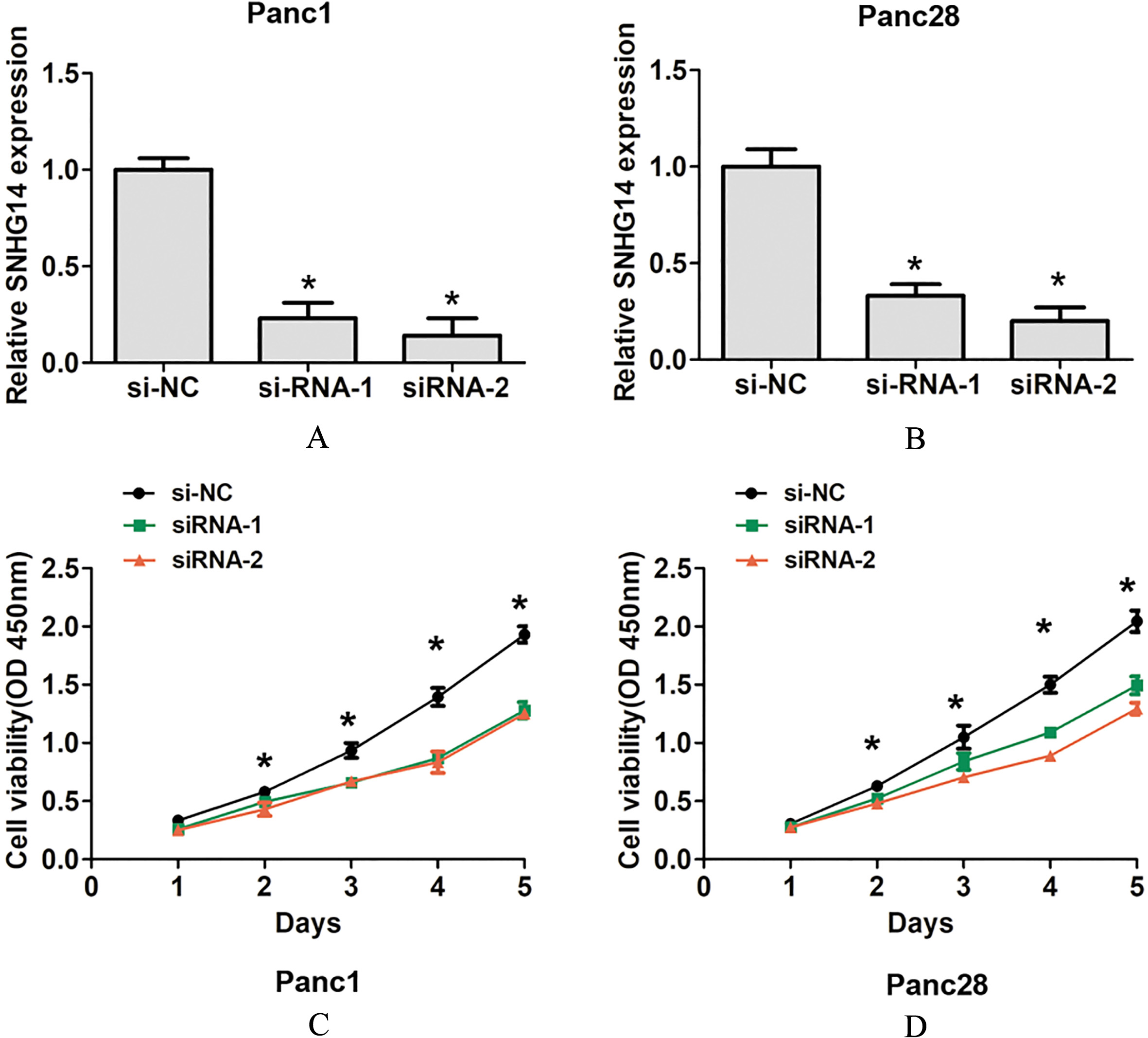
The effects of SNHG14 knockdown on cell colony forming and cell invasion ability. (A) and (B) Cell colony formed assays results in si-SNHG14-transfected and control siRNA-transfected Panc1 and Panc28 cells. (C) and (D) Transwell cell invasion assays results in si-SNHG14-transfected and control siRNA-transfected Panc1 and Panc28 cells, 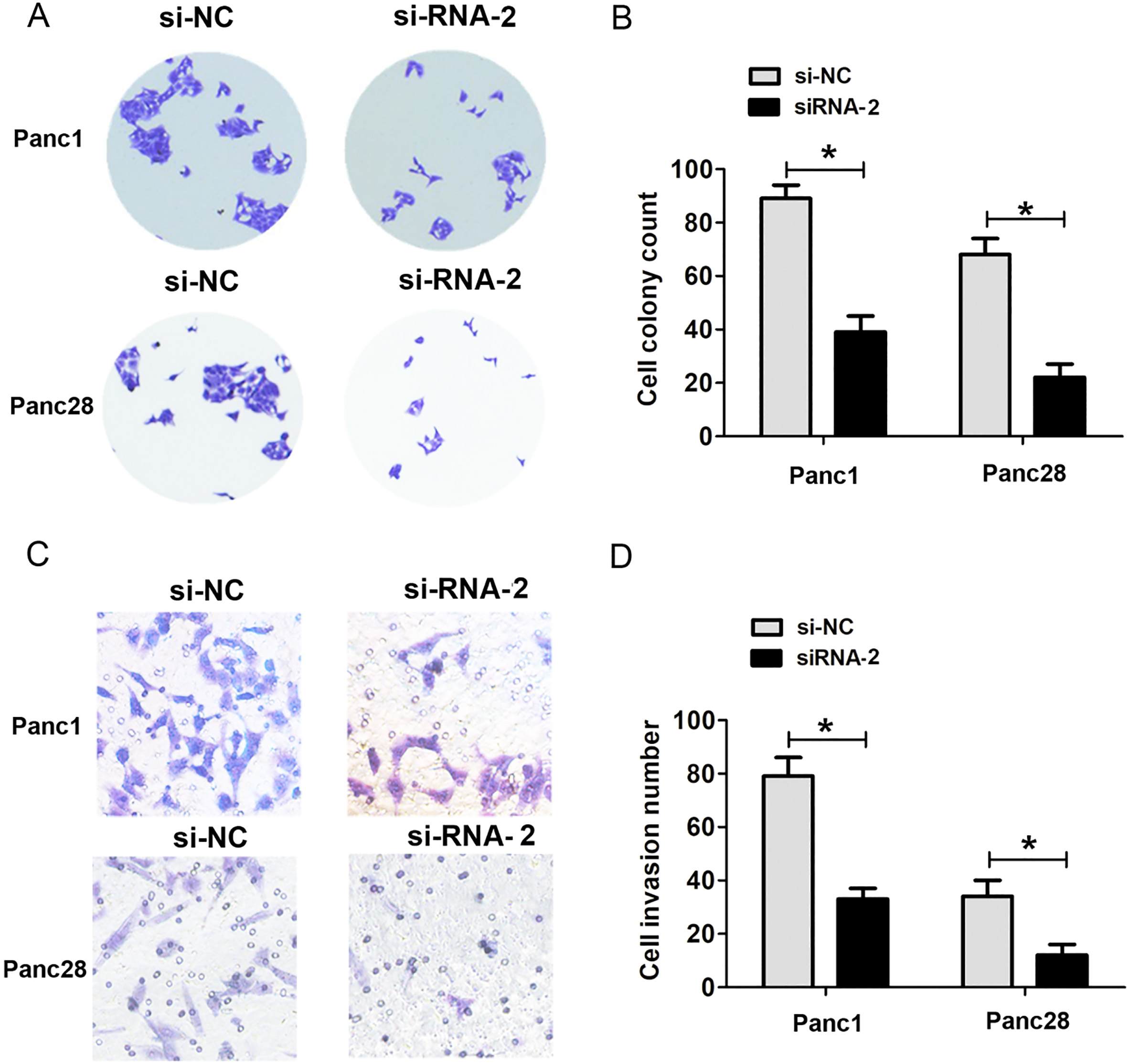
SNHG14 expression regulated cell invasion and EMT pathway by interacting with EZH2. (A) and (B) Western blot analysis results showed the protein expression of E-cadherin and Vimentin in si-SNHG14-transfected and control siRNA-transfected Panc1 and Panc28 cells. (C) QRT-PCR analysis results showed the mRNA expression of E-cadherin in si-SNHG14-transfected and control siRNA-transfected Panc1 and Panc28 cells. (D) The RIP results showed that lncRNA SNHG14 could interact with EZH2, 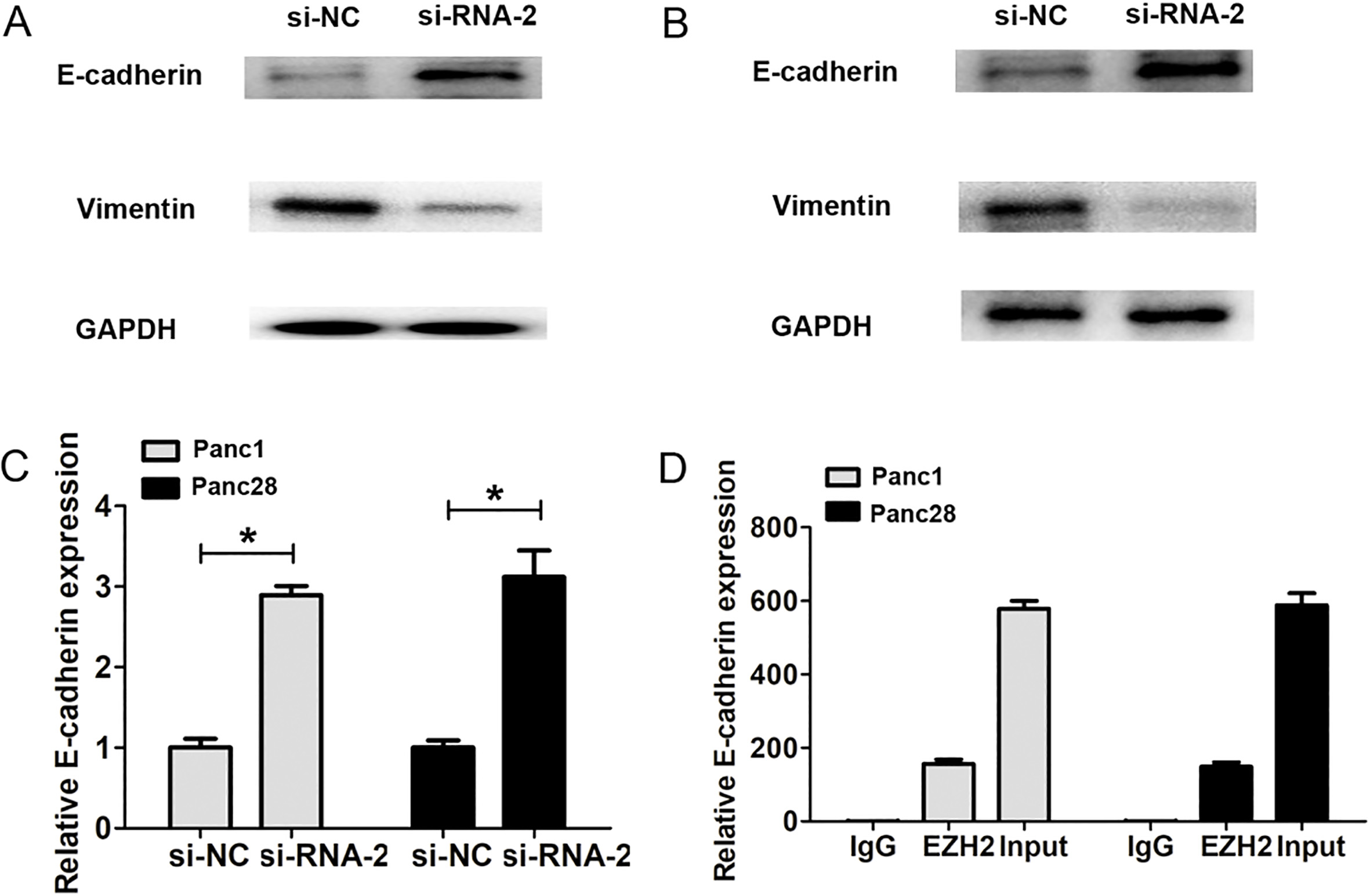
Moreover, we also detected the expression of lncRNA SNHG14 in four human pancreatic cancer cell lines Panc1, Panc28, AsPC1 and BxPC3 and a human pancreatic ductal epithelial cell line HPDE. Our results indicated that lncRNA SNHG14 expression was also higher in PDAC cells compared to that in the HPDE (Fig. 1D). To detect the effects of lncRNA SNHG14 expression on PDAC cells proliferation capacity, we performed SNHG14 knockdown assays in two tumor cell lines including Panc1 and Panc28 cells. The two siRNAs of SNHG14 were used to significantly downregulate the expression of lncRNA SNHG14 in PDAC cells lines (Fig. 2A and B). Furthermore, we performed functional assay to detect the effects of SNHG14 expression on cell proliferation and invasion using siRNA-1, due to its higher knockdown efficiency in PDAC cells. The results of CCK8 cell proliferation assays showed that SNHG14 knockdown significantly reduced cell proliferation ability compared to corresponding control groups in Panc1 and Panc28 cells (Fig. 2C and D). Cell colony formed assays showed that SNHG14 knockdown significantly decreased cell colony number, compared to corresponding control groups in Panc1 and Panc28 cells (Fig. 3A and B). Thus, these results indicated that lncRNA SNHG14 knockdown could inhibit cell proliferation ability of PDAC.
Downregulation of SNHG14 inhibits cell invasion ability and EMT in PDAC
Next, transwell invasion assays showed that SNHG14 knockdown significantly decreased cell invasion number, compared to corresponding control groups in Panc1 and Panc28 cells (Fig. 3C and D). We also detected the epithelial-mesenchymal transition (EMT) related makers after SNHG14 knockdown in Panc1 and Panc28 cells. The results showed that SNHG14 knockdown significantly increased the E-cadherin expression, but decreasing the expression of Vimentin compared to corresponding control groups in Panc1 and Panc28 cells (Fig. 4A and B). These results indicated that lncRNA SNHG14 knockdown could inhibit cell invasion ability and EMT by increasing the E-cadherin expression, but decreasing the expression of Vimentin in PDAC cells.
LncRNA SNHG14 affects cell invasion ability by interacting with EZH2 in PDAC
Recently, studies have showed that LncRNA can function as molecular scaffold to help epigenetic enzymes to bind on promoters of target genes such as, EZH2, a core component of polycomb repressive complex 2 (PRC2), which catalyzes trimethylation of H3K27me3 and leads to subsequent gene silencing, such as, LncRNA FEZF1-AS1 enhances epithelial-mesenchymal transition (EMT) through suppressing E-cadherin via interacting with EZH2 [13]. To explore their interaction in PDAC cells, we found that E-cadherin mRNA expression is upregulated after SNHG14 knockdown in Panc1 and Panc28 cells (Fig. 4C). Moreover, we performed the RIP assay. As shown in Fig. 4D, the RIP results showed that lncRNA SNHG14 was detected in the immunoprecipitates using qRT-PCR by using EZH2 antibody. Thus, we demonstrated that LncRNA SNHG14 affected cell invasion by regulating E-cadherin via interacting with EZH2.
Discussion
Accumulating evidence has revealed that lncRNAs play crucial roles in human diseases including cancers [14]. Recent studies have reported the relationship between SNHG14 expression and different aspects of disease and tumorigenesis including proliferation, migration, invasion and metastasis [15]. For example, long non-coding RNA SNHG14 promotes microglia activation by regulating miR-145-5p/PLA2G4A in cerebral infarction. Long non-coding RNA SNHG14 contributes to gastric cancer development through targeting miR-145/SOX9 axis [16]. Exosome-mediated transfer of lncRNA-SNHG14 promotes trastuzumab chemoresistance in breast cancer. Long non-coding RNA SNHG14 exerts oncogenic functions in non-small cell lung cancer through acting as a miR-340 sponge [10]. LncRNA SNHG14 promotes inflammatory response induced by cerebral ischemia/reperfusion injury through regulating miR-136-5p/ROCK1 [17]. In present study, our results found that long non-coding RNA SNHG14 was higher expression in PDAC tissue compared to adjacent non tumor tissues by quantitative real-time PCR. Higher SNHG14 expression was significantly higher in patients with lymph node metastasis. Moreover, we demonstrated that higher SNHG14 expression predicted a poor prognosis in PDAC patients. Of courses, the expanded sample analysis could demonstrate that SNHG14 could be a clinical prognostic maker for PDAC patients in the further.
To further explore whether the abnormal expression of SNHG14 could affect cell proliferation and invasion in PDAC progression. By performing the cell proliferation and cell invasion assays, we showed that downregulation of SNHG14 expression significantly inhibited cell proliferation, invasion and EMT pathway. In HCC progression, researchers showed that upregulated SNHG14 expression could accelerate HCC cell proliferation and colony formation bur inhibit cell apoptosis [18]. Similarly. Our results showed that downregulation of SNHG14 expression significantly affect cell proliferation, invasion and EMT pathway in PDAC progression.
Recently, studies also have showed that lncRNAs function as molecular scaffold to help epigenetic enzymes to bind on promoters of EZH2, a core component of polycomb repressive complex 2 (PRC2) [19]. LncRNAs affects cell proliferation and invasion by interacting with EZH2 in some tumors, such as, lncRNA MALAT1 promotes epithelial-to-mesenchymal transition of esophageal cancer through Ezh2-Notch1 signaling pathway [20]. Long noncoding RNA linc-UBC1 promotes tumor invasion and metastasis by regulating EZH2 and repressing E-cadherin in esophageal squamous cell carcinoma [21]. LncRNA FEZF1-AS1 enhances epithelial-mesenchymal transition (EMT) through suppressing E-cadherin and regulating WNT pathway in non-small cell lung cancer (NSCLC) [13]. Long non-coding RNA UCA1 promotes gallbladder cancer progression by epigenetically repressing p21 and E-cadherin expression [22]. In the study, we speculated that LncRNA SNHG14 may regulating cell invasion and EMT pathway by interacting with EZH2. To explore their interaction in PDAC cells, we found that E-cadherin mRNA expression is upregulated after SNHG14 knockdown in Panc1 and Panc28 cells. Furthermore, we performed the RIP assay. The RIP results showed that lncRNA SNHG14 could interact with EZH2. Thus, we demonstrated that SNHG14 affects tumor progression by interacting with EZH2 in PDAC. In the further, we will explore the underlying mechanism for SNHG14 affecting the EMT progression in PDAC.
In conclusion, our results indicated that LncRNA SNHG14 expression was higher in PDAC and predicted a poor prognosis. LncRNA SNHG14 knockdown affect cell proliferation and invasion ability. Moreover, we showed that SNHG14 expression regulated cell invasion and EMT pathway by interacting with EZH2. Thus, these results indicated that SNHG14 expression acts as prognostic maker for PC and potential target of PC.
Footnotes
Acknowledgments
The study was supported by Science and Technology Research Projects in Anhui Province (1804h08020 277).
