Abstract
Background:
Chemotherapy options in patients with advanced pancreatic ductal adenocarcinoma (PDAC) after failure of standard chemotherapies are limited.
Objectives:
We aimed to report the efficacy and safety of the leucovorin and 5-fluorouracil (LV5FU2) and carboplatin combination in this setting.
Design:
We performed a retrospective study including consecutive patients with advanced PDAC who received LV5FU2–carboplatin between 2009 and 2021 in an expert center.
Methods:
We measured overall survival (OS) and progression-free survival (PFS), and explored associated factors using Cox proportional hazard models.
Results:
In all, 91 patients were included (55% male, median age 62), with a performance status of 0/1 in 74% of cases. LV5FU2–carboplatin was mainly used in third (59.3%) or fourth line (23.1%), with three (interquartile range: 2.0–6.0) cycles administered on average. The clinical benefit rate was 25.2%. Median PFS was 2.7 months (95% CI: 2.4–3.0). At multivariable analysis, no extrahepatic metastases (p = 0.083), no ascites or opioid-requiring pain (p = 0.023), <2 prior treatment lines (p < 0.001), full dose of carboplatin (p = 0.004), and treatment initiation >18 months after initial diagnosis (p < 0.001) were associated with longer PFS. Median OS was 4.2 months (95% CI: 3.48–4.92) and was influenced by the presence of extrahepatic metastases (p = 0.058), opioid-requiring pain or ascites (p = 0.039), and number of prior treatment lines (0.065). Prior tumor response under oxaliplatin did not impact either PFS or OS. Worsening of preexisting residual neurotoxicity was infrequent (13.2%). The most common grade 3–4 adverse events were neutropenia (24.7%) and thrombocytopenia (11.8%).
Conclusion:
Although the efficacy of LV5FU2–carboplatin appears limited in patients with pretreated advanced PDAC, it may be beneficial in selected patients.
Introduction
Pancreatic ductal adenocarcinoma (PDAC) has a constantly increasing incidence, estimated to be of 4.8 per 100,000 worldwide in 2018. 1 Accordingly, its mortality rate has more than doubled from 1990 to 2017 2 and it is expected to become the second most common cause of cancer death in Europe and in the United States by 2030. 3 Its prognosis remains poor, with an all-stage 5-year overall survival (OS) rate hardly reaching 10%. 4
Approximately two-thirds of patients with PDAC are diagnosed at a metastatic stage. 5 In this setting, the treatment relies on systemic chemotherapy, but options are limited. Following more than 15 years of use of gemcitabine alone as standard, the FOLFIRINOX (5-fluorouracil administered as leucovorin and 5-fluorouracil (LV5FU2), irinotecan, and oxaliplatin) and gemcitabine plus nab-paclitaxel combinations have become references as -first line treatments.6,7 Nevertheless, the emergence of chemotherapy-resistant cancer cells over time and the occurrence of cumulative toxicity hamper their prolonged antitumor efficacy. As quality of life remains a key objective in these patients, protocol adaptations are frequent, notably regarding oxaliplatin to limit the cumulative peripheral neurotoxicity. 8 This leads to dose reductions or even early discontinuation, 9 which may reduce their impact on survival. 10 The use of a different platinum agent could have relevant efficacy to maintain antitumor efficacy in case of discontinuation or failure of oxaliplatin.
It is estimated that only half of the patients with advanced PDAC are fit enough to receive a second-line chemotherapy. 11 In this case, treatment options are limited and there are no recommendations on the use of a further-line chemotherapy after failure of standard regimens, except NALIRI (LV5FU2 and nanoliposomal irinotecan) after gemcitabine-based first-line chemotherapy in selected patients and depending on its availability.11–14 Although oxaliplatin and cisplatin are recommended options by National Comprehensive Cancer Network guidelines, 15 data about the use of carboplatin in PDAC are lacking. While carboplatin may share some of its antitumoral activity with oxaliplatin, it also has specific mechanisms of action and yields negligible neurotoxicity.16,17 Hence, the use of carboplatin-containing chemotherapy could be relevant in patients with advanced PDAC, as previously reported in combination with gemcitabine, 18 and in other pancreatic neoplasms including neuroendocrine carcinomas 19 and mucinous cystadenocarcinomas. 20 The aim of this study was to report the efficacy and tolerance of the LV5FU2–carboplatin combination in a large and comprehensive consecutive cohort patients with advanced PDAC after failure of standard chemotherapies.
Methods
Patients
We conducted a retrospective study at Beaujon University Hospital (Clichy, France). We studied the medical records of all consecutive patients who received at least one cycle of LV5FU2–carboplatin chemotherapy for inoperable, locally advanced or metastatic PDAC, between July 2009 and August 2021. Patients were identified from the Chimio® software (Computer Engineering, Paris, France). Patients without pathologically proven PDAC were not included. Exclusion criteria comprised other concomitant active malignancies.
All indications of LV5FU2–carboplatin combination were decided during weekly dedicated multidisciplinary tumor boards. The choice of this combination was applied as a systematic policy as third/fourth line proposed in our institution during this period. The protocol consisted in carboplatin at an area under the curve (AUC) of 4 mg/ml/min and folinic acid at 400 mg/m2 followed by a bolus of 5-fluorouracil at 400 mg/m2, then 5-fluorouracil was infused at 2400 mg/m2 continuously over 46 h, in a 14-day cycle. All patients received an aprepitant of 125 mg at Day 1 and 80 mg at Days 2 and 3, 80 mg of methylprednisolone (Day 1), and 8 mg of ondansetron (Days 1–5), for prevention of acute nausea and vomiting. Patients underwent standardized clinical and biological evaluation before every chemotherapy cycle, and morphological computed tomography (CT) scan was performed every six cycles, or earlier in case of clinical suspicion of progression, as it was done in routine practice in all patients treated for PDAC in our center.
Data collection
Data were collected from medical records and included sex, date of diagnosis and last follow-up, genetic predisposition when available, tumor localization (head or body/tail), tumor stage, and localization of metastatic sites. We recorded previous treatments received including any surgical resection and perioperative treatments. When neoadjuvant and adjuvant chemotherapy had been administered, these were considered as one single line of treatment. We also collected data regarding previous oxaliplatin chemotherapy, including the total number of cycles, oxaliplatin dose reductions, causes of discontinuation, and best tumor response according to Response Evaluation Criteria in Solid Tumors (RECIST) v 1.1. 21
We recorded the indication of LV5FU2–carboplatin and the following data at treatment initiation: World Health Organization (WHO) performance status, body mass index, oxaliplatin-induced sensory neurotoxicity, the presence of opioid-requiring pain or ascites, and the main laboratory findings. Adverse events were evaluated according to the Common Terminology Criteria for Adverse Events v 4.0. 22 Dose reductions were collected, including their causes and the cumulative dose of carboplatin administered in AUC. Best tumor response under treatment and progression were centrally measured by an experienced radiologist. To this end, disease evolution was evaluated according to RECIST v 1.1 as complete response, partial response, stable disease, or progressive disease, 21 on each CT scan performed under treatment, in comparison with baseline CT scan. The clinical benefit rate was calculated as the percentage of patients experiencing objective response or stabilization among the whole population.
Statistical analyses
Continuous variables were described as medians with their 25–75 interquartile range (IQR). Categorical variables were described as frequencies and percentages. Progression-free survival (PFS) was measured from the time of LV5FU2–carboplatin initiation to the date of RECIST-defined progression or death. OS was measured from the time of LV5FU2–carboplatin initiation to the date of death. Patients alive without event at their last follow-up were censored at that date. Median PFS and OS were estimated using the Kaplan–Meier method and were compared using the log-rank test. Variables potentially associated with PFS and OS were explored using Cox proportional hazard regression models. All clinically relevant, non collinear variables with p < 0.25 at univariable analyses were used for backward stepwise multivariable models, in addition to progression during prior oxaliplatin. Values of p < 0.05 were considered to be statistically significant. All statistical tests were bilateral. Statistical analyses were performed with the SPSS software (version 20, IBM).
Results
Patients
We identified 111 patients treated with LV5FU2–carboplatin between July 2009 and August 2021. After a review of medical records, we excluded 15 patients who did not have PDAC, and five additional patients for not having received LV5FU2–carboplatin (n = 2), concomitant lung cancer (n = 1), or major missing data (n = 2) (Figure 1). Among the 91 patients finally included, 54.9% were male, and median age at baseline was 61.6 years (IQR: 55.7–65.6) (Table 1). A genetic predisposition to PDAC was noted in 7.7% of patients, consisting in germline mutations in the BRCA2 (n = 5), BRCA1 (n = 1), or PRSS1 (n = 1) genes.
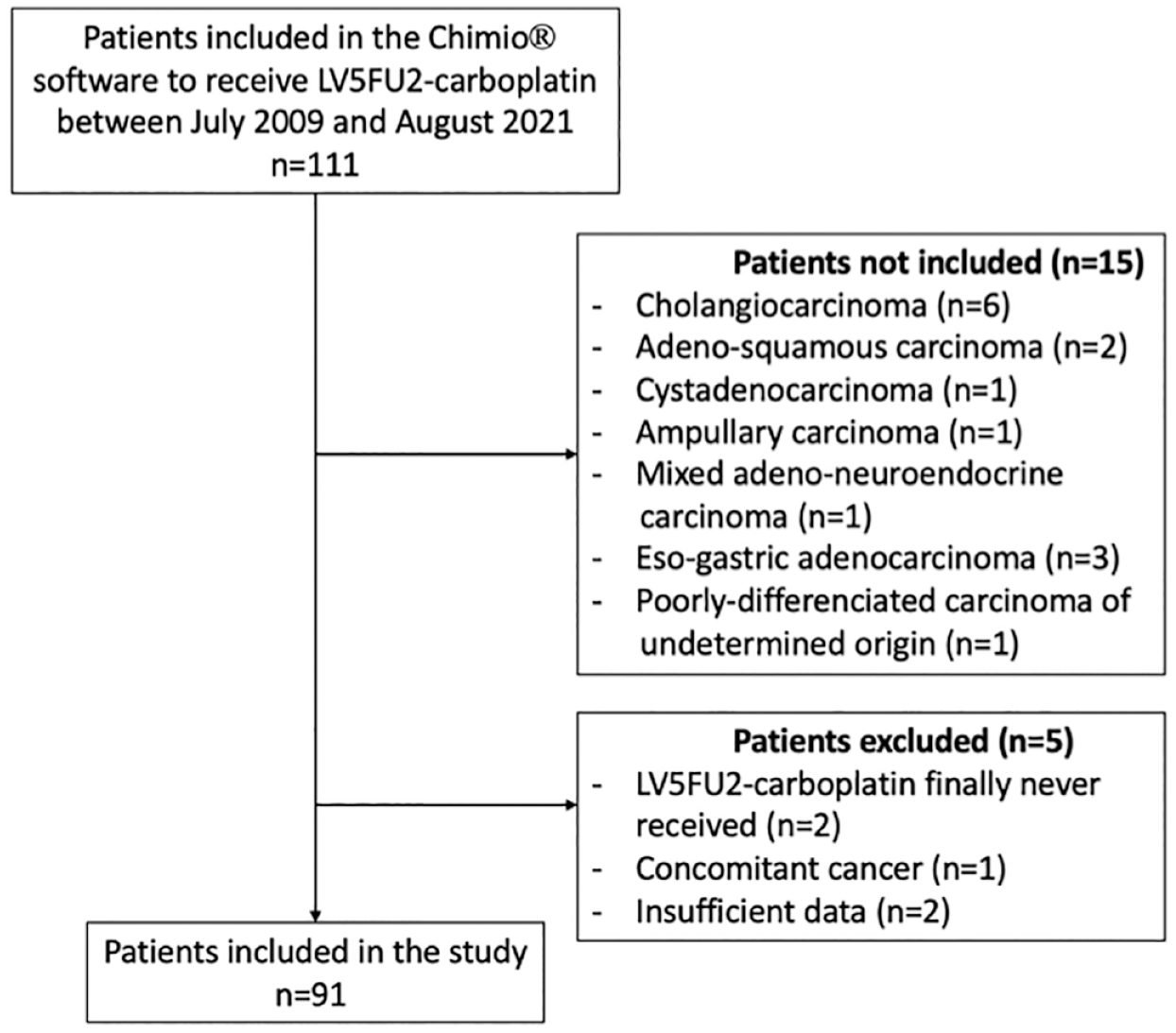
Flow chart of the study.
Baseline characteristics of the 91 patients included in the study.

IQR, interquartile range; LV5FU2, leucovorin and 5-fluorouracil; WHO, World Health Organization.
LV5FU2–carboplatin was introduced 17.7 months (IQR: 13–24) after initial diagnosis on average. It was indicated for failure of previous lines in 82.4% of cases (third line: 59.3%, fourth line: 23.1%), or reintroduction of platinum in patients who had oxaliplatin limiting toxicity during first or second line of treatment including peripheral neurotoxicity or severe allergy in 13 and 3 patients, respectively. In all, 18 patients (19.8%) had previously undergone PDAC surgical resection followed by adjuvant chemotherapy; half of them had received neoadjuvant treatment. The most frequent previous chemotherapy regimens administered were FOLFIRINOX (70.3%) and gemcitabine plus nab-paclitaxel (57.1%) (Table 1). Overall, all patients had previously received oxaliplatin-based chemotherapy, with a median of 10 (IQR 6–13) cycles. Tumor response to oxaliplatin was previously observed in 40 patients (44.9%), and 15 patients (16.5%) had previously progressed while treated with oxaliplatin. In total, 14 patients did not experience previous disease progression on LV5FU2, all corresponding to patients who received LV5FU2–carboplatin because of oxaliplatin-related allergy or limiting neuropathy.
At baseline, most patients (90.1%) had distant metastases, mainly in the liver (74.7%), and 45.1% of patients had extra-hepatic metastases (Table 1). Most patients (73.6%) had WHO performance status of 0 or 1, 23.1% of 2, and 2.2% of 3. In addition, 57.1% of patients had opioid-requiring pain and 13.2% of patients had ascites. Residual peripheral neurotoxicity due to prior oxaliplatin or nab-paclitaxel was present in 70 (76.9%) and 2 (2.2%) patients, respectively. Median serum albumin level was of 36.6 g/l (IQR: 33.0–39.7).
Treatment received and toxicity
Patients received a median number of three cycles (IQR: 2.0–6.0) of LV5FU2–carboplatin cycles. The median cumulative dose of carboplatin administered was 11.2 AUC (IQR: 4.0–18.2) with a median dose of 3.5 AUC per cycle (IQR: 3.2–4.0). In all, 60 patients (65.9%) had at least one cycle of carboplatin administered at reduced dose, because of asthenia or worsening of general condition (56.7%), hematologic toxicity (38.3%), or worsening of peripheral neurotoxicity (5%). Regarding LV5FU2, 58 (63.7%) and 23 (25.3%) patients received at least one chemotherapy cycle with reduced dose of the bolus or the continuous infusion, respectively, because of asthenia or worsening of general condition (55.2% and 52.2%, respectively) or for adverse events (44.8% and 47.8%, respectively). Overall, 72.5% of patients had dose reductions in LV5FU2 and/or carboplatin. The most frequent reasons of discontinuation of LV5FU2–carboplatin were documented tumor progression (41.8%) and terminal worsening of general condition (48.4%).
The most common severe (grade 3/4) adverse events that occurred during treatment were neutropenia (24.7%), thrombocytopenia (11.8%), anemia, and nausea/vomiting (7.1% each) (Table 2). Preexistent peripheral neurotoxicity remained absent, stable, improved, or worsened in 18.7%, 60.4%, 7.7%, and 13.2% of patients, respectively. Granulocyte-colony stimulating factor (GCS-F) and erythropoietin were used in 42.9% and 35.2% of patients, respectively. There was no death due to toxicity.
Adverse events reported in patients receiving LV5FU2–carboplatin chemotherapy (available in 85 patients).

68 patients (80%) had preexisting peripheral neuropathy.
LV5FU2, leucovorin and 5-fluorouracil.
Best morphological response and PFS
The best morphological response on LV5FU2–carboplatin was evaluated in 68 patients (23 patients did not have a morphological imaging after the last cycle of chemotherapy, because of prompt terminal deterioration of general status). Among all the 91 patients, the rates of patients achieving objective response or tumor stabilization were 3.3% (3/91) and 22% (20/91), respectively, leading to a clinical benefit rate (response or stabilization) of 25.2%. There were no specific features to the three patients experiencing objective response. None of them had progressed under prior oxaliplatin-based regimen, although these very limited figures hamper to observe some clear pattern.
The median PFS after initiation of LV5FU2–carboplatin was 2.7 months (95% CI: 2.4–3.0). The 3-, 6-, 9-, and 12-month PFS rates were 41.8 ± 5.2%, 19.8 ± 4.2%, 12.1 ± 3.4%, and 8.8 ± 3%, respectively. Neither prior objective response to oxaliplatin [hazard ratio (HR): 0.86; 95% CI: 0.56–1.31; p = 0.48), progression with oxaliplatin (HR: 1.10; 95% CI: 0.63–1.93; p = 0.73) nor time elapsed between last cycle of oxaliplatin and beginning of LV5FU2–carboplatin (HR: 1.00; 95% CI: 0.98–1.02; p = 0.86) did influence PFS (Figures 2(a) and (b)).
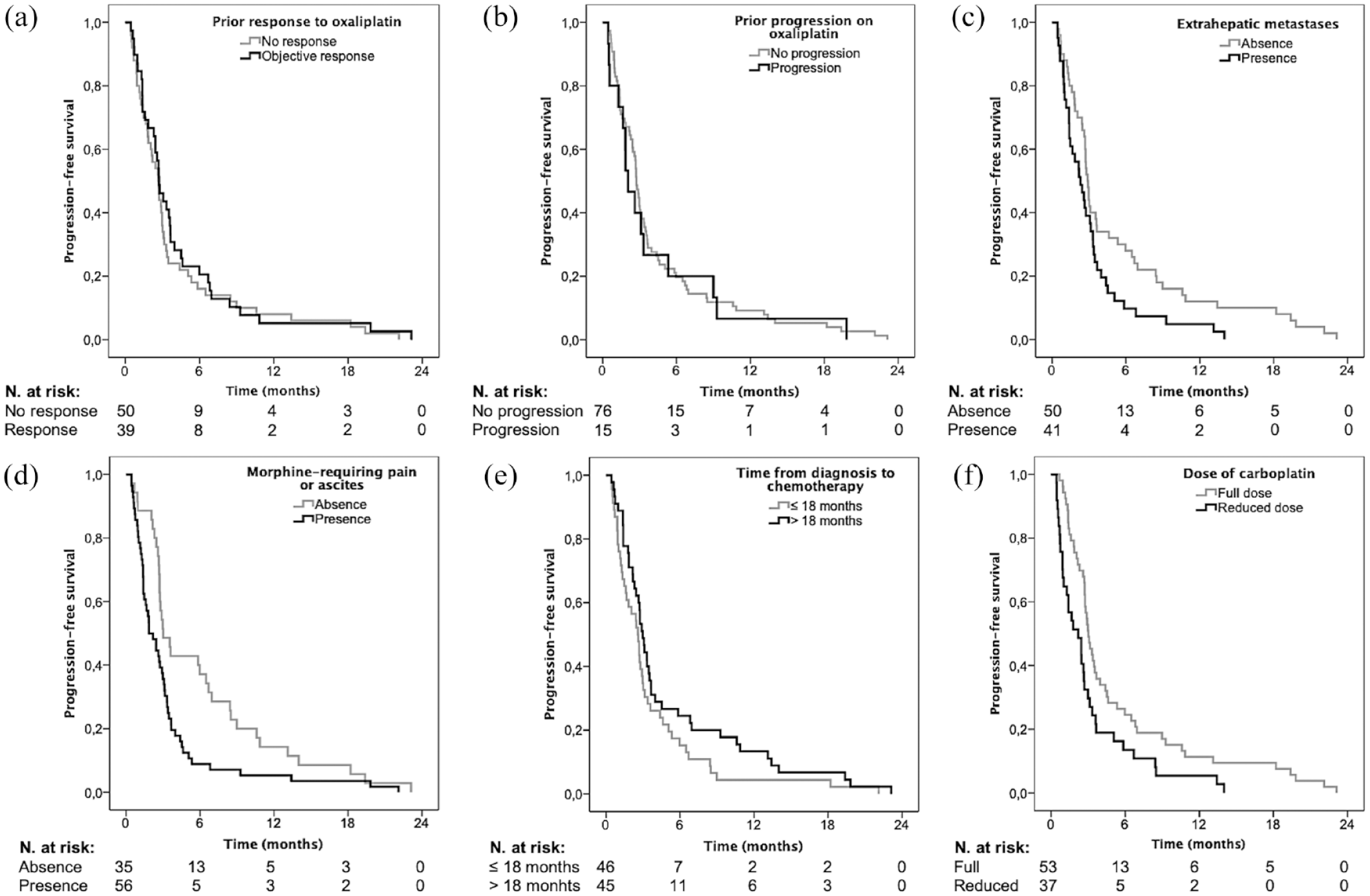
PFS curves of patients with pretreated advanced PDAC treated with LV5FU2–carboplatin, depending on variables of interest. (a) prior objective response to oxaliplatin or not (p = 0.48); (b) prior progression on oxaliplatin or not (p = 0.73); (c) presence of extrahepatic metastases or not (p = 0.022); (d) ascites or opioid-requiring pain or not (p = 0.007); (e) time from diagnosis to chemotherapy ⩽ or >18 months (p = 0.099); and (f) carboplatin administered at reduced or full dose (p = 0.008).
On multivariable analysis (Table 3), ascites or opioid-requiring pain (HR: 1.73; 95% CI: 1.08–2.78; p = 0.023), ⩾2 prior treatment lines (HR: 3.58; 95% CI: 1.80–7.13; p < 0.001), and carboplatin administered at reduced dose (HR: 1.92; 95% CI: 1.23–2.98; p = 0.004) were independently associated with significantly shorter PFS (Figure 2(c)–(f)). On the contrary, LV5FU2–carboplatin introduction >18 months after initial diagnosis (HR: 0.36; 95% CI: 0.22–0.59; p < 0.001) was associated with longer PFS.
Multivariable analysis of variables associated with PFS.

HR, hazard ratio; PFS, progression-free survival.
Overall survival
The median total follow-up from initiation of LV5FU2–carboplatin was 3.9 months (95% CI: 3.39–4.55). After the discontinuation of LV5FU2–carboplatin, 17 (18.7%) and 12 (13.2%) patients received one or more subsequent treatment lines. Median OS from initial PDAC diagnosis was 24.4 months (95% CI: 20.0–28.9).
Median OS after initiation of LV5FU2–carboplatin was 4.2 months (95% CI: 3.5–4.9) and the 3-, 6-, 9- and 12-month OS rates were 74.1 ± 4.7%, 33.7 ± 5.1%, 27.6 ± 4.8%, and 19.9 ± 4.4%, respectively. Neither previous morphological response to oxaliplatin (HR: 1.04; 95% CI: 0.67–1.62; p = 0.85) nor prior progression while on oxaliplatin (HR: 1.09; 95% CI: 0.61–1.95; p = 0.78) did influence OS (Figures 3(a) and (b)). Interestingly, median OS was 13.8 months (95% CI: 0.34–24.34) in patients achieving clinical benefit, compared to 3.5 months (95% CI: 3.26–3.70) in patients who did not (p < 0.001).

OS curves of patients with pretreated advanced PDAC treated with LV5FU2–carboplatin, depending on variables of interest. (a) Prior objective response to oxaliplatin or not (p = 0.85); (b) prior progression on oxaliplatin or not (p = 0.78); (c) presence of extrahepatic metastases or not (p < 0.001); (d) ascites or opioid-requiring pain or not (p = 0.006); (e) <2 or ⩾2 prior treatment lines (p = 0.013); and (f) carboplatin administered at reduced or full dose (p = 0.058).
On multivariate analysis (Table 4), opioid-requiring pain or ascites (HR: 1.71; 95% CI: 1.03–2.86; p = 0.039) was significantly associated with shorter OS. Moreover, extrahepatic metastases (HR: 1.62; 95% CI: 0.98–2.68; p = 0.058), a number of previous lines ⩾2 (HR: 1.89; 95% CI: 0.96–3.71; p = 0.065) and carboplatin administered at reduced dose (HR: 1.51; 95% CI: 0.92–2.47; p = 0.10) influenced OS without reaching the threshold of statistical significance (Figure 3(c)–(f)).
Multivariable analysis of variables associated with worse OS.

HR, hazard ratio; OS, overall survival; WHO, World Health Organization.
Of note, among the six patients with known BRCA1/2 germline mutation, four achieved tumor control (66%), median PFS was 5.9 months, and median OS was 12.1 months.
Discussion
The FOLFIRINOX and gemcitabine plus nab-paclitaxel combinations are well-established standards for first-line treatment in patients with advanced PDAC.6,7 With the exception of NALIRI after gemcitabine-based first-line chemotherapy, 13 further-line chemotherapies are not standardized, although their use has been associated with prolonged survival in fit patients. 23 We report here that LV5FU2–carboplatin allowed to achieve median OS and PFS of 4.2 and 2.7 months, respectively, in patients with heavily pretreated advanced PDAC. While these figures appear limited, they are consistent with previously published data. 23
In our study, LV5FU2–carboplatin was prescribed in 82.4% of cases in second line of treatment, after failure of previous recommended regimens, 17.7 months after initial diagnosis of PDAC on average. Patients had preserved general and nutritional status (WHO performance status of 0 or 1 in approximately 75% of cases and a median albumin level of 36.6 g/l), which are important prognostic indicators. 24 Of note, a positive selection bias toward patients with good overall prognosis existed in this cohort, reflected by a median OS from initial PDAC diagnosis of 24.4 months (95% CI: 20.0–28.9).
This study could not assess whether LV5FU2–carboplatin could be more beneficial than best supportive care alone for patients with advanced PDAC after failure of standard chemotherapies as it was nonrandomized and retrospective. Although outcomes were limited, 9-month OS rate was close to 30%, which is worth underlining for a setting with such dismal prognosis. Our results indicate that beyond the number of previous treatment lines, the absence of ascites or opioid-requiring pain and the absence of extrahepatic metastases might be criteria for selecting the best candidates for LV5FU2–carboplatin. Consistently, several studies reported second-line chemotherapy to be less efficient in patients with higher metastatic extent, particularly peritoneal metastases.25,26 In addition, general condition compatible with full chemotherapy doses and slower disease evolution (diagnosis > 18 months before treatment initiation) was associated with prolonged tumor control under LV5FU2–carboplatin. Similarly, in a previous series, second-line chemotherapy achieved longer OS in patients who had longer first-line treatment duration. 27 In our analyses, only prognostic factors in the setting of advanced disease were associated with survival. Hence, it is possible that these variables translate disease behavior rather than true treatment effectiveness, notably considering the long survival of this selected group of patients who were able to undergo multiple lines of therapy. Overall, this underlines the prognostic importance of PDAC natural history, that is, a disease with slower progression should be better controlled with subsequent lines of treatment.
Interestingly, previous efficacy of oxaliplatin-based chemotherapy – or previous progression while under oxaliplatin – did not influence PFS or OS using LV5FU2–carboplatin chemotherapy. Consistently, antitumor activities of oxaliplatin and carboplatin share some similarities, but had also different mechanisms. Notably, and conversely to carboplatin, the cytotoxicity of oxaliplatin is not mediated by DNA damage response but through ribosome biogenesis stress. 17 Hence, previous efficacy of oxaliplatin may not be used for selection of candidates to further-line LV5FU2–carboplatin. Especially, prior limited efficacy of oxaliplatin might not discourage the use of further-line LV5FU2–carboplatin in otherwise good candidates.
We observed a possible higher efficacy of LV5FU2–carboplatin in patients with germline BRCA1/2 mutations, although the low number of patients did not allow for inferential analyses. These results must be indicated with caution as all patients did not undergo germline testing but only in case of suggestive personal or family past history. Still, homologous recombination deficiency is usually associated with increased efficacy of platinum salts in malignancies including PDAC. 28 Hence, patients with germline BRCA1/2 mutations might be good candidates for further carboplatin-based chemotherapy, as previously shown in patients with BRCA-mutated gynecologic cancers. 29
In our study, 72.5% patients received reduced doses of LV5FU2–carboplatin due to poor tolerance to previous treatments and/or progressive impairment of their general condition. Of note, the use of reduced doses of carboplatin was associated with reduced PFS, even after adjustment on other potentially cofounding variables. Keeping in mind that LV5FU2–carboplatin may achieve no or limited efficacy in most patients, it seems reasonable to reserve its use for patients fit enough to receive it at full doses. Residual peripheral neurotoxicity might not lead to systematically reduce carboplatin doses. Indeed, carboplatin-induced worsening of cumulative toxicity was infrequent in our series (13%), which is consistent with previous literature. 30 Finally, the tolerance of LV5FU2–carboplatin was acceptable despite hematological toxicity that was non-negligible with 18% of grade 3 neutropenia. This could however be prevented by the use of GCS-F.
The main limitation of our study is its retrospective design, although data collection was standardized, and missing data were limited. Notably, this retrospective study could not explore quality of life, which is of upmost importance in the context of palliative chemotherapy. As previously underlined, the absence of a control group consisting in best supportive care alone did not enable to evaluate the magnitude of benefit using LV5FU2–carboplatin, in this clinical setting associated with dismal prognosis. As discussed above, the inclusion of patients able to receive further-line chemotherapy inevitably implied a positive selection bias toward patients with good overall prognosis. Finally, while the therapeutic management of PDAC has changed during the 2009–2021 period, the most significant changes have concerned perioperative chemotherapy in non-metastatic setting, hence with limited expectable influence on our results.
On the other hand, this is the largest single series on LV5FU2–carboplatin chemotherapy in patients with pretreated advanced PDAC. In fact, a still limited number of studies have addressed the question of further-line treatment in advanced PDAC with results similar to those of our study, underlining the little activity of treatments for most patients in this setting. The clinical deterioration and poor prognosis of refractory patients greatly limit the number of candidates for further-line treatment.31–36 Nevertheless, the availability of more effective standard first- and second-line chemotherapies (i.e. FOLFIRINOX and nab-paclitaxel in first line, NALIRI after gemcitabine-based first-line chemotherapy) and improvements in supportive care during the last decade have improved OS of PDAC patients, thus progressively increasing the number of patients who are eligible for further-line treatments.6,7,13,37
In summary, LV5FU2–carboplatin combination has an acceptable tolerance but limited overall efficacy in patients with advanced PDAC after failure of standard chemotherapies. Nonetheless, this treatment showed some efficacy in selected patients without ascites or opioid-requiring pain, longer disease evolution and who are clinically fit to receive full-dose carboplatin. Prospective studies are needed to assess the role of LV5FU2–carboplatin combination in this specific subpopulation, using predictive tumoral biological markers such as an enlarged testing of DNA-repair genes.
