Abstract
BACKGROUND:
Radioresistance leads to treatment failure in patients with nasopharyngeal carcinoma (NPC). Thus, enhancing the radiosensitivity of NPC cells would likely increase the effectiveness of radiotherapy. Annexin VII (Annexin A7, ANXA7) might be a tumor promoter in NPC but its functions in radiosensitivity remain unclear.
METHODS:
NPC cell lines CNE2-shANXA7 and CNE2-pLKO.1 were generated and CNE2-shANXA7 nude mice xenograft tumor models were established. The main effects and molecular mechanisms of ANXA7 knockdown in NPC radiosensitivity were studied in vitro and in vivo by analyzing cell viability, clonogenicity, apoptosis, cell cycle distribution, tumor radioresponse and immunohistochemistry assay.
RESULTS:
ANXA7 knockdown revealed potentially enhanced NPC cell radiosensitivity via apoptosis and increased the cell number at the G2/M phase. In the xenograft model, NPC cells with ANXA7 knockdown were dramatically sensitive to irradiation and tumor growth was significantly suppressed. Compared to CNE2-pLKO.1 xenografts, CNE2-shANXA7 showed more
CONCLUSIONS:
ANXA7 knockdown increased the radiosensitivity of NPC by enhancing apoptosis, modulating the cell cycle distribution into more radiosensitive phases, promoting DNA damage, and inhibiting repair. We showed that decreased ANXA7 levels enhanced radiosensitivity and provided insights into the therapeutic targets for NPC radiotherapy.
Introduction
Nasopharyngeal carcinoma (NPC) is characterized by distinct geographical distributions and is one of the most common malignant tumors in southern China and Southeast Asia, with an incidence ranging from 20 to 50/100,000 [1, 2], and a 5-year survival of 70–80% after appropriate therapy [3]. Radiotherapy (RT) remains the preferred treatment for patients with NPC because of its high sensitivity. However, radioresistance leads to treatment failure in patients receiving radiotherapy for NPC [4, 5]. Hence, preventing radioresistance and predicting and increasing radiosensitivity are critical for further improving patient prognosis and increasing their 5-year survival rates. Annexin VII (Annexin A7, ANXA7), a member of the annexin family, is a calcium and phospholipid-binding protein with diverse properties. The protein was overexpressed in NPC compared to normal tissues [6] and is closely related to apoptosis and metastasis [7]. It might also act as a tumor promoter in NPC [8]. However, the function of ANXA7 in the radiosensitivity of NPC remains poorly understood. Therefore, we investigated the role of ANXA7 and the underlying molecular mechanisms of NPC radiosensitivity.
Materials and methods
Materials
pLKO.1 vector and pLKO.1-ANXA7-shRNA plasmids were obtained from RiboBio Co. Ltd (Guang-zhou, China). Fetal bovine serum (FBS), Dulbecco’s modified Eagle’s medium (DMEM), and Lipofectamine 2000 were bought from Invitrogen (Invitrogen, Carlsbad, CA, USA). Microporous polyvinylidene fluoride (PVDF) transfer membranes were bought from Millipore (Billerica, MA, USA). The anti-ANXA7 monoclonal antibody was obtained from Abcam (ab197586, Cambridge, UK).
Human NPC tissue samples
Eighty-two human NPC tissue samples (42 radiosensitive NPC tissues and 40 radioresistant NPC tissues) were obtained from the First Affiliated Hospital of the University of South China, China. The patients were pathologically diagnosed as having NPC by experts from the Department of Pathology. Three months after the diagnosis, cancer regression in the neck and nasopharynx (nasal endoscopy and nasopharyngeal computed tomography [CT] examination) was assessed in all patients who completed RT. The short-term therapeutic effect of RT was evaluated according to the international standard, in which progression of disease (PD) was defined as increased tumor volume; stable disease (SD) as a tumor volume decrease by
Cell lines
CNE2 and CNE2-IR (a radioresistant subclone cell line derived from CNE2) cells used in our previous study [9] were cultured in DMEM including 10% FBS in a 37
CNE2 cell transfection with ANXA7 shRNA plasmids
To establish a stable knockdown of the ANXA7-expressing NPC cell line, CNE2 cells were transfected with pLKO.1 or pLKO.1-ANXA7-shRNA plasmids by Lipofectamine 2000 transfection reagent according to the manufacturer’s instructions. After selection with 1.0
Western blotting
CNE2-pLKO.1 and CNE2-shANXA7 cell lysates were examined by western blotting as previously described [10]. In brief, the lysates (30
Clonogenic assay
After exposure to ionizing radiation (IR), the radiosensitivities of CNE2-shANXA7 and CNE2-pLKO.1 cells were evaluated by the clonogenic assay as previously reported [10]. In brief, the cells were cultured in 6-well plates and exposed to IR at doses ranging from 0 to 8 Gy. After being cultured for 2 weeks, holoclones with
Cell viability after irradiation
The cells were seeded into sterile 96-well microtiter plates (2,000 cells/well), cultured for 12 h, and irradiated (5 Gy). Cell viability was assessed by the 3-(4,5)-dimethylthiahiazo-(-z-y1)-3,5-di-phenytetrazolium-romide (MTT) assay. After 24 h, 5 g/L MTT (20
Apoptosis and cell cycle post-irradiation with flow cytometry
The cells were cultured for 12 h in T-100 flasks and exposed to IR (5 Gy). After culturing for another 24 h, the cells were collected, fixed with chilled 70% ethanol for 1 h at
Hoechst 33258 staining of irradiated cells
After culturing for 12 h in 6-well plates, the cells were exposed to 5 Gy of IR. After 72 h, the culture was fixed at 4
Expressions of ANXA7 in the radiosensitive and radioresistant NPC tissues
Expressions of ANXA7 in the radiosensitive and radioresistant NPC tissues
NOTE: *
All experimental protocols involving animals in this study were approved by the local ethical committee. Male athymic Balb/c nude mice (18–24 g, 5–6 weeks of age) were bought from Shanghai Laboratory Animal Center (Shanghai, China) and were adaptively bred in a laminar airflow chamber under specific-pathogen-free conditions. CNE2-shANXA7 and CNE2-pLKO.1 cells were grown at 37
Immunohistochemistry
For immunohistochemistry, 40 radioresistant and 42 radiosensitive NPC formalin-fixed and paraffin-embedded tissue sections were incubated with anti-ANXA7. Tumor tissues from the nude mice with NPC were assayed by immunohistochemistry for phosphorylated H2A histone family member X (
TUNEL assay in situ
Apoptotic cells were detected by TUNEL assay in paraffin-embedded tissue sections obtained from the xenograft tumors after IR according to a standard protocol. The TUNEL-positive cells, indicated by a diffuse distribution of fine brownish-yellow particles or the intracellular distribution of coarse brown particles, were counted under a microscope. To quantitatively evaluate the apoptotic cells, the sections were examined and the number of TUNEL-positive cells per 1,000 cancer cells in 10 random microscopic fields was determined by light microscopy.
Statistical analysis
The mean
Results
ANXA7 expression in human NPC tissues and cell lines
As shown in Fig. 1A, immunohistochemical analysis showed ANXA7 mainly in the nuclei, with mark-edly lower expression in radiosensitive tissues than that in radioresistant tissues. ANXA7 overexpression was observed in 52.4% (22/42) of radiosensitive human NPC tissue and 77.5% (31/40) of radioresistant human NPC tissue samples (Table 1,
The expressions of ANXA7 in NPC cell lines and human NPC tissues. (A) Immunohistochemistry was performed to examine the expressions of ANXA7 proteins in human NPC tissues. (B) Western blotting was used to analyze the expressions of ANXA7 in CNE2 and CNE2-IR cell lines. (C) The histogram showed the expressions level of ANXA7 in CNE2 and CNE2-IR cells as determined using densitometric analysis. 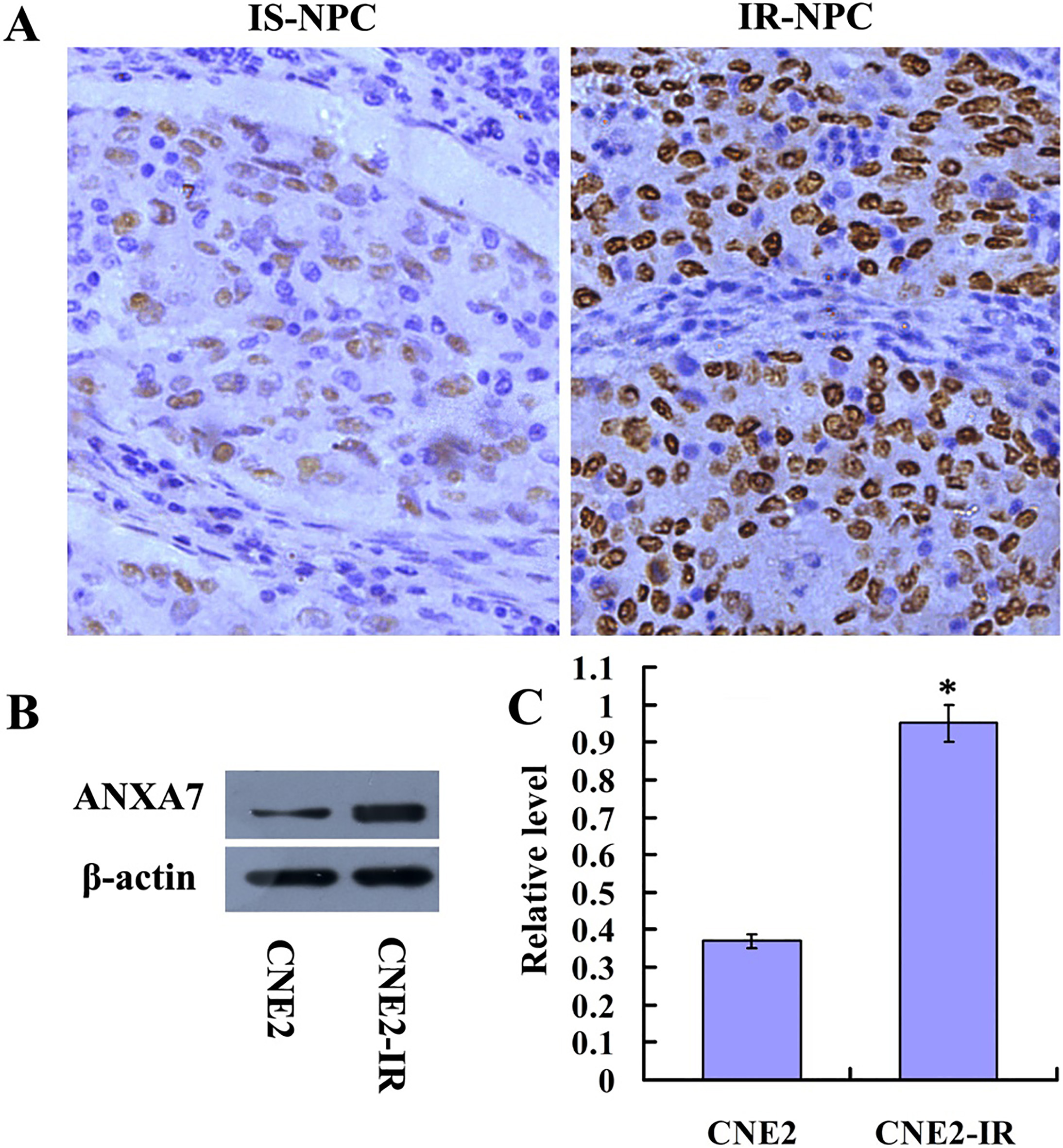
Construction of CNE2-shANXA7 cell line with overexpression of ANXA7. (A) A representative result showed the establishment of CNE2-shANXA7 cell line with knockdown of ANXA7, pLKO.1-ANXA7-shRNA-tansfected CNE2 cells (left), the vector pLKO.1-transfected CNE2 cells (Middle), and non transfected CNE2 cells (right). (B) The histogram showed the transfection rate of CNE2-shANXA7, CNE2-pLKO.1 and CNE2 cells. (C) Immunohistochemistry analysis was used to examine the expressions of ANXA7 in pLKO.1-ANXA7-shRNA-tansfected, pLKO.1-transfected and untransfected CNE2 cells. (D) Western blot analysis was used to examine the expressions of ANXA7 in pLKO.1-ANXA7-shRNA-tansfected, pLKO.1-transfected and untransfected CNE2 cells. (E) The histogram showed the expressions of ANXA7 in CNE2, CNE2-pLKO.1 and CNE2-shANXA7 cells as determined using densitometric analysis (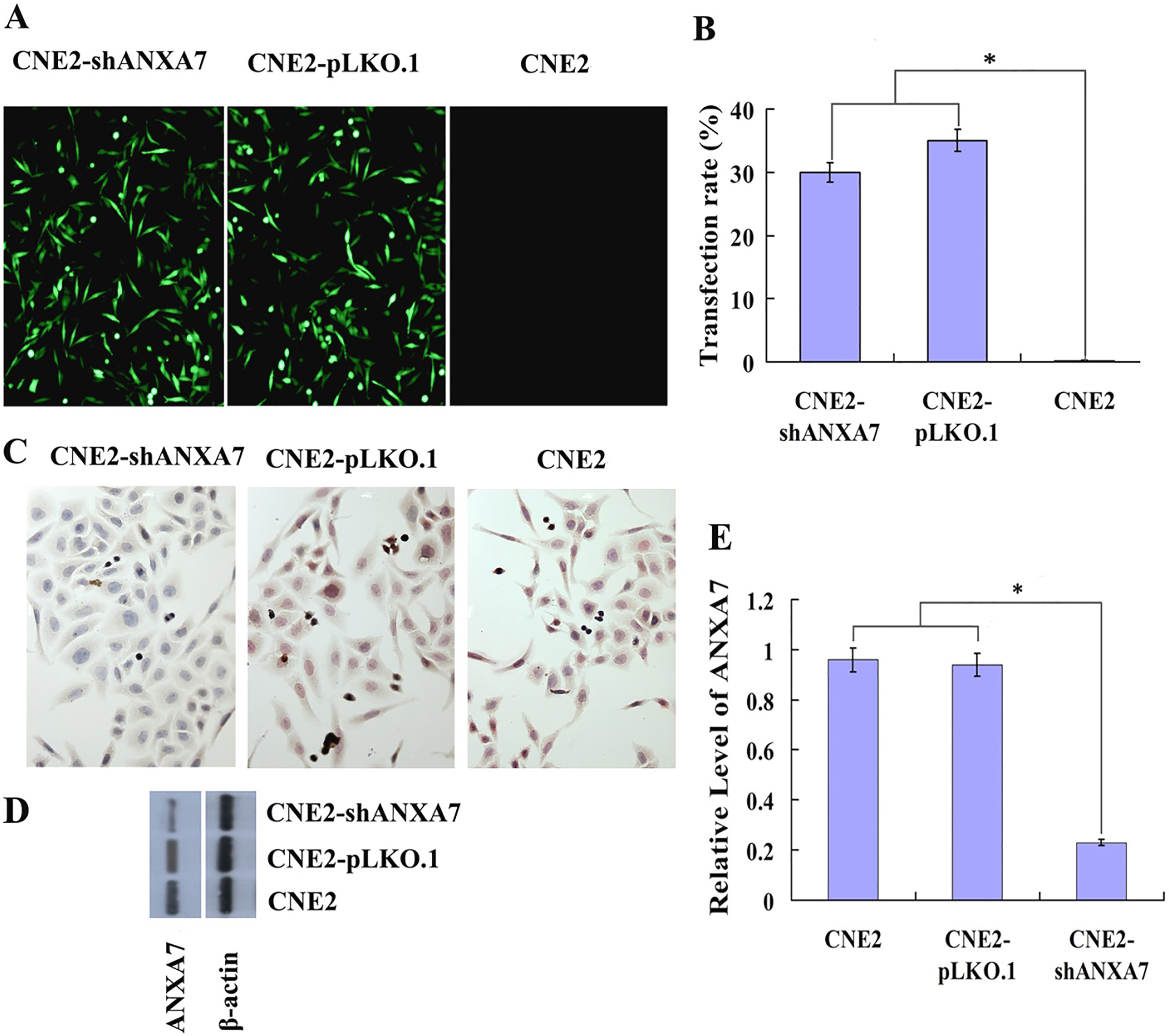
To explore the association of ANXA7 expression in NPC cells with radiosensitivity, the pLKO.1 and pLKO.1-ANXA7-shRNA plasmids were transfected into CNE2 cells with puromycin screening. Fourteen days later, the colonies were picked and expanded and the ANXA7 expression was detected by immunohistochemistry and western blotting. As shown in Fig. 2, the ANXA7 protein expression level in CNE2-shANXA7 cells was markedly lower than that in CNE2 and CNE2-pLKO.1 cells (
Cell cycle distribution of CNE2-shANXA7 and CNE2-PLKO.1 cell lines at 24 h after irradiation with 5 gray
Cell cycle distribution of CNE2-shANXA7 and CNE2-PLKO.1 cell lines at 24 h after irradiation with 5 gray
Note:
CNE2-pLKO.1 and CNE2-shANXA7 cells were assessed by the clonogenic survival assay after IR with doses from 0 to 8 Gy to investigate the relationship between ANXA7 knockdown and radiosensitivity in NPC cells. As shown in Fig. 3A, the surviving colonies of CNE2-shANXA7 cells were remarkably fewer and smaller than those of CNE2-pLKO.1 cells (Fig. 3B). The DMFs were 0.67 and 0.53 at 1% and 10% of isosurvival levels for CNE2-shANXA7, respectively. The SF2 (SF at 2 Gy) was 0.41 for CNE2-shANXA7 cells and 0.76 for CNE2-pLKO.1 cells. Furthermore, following the exposure of CNE2-shANXA7 and CNE2-pLKO.1 cells to 5 Gy radiation to analyze the effects on cell growth, we observed that the cell viability decreased more slowly in CNE2-pLKO.1 cells than in CNE2-shANXA7 cells (Fig. 3C). Together, these findings showed a markedly higher radiosensitivity of CNE2-shANXA7 cells than of CNE2-pLKO.1 cells, indicating that ANXA7 expression decreased their radiosensitivity.
Effects of ANXA7 knockdown on radiosensitivity of NPC cells after irradiation in vitro. (A and B) Clonogenic survival assay. CNE2-shANXA7 and CNE2-pLKO.1 cells were plated in six-well plate and were irradiated with radiation doses of 0–8 gray, and the survival fraction was calculated by counting the amount of colonies after 12 days of incubation. (C) Cell viability analysis. CNE2-shANXA7 and CNE2-pLKO.1 cells were plated into 96-well plate, after 12 h of culture, cells were irradiated (5 gray). MTT assays was used to monitor the cell viability at different time intervals. CNE2-shANXA7 vs. CNE2-pLKO.1. 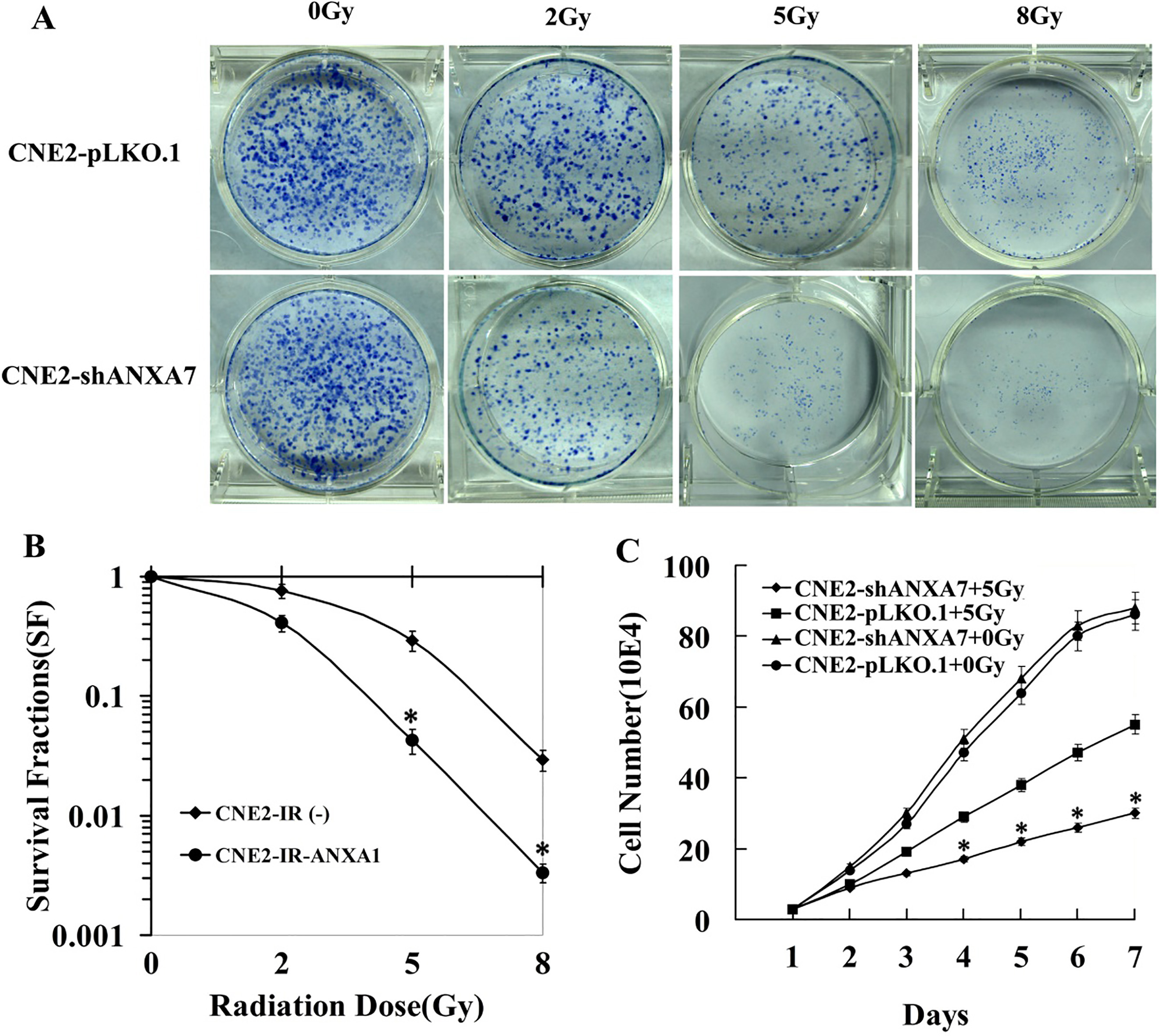
The differences in apoptosis induced by IR between CNE2-shANXA7 and CNE2-pLKO.1 cells were studied by FCM analysis and Hoechst staining assay. We observed more apoptotic CNE2-shANXA7 cells than control cells.
Effects of ANXA7 knockdown on cell cycle distributions in NPC cells post-IR
Cell cycle progression at various phases may determine the radiation sensitivity of NPC cells. The cell cycle distributions in CNE2-shANXA7 and CNE2-pLKO.1 cells were examined by FCM 24 h after exposure to 5 Gy IR. The results are shown in Table 2. No differences between the two groups were noted in G0/G1 phases at 24 h post-IR. However, as compared to CNE2-pLKO.1 cells, fewer CNE2-shANXA7 cells were detained in the S and G2/M phases (
Effects of ANXA7 knockdown on apoptosis of NPC cells post-irradiation. (A) Hoechst staining assay. CNE2-shANXA7 and CNE2-pLKO.1 cells were irradiated with 5 gray of irradiation, after culturing for 72 h, apoptotic cells were identified by hoechst 33258. Apoptotic nuclei (arrow) being stained with intense fluorescence were indicated. (B) The histogram showed the apoptotic rate of CNE2-shANXA7 and CNE2-pLKO.1 cells. The experiments were repeated three times; mean, points, bars, SD. (C) FCM analysis. CNE2-shANXA7 and CNE2-pLKO.1 cells were irradiated with 5 gray of irradiation, after culturing for 24 h, the apoptosis were assessed with FCM. CNE2-shANXA7 vs. CNE2-pLKO.1, 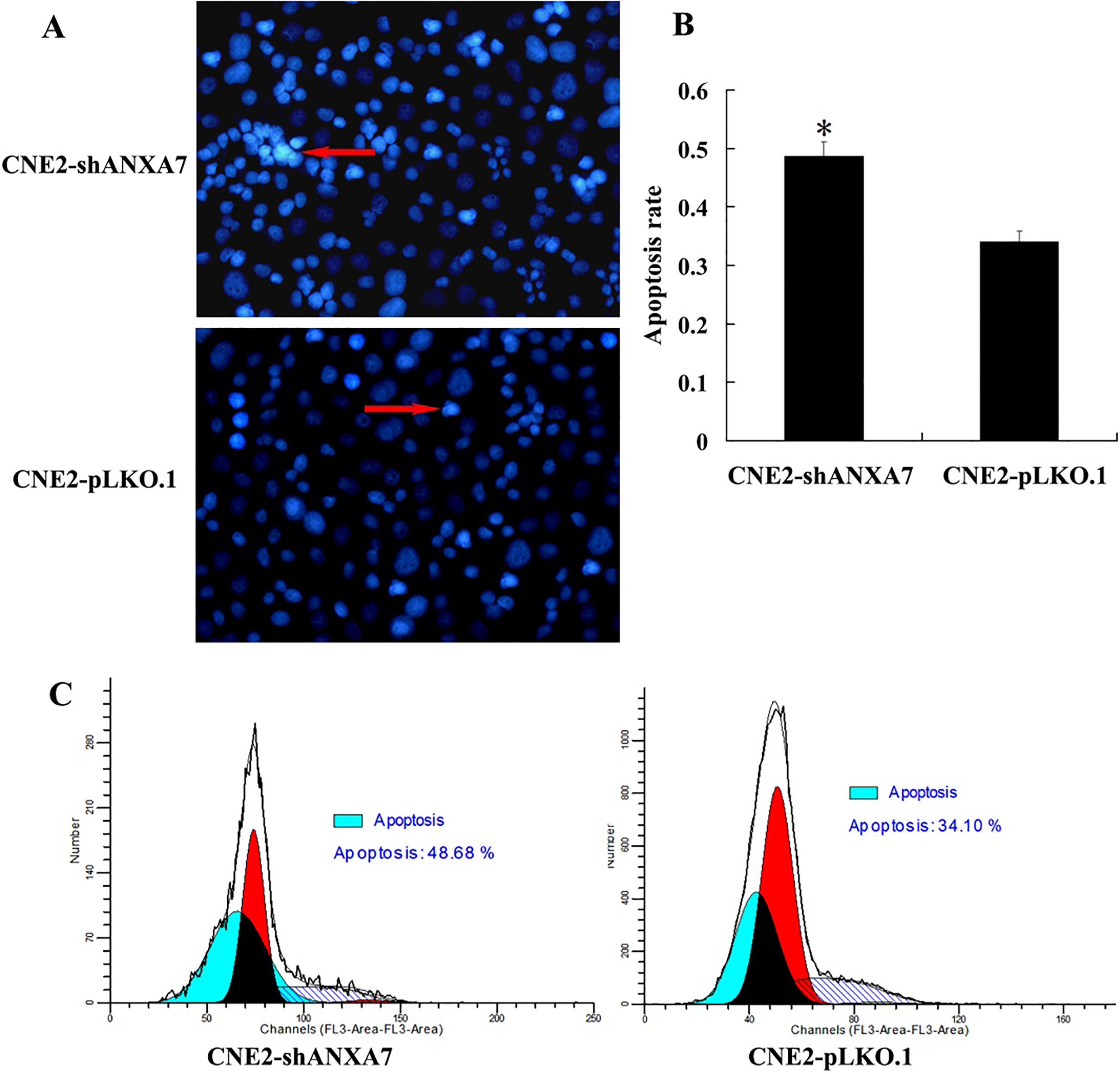
Xenograft nude mice models were developed using ANXA7 knockdown NPC cells to study the effects of ANXA7 on NPC radiosensitivity in vivo. The radioresponses of the tumors were estimated after irradiation with total doses of 10 Gy. Slower tumor growth was observed in the ANXA7 knockdown mice. The subcutaneous xenografts in the CNE2-shANXA7 group were significantly smaller and lighter than those in the CNE2-pLKO.1 group. The results showed markedly higher radiosensitivity in CNE2-shANXA7 cell tumors with ANXA7 knockdown than that in CNE2-pLKO.1 cells. TUNEL assay revealed that the radiation-induced apoptosis in CNE2-pLKO.1 xenografts was much less than that in CNE2-shANXA7 xenografts, indicating that ANXA7 knockdown markedly increased tumor cell apoptosis in xenografts. In addition, immunohistochemical staining showed that ANXA7 knockdown increased the ratio of
CNE2-shANXA7 increases NPC cell radiosensitivity in vivo. (A) The weight and volume of tumors generated by CNE2-shANXA7 cells and CNE2-pLKO.1 cells following 10 gray irradiation. (right) The growth curves of the tumors post-irradiation; (middle) the mean weights of the tumors post-irradiation at the sacrifice; (left) the mice were put to death, and the tumors were photographed 2 weeks post-irradiation. (B) (left) representative image of TUNEL detection of apoptosis in situ in the tumors after irradiation; (right) histogram indicated the percent of apoptotic cells. (C) (left) Representative immunohistochemical staining of 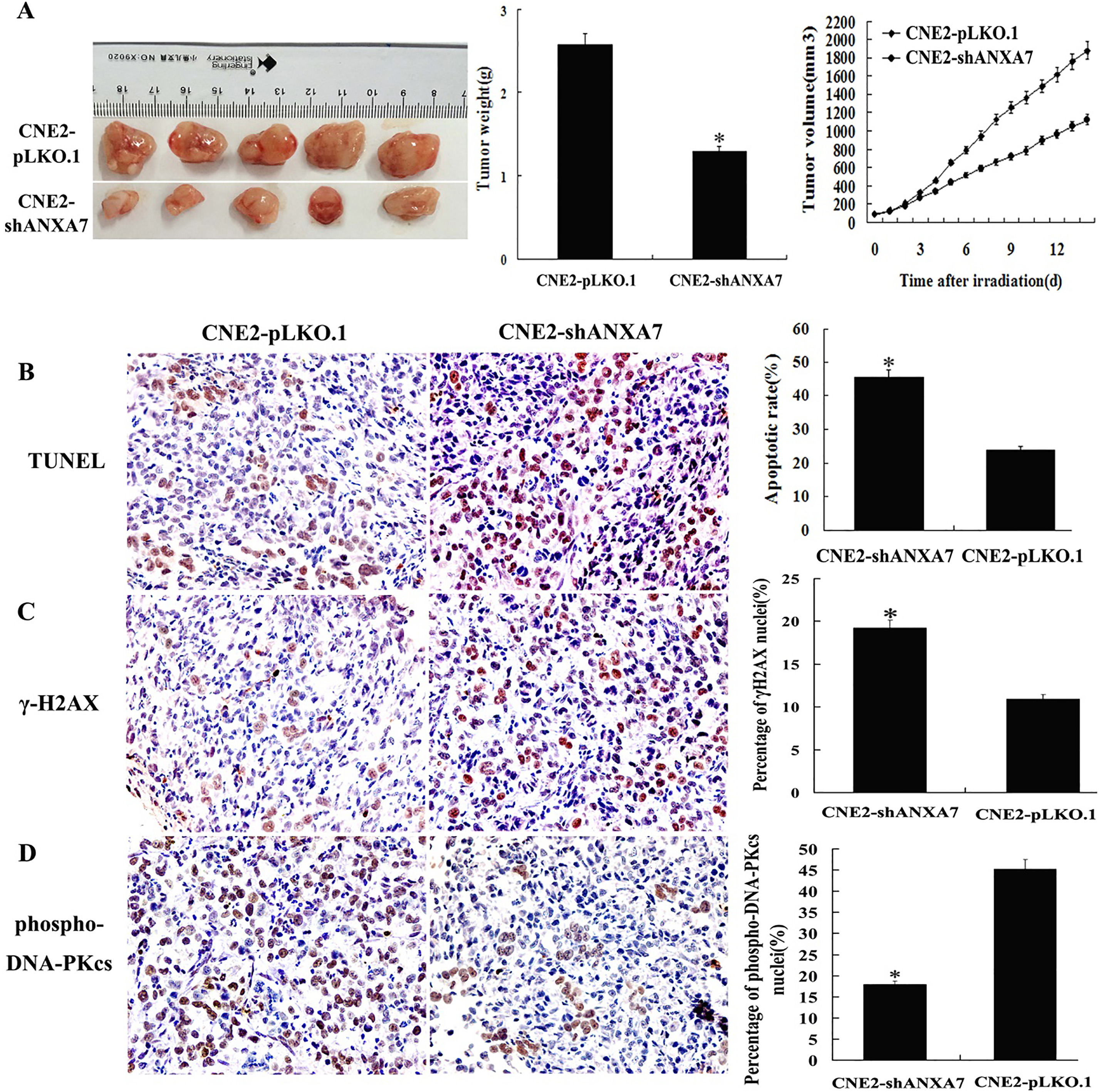
RT is the mainstay of therapy for most patients with NPC because of its high sensitivity. Improving the radiosensitivity of cancer cells is key for strengthening the therapeutic effect of RT. The radiosensitivity of NPC is related to factors such as cell growth and proliferation, apoptosis, cell cycle, and repair of radiation damage. Accumulating evidence indicates that ANXA7 proteins play an important role during tumor development and progression. The deregulation of ANXA7 proteins is associated with differentiation, apoptosis, metastasis, and poor prognosis in various tumors [12, 13, 14]. To elucidate the role of ANXA7 and its underlying mechanism in NPC radioresistance, we constructed stable ANXA7 knockdown (CNE2-shANXA7) and control (CNE2-pLKO.1) cell lines and performed radioresponse tests in vitro. The clonogenic survival assay findings demonstrated remarkably fewer and smaller surviving CNE2-shANXA7 cell colonies than those of CNE2-pLKO.1 cells. The results of the MTT assay showed significantly reduced CNE2-shANXA7 cell viability following IR compared to that in CNE2-pLKO.1 cells. These results suggested that ANXA7 knockdown could promote cell growth and proliferation, as the radiosensitivity of CNE2-shANXA7 cells was higher than that of CNE2-pLKO.1 cells. To elucidate the effects of ANXA7 knockdown on cellular radiosensitivity of NPC in vivo, CNE2-shANXA7 and CNE2-pLKO.1 mouse xenograft models were established. The subcutaneous tumors from the CNE2-shANXA7 group were significantly smaller and lighter than those from the CNE2-pLKO.1 group post-irradiation in vivo. The data showed significantly inhibited xenograft tumor growth post-radiotherapy in the ANXA7 knockdown group and markedly higher radiosensitivity of tumors from CNE2-shANXA7 cells than that for tumors from CNE2-pLKO.1 cells. Hoechst staining and FCM showed higher radiation-induced apoptosis in CNE2-shANXA7 cells than in CNE2-pLKO.1 cells. TUNEL assay in tumor tissue sections showed markedly increased cellular apoptosis in xenografts from ANXA7 knockdown cells. Cell cycle in CNE2-pLKO.1 and CNE2-shANXA7 cells measured by FCM at 24 h post-IR with 5 Gy showed no difference with respect to G0/G1 phases, but more G2/M phase and less S phase arrests were observed in CNE2-shANXA7 cells than in CNE2-pLKO.1 cells. Taken together, our findings demonstrated that ANXA7 knockdown remarkably increased NPC cell radiosensitivity in vivo and in vitro; thus, ANXA7 proteins may participate in the cellular response to irradiation in NPC.
Apoptosis plays a vital role in the cell death pathway after IR; thus, radiation-induced apoptosis is critical in radiation therapy. Tumor cell radiosensitivity is associated with cell cycle checkpoints, apoptosis, and DNA damage repair [15, 16]. Radiation causes DNA damage, triggering the apoptotic pathway in tumor cells. Cells with repairable damage enter the repair program, while cells with un-repairable damage enter the apoptotic process [17]. Furthermore, apoptosis plays a crucial role in RT as a part of the cell death pathway after irradiation [18].
ANXA7, which co-locates with B-cell lymphoma 2 (Bcl2) in the mitochondria and cytoplasm, was associated with Hca-P cell apoptosis via alteration of the mitochondrial membrane potential and Bcl2 expression [19]. Inhibition of ANXA7 expression enhanced apoptosis of BGC823 cells in vivo and in vitro, and markedly inhibited the growth of tumor xenografts [20]. In the present study, ANXA7 knockdown increased IR-induced apoptosis in NPC cells in vivo and in vitro, indicating that this knockdown heightened NPC radiosensitivity by increasing apoptosis. The cell-cycle distribution is one of the most important determinants for tumor radiosensitivity [21]. The cell’s sensitivity to RT varies at different stages of the cell cycle. Cells arrested at the G2/M phase are more sensitive to radiation, whereas cells arrested at the S phase are more resistant to radiation [22]. Zheng et al. reported that miRNA-200c increased esophageal cancer radiosensitivity through sub-G1 and G2/M phase arrest [23]. Cao and colleagues reported a decreased proportion of A549S1 (radioresistant A549) cells in the G0/G1 phase and a consequent increase in the number of cells in the S phase as compared the proportion of A549 cells in these phases [24]. In this study, we found that the ratio of cells at the G2/M phase was remarkably increased in CNE2-shANXA7 cells in comparison with CNE2-pLKO.1 cells after IR. These phases corresponded to the times in which cells are most sensitive to the effects of IR. An inverse relationship was observed in S-phase fraction analysis. Generally, the cell cycle is arrested at certain phases to repair damaged DNA. The cell cycle progresses when the damaged DNA is successfully repaired; otherwise, apoptosis occurs. This process requires many cytokines to repair the cells blocked at the G2/M phase following IR-induced DNA damage [25]. Our findings indicated that ANXA7 knockdown arrested the cell cycle at the G2/M phase, participated in the regulation of radiosensitivity through cell cycle arrest, and increased CNE2-shANXA7 cell radiosensitivity by changing the distribution of the cell cycle.
IR primarily results in DNA damage of cells and activates the DNA damage repair pathways [26]. The DNA damage is closely associated with cell radiosensitivity and is the main cause of tumor cell death after RT [27, 28]. Phosphorylation of histone H2AX is associated with the accumulation of damage repair proteins and chromatin damage [29] and, thus, is a sensitive reporter for DNA damage [27, 30]. Meanwhile, phosphorylation of histone H2AX is positively related to increased DSBs and increased numbers of
Conclusion
In summary, ANXA7 was crucial for radiation sensitivity of NPC cells both in vivo and in vitro; therefore, downregulated ANXA7 protein expression may improve NPC cell radiosensitivity by reducing colony growth, blocking the cell cycle at the G2/M phase, inducing increased apoptosis, increasing
Footnotes
Acknowledgments
We would like to thank Editage (www.editage.cn) for English language editing.
