Abstract
Gastric cancer is one of the most common cancers in the world. However, current medical technologies have not identified a reliable method to cure advanced gastric cancer, and early gastric cancer is difficult to diagnose. Therefore, we focused on circular RNAs (circRNAs) that have been proven to be involved in the carcinogenesis of gastric cancer. We first used quantitative reverse transcription-polymerase chain reaction (qRT-PCR) to evaluate the expression levels of hsa_circ_0005654 in 301 tissues, including 122 healthy gastric mucosa samples, 68 paired tissues from early gastric cancer and adjacent nontumor mucosae obtained by submucosal dissection, and 43 chronic gastritis tissues. Then, we analyzed the relationship between the expression levels of hsa_circ_0005654 and the clinicopathological characteristics of patients with early gastric cancer. We ultimately confirmed the clinical diagnostic value of hsa_circ_0005654 through generating receiver operating characteristic (ROC) curves and comparing the areas under the ROC curves (AUCs).
Our data revealed that hsa_circ_0005654 was significantly downregulated in early gastric cancer tissues compared with matched normal mucosae (
Introduction
Cancer incidence and mortality are rapidly growing in the world [1, 2]. In spite of chemotherapy having a crucial therapeutic value, surgery, which causes substantial patient suffering, is the only known form of curative treatment for most patients with advanced gastric malignancy and soft-tissue sarcomas [3, 4, 5]. In other words, modern medicine still lacks effective treatments for advanced gastric cancer. Accordingly, it is urgently necessary for us to seek new biomarkers of early gastric cancer.
Circular RNAs (circRNAs) that have higher tissue specificity and more stable structures compared with linear RNAs are a unique set of noncoding RNAs [6]. Basically, circRNAs are synthesized by a proactive back-splice event as loops without 5’ caps and 3’ poly (A) tails, and as a consequence, they are difficult to be degraded by ribonucleases including RNase R and exonucleases and more likely to maintain stable expression and thus can be used as biomarkers in diagnosis [7, 8].
CircRNAs play a vital role in gene regulation, neural development and in the initiation and progression of numerous diseases such as atherosclerotic vascular diseases, neurological disorders, cardiovascular diseases and most heart diseases [9, 10, 11, 12, 13]. In addition, some researchers found that circRNAs in human saliva can be used as disease biomarkers [14]. Additionally, some findings indicated that circ_10720, hsa_circ_0064428 and circMTO1 may serve as potential new biomarkers of hepatocellular carcinoma; plasma circ_002453 was also identified as a diagnostic biomarker of lupus nephritis; and hsa_circ_0036877 as a potential novel blood biomarker for early pre-eclampsia was discovered by some scientists [15, 16, 17, 18, 19]. What’s more, fusion circRNAs (f-circRNAs) that are produced from transcribed exons of distinct genes are able to increase the cellular proliferation rate and contribute to cellular transformation and tumorigenesis [20].
Gastric cancer is one of the most common malignant tumors in the world and the second leading cause of cancer-related death worldwide, especially in Eastern Asia, Eastern Europe, and South America [21, 22]. Although surgical techniques integrated with chemotherapy strategies have developed at a rapid pace in recent years, the treatment effectiveness of advanced gastric cancer has not shown any improvement. This is mainly because most patients with gastric cancer cannot be diagnosed in the early phase and thus lose their best curative chance; a recent study suggested that 90% of early gastric cancer patients can be cured [23]. Therefore, the key to overcoming the gastric cancer problem is to diagnose it earlier. Early gastric cancer is gastric cancer limited to the mucosal or superficial submucosal layers regardless of lymph node metastasis, with 0-IIa, 0-IIb, 0-IIc, 0-IIa
Endoscopy followed by pathological diagnosis is still the gold standard for gastric cancer diagnosis. As we all know, endoscopy is a subjective method of diagnosis that may result in misdiagnoses, and whether we can find gastric cancer correctly relies entirely on the experience of the endoscopists. The traditional biomarkers, for example, carbohydrate antigen 19-9 (CA19-9), whose sensitivity and specificity are 60.5% and 55.9%, respectively, cannot diagnose early gastric cancer effectively [26]. Thus, to improve the diagnostic rate of early gastric cancer, finding high-efficiency biomarkers is crucially important.
New types of biomarkers have successfully been identified in recent years [27, 28, 29]. More interestingly, Li et al. found that a combination of different circRNAs that have been identified as cancer biomarkers may increase the specificity and sensitivity of detecting a variety of malignancies as compared with a single circRNA [30].
On account of circRNAs as a category of new biomarkers being related to a large variety of diseases, we began to focus our attention on investigating the roles of circRNAs in gastric cancer. We have explored the global circular RNA expression profile of human gastric cancer [31]. A total of 308 circRNAs, including 201 downregulated circRNAs and 107 upregulated, were found significantly aberrantly expressed in gastric cancer tissues [31]. In this study, we focused our attention on investigating the role of hsa_circ_0005654 (
Materials and methods
Patients’ samples
In this study, a total of 301 tissues were collected. Among them, sixty-eight early gastric cancer mucosae and paired adjacent mucosae were collected from early gastric cancer patients who received treatments through endoscopic submucosal dissection (ESD) procedures, which are small endoscopic surgeries conducted by experienced endoscopists to entirely remove early gastric cancer tissues under endoscopy, at the Affiliated Hospital of Medical School of Ningbo University between September 2016 and February 2018. Furthermore, 122 healthy gastric mucosa samples and forty-three chronic gastritis tissues were obtained from biopsy specimens. All tissues were immediately stored in RNA-fixer Reagent (Bioteke, Beijing, China) after removal from these patients’ stomachs and preserved at
The diagnosis of every sample was confirmed histo-pathologically. Written informed consent forms were obtained before the experiments. Histologic grade was assessed following the National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines for Oncology (V. 2.2018). All of these specimens and data were collected in a double-blind manner. The Human Research Ethics Committee of Ningbo University (IRB No. 20100303) approved all aspects of this research.
Total RNA isolation
Total RNA was isolated from all tissues covering the frozen early gastric cancer mucosae, healthy gastric mucosa samples, chronic gastritis tissues and adjacent nontumor mucosae using TRIzol reagent (Invitrogen, Karlsruhe, Germany) following the manufacturer’s instructions. Then, 20
Electrophoresis of total RNA
To assess the RNA integrity, 1.5% agarose/ethidium bromide gel electrophoresis was used.
Reverse transcription
cDNA was synthesized from 2
Quantitative detection of hsa_circ_0005654
The real-time quantitative reverse transcription-polymerase chain reactions (qRT-PCR) were carried out using GoTaq qPCR Master Mix (Promega) on an Mx3005P Real-Time PCR System (Stratagene, La Jolla, CA, USA) in a 25-
Electrophoresis and sequencing of qRT-PCR products
To identify the qRT-PCR products integrity, gel electrophoresis was used. After that, the qRT-PCR products of hsa_circ_0005654 were purified by using a UNIQ-10 PCR Product Purification Kit (ThermoFisher Scientific, Waltham, MA, USA) and then cloned into the pUCm-T vector (ThermoFisher Scientific). Subsequently, DNA sequencing was performed by the ThermoFisher Scientific Company, Ltd.
Statistical analysis
Statistical analyses were performed using Statistical Program for Social Science version 22 (SPSS 22.0) software (SPSS Inc., Chicago, IL, USA) and GraphPad Prism 8.0 (GraphPad Software, La Jolla, CA, USA). Student’s t-test (two tailed) was used for comparisons between two groups and one-way analysis of variance (ANOVA) was used for multigroup comparisons. A
Results
General characteristics of the subjects
About half of patients in the early gastric cancer group were men (52.9%). The average age of diagnosis of early gastric cancer was 66 years old (Table 1).
The relationship between hsa_circ_0005654 expression levels (
in early gastric carcinomatous mucosae from ESD operations and the clinicopathological factors of patients with early gastric cancer
The relationship between hsa_circ_0005654 expression levels (
ESD: endoscopic submucosal dissection.
The ratio of A
Hsa_circ_0005654 was downregulated in early gastric cancer mucosae and chronic gastritis tissues
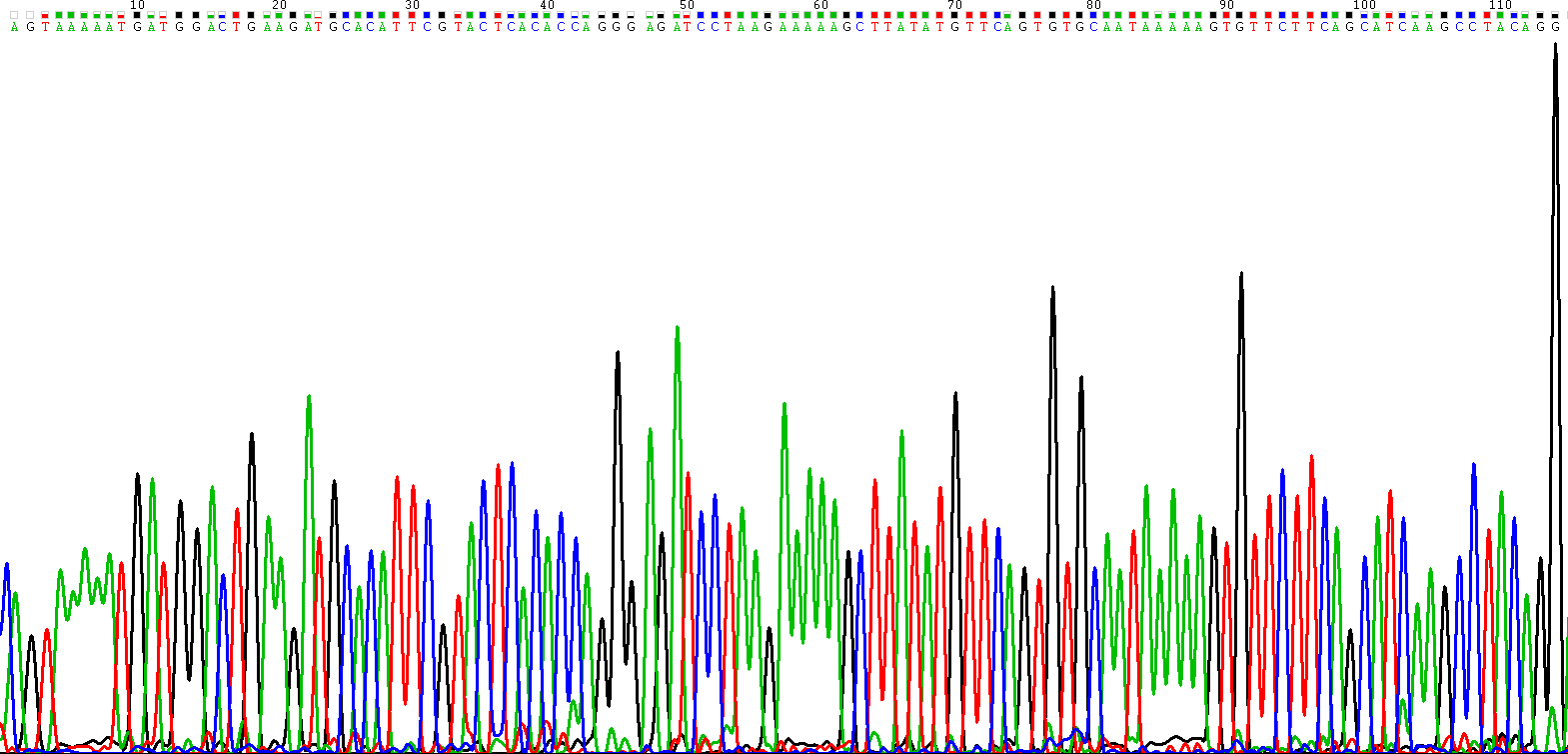
Since no previous studies have evaluated the expression of hsa_circ_0005654 in early gastric cancer, the qRT-PCR method was used to measure hsa_circ_00056 54 expression levels in tissues from early gastric cancer mucosae, healthy gastric mucosa samples and chronic gastritis tissues. After electrophoresis of qRT-PCR products (Supplementary Fig. S2), we confirmed the head-to-tail splicing junction of hsa_circ_0005654 through sequencing of the product of qRT-PCR (Supplementary Fig. S3), which was consistent with the sequence of that from the circBase (
Hsa_circ_0005654 showed a statistically downregulated expression in early gastric cancer mucosae and matched adjacent nontumor gastric mucosae. Real-time qRT-PCR was used to determine the expression level. The 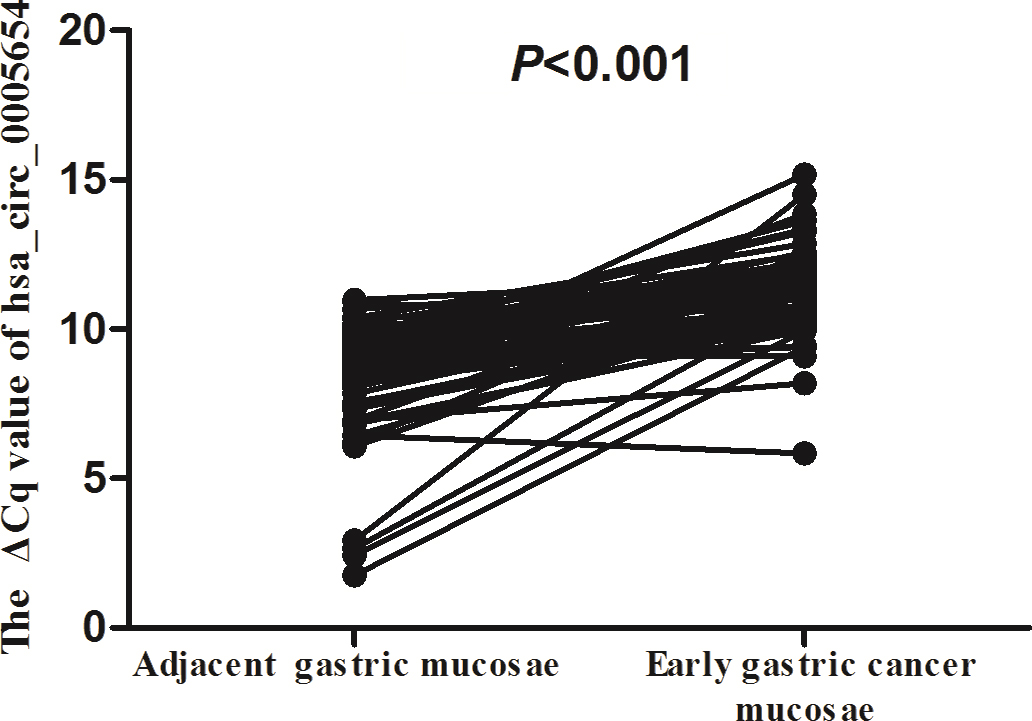
Expression of hsa_circ_0005654 in mucosae samples from 122 healthy participants, 43 chronic gastritis patients and 68 patients with early gastric cancer was detected by qRT-PCR. The expression of hsa_circ_0005654 in mucosae samples of early gastric cancer (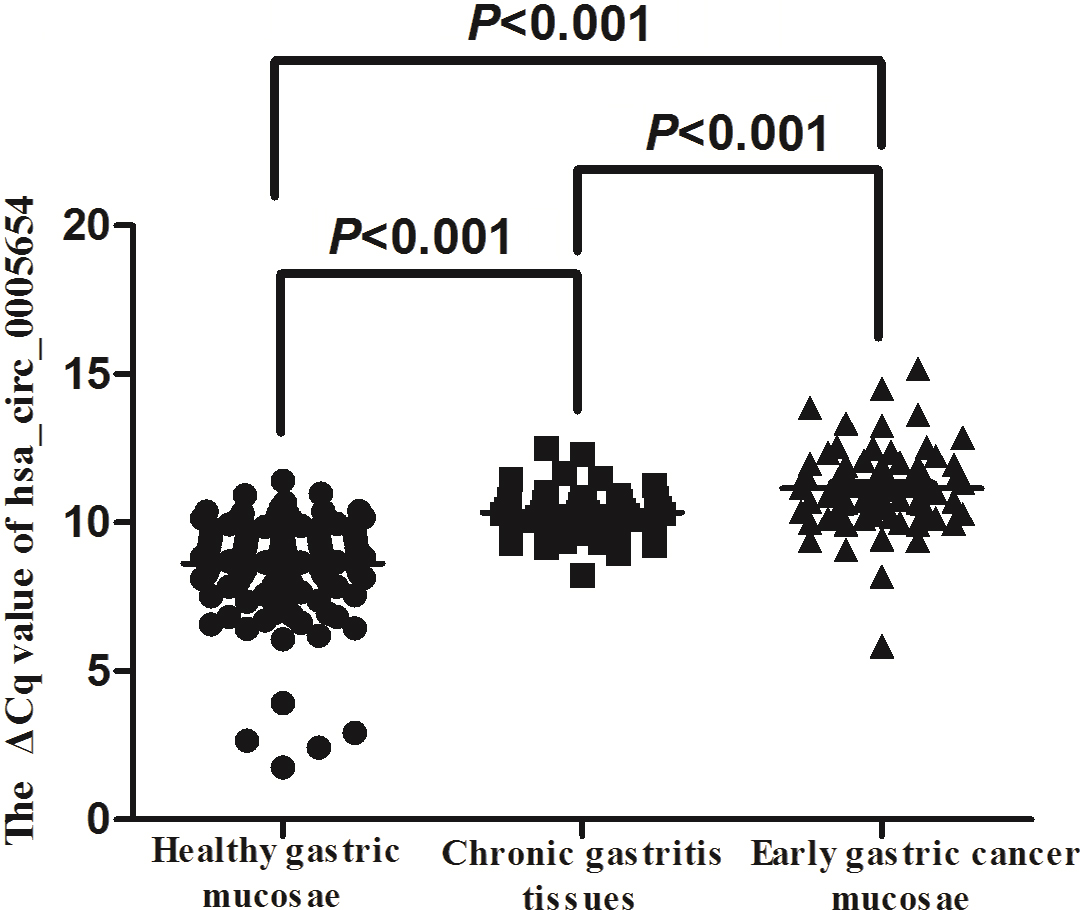
We found that hsa_circ_0005654 expression levels were significantly lower in early gastric cancer mucosae vs. matched nontumor mucosae (Fig. 1). Meanwhile, hsa_circ_0005654 expression levels were also downregulated in early gastric cancer compared with healthy tissues (Fig. 2). Later, we used ROC curves to investigate the diagnostic value of hsa_circ_0005654 in distinguishing early gastric cancer tissues andmatched normal adjacent nontumor mucosae. The area under the ROC curve (AUC) was 0.927 (
Receiver operation characteristics curve was constructed for differentiating early gastric carcinomatous from adjacent nontumor tissues.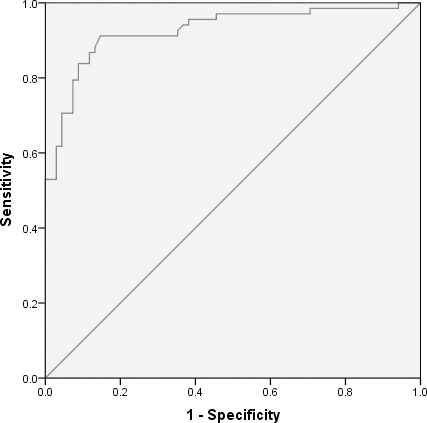
Receiver operation characteristics curve was constructed for differentiating early gastric carcinomatous from healthy controls.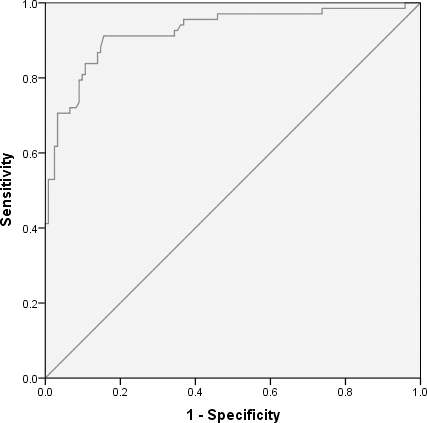
Gastric cancer is the most aggressive digestive system neoplasm [32]. Despite the great efforts that have been made to explore better curative strategies in recent years, combined therapy of surgery and radiotherapy or chemotherapy are still unable to prolong the survival of patients with advanced gastric cancer, and targeted agents have not provided patients with advanced gastric cancer satisfactory outcomes [33, 34]. In contrast, the 5-year survival rate of early gastric cancer can reach 90–95% [35]. Therefore, it’s essential for us to discover gastric cancer and cure it at an early stage. However, present clinical diagnostic biomarkers including CEA and CA19-9, which have low sensitivity and specificity, cannot satisfy the need of early diagnosis of gastric cancer [26]. Thus, it’s necessary and meaningful for us to find a new high-efficient biomarker to discover early gastric cancer.
CircRNAs are abundant and evolutionarily conserved molecules [36, 37]. In recent years, with the development of novel bioinformatics approaches and high-throughput RNA deep sequencing (RNA-seq) technology, a large number and variety of circRNAs have been found [38, 39]. To date, because circRNAs are covalently closed, single-stranded circular transcripts and possess a longer half-life compared with ordinary noncoding RNA, they have become hotspots in the noncoding RNA research field [20, 40]. Despite an increasing number of studies on circRNAs, the expression profiles and functions of circRNAs in early gastric cancer are still unknown. CircRNAs have some important characteristics such as unique structures, conservation across species, cell type-specific and tissue-specific expression patterns, and stability in blood, saliva and exosomes [14, 28, 41]. Moreover, circRNAs’ expression in human cells are sometimes more extensive than their linear subclasses [42]. Because of these characteristics, circRNAs may potentially be used as biomarkers and therapeutic targets of early gastric cancer.
In this study, we investigated whether hsa_circ_000 5654 could be an early biomarker of gastric cancer. To achieve this goal, we used samples taken from patients during ESD operations. These gastric cancer mucosae are samples of early gastric cancer and are good materials for screening early biomarkers of gastric cancer.
Our qRT-PCR results showed that the expression of hsa_circ_0005654 was significantly lower in early gastric cancer mucosae than in adjacent nontumor mucosae and healthy gastric mucosa samples (Fig. 1). We further found that hsa_circ_0005654 was downregulated in chronic gastritis tissues (Fig. 2). In addition, we also discovered that there was a significant difference in expression levels of hsa_circ_0005654 between early gastric cancer mucosae and chronic gastritis tissues (Fig. 2). Additionally, we have explored the early diagnostic value of hsa_circ_0005654 (Figs 3 and 4). To our knowledge, this is the first report on hsa_circ_0005654 expression in early gastric cancer.
In recent studies, several laboratories have proven that compared with ordinary protein-based diagnostic methods, some circRNA-based diagnostic methods have remarkably higher diagnostic values [43, 44, 45, 46]. Based on our data, hsa_circ_0005654 can be used as a new potential biomarker in the diagnosis of early gastric cancer. Its sensitivity and specificity were 91.2% and 85.3%, respectively. The false positive rate and false negative rate were 14.7% and 8.8%, respectively. The positive predictive value and negative predictive value were 86.11% and 90.63%, respectively. Compared with the current clinical diagnostic biomarkers CEA and CA19-9, hsa_circ_0005654 had a higher sensitivity and specificity for screening for early gastric cancer.
Conclusion
Due to its high stability, hsa_circ_0005654 could be used as a novel early diagnostic biomarker of gastric cancer.
Footnotes
Acknowledgments
This work was supported by the National Natural Science Foundation of China (no. 81772279), the Scientific Innovation Team Project of Ningbo (no. 2017C110019), the National Undergraduate Training Program for Innovation and Entrepreneurship (no. 201811646025), the Student Research and Innovation Program of Ningbo University (no. 2018SRIP2507 and no. 2019SRIP1902), and the K. C. Wong Magna Found in Ningbo University.
Conflict of interest
The authors declare that no competing interests exist.
Supplementary data
Representative results of 1.5% agarose/ ethidium bromide gel electrophoresis of total RNA from three representative early gastric cancer mucosae samples.
Representative results of electrophoresis of qRT-PCR products. M, DNA ladder. 1 and 2, early gastric cancer mucosae; 3 and 4, adjacent nontumor mucosae; 5, healthy gastric mucosa; 6, chronic gastritis tissues.
Representative sequencing results of the products of qRT-PCR for the detection of hsa_circ_0005654 in early gastric carcinomatous mucosae.