Abstract
Long non-coding RNAs (lncRNAs) are important to the occurrence and advancement of human cancers. We found through GEPIA that LINC00893 was lowly expressed in thyroid carcinoma (THCA) tissues, whereas the specific functions of LINC00893 has never been reported in PTC. In the current study, we confirmed that LINC00893 was expressed at a low level in PTC cells. Through gain-of-function assays, we determined that LINC00893 overexpression abrogated proliferation and migration abilities of PTC cells. Through signal transduction reporter array we found that LINC00893 potentially modulated the signals of phosphatase and tensin homolog (PTEN)/AKT pathway. In addition, overexpression of LINC00893 increased the expression of PTEN but reduced the levels of phosphorylated AKT in PTC. Additionally, mechanism assays unveiled that LINC00893 stabilized PTEN mRNA via recruiting Fused in sarcoma (FUS) protein. Finally, rescue assays demonstrated that LINC00893 hampered the proliferation and migration of PTC cells via PTEN/AKT pathway. Together, our study first clarified that LINC00893 functions as a tumor suppressor in PTC by blocking AKT pathway through PTEN upregulation.
Introduction
Thyroid carcinoma (THCA) accounts for 1% of all human cancers. It takes the largest proportion in endocrine-related malignancies [1]. Papillary thyroid cancer (PTC) is the most common subtype of THCA, which has achieved extensive attention over the past years [2]. Exposure to ionizing radiation and family history are risk factors for PTC [3, 4]. Conservative radioactive iodine therapy and thyroidectomy are typical therapeutic methods for PTC. However, there are still few PTC patients showing frustrating prognosis due to recurrence and distant metastasis [5, 6]. Thus, exploration of molecular mechanism underneath PTC progression is important to develop more efficient therapeutic targets.
Long non-coding RNAs (lncRNAs), a category of noncoding RNAs (ncRNAs), are initially defined as transcriptional noises [9, 10]. Recent years, more and more dysregulated lncRNAs have been reported to be related to the progression of human cancers, including PTC [10, 11]. For example, LINC00514 can induce the malignant progression of PTC through miR-204-3p/CDC23 [12]. LncRNA FOXD2-AS1 facilitates PTC progression via miR-485-5p/KLK7 [13]. LncRNA AB074169 hampers PTC cell proliferation by affecting KHSRP-modulated CDKN1A expression [14]. In the current study, long intergenic non-protein coding RNA 893 (LINC00893) was chosen as the research object based on the search results of online GEPIA database. To our knowledge, this study is the first one to unveil the role of LINC00893 in PTC.
Through Signal Transduction Reporter Array, we found that LINC00893 was correlated with PTEN/AKT in PTC. Phosphatase and tensin homologue (PTEN) is a well-known tumor suppressor [15]. High PTEN expression is crucial for maintaining normal cellular function [16]. As the upstream gene of AKT, PTEN contributes to the inactivation of AKT signaling pathway to repress cancer development [17, 18, 19]. Moreover, previous studies have revealed that lncRNAs can regulate tumorigenesis and tumor development through regulating PTEN/AKT pathway. For example, lncRNA GAS5 can enhance PTEN expression by absorbing miR-222-3p [20]. Romina Sepe et al. demonstrated that downregulation of lncRNA RP5-1024C24.1 and lncRNA MPPED2 activates PTEN/AKT pathway in thyroid cancer [21]. However, no previous study demonstrated the association between PTEN/AKT and LINC00893 in PTC. Thus, we focused on the association between PTEN/AKT and LINC00893 in PTC progression.
To summarize, the current study focused on the functions of LINC00893 in PTC progression and its association with PTEN/AKT pathway.
Materials and methods
Cell culture
Human PTC cell lines (TPC-1, MDA-T120 and BCPAP) and normal thyroid epithelial cell line Nthy-ori-3-1 were obtained from the American Type Culture Collection (APTCC; Manassas, VA, USA). All cell lines were certified via STR profiling before this study. Cell lines were cultured in RPMI-1640 medium (Gibco, Carlsbad, CA, USA) added with 10% fetal bovine serum (Invitrogen, Carlsbad, CA, USA), 100 U/ml penicillin (Sigma-Aldrich, St. Louis, MO, USA) and 100 U/ml streptomycin (Sigma-Aldrich). All cell lines were maintained in a humidified air fulfilled with 5% CO
Cell transfection
Cell transfection was performed via Lipofectamine 3000 Reagent (Invitrogen) in line with the protocols. In order to silence LINC00893, specific short hairpin RNAs targeting LINC00893 (sh/LINC00893#1 and sh/LINC00893#2) and corresponding controls (sh/NC) were synthesized. In order to overexpress genes, the whole sequences of LINC00893, PTEN and FUS were separately cloned into pcDNA3.1 vectors, and pcDNA3.1 empty vector was utilized as negative control (NC). All plasmids were purchased from RiboBio (Guangzhou, China).
Quantitative real-time PCR (qRT-PCR)
Total RNA was extracted from PTC cells by routinely using Trizol reagent (Invitrogen). RNA integrity was examined by 1% AGE (agarose gel electrophoresis), and RNA purity was detected at 260 nm and 280 nm using spectrophotometer (Bio-Rad, Hercules, CA). The RNA was then reversely-transcribed into cDNA via PrimeScript RT reagent Kit (Takara, Dalian, China). SYBR Premix EX Taq II Kit (Takara) were applied to perform qRT-PCR. Relative expression was calculated by the 2
Cell counting kit 8 (CCK-8) assay
CCK-8 kit (CCK-8; Beyotime, Haimen, China) was applied to measure the proliferation ability of cells that were seeded in 96-pore plates (1
5-ethynyl-2’-deoxyuridine (EdU) assay
Cell-Light EdU DNA Cell Proliferation Kit (RiboBio) was used to measure cell proliferation property under manufacturer’s instructions. After incubation, cells were fixed by formaldehyde and incubated for half an hour with Apollo
Transwell assay
Cell migration was determined by using transwell migration assay in a 24-well transwell chamber with 8 mm-pore size (BD Biosciences, San Jose, CA). Cells in serum-free medium (1
Signal transduction reporter array
Fifty typical signal pathways were detected simultaneously in PTC cells with LINC00893 overexpression using Signal Transduction Reporter Array (Qiagen). The transcription factor-responsive firefly luciferase reporters or the Renilla reporter were co-transfected with pcDNA3.1/LINC00893 or pcDNA3.1 into PTC cells for 48 h. Then, the luciferase activities of all pathways relative to Renilla activity were tested.
Nucleus-cytoplasm separation assay
In line with the manufacturer’s directions, the nucleus-cytoplasm separation of BCPAP and TPC-1 cells was conducted using a PARIS Kit (Invitrogen). RNAs extracted from both nuclear and cytoplasmic fractions were analyzed via qRT-PCR. GAPDH served as the cytoplasmic control and U6 as the nuclear control.
Western blot
Equal quantities of extracted protein from cells (50
RNA immunoprecipitation (RIP)
RIP was manipulated with an EZ Magna RNA immunoprecipitation Kit (Millipore). Cells were first lysed in RIP lysis buffer. Second, magnetic beads were pre-incubated with anti-SNRNP70 (Millipore), anti-FUS (Abcam) or anti-IgG (Millipore) at room temperature for 30 minutes. Then the cell lysates were immunoprecipitated with beads for 4 h at 4
Statistical analysis
All experiments were implemented in 3 bio-replicates. Data from at least triplicate independent experiments were dissected by using GraphPad Prism 6.0 (GraphPad Prism, San Diego, CA, USA), which were expressed as the mean
Results
LINC00893 is downregulated in PTC cells and its overexpression impedes PTC cell proliferation and migration
According to GEPIA dataset, LINC00893 was downregulated in THCA samples versus normal samples (Fig. 1A). Additionally, starBase-pan cancer further confirmed the downregulation of LINC00893 in PTC specimens (
LINC00893 is downregulated in PTC cells and overexpression of LINC00893 inhibits cell proliferation and migration. (A) GEPIA data showed that LINC00893 was downregulated in THCA samples. (B) qRT-PCR data of LINC00893 (transcript NONHSAT223619.1) level in PTC cells (TPC-1, MDA-T120 and BCPAP) and the normal thyroid epithelial cell (Nthy-ori 3-1). (C) LINC00893 level under pcDNA3.1/LINC00893 transfection in PTC cells was measured by qRT-PCR. (D) CCK-8 data of PTC cell viability under LINC00893 overexpression. (E) EdU image and quantification of EdU positive cell ratio under LINC00893 overexpression in PTC cells. (F) The migrated cell number was calculated according to the images obtained from transwell assays. All experiments were implemented in 3 bio-replicates. 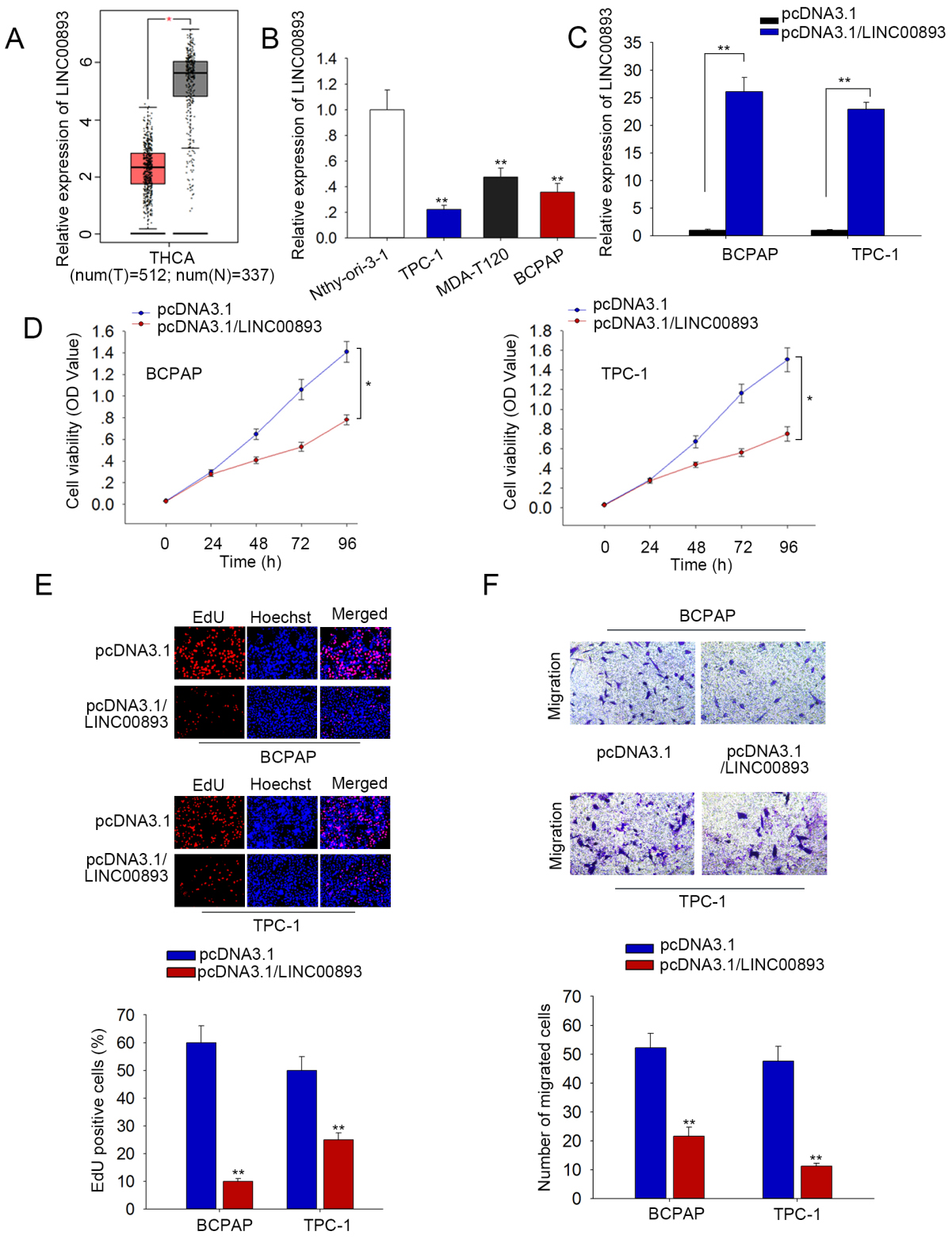
LINC00893 inactivates PTEN/AKT pathway in PTC. (A) Nucleus-cytoplasm separation assay was utilized to determine the subcellular position of LINC00893. GAPDH served as the internal control for cytoplasm, while U6 served as a nuclear control. (B) Activity of 50 signal pathways in PTC and BCPAP cells was measured by Signal Transduction Reporter Array under LINC00893 overexpression. (C) The protein levels of PTEN, p-AKT and AKT in PTC cells and the normal cell were measured by western blot. (D) Western blot analysis of PTEN, p-AKT and AKT in PTC cells with LINC00893 overexpression. All experiments were implemented in 3 bio-replicates.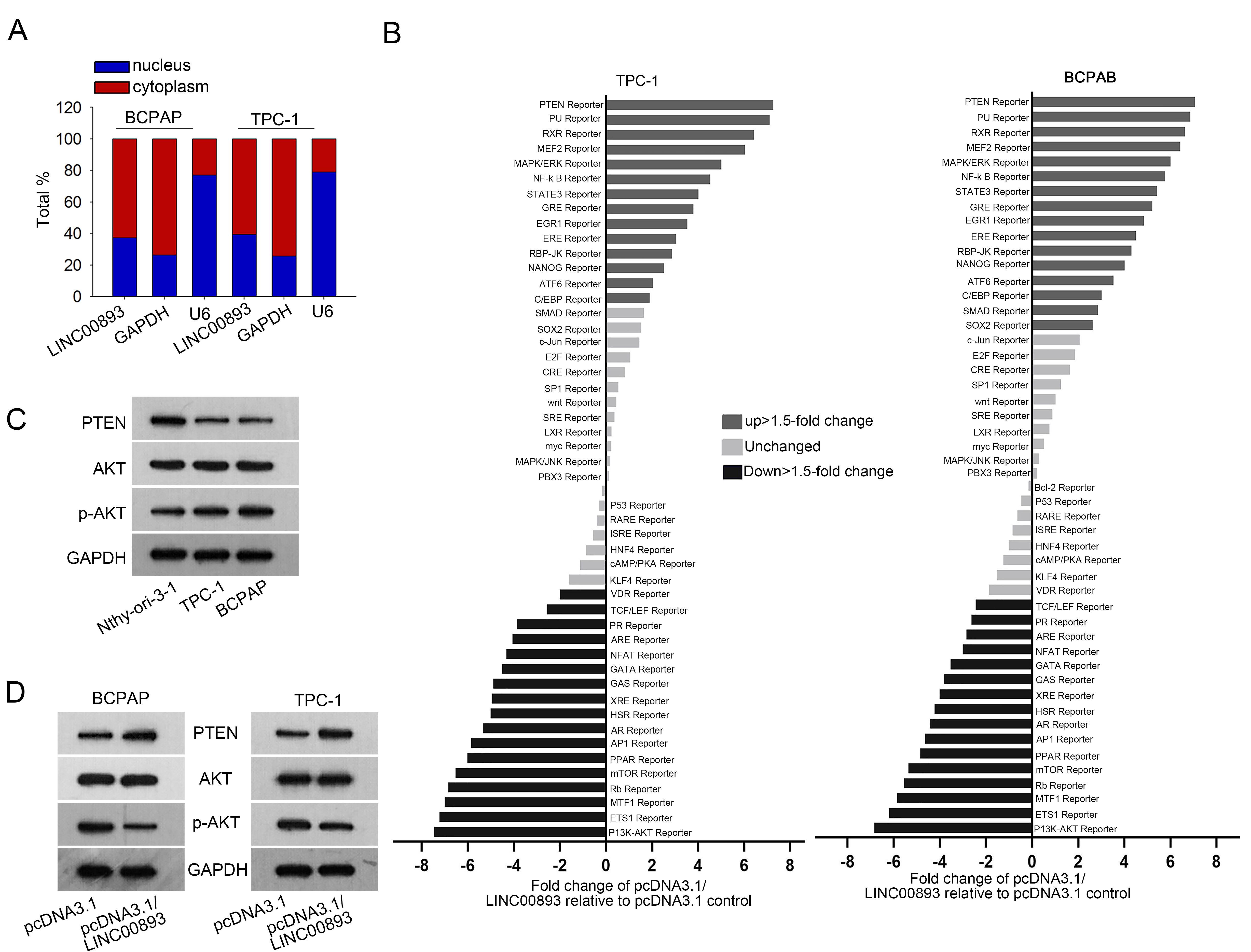
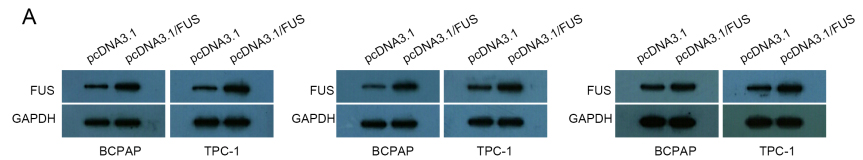
LINC00893 stabilizes PTEN mRNA by recruiting FUS. (A) and (B) qRT-PCR data of PTEN and LINC00893 enrichment in anti-FUS group, with IgG as negative control whereas SNRNP70 and U1 as positive controls. (C) and (D) qRT-PCR and western blot results of FUS level in PTC cells transfected with pcDNA3.1/FUS. (E) qRT-PCR data of PTEN level in PTC cells under FUS overexpression. (F) Interference efficiency of sh/LINC00893#1/2 was detected by qRT-PCR. (G) Co-influences of LINC00893 and FUS on PTEN mRNA half-life after ActD treatment. 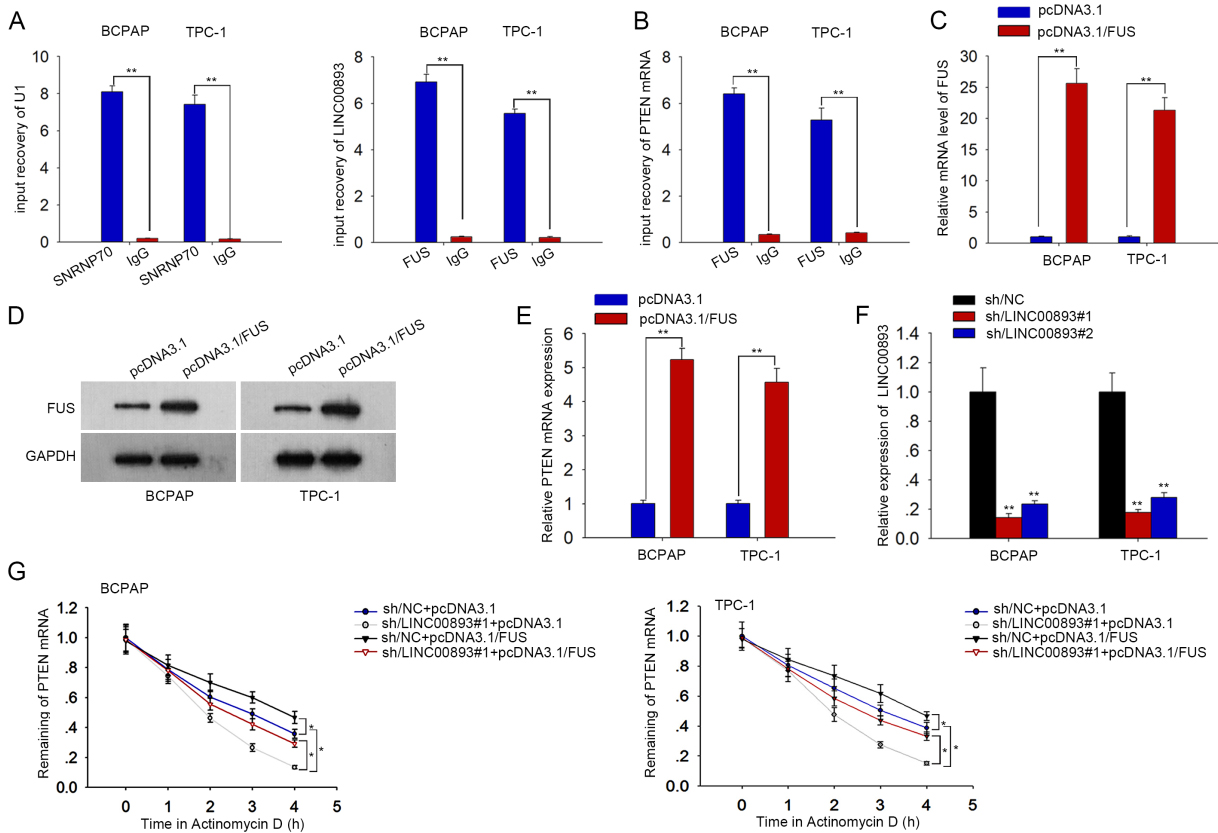
LINC00893 regulates PTEN mRNA expression through FUS. (A) qRT-PCR data of PTEN mRNA enrichment in FUS RIP in PTC cells with LINC00893 overexpression. (B) qRT-PCR and western blotting analyses of FUS level in PTC cells under LINC00893 overexpression. (C) qRT-PCR and western blotting of PTEN expression under different transfections. All experiments were implemented in 3 bio-replicates. 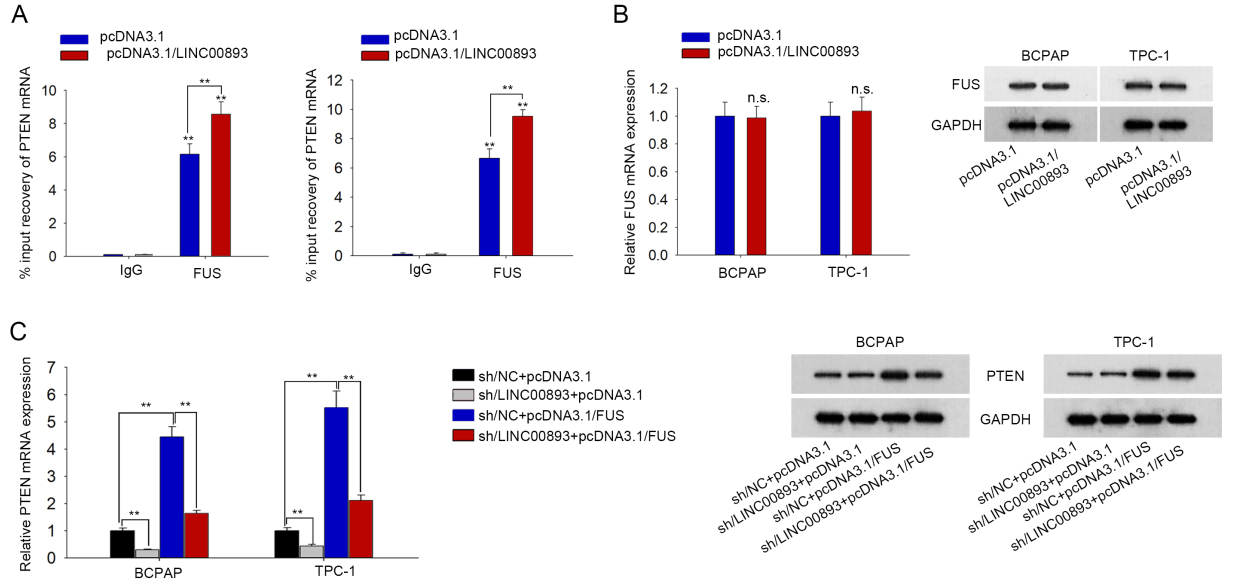
Then, we detected the functions of LINC00893 in PTC cells. qRT-PCR data showed that LINC00893 exhibited the lowest level in TPC-1 and BCPAP cells, so we overexpressed it in these two cell lines for gain-of-function experiments. We confirmed that LINC00893 level was increased under pcDNA3.1/LINC00893 transfection in TPC-1 and BCPAP cells (Fig. 1C). Later, data of CCK-8 assay illustrated that overexpression of LINC00893 reduced the viability of PTC cells (Fig. 1D). Accordingly, the EdU-positive PTC cells was decreased under LINC00893 overexpression (Fig. 1E). Besides, number of migrated PTC cells was declined under LINC00893 overexpression according to data from tranwell assays (Fig. 1F). Together, LINC00893 is downregulated in PTC cells and overexpression of LINC00893 impedes PTC cell proliferation and migration.
Later, we inquired the mechanism of LINC00893 in PTC cells. First, we identified the major distribution of LINC00893 was in the cytoplasm (Fig. 2A). Then, we conducted signal transduction reporter array to test the influence of LINC00893 on 50 typical signaling pathways. Consequently, we found that LINC00893 overexpression stimulated PTEN reporter activity but declined PI3K/AKT reporter activity (Fig. 2B). PTEN/AKT pathway is known as a regulator in cellular activities, including proliferation and migration [17, 19]. Therefore, we assumed that LINC00893 modulated PTEN/AKT pathway in PTC cells. Western blotting data showed that PTEN level was lower, whereas phosphorylated AKT (p-AKT) level was higher in PTC cells in contrast to the normal cell (Fig. 2C). Furtherly, we verified that overexpression of LINC00893 markedly boosted PTEN expression and evidently decreased p-AKT level (Fig. 2D). Collectively, LINC00893 inhibits PTEN/AKT pathway in PTC cells.
LINC00893 stabilizes PTEN mRNA by recruiting FUS in PTC cells
RNA-binding proteins (RBPs) are significant regulators of cancer phenotypes [22, 23]. Here, we inquired whether LINC00893 regulated PTEN expression through interacting with a certain RBP. We searched starBase v3.0 (
PTEN involves in LINC00893-mediated PTC cell proliferation and migration
To determine whether PTEN mediated LINC00893 function in PTC, we applied the in vitro rescue assays. First, PTEN was overexpressed by pcDNA3.1/PTEN in BCPAP cells (Fig. 5A). As presented by western blotting, the effects of LINC00893 knockdown on the protein levels of PTEN and p-AKT were attenuated by PTEN overexpression (Fig. 5B). Both CCK-8 and EdU assays demonstrated that overexpression of PTEN abolished the stimulating effect of LINC00893 silencing on cell proliferation (Fig. 5C and D). Moreover, migration of PTC cells was strengthened by LINC00893 inhibition, but this effect was abolished by PTEN overexpression (Fig. 5E). In brief, PTEN reverses the effect of LINC00893 silencing on PTC cell proliferation and migration.
LINC00893 inhibits proliferation and migration of PTC cells via PTEN/AKT pathway. (A) PTEN level in BCPAP cells with pcDNA3.1/PTEN transfection was measured through qRT-PCR. (B) Levels of PTEN, p-AKT, and AKT in indicated transfection groups were measured by western blotting. (C) and (D) Cell proliferation under different transfections was evaluated through CCK-8 and EdU experiments. (E) Cell migration was analyzed through transwell assay. All experiments were implemented in 3 bio-replicates. 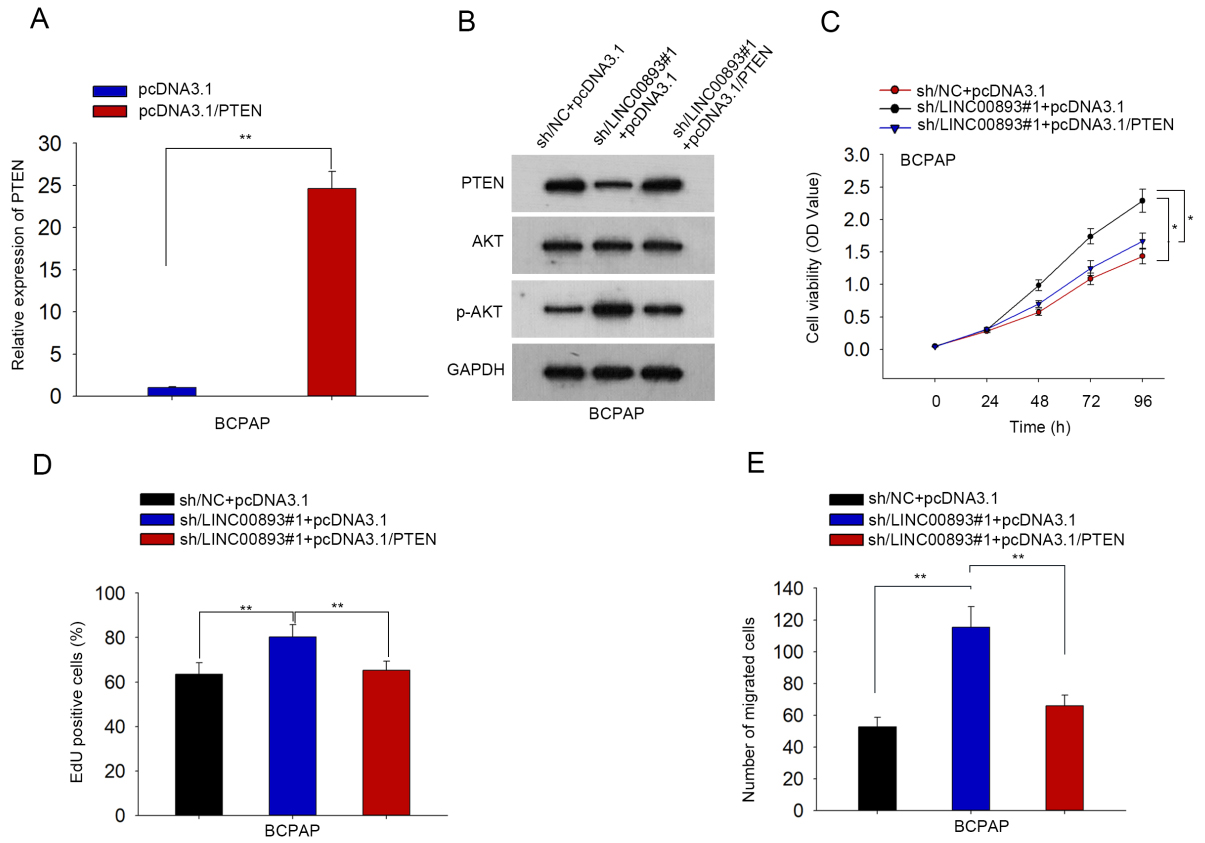
Referring to past reports, lncRNAs are well- established molecules implicated in the development of various human cancers, including liver cancer [24], non-small cell lung cancer [25] and colon cancer [26]. LncRNAs have been increasingly identified as tumor suppressor in PTC. For instance, WT-1-AS1 is lowly expressed in PTC cells and represses Survivin expression [27]. LncRNA RPL34-AS1 prohibits proliferation but drives apoptosis in PTC cells [28]. These findings indicated that the essential tumor-suppressing roles of lncRNAs in PTC progression.
This study analyzed TCGA data through GEPIA tool and found that LINC00893 was downregulated in THCA tissues and obtained similar results through starBase-pan cancer. Hence, we identified the potential correlation between LINC00893 and PTC progression. Interestingly, we found that LINC00893 possesses 3 transcripts, and only transcript NONHSAT223619.1 was downregulated in PTC cells, so we referred to NONHSAT223619.1 as LINC00893 in this study and suggested that this transcript participated in PTC progression. Gain-of-function assays exhibited that LINC00893 overexpression inhibited cell proliferation and migration, indicating the tumor-suppressing role of LINC00893 in PTC.
As reported previously, lncRNAs modulate gene expression in both cytoplasm and nucleus [29]. This study found that LINC00893 was a cytoplasmic lncRNA in PTC cells. Numerous pathways can mediate the function of lncRNAs in PTC. For example, LINC01050 modulates Hedgehog pathway to enhance PTC progression [30]. LncRNA SLC26A4-AS1 represses EMT process in PTC via MAPK pathway [31]. Herein, we detected 50 canonical pathways related to the malignant phenotypes of human cancers through signal transduction reporter array. As a result, LINC00893 overexpression stimulated PTEN and repressed AKT in PTC cells, indicating that LINC00893 was related to PTEN/AKT pathway in PTC. PTEN/AKT pathway has been probed in multiple researches, influencing cellular activities including proliferation and migration [17, 18, 19]. Several lncRNAs have been reported to be regulators for PTEN/AKT pathway in PTC as evidenced by former works, such as lncRNA GAS5 and RP5-1024C24.1 [20, 21]. Consistently, we proved that LINC00893 induced PTEN level and inactivated AKT by downregulating p-AKT. Thus, we suggested that LINC00891 might regulate AKT activation through PTEN upregulation in PTC cells.
Then, we preliminarily explained how LINC00893 regulated PTEN. Based on the cytoplasmic location of LINC00893 in PTC cells, we deduced that LINC00893 regulated PTEN post-transcriptionally. It is noted that RNA-binding proteins are key post-transcriptional regulators of genes [32]. It is known that lncRNAs can recruit RNA binding proteins and affect functional genes. For example, LEF-AS1 interacts with HNRNPL to stabilize LEF1 mRNA in osteosarcoma [33]. NEAT1 stabilizes ELF3 via recruiting IGF2BP1 [34]. Herein, we predicted through the bioinformatics tool starBase that FUS bound to both LINC00893 and PTEN. FUS is known as a DNA/RNA binding protein existing in both nucleus and cytoplasm [35, 36]. Moreover, cytoplasmic FUS can bind to mRNAs to strengthen mRNA stability [37]. Herein, FUS was first linked to LINC00893 and PTEN in PTC. We proved that LINC00893 strengthened the interplay between FUS and PTEN mRNA, indicating that LINC00893 recruited FUS to stabilize PTEN mRNA in PTC cells. Lastly, rescue experiments validated that PTEN abrogated the positive effects of LINC00893 knockdown on PTC cell proliferation and migration, indicating that PTEN/AKT involved in LINC00893-mediated PTC cell functions.
In conclusion, this study discovered a novel oncogenic lncRNA LINC00893 in PTC cells by demonstrating that LINC00893 was downregulated in PTC cells and repressed cell proliferation and migration. We illustrated the mechanism that LINC00893 recruited FUS to stabilize PTEN mRNA to activate PTEN/AKT pathway. These data potentially provided a novel therapeutic target for PTC. However, lack of clinical data and in vivo animal models is the main limitations of our current study. We will put more effort to validate the function of LINC00893 in PTC through clinical and in vivo studies.
Footnotes
Acknowledgments
We faithfully thank all participators in this research.
Conflict of interest
None.
Supplementary data
LINC00891 level in THCA and normal tissues. (A) LINC00893 level in THCA samples versus normal ones were analyzed in starBase pan-cancer. (B) LINC00893 level in normal thyroid tissues was obtained from NCBI database. (C) qRT-PCR of transcript NONHSAT258993.1 and NONHSAT258994.1 in PTC cells versus normal cells. n.s. means no significance.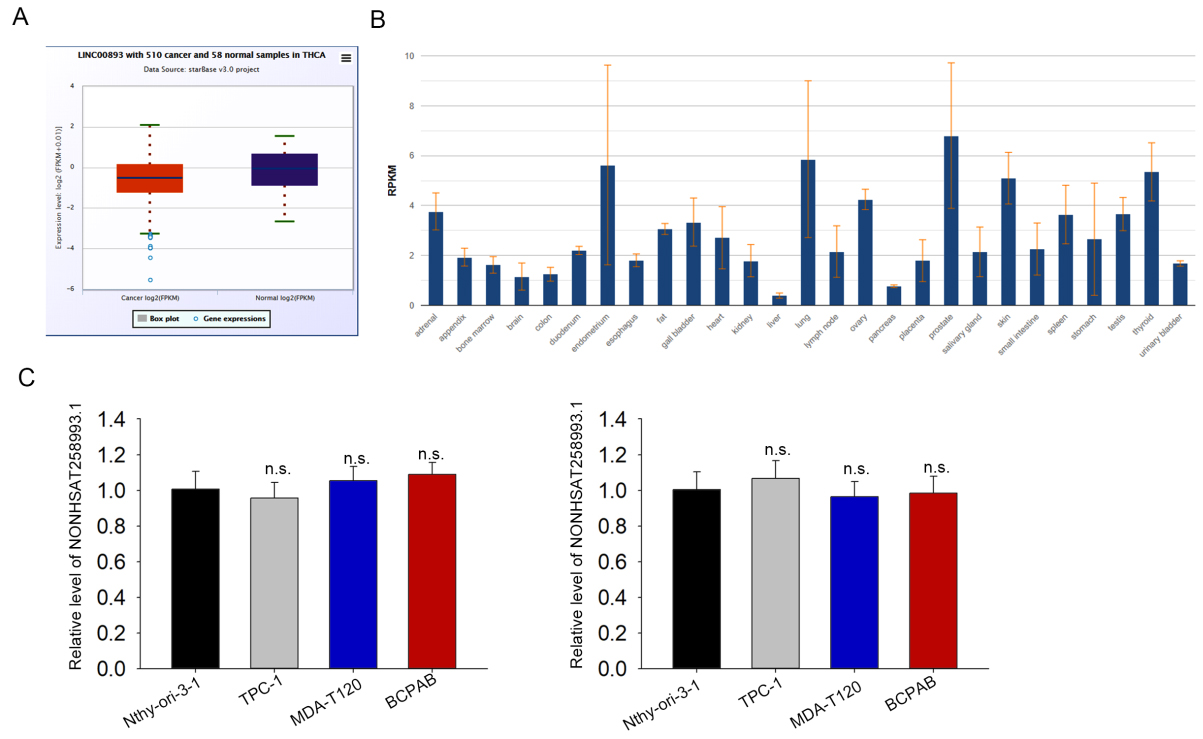
Three repeated results for western blot data in Fig. 3D.