Abstract
BACKGROUND:
Cancer recurrence for patients with early breast cancer is significant. Patients will benefit from more non-invasive modes of monitoring and we aim to study the feasibility of urinary circulating tumor DNA (ctDNA) to monitor for residual disease (MRD).
METHODS:
In this longitudinal study, 300 early breast cancer patients were recruited prospectively. Measurements were taken prior to treatment and at different time points thereafter for a total of 8 measurements. Comparisons were made with healthy volunteers and patients without detectable mutations in urine specimens. Disease free relapse were correlated to both urinary DNA quantity and ctDNA concentration.
RESULTS:
Baseline index measurements showed 38% of patients with detectable mutations. The concordance with biopsy tissues was 97.3%. Overall, breast cancer patients had higher urinary DNA compared with healthy volunteers. Over time, fluctuations in urinary DNA was negligible in healthy volunteers, indicating the stability of the marker. Among the patients with detectable mutations, we observed that higher urinary DNA quantity measurements at 6-month and patients with positive mutations were associated with greater risk of relapse. Hazard ratios for patients in this category was 1.65 (95% CI 1.26–2.16) and 1.98 (95% CI 1.48–2.63) respectively.
CONCLUSION:
Urinary DNA offers non-invasive probing and real-time monitoring of breast cancer relapse. Our results demonstrated clear clinical relevance in breast cancer and significant risk profiling of early breast cancer patients. This potentially aids to complement current cancer relapse monitoring and may help in early intervention.
Introduction
Breast cancer is one of the most prevalent cancer [1] globally and warrants greater attention. Numerous challenges remain in the clinical management especially in early disease. With the advent of better imaging devices and greater awareness for early screening, it is possible to detect more breast cancer cases in the population [2]. In early disease, these patients were not detected with any significant evidence of macroscopic metastasis, yet significant disease relapse have been associated [3, 4]. One of the main difficulties for better disease management is that current imaging modalities may be limiting to detect small tumors. The identification of patients with residual micro-metastasis or minimal residual disease that is not eradicated in therapy will allow clinicians to address these high risk patients.
Utility of circulating tumor DNA (ctDNA) has been effective in patients with advanced cancers for monitoring and prognostication [5, 6]. Plasma or serum derived ctDNA derived from apoptotic or necrotic tumor cells [7] present in the systemic circulation addresses the critical challenge for serial monitoring of cancer patients. Sample extraction is performed via peripheral blood draw that is less invasive than conventional solid tumor biopsy. In earlier studies, absolute amount of circulating DNA has been correlated to tumor burden [8]. This demonstrates close correlation to cancer and may aid in detecting micro-metastasis, which are hard to detect via radiographic imaging and requires alternative detection techniques [9]. In observational studies for lung cancer over an extended period of time, studies have demonstrated that charting the variations in ctDNA concentrations are good early predictors for overall survival [10]. Technology advances have brought about more sensitive methods for ctDNA detection [11]. This potentially may aid to address early cancer, which has been difficult with presumably low tumor burden. Alternative methods for sampling ctDNA via urine has been proposed [12] with excellent results and cost effectiveness shown in advanced cancer [13] but limited available data in early disease.
Here, we assess systematically the potential for urinary ctDNA detection in early disease. Detecting somatic mutations linked to primary breast cancer of these patients, this can be used to link to the presence of minimal residual disease (MRD) for risk profiling and prognostication. Monitoring closely the changes in circulating DNA concentrations, our study correlated the cancer relapse rates of these patients. In addressing the needs for better and quickly cancer detection, urinary ctDNA detection is truly non-invasive and easy to collect. The assay presents an attractive monitoring tool that may be extended for early clinical intervention of early breast cancer that are at higher risk of relapse.
Methods and materials
Study design
Serial urine samples were recovered from early breast cancer patients and healthy volunteers at different time intervals. Patients were recruited at the Xiangyang Central Hospital and treated with standard therapy. Healthy volunteers were recruited from individuals from health screening programs and were deem disease free from mammograms. All procedures were approved by the institutional review board and strictly follows the Declaration of Helsinki. Written informed consent was obtained from all trial participants. A total of 300 patients and 50 healthy volunteers were recruited. Healthy controls were recruited at healthcare screening programs with no abnormalities detected via a standard mammogram. For early breast cancer patients, majority were walk-in patients with complains of lumps or pain in their breast region. Mammogram showed positive results and followed up by CT scans as part of routine diagnostic workup. As per the adjudication of the attending physician, no metastatic disease was detected, and these patients were then recruited. Patients with metastatic disease were excluded. Breast cancer patients were treated with neoadjuvant chemotherapy. After completion of surgery, patients were provided with post-operative radiotherapy. Urinary ctDNA was assessed prior to treatment and compared with tumor biopsy results. Post-surgery samples were also taken. Thereafter, measurement time points were taken at monthly intervals for 6 months. Patient follow up via clinic visits or phone calls were conducted thereafter for a period of 3 years at monthly intervals for correlation of relapse. Throughout the study, the attending physician and the study subject were not made known of the results and standard of care for patients were not altered in any way for the best interest of the patients.
Clinical sample preparation
Core biopsy were taken as per routine practice for histopathological analysis. For urine samples, the first morning midstream urine were collected and processed within 3 hours of collection. 50 mL of samples were taken in a sterile receptacle. For urine processing, urine samples were transferred to a 50 mL centrifuge tube and spun down at 4,000 g for 10 minutes at 4
Purification of urinary DNA and ddPCR
Molecular profiles of PIK3CA and TP53 for each cancer patient was established from tissue biopsies. For urinary ctDNA comparison, we utilized ddPCR to sensitive readout. For nucleic acid purification from urine specimens, the Qiagen QIAamp cell free Circulating Nucleic Acid kit (Qiagen Inc, USA) was used. The kit utilizes a series of centrifugation steps and spin columns and processes strictly followed manufacturer’s instructions. The elute containing urinary nucleic acid was collected in 20
Statistical analysis
The primary goal of the study is to address early breast cancer monitoring especially in MRD and chart its variations during treatment. A direct comparison using Student t test was made between the categorical variables of healthy controls and cancer patients for detecting urinary DNA. This demonstrates the clinical significance in circulating DNA released by the body. One way analysis of variance (ANOVA) was performed to compared the results of quantifiable DNA at different time points. Concordance measure, sensitivity, specificity, positive predictive value and negative predictive value were tabulated for the assay. Multivariate COX regression model and the disease-free relapse curve were computed that will address any confounding factors. The hazard ratio (HR) was determined to assess various parameters linked to risk prediction for early breast cancer.
Urinary DNA participants and early breast cancer patients’ demographics
Urinary DNA participants and early breast cancer patients’ demographics
ER: Estrogen receptor; PR: Progesterone receptor; WT: wildtype.
Baseline characterization of Early breast cancer patients and healthy volunteers shows significant differences in urinary DNA. (A) Agreement of urinary DNA profiling with tumor tissue biopsies. (B) Comparison of recovered urinary DNA demonstrating significantly higher levels associated with patients. (C) Comparison of recovered urinary DNA among mutation positive and wildtype patients showing a slightly elevated level of mean DNA concentration.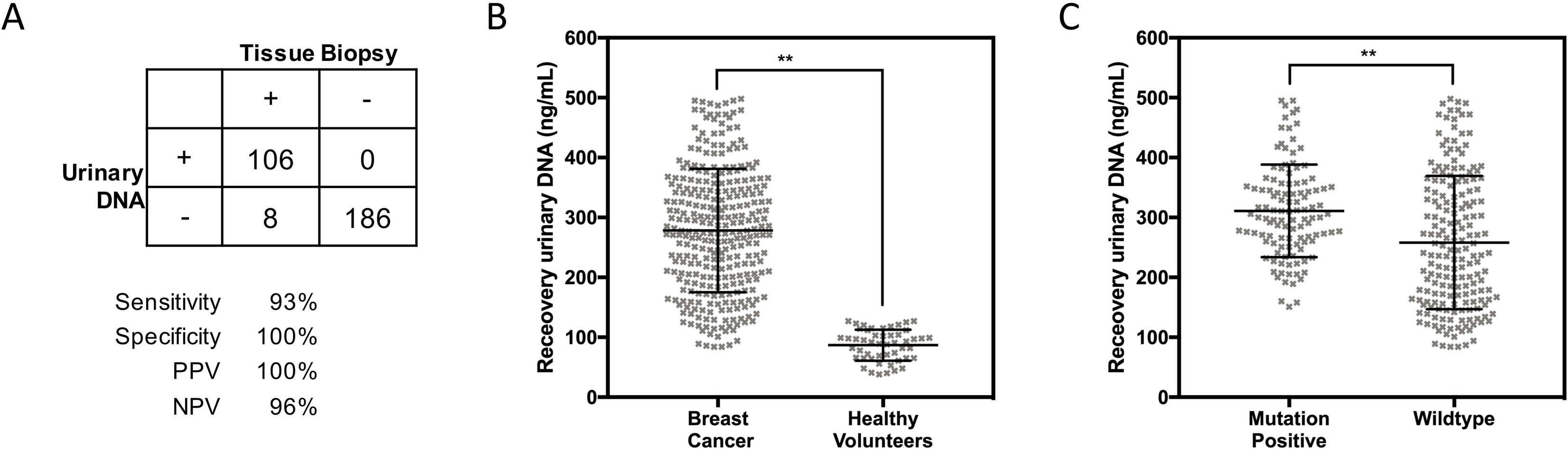
Urinary DNA profile at baseline
In a prospective analysis of 350 participants, we investigate the potential utility of urinary ctDNA in early breast cancer. Details of trial participants are shown in Table 1. Among the study group, mean age for patients and healthy volunteers were 53 and 50 years old respectively. Breast cancer patients were profiled for estrogen receptor (ER), progesterone receptor (PR) and HER2 status. The proportion of patient harbouring these key genetic changes (ER/PR+; HER2+; Triple negative) were 44.0%, 32.3% and 23.7% respectively. Additional molecular profiling for PIK3CA and TP53 mutations were conducted on clinical specimens for this current evaluation on urinary ctDNA. Of the 300 clinical samples at baseline, 38% percent of the samples had either one or both mutations. Figure 1A highlights the comparison with tumor biopsy results. Agreement with tumor tissues results was 97.3%. The high degree of concordance with clinical specimens demonstrated the excellent accuracy of the assay for early breast cancer patient profiling. All 50 healthy volunteers showed wildtype characteristics for PIK3CA and TP53. Comparisons were made in different study groups on the recovered circulating DNA quantity. Figure 1B shows the results that demonstrated an elevated level within early breast cancer patients. The quantity obtained from patients were 19.2 folds higher than healthy volunteers. Among the patient group, it was interesting to note that patients that harboured PIK3CA and TP53 mutations from urinary ctDNA had higher levels (Fig. 1C) than wildtype patients. A Student shows statistical significance between these two groups with
Serial Measurements and tracking urinary circulating DNA
Longitudinal measurements allowed comparison of recovered circulating DNA at different time points post-surgical procedures. We concurrently performed the analysis on healthy controls to ascertain the inherent urinary circulating DNA stability. Molecular profiling was also performed as a means to understand MRD within early breast cancer patients after potential curative surgical procedures. Figure 2A shows the measurements of healthy controls that indicated a fairly uniformly mean extracted urinary DNA over a 6-month period. There were three dropouts for this control group and their results were not included in this analysis. A one-way analysis of variance showed no significant differences in measured background quantity of urinary DNA. We do not also see any significant variability within each time point with the highest and lowest CV at 32% and 30% respectively. For patient measurements highlighted in Fig. 2B, a pairwise ANOVA showed significant changes in the mean recovered urinary DNA. Of note, a sharp decline in mean urinary DNA was seen immediately post-surgery. We compared the mean values to healthy controls at each time point and patients’ urinary DNA levels were statistical significant to be higher using a Student
Serial tracking of urinary DNA and concordance with mutation profiles from primary tumor tissues. (A) Serial measurements of healthy volunteers that demonstrated a stable measurement over a period of 6 months. (B) Serial measurements of early breast cancer patients that recorded strong changes post surgery and increasing mean levels thereafter. (C) Concordance with primary tumor tissues from urinary DNA profiling. (D) Urinary DNA levels between mutation positive patients (black bars) and patients with wildtype status (grey bars).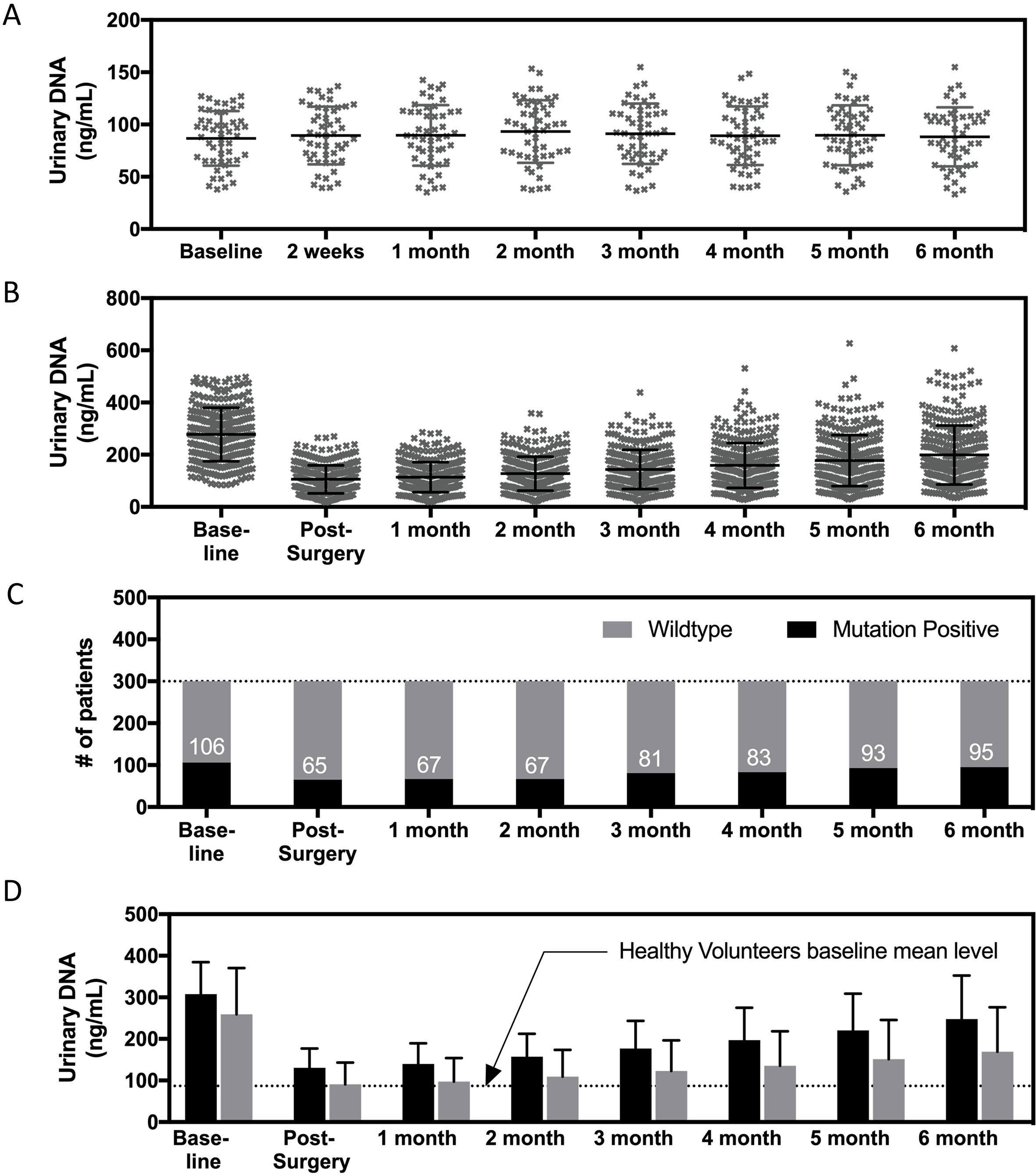
Kaplan Meier analysis of disease free survival among early breast cancer patients. (A) Baseline detected levels of urinary DNA and the cohort is equally split by the levels of detected urinary DNA. (B) Baseline separation of mutational status from urinary DNA testing. (C) Measurement post-surgery of the levels of urinary DNA that demonstrated a more significant separation of patient groups compared with baseline results. (D) Measurement post-surgery and comparison of patient groups by the mutational profiles. (E) 6-month measurement of detected urinary DNA that showed better separation of patient groups within the entire monitoring period. (F) Disease free analysis and comparison with patient groups using the mutational profiles at 6-month. 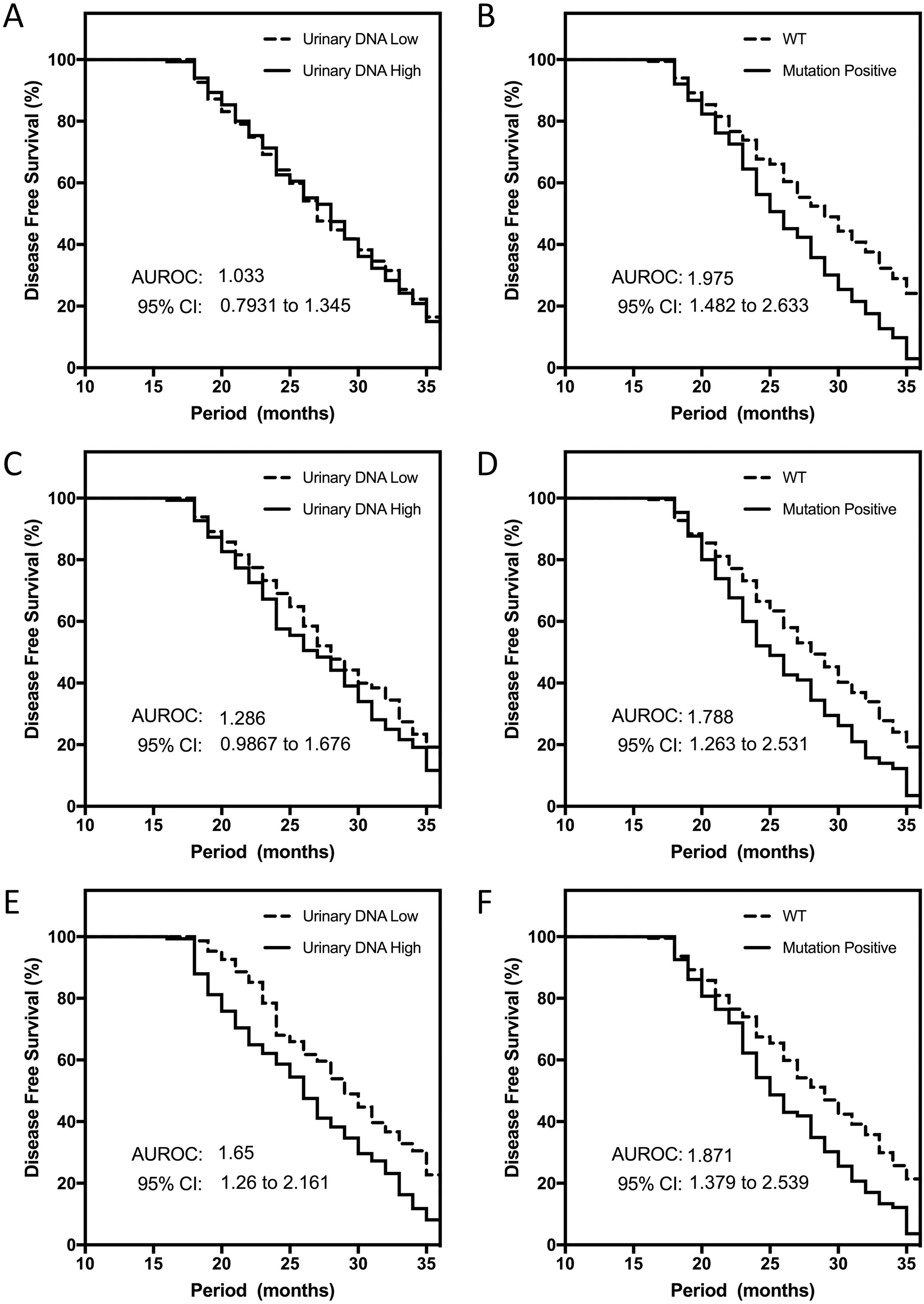
The clear differences in various patient groups highlighted the potential to further explore the parameter to address critical challenges in early breast cancer patients. Follow up contact tracings with patients and collecting data of disease relapses allowed the correlations with urinary circulating DNA to be established. We compared data retrieved at baseline and dividing the patient cohort equally based on the level of urinary circulating DNA. Figure 3A shows no significant differences in disease free survival and relapsed cases in both patient groups that were separated by the level of high and low urinary DNA. The hazard ratio (HR) determined for this comparison was 1.03 (95% CI 0.79 to 1.34). For patients with detectable mutations and those of wildtype TP53 and PIK3CA, we observed that the relapsed rates were higher in the former (Fig. 3B). The HR established in the comparison was 1.98 (95% CI 1.48 to 2.63). We repeated the analysis for post-surgery results and observed stronger separations with urinary circulating DNA recovered at this measurement time point (Fig. 3C). The HR as determined was 1.29 (95% CI 0.99 to 1.68) for patient’s disease-free survival cases. Significant statistical difference was consistently observed with patients grouped by their mutational status (Fig. 3D). HR was 1.79 (95% CI 1.26 to 2.53) and slightly lower to the measurements prior to treatment. Given significant changes was also observed at the end of monitoring, we analysed the disease-free survival rates at 6-month. The results indicated that urinary circulating DNA recovered from patients were significantly correlated. The patient group with higher recovered urinary DNA had worse outcomes and HR was 1.65 (95% CI 1.26 to 2.16) as shown in Fig. 3E. For patients with positive mutations detected from urine samples, we consistently observed clear statistical significance (
Discussion
Here, the study investigated the relevance and utility of urinary DNA in early breast cancer patients undergoing treatment. The challenges for these patients is to assess MRD and identify those that may be at higher risk of relapse. Probing the disease using urine samples is attractive as it is non-invasive and circulating DNA from plasma has proven to be a good substitute for primary tumor tissues in different cancers [7]. Plasma ctDNA has traditionally been the main focus in many studies but blood draws required trained phlebotomists and this will likely restrict the procedure to primary care settings. In certain populations such as geriatric patients, drawing relatively large volume of blood is equally challenging. Previous studies have highlighted that circulating DNA exist within urine samples as a result of ultrafiltration in the kidneys from systemic circulation [15]. This draws the interest to better understand the use of urine samples in cancer management. Screening of patients using radiographic imaging has been the key success factor in raising the detection rates of early breast cancer but this is less useful post treatment as micro-metastases may be missed. Our study presents an investigation into probing early disease that may significantly lead to changes in post treatment monitoring of early breast cancer patients.
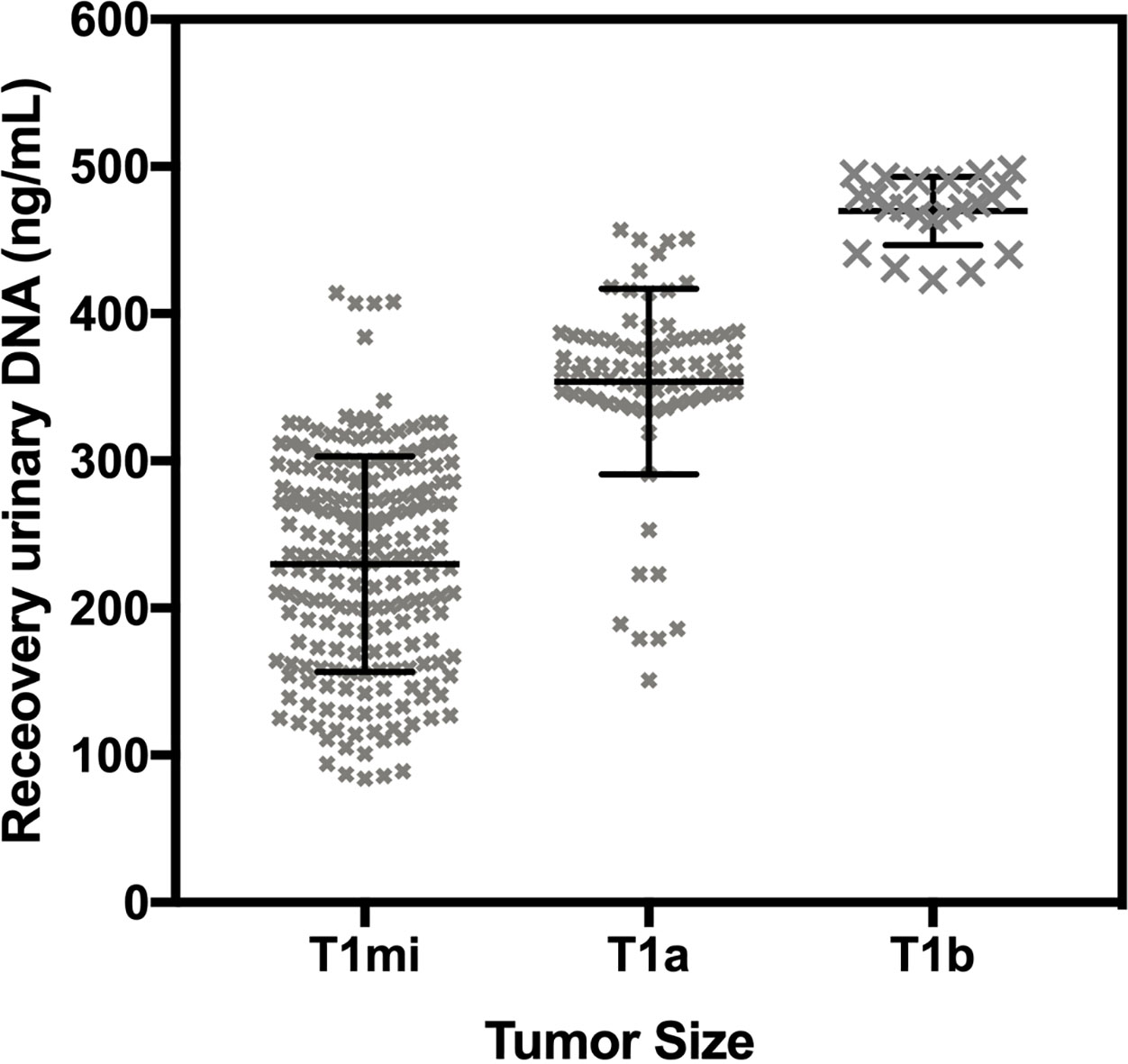
At baseline measurements, our data highlighted the fundamental clinical relevance of urinary circulating DNA using certain genetic anomalies typically found in breast cancer patients. Essentially for baseline measurements, urinary ctDNA demonstrated significant results for mutation detection and clinical relevance. The quantity of urinary DNA was observed to be clearly higher in patients. Further analysis addressing tumor size based on initial CT scans showed higher urinary DNA associated with larger tumors (Supplementary Fig. 1). PIK3CA and TP53 are two common types of mutations and PIK3CA had been predictive of recurrence in primary breast cancer [14, 16]. In earlier studies, TP53 mutations associates with an elevated risk of breast cancer-specific death, irrespective of tumor size, nodal status, and hormone receptor status [17, 18]. We observed that the assay has good clinical specificity with all healthy controls and patients with wildtype characteristics to be concordant. PIK pathways have also been closely studied for treatment response measures [19, 20]. Hence the ability to detect this mutation in urine samples provide additional source to probe breast cancer patients. In earlier studies, PIK mechanism analysis showcased the significance of these related mutations in AR phosphorylation for triple negative breast cancer and this was demonstrated as a strong predictor for prognosis [16].
We closely monitored these patients via serial sampling to trace the changes in mutation detection among the study group and record the quantities of recovered urinary DNA. For post-surgery measurements, we observed a significant drop in all patients while healthy volunteers maintained its baseline levels. This suggests that the surgical removal of the tumor source directly impacted the levels of urinary DNA. This is consistent with the genetic profiling as a number of patients with positive mutations could not be detected post-surgery using ddPCR. Interestingly and significantly, a large proportion of patients remains positive with these mutations, presumably with the presence of MRD that were not picked up in radiographic scans. The persistence of residual tumor cells after surgery and adjuvant therapy highlights the difficulties in detection and monitoring of breast cancer patients [9]. Given these results, we follow up with the patients to understand the prognostic utility of urinary DNA. Measurements were compared with disease free survival for a number of time points. At baseline measurements, we observed a strong correlation to PIK3CA and TP53 presence but not the detected levels of urinary circulating DNA. This is consistent with the study conducted by Isaac et al. [21] that noted baseline ctDNA from plasma was also not predictive of disease free survival. Possibly, this can be attributed to the treatment efficacy and response of patients that were not taken in consideration at baseline. For both post-surgery and at 6-month time points, we observed that the levels of detected urinary DNA became significant and the predictive response was better at later time intervals. For both mutation positive patients, the results were significant at different time points. Despite the encouraging findings for improved patient monitoring in early breast cancer, further investigations are needed to address patients with relatively lower urinary DNA but present with recurrent disease. We also observed a few discordant cases of mutation detection with tissue biopsies that may be limited by the sensitivity of urinary DNA. More work is needed to better establish the sensitivity and specificity of the assay with a larger patient cohort. Other complementary assays that demonstrated excellent sensitivity may be exploited that draws upon the non-invasive probing of cancer cells [3, 22, 23]. Though the current results demonstrated significant correlations to disease outcome, further clinical research needs to be conducted. The potential of the study aids to complement existing monitoring regimes and is exploratory to provide risk stratifications. A larger cohort is required to validate the use of the marker and subsequent clinical interventions will allow better assessment of clinical impact.
Conclusions
In conclusion, we have shown urinary DNA can be a useful non-invasive procedure for early breast cancer. The challenges in current testing methods are picking up MRD after standard therapy and identifying high risk patients with recurrent disease. Urinary DNA presents an attractive means to probe disease changes and has demonstrated strong clinical relevance in this current study. This fills a critical gap for continual monitoring of early breast cancer. The new approach can help to better provide risk stratifications and drive further genetic testing to understand early breast cancer. This will also aid in future early interventions that may reduce the rates of recurrent disease.
Footnotes
Acknowledgments
This work was supported by research grants provided by Xiangyang Central Hospital.
Conflict of interest
All authors declare no conflict of interest.
Supplementary data
Correlation of tumor size to recovered urinary DNA. ANOVA shows significant difference in measured quantity.