Abstract
Despite initial treatment, breast cancer recurrence affects approximately 30% of patients. Currently, there exists no standardized approach to detect recurrence before clinical or radiologic signs manifest. Circulating tumor DNA (ctDNA) is a minimally invasive blood test that offers potential to monitor molecular disease and individualize care. This study explores the utility of ctDNA in recurrence monitoring and clinical decision-making for high-risk breast cancer cases within a community setting. Seventy-two patients with high-risk breast cancer features—defined as stage III disease, triple-negative or HR−/HER2+ following neoadjuvant treatment, metastatic breast cancer without evidence of disease, bilateral breast cancer history, high-risk genetics (BRCA1/BRCA2 mutations), <40 years old at diagnosis, or history of breast cancer recurrence—were offered tumor-informed ctDNA assays at 3- to 6-month intervals. Analysis was conducted on 67 cases with a mean diagnostic age of 52.69 at diagnosis. The cohort was ethnically diverse, including White (n = 21, 31.82%), Japanese (n = 15, 22.73%), Native Hawaiian (n = 11, 16.67%), and Filipino (n = 7, 10.61%) patients. Seven (10.45%) tests were positive: six predicted recurrence despite four with initially negative radiological findings, and one prompted treatment resumption following prior non-adherence. However, one negative result was false and later showed a contralateral breast recurrence, and another negative test coincided with a new primary cholangiocarcinoma. In two cases, ctDNA negativity was utilized to monitor treatment response in metastatic disease and inform therapeutic adjustments. In real-world settings, ctDNA served as a valuable tool for earlier recurrence prediction, expediting radiological confirmation, and influencing treatment. Nevertheless, false results carry the risk of hindering effective care and inducing considerable patient anxiety. Future large-scale studies are warranted in high-risk breast cancer populations to evaluate ctDNA’s impact on patient survival outcomes, treatment monitoring, and patients’ emotional experiences.
Plain language summary
Keywords
Background/Introduction
Breast cancer is a globally prevalent cancer diagnosis, ranking second in cancer-related deaths among women in the United States. 1 Despite initial treatment success, up to 30% of patients with breast cancer will experience recurrence.2,3 Recurrence often is highest within the first 5 years after diagnosis, either returning locally at the site of the original tumor, regionally into neighboring lymph nodes, or distantly into tissues and organs beyond the initial site as metastasis. 4 About 20% of patients develop recurrence at a later stage, 5 and if metastatic, suffer a poor prognosis with a 5-year survival of about 68%. 6 Risk factors for recurrence vary by subtype but include lymph node involvement, large tumor size, younger age (<40) at the time of diagnosis, and obesity. 7
To identify possible recurrence early and improve overall survival, current National Comprehensive Cancer Network (NCCN) surveillance protocols suggest regular clinical exams 1–4 times per year, continued annual breast imaging with mammography, breast MRI when applicable, and lifestyle education. Screening for metastasis with laboratory or imaging studies is not recommended, and these are indicated only if clinical signs and symptoms are suggestive of recurrent disease. 8 No standardized tool exists to test for early recurrence risk before clinical signs manifest and radiologic tests confirm suspicious clinical symptoms.
Circulating tumor DNA (ctDNA) has emerged as a promising biomarker for residual disease monitoring and predicting breast cancer recurrence trends. Various commercial platforms are available, including both tumor-informed and tumor-agnostic ctDNA, for current use in patients with high-risk breast cancer. ctDNA assays utilize liquid biopsies to detect tumor-specific mutations that may indicate minimal residual disease (MRD) and impending recurrence.9–11 Tumor-informed ctDNA tests sequence apoptotic tumor DNA in the bloodstream for somatic variants through whole-exome sequencing of the primary tumor sample. Early mutations that occur at the tumor origin are identified to create a patient-specific panel that can detect early mutations.
The benefits of tumor-informed ctDNA include its minimally invasive approach to molecular tumor profiling and a 90% sensitivity and reproducibility of mutated allele detection. 12 Since ctDNA has a short half-life of up to 150 min, it can effectively monitor a tumor’s response to therapy in real time and consequently enhance treatment efficacy standards. 13 Clinical applications of ctDNA testing have been described, including the identification of tumor mutations, screening for the early detection of cancer, tracking of therapeutic responses, predicting MRD, and monitoring patients for recurrence.9–11 In studies assessing breast cancer recurrence, ctDNA positivity has been reported to demonstrate high sensitivity and specificity, 14 with median lead times from clinical relapse ranging from 8.9 15 to 10.7 months, even across all four subtypes. 16
Studies involving patients with high-risk breast cancer emphasize ctDNA’s negative prognostic capabilities and the importance of frequent surveillance to optimize lead time. The I-SPY2 trial assessing ctDNA following neoadjuvant chemotherapy (NACT) in patients with high-risk early-stage luminal A breast cancer and patients with Triple Negative Breast Cancer (TNBC) found that the persistent presence of ctDNA at any time point of NACT (before, during, and after) predicted poorer response and metastatic recurrence, regardless of pathological complete response (pCR). 17 They also found that early clearance of ctDNA significantly predicted pCR, implying its utility as a surrogate measure of tumor burden comparable to radiological imaging, particularly in aggressive subtypes like TNBC. Results from the c-TRAK TN trial, which prospectively tracked molecular residual disease and pembrolizumab activity in 161 patients with moderate-to-high-risk early-stage TNBC, further theorized that frequent testing at less than 3-month intervals may allow earlier interventions to expedite ctDNA clearance, especially given its median 10.7-month lead time. 18
The phase III monarchE trial, which demonstrated the efficacy of adjuvant abemaciclib (CDK4/6 inhibitor) targeted therapy with endocrine therapy (ET) in patients with HR+/HER2+ node-positive high-risk early-stage breast cancer, also assessed the prognostic capabilities of ctDNA (Signatera, Inc., Austin, TX, USA). This trial affirmed the negative prognostic value of ctDNA, with patients who persistently tested positive tending to develop more distant recurrence and longer lead times (median of 15 months). In addition, results contended that ctDNA’s potential to monitor treatment response, as ctDNA positivity mirrored favorable treatment responses in concordance with the original trial findings, with 40% of participants initially ctDNA positive becoming undetectable and 42% experiencing an invasive disease-free survival event following ET + abemaciclib treatment upgrading. 19
Overall, studies have demonstrated the potential of ctDNA across various subtypes, with lead times as low as 9–11 months, offering insights into its clinical implications for prognosis and monitoring treatment response. Although multiple uses of ctDNA have been speculated, Medicare and Medicaid Services approved coverage reimbursement for the tumor-informed Signatera assay strictly for adjuvant and recurrence monitoring in stage IIB and III breast cancers. 20
With few studies investigating molecular recurrence in patients with high-risk breast cancer, our case series aims to assess how ctDNA is currently being used in clinical practice, with a focus on clinician decision-making and outcomes, incorporating insights from breast cancer populations in a community setting in Hawaii, where breast cancer incidence rates remain the highest in the nation. 21
Methods: Patient information, diagnostic assessment, and therapeutic intervention
We conducted a case series of 72 patients with high-risk breast cancer managed at a single outpatient center under the Signatera early access program to investigate how ctDNA is utilized in the clinic. High-risk status was defined by the following criteria: stage III disease, TNBC, or HR−/HER2+ breast cancer following neoadjuvant treatment, metastatic breast cancer without evidence of disease, bilateral breast cancer history, high-risk genetics (i.e., BRCA1 or BRCA2 mutations), young age at diagnosis (<40 years old), or a history of breast cancer recurrence of any subtype.
Patients eligible for the study were prospectively enrolled on a rolling basis from March 2021 to August 2024. All participants underwent personalized tumor-informed ctDNA assays (Signatera™; Natera, Inc., Austin, TX, USA), which involved whole-exome sequencing of the primary tumor and matched normal blood samples to identify somatic clonal variants. For each patient, a personalized panel of 16 tumor-specific variants was selected based on clonality, detectability, and variant allele frequency. This bespoke panel served as a molecular fingerprint to monitor for molecular recurrence throughout the patient’s disease course. Plasma cell-free DNA was isolated, PCR-amplified, and sequenced, with a lower limit of 0.01% variant allele frequency.
Blood samples were taken at baseline and 3-to-6-month intervals for up to 2 years. Those with detectable positive ctDNA results were subsequently offered diagnostic imaging for further evaluation. Through the early access program, participants with undetectable negative ctDNA results had the option to continue testing for up to 5 years from their initial test, with the same option extended to patients with positive ctDNA results. Participants could choose to discontinue testing at any time for any reason.
Demographic data (age at time of diagnosis, reported race and ethnicity, gender), tumor clinical/pathological characteristics (tumor size, grade, node status, hormone receptor status, recurrence, metastasis), treatment history (surgery, chemotherapy, radiation, ET), pCR, tumor-infiltrating lymphocytes, and biomarker test results (genetic testing, genomic testing, and liquid biopsy) were recorded for each patient. Particular attention was given to illustrative or clinically impactful cases, with a focus on the predictive value of ctDNA as well as its influence on treatment decisions made by clinicians.
The reporting of this study conforms to the Consensus-based Clinical CAse REporting (CARE) Guideline statement 22 (CARE-checklist).
Results: Clinical findings, follow-up, and outcomes
A total of 72 patients registered between March 2021 and August 2024 were offered ctDNA tumor sequencing. Of these, five patients did not obtain ctDNA testing and were excluded from further analysis, leaving 67 patients for final review. The age of patients ranged from 28 to 83 years with an average age of 52.69 years old. The cohort was racially and ethnically diverse: White (n = 21, 31.82%), Vietnamese (n = 1, 1.52%), Korean (n = 2, 3.03%), Japanese (n = 15, 22.73%), Chinese (n = 5, 7.58%), Filipino (n = 7, 10.61%), Native Hawaiian (n = 11, 16.67%), Black (n = 2, 3.03%), and Hispanic (n = 3, 4.55%).
An average of four ctDNA tests were conducted per patient over 2 years, ranging from 1 to 10 tests. Fifty-five patients completed at least one ctDNA test. Seven (10.45%) tests were positive: three predicted metastatic diagnosis despite initially negative radiological findings (
Notably, two negative ctDNA cases showed unique results: one discovered contralateral recurrent disease despite two negative ctDNA tests (
Testing was also used to monitor metastases in two cases: 1 metastatic bone lesion with 8 negative draws that led to treatment cessation (
Discussion
Metastatic treatment monitoring without evidence of disease
Our study revealed several important uses of ctDNA in the clinical setting. An occasional, challenging clinical scenario includes patients who have initially presented with metastatic disease, completed aggressive treatments with curative intent, and are now without any evidence of disease. In our study, ctDNA was used to inform treatment response and help with decision-making in patients with negative ctDNA results following a metastatic breast cancer diagnosis (
Unique breast cancer cases and trajectories.
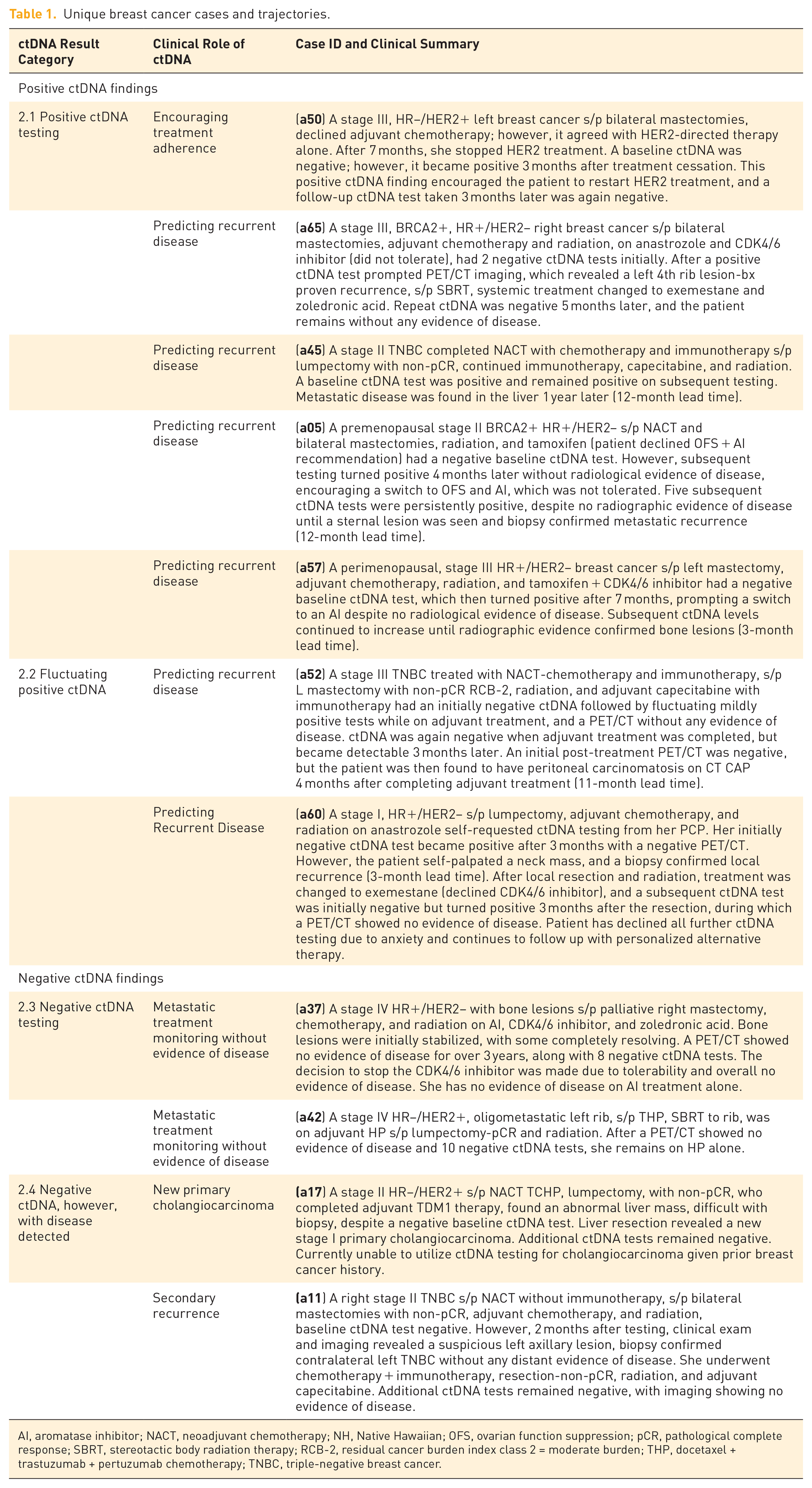
AI, aromatase inhibitor; NACT, neoadjuvant chemotherapy; NH, Native Hawaiian; OFS, ovarian function suppression; pCR, pathological complete response; SBRT, stereotactic body radiation therapy; RCB-2, residual cancer burden index class 2 = moderate burden; THP, docetaxel + trastuzumab + pertuzumab chemotherapy; TNBC, triple-negative breast cancer.
Predicting recurrent disease and treatment upgrading
Our study demonstrated the role of ctDNA positivity in expediting metastatic diagnoses and exploring early treatment approaches, potential uses speculated by previous trials due to its negative prognostic value.14,17 Diagnostic imaging was offered to those with positive ctDNA tests at any time point. This allowed patients and clinicians to consider potential treatment options sooner. Positive ctDNA testing helped guide earlier diagnostic imaging, which led to subsequent metastatic diagnoses in two cases (

Clinical trajectories of patients with positive ctDNA. (
In two additional cases with ctDNA positivity (
While current NCCN guidelines rely on clinical signs as the primary indication for imaging, ctDNA’s lead time may offer an additional way to detect asymptomatic metastases at a possibly more dormant phase. With the latest American Society of Clinical Oncology (ASCO) recommendations on the use of tumor markers dating back to 2015, long-term studies on detecting and treating recurrence earlier with ctDNA may discover new insights into how early detection and intervention affect patient outcomes. 23 Until this is established, ctDNA can still serve as a tool to characterize disease burden and provide clarity to patients with high-risk breast cancer, who often contend with the unpredictable nature of their disease progression.
Discrepant results
Our findings demonstrate both the potential and challenges associated with ctDNA in clinical practice. Although trials have demonstrated the high sensitivity of ctDNA,
14
our study encountered several instances of fluctuating findings, such as discrepancies with radiological results and clinical symptoms. In two cases (
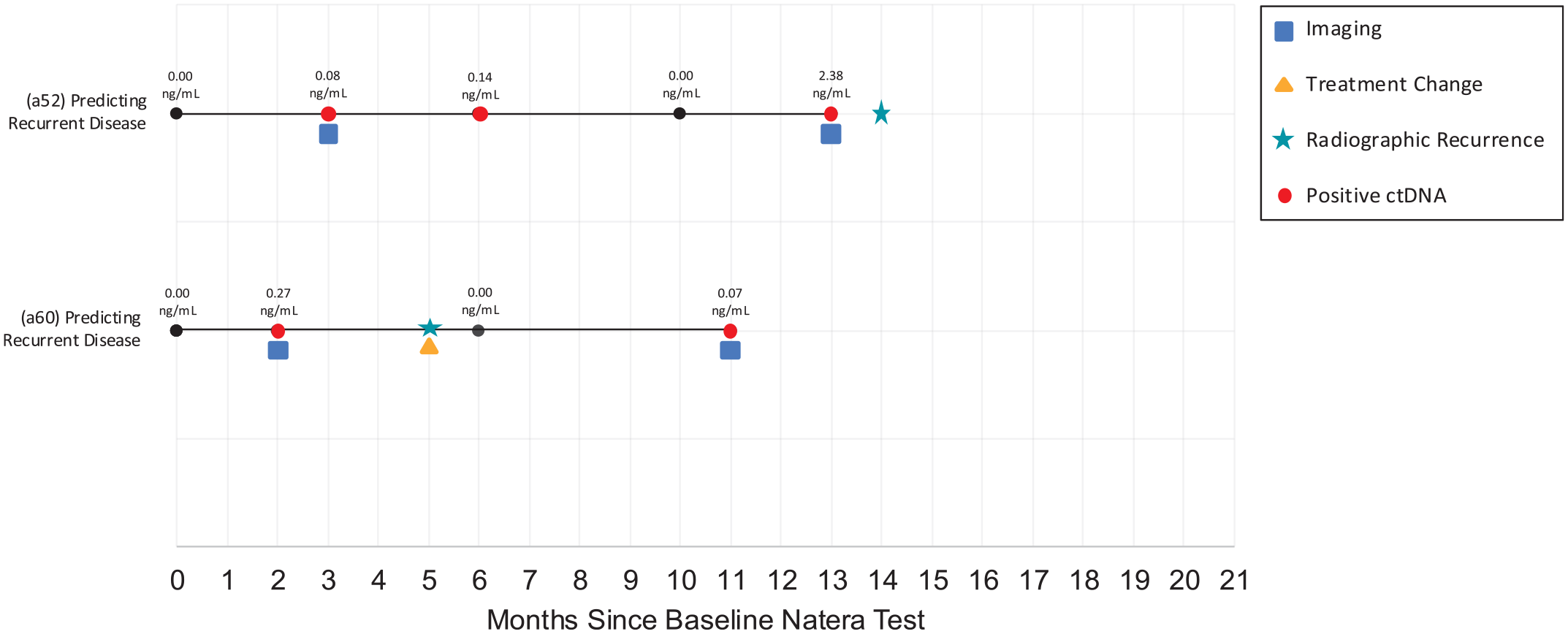
Clinical trajectories of fluctuating patients with positive ctDNA. (

Clinical trajectories of patients with negative ctDNA. (
Similarly, one case with negative ctDNA results (

Clinical trajectories of patients with negative ctDNA with disease detected. (
Currently employed tumor-informed ctDNA tests remain inherently limited to monitoring mutations based on the original tumor and may not be able to detect recurrences arising in new locations or tissue, such as in our case of contralateral recurrence. As such, these assays would not be expected to detect a new primary malignancy, such as the cholangiocarcinoma in this case (
To maximize the clinical utility of ctDNA testing, clinicians should ensure that patients understand the capabilities and limitations of these assays. Future research directions, potentially integrating tumor-informed assays with tumor-agnostic approaches, may enhance the sensitivity of recurrent disease detection, offering a more comprehensive strategy for early detection regardless of the original tumor’s biology.
Patient experience with ctDNA testing
While ctDNA poses a risk of discrepant results and associated anxiety, it can also promote treatment adherence, particularly in those undergoing treatment for earlier-stage cancers. In one case (
Overall, ctDNA can lead to earlier detection of recurrence and intervention, providing a tailored disease evaluation to patients and even promoting adherence in some cases. However, its consequences include the risk of unnecessary workup and treatment, especially in discrepant findings, which can especially burden patients emotionally. With a 30% recurrence rate,2,3 patients with breast cancer who are in remission may experience uniquely heightened levels of anxiety, especially those at high risk. Studies have shown that breast cancer survivors living with cancer for at least 2 years exhibit significantly higher incidence of anxiety disorders than the general population, with 17.9% meeting clinical criteria for anxiety.
25
The emotional toll of scanxiety, characterized by distress and anxiety linked to cancer-related imaging and tests,
26
compounds these feelings. As ctDNA gains notoriety for its potential in detecting breast cancer recurrence, it may expand the repertoire of diagnostic tests available to patients with breast cancer. However, the possibility of discrepant results, as observed in our study, may worsen patients’ feelings of unease, mistrust, and panic. These very harms of serial ctDNA monitoring, in addition to increased diagnostic procedures and costs, unnecessary treatment upgrading, and heightened patient anxiety, support the current ASCO and National Clinical Trials Network guidelines, which do not endorse ctDNA testing for early-stage breast cancer.
21
As radiological relapse is often used to validate ctDNA positivity, the experience of receiving liquid biopsies and imaging is closely intertwined, and conflict between the two can exacerbate anxiety as demonstrated in our study (
Racial and ethnic disparities
Our study is inclusive of a wide range of ethnic and racial diversity without restriction to specific subtypes. The majority of our cohort consisted of Asian (45.45%), White (31.82%), and Native Hawaiian/Pacific Islander (NHPI; 16.67%) individuals, addressing significant gaps in breast cancer research (Table 2). NHPI individuals exhibit some of the highest cancer mortality rates, 27 and Hawaii breast cancer incidence rates remain the highest in the nation. 21 National ctDNA databases have shown that patients who identify as Asian and NHPI utilizing ctDNA are the least represented (4.7%) compared to patients who identify as White (71.5%), Black (12.1%), and Hispanic (9.1%). In addition, the aggregation of these groups into broader AAPI categories 28 may obscure substantial health disparities among Pacific Islanders, with differences in 10-year survival outcomes as wide as 9% between NHPI and East Asian patients with breast cancer. 29 Studies investigating racial inequities of ctDNA similarly conflate NHPI populations with “Asian/other/unknown,” with similarly low participation (9.7%) compared to patients who identify as White (24.8%), Black (32.6%), and Hispanic (33%). 30 Aggregating Native Hawaiian and Asian American race categories could similarly impact reports on ctDNA’s negative prognostic utility, which has become evident in studies. Of the six cases in our study with positive ctDNA whose findings led to earlier radiological confirmation or intervention (2.1 and 2.2, Table 1), five were white. The one case that failed to detect contralateral recurrence was Filipino (2.4, Table 1). In larger studies, appropriate demographic reports are crucial to equitably advance this field of personalized medicine in diverse patient populations.
Demographic characteristics: Age, race/ethnicity, and breast cancer subtype.

TNBC, triple negative breast cancer.
Study limitations
While this study provides valuable real-world insights, it is not without limitations. The early-access program allowed clinicians to be more experimental than systematic with ctDNA orders, which led to earlier detection and intervention in several cases. No ctDNA timepoints were required beyond baseline assessments, and collections were not randomized. In addition, baseline ctDNA tests were established throughout different stages of the disease course. Nevertheless, studies involving patients with high-risk breast cancer have previously established ctDNA’s negative prognostic capability at any time point, whether persistent positivity occurs before, during, or after neoadjuvant therapy. 17 Our relatively small sample size and lack of randomization constrain the generalizability of our findings across broader populations and limit our ability to confirm our positive ctDNA results and earlier intervention as a surrogate for improvement in overall survival, a gap yet to be addressed in ongoing trials. However, our results showcase how ctDNA has been used in real-life clinical situations, affecting both patient and clinician decision-making, ultimately showcasing its utility and harms.
Future directions
While persistent ctDNA positivity has consistently predicted recurrence,17,19 it is important to acknowledge ongoing criticism that the earlier detection of molecular residual disease has not been shown to improve overall survival in metastatic breast cancer. This concern reflects a broader skepticism toward intensive surveillance strategies, particularly when interventions are unlikely to alter the overall disease trajectory and thus may introduce potential harms, including limited therapeutic options later in the disease course, increased healthcare costs, and heightened patient anxiety. 20
Ongoing trials, such as the SURVIVE study (NCT05658172), are currently assessing potential survival benefit in a surveillance arm utilizing ctDNA in patients with intermediate-to-high-risk breast cancer receiving follow-up care. 31 Although survivorship bias has not yet been clinically proven in ctDNA studies, machine learning models have been validated in external cohorts using ctDNA metrics, indicating the potential of ctDNA to impact risk stratifications and thus treatment decisions. Initial predictive models for survival integrate factors such as the number of detected variants and the total cell-free DNA extracted from plasma, posing future avenues to further explore in larger cohorts and trials. 32
While our study does not address survival benefit directly, it highlights that ctDNA can still meaningfully guide clinical decision-making, particularly in cases where persistent ctDNA negativity may support treatment de-escalation and improved quality of life in patients facing the psychological and physical toll of metastatic treatment. These real-life clinical cases highlight that utility may not only lie in prolonging survival, but also in enabling patient-centered, individualized care in the metastatic setting.
Summary
Our findings represent a real-world evaluation of clinician practices in response to testing as well as patient experience, demonstrating ctDNA’s potential and its challenges. Nevertheless, our study showed that ctDNA can support clinicians in personalizing treatment regimens and improving treatment adherence in patients with high-risk breast cancer. This testing can serve as a valuable tool in managing high-risk breast cancer with appropriate discussion of limitations and clear communication of both its potential benefits and harms.
Conclusion
Our findings demonstrate cases where ctDNA serves as a valuable tool for disease evaluation. In some cases, earlier recurrence detection in patients with persistently positive ctDNA allowed clinicians to perform proactive treatment upgrades. In addition, ctDNA negativity was used to monitor metastatic treatment response and the potential for de-escalation. While ctDNA can characterize disease burden and facilitate earlier diagnosis of recurrence and potential intervention, it is unclear whether this ultimately changes the disease course outcome. There is a need for more research to refine its clinical applications. While the emotional burdens of positive ctDNA results have been seen in this study, ctDNA positivity has also demonstrated potential in aiding treatment adherence. Future studies are ongoing to discern whether early treatment of molecular breast cancer relapse delays or prevents subsequent clinical relapse. This study also emphasizes the need to include diverse populations in future ctDNA studies and the importance of considering the emotional impact of ctDNA testing on patients.
