Abstract
BACKGROUND:
Ovarian cancer remains one of the most lethal malignancies in women and the unfavorable prognosis and frequent recurrence are mainly due to the chemoresistance. However, the main mechanism underlying chemoresistance is still elusive.
OBJECTIVE:
To determine the role and biological function of ITGBL1 in ovarian cancer chemoresistance.
METHODS:
Immunohistochemical staining was used to determine the expression of ITGBL1 in ovarian cancer tissues. The association between ITGBL1 expression and clinicopathological features and survival was determined. Functional analysis including cell viability, apoptosis assays were performed after chemo drugs treatment to confirm the role of ITGBL1 in chemoresistance. In vivo tumor growth assay was used to detect the chemosensitivity of tumor cells. Western blot was used to detect the expression of indicated proteins.
RESULTS:
We noticed that ITGBL1 expression was significantly upregulated in ovarian cancer tissues compared to that in adjacent non-cancer tissues and high expression of ITGBL1 was significantly associated with lymph node invasion and advanced FIGO stage. More importantly, high ITGBL1 was an independent prognostic factor of ovarian cancer. Further experiments demonstrated that ITGBL1 promoted tumor cell resistant to chemo drugs both in vitro and in vivo. Mechanically, we found that ITGBL1 could activate PI3K/Akt signaling and using PI3K/Akt inhibitor could abrogate ITGBL1 induced chemoresistance.
CONCLUSIONS:
Our findings indicate that upregulation of ITGBL1 has important clinical significance and drives chemoresistance in ovarian cancer. Detection and depletion of ITGBL1 might be the potential approaches for diagnosis and therapy for ovarian cancer patients.
Introduction
Ovarian cancer (OC) is one of the most lethal disease with high mortality in women worldwide [1]. The overall 5-year survival of ovarian cancer patients is about 46% [2, 3]. However, patients in Stage I/II have a much higher survival rate than them in Stage III/IV (74% vs 27%), due to the frequent recurrence and chemoresistance [4, 5]. Recently, studies on the understanding of mechanisms underlying ovarian cancer progression and chemoresistance have identified several genetic and epigenetic alternations, such as genomic breakage, CCNE1 amplifications and MICU1-induced glycolysis [6, 7, 8]. However, the molecular regulation associated with ovarian cancer remains elusive. Hence, it is urgently needed to identify the features of ovarian cancer and new biomarkers for predicting the prognosis and therapy effects.
Integrin subunit beta-like 1 (ITGBL1) was first cloned and reported in 1999 and characterized by the integrin-like cysteine-rich repeat, and was belong to the EGF-like family [9]. It was also known as TIED (Ten integrin EGF-like repeat domain-containing protein) [10]. However, ITGBL1 has no canonical transmembrane domains or RGD motif, indicating the distinct regulation pattern of ITGBL1 [11]. ITGBL1 could be secreted and has been demonstrated to regulate several critical biological functions such as cell adhesion, migration and invasion [12, 13]. ITGBL1 is overexpressed in bone metastatic subclone cells compared to the parental cell, which is coincident with the fact that ITGBL1 is frequently co-expressed with genes that associated with bone remodeling and metastasis [14]. ITGBL1 also has peak expression in mid-gestational placenta [15]. Recently, ITGBL1 was demonstrated to promote cell migration through FAK/SRC signaling In ovarian cancer [16]. However, the prognostic value of ITGBL1 and its role in ovarian cancer chemoresistance are largely unknown.
In the present study, we explored the expression and clinical significance of ITGBL1 in ovarian cancer. We found that high expression of ITGBL1 could be an independent prognostic factor of ovarian cancer. Moreover, we noticed that ITGBL1 confer tumor cell chemoresistance both in vitro and in vivo via upregulating PI3K/Akt signaling. These results provided new insight to the ovarian cancer treatments.
Materials and methods
Ethics statements and clinical samples
The study was approved by the Ethics Committees of The Affiliated Hospital of Inner Mongolia Medical University. 210 cases of ovarian cancer specimens and corresponding adjacent non-cancer tissues were surgically removed and restored in Department of Obstetrics and Gynecology, The Affiliated Hospital of Inner Mongolia Medical University from Match 2006 to June 2013. Written informed consent was obtained from all patients involved in this study and none of the patients had received chemotherapy, radiotherapy or related therapies before surgery. The histological and clinical stages were classified according to FIGO [17].
Immunohistochemical staining (IHC)
Immunohistochemical staining was performed as described previously [18]. Briefly, the slides were deparaffinized in xylene and rehydrated in gradient ethanol. After retrieving the antigen in citrate buffer, the slides were incubated with 0.3% H
All slides were determined by two independent pathologists. To evaluate the results of immunohistochemical staining, semiquantitative score system for the staining intensity and positive cell percentage was used as described previously [18]. Briefly, the intensity was scored as 1 (no staining), 2 (weak), 3 (moderate), 4 (strong) and the percentage was scored as 1 (0–15%), 2 (15–50%), 3 (50–75%), 4 (
Reagents, cell lines and transfection
Cisplatin (Cis, #S1166) and paclitaxel (Taxol, #S11 50) were purchased from Selleck Inc (Houston, TX, USA). Ovarian cancer cell lines (OV90, SKOV3, ES2, CAOV3 and OVCAR4) were came from the American Type Culture Collection (ATCC) and maintained according to the protocol.
Small interfering RNAs (siRNAs) targeting ITGBL1 were purchased from Genepharma (Shanghai, China). The sequences were as followed: 5
ITGBL1 is upregulated in ovarian cancer and associated with shorter survival and recurrence. (A) The representative images of IHC staining in 210 cases of paired ovarian cancer tissues and non-cancer tissues (left panel), and statistical analysis (right panel). Bar, 100 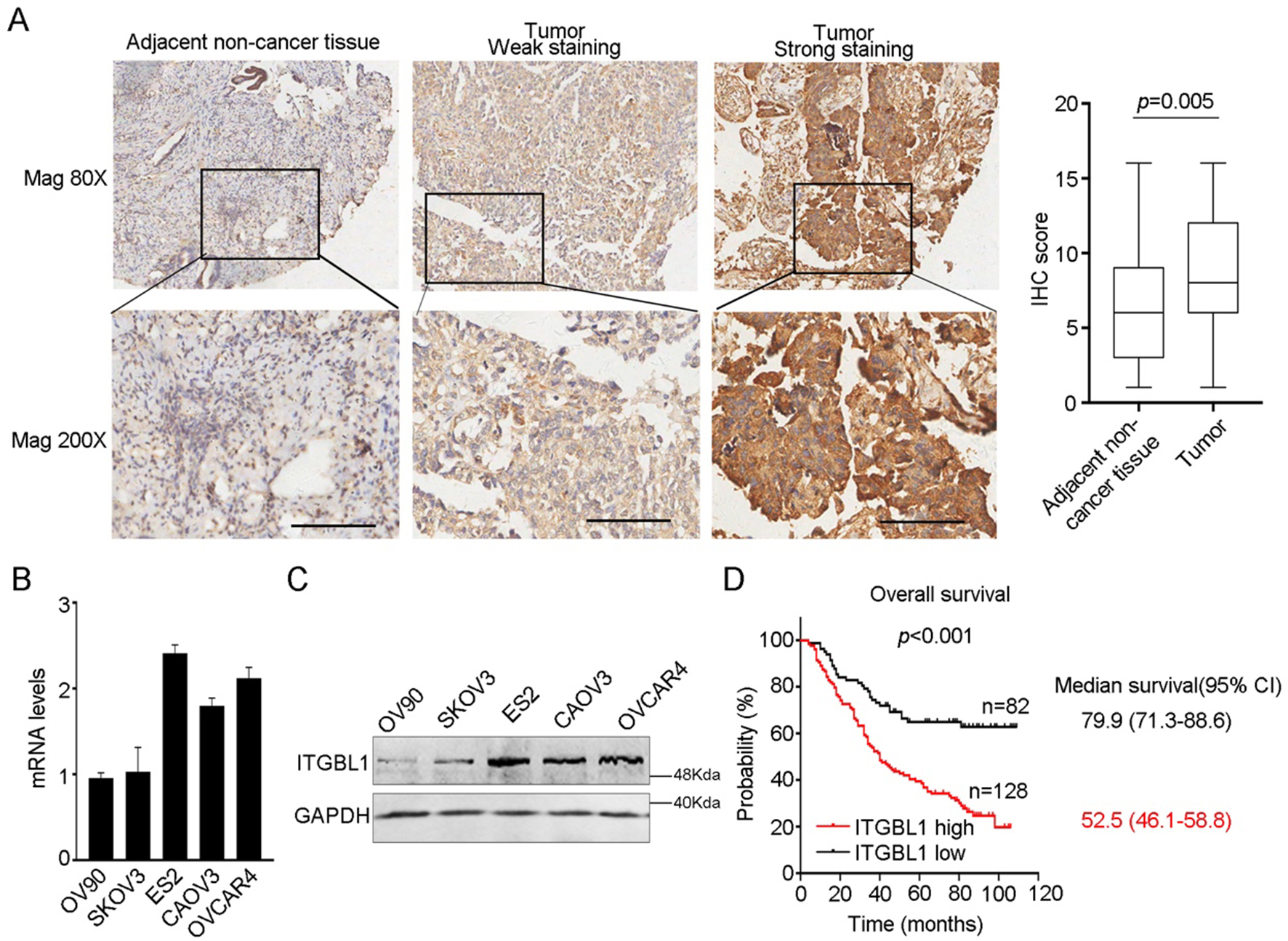
Total RNA was extracted from primary tumour and adjacent tumour tissues or cells using Trizol reagent. The first strand cDNA was synthesized by PrimeScript RT-PCR kit (Takara, Dalian, China) according to the manufacturer’s recommendation. Quantitative real-time PCR was performed using 7500 Real-time PCR system (Applied Biosystem, Foster City, CA, USA) with SYBR Green method as described previously [20]. GAPDH gene was used as internal control. The primers used were as follows: ITGBL1, forward, 5
Western blot
Total proteins from fresh tissues were lysed in RIPA buffer containing proteinase inhibitors and phosphatase inhibitors cocktail according to the manufacturer’s recommendation. The protein concentration was determined by BCA methods. 80
Knocking down of ITGBL1 promotes chemodrugs induced apoptosis. A: The efficiency of siRNAs targeting ITGBL1 in CAOV3 and ES2 cells was examined by western blot. The expression value was calculated by ImageJ according to the density of each band. B: Cell apoptosis was examined by FACS after chemodrugs treatment. C: The viability of CAOV3 and ES2 knocking down of ITGBL1 cells and control cells after gradient Cis treatment. The IC50 value was calculated. D: The TUNEL assay of CAOV3 and ES2 knocking down of ITGBL1 cells and control cells after Cis treatment. 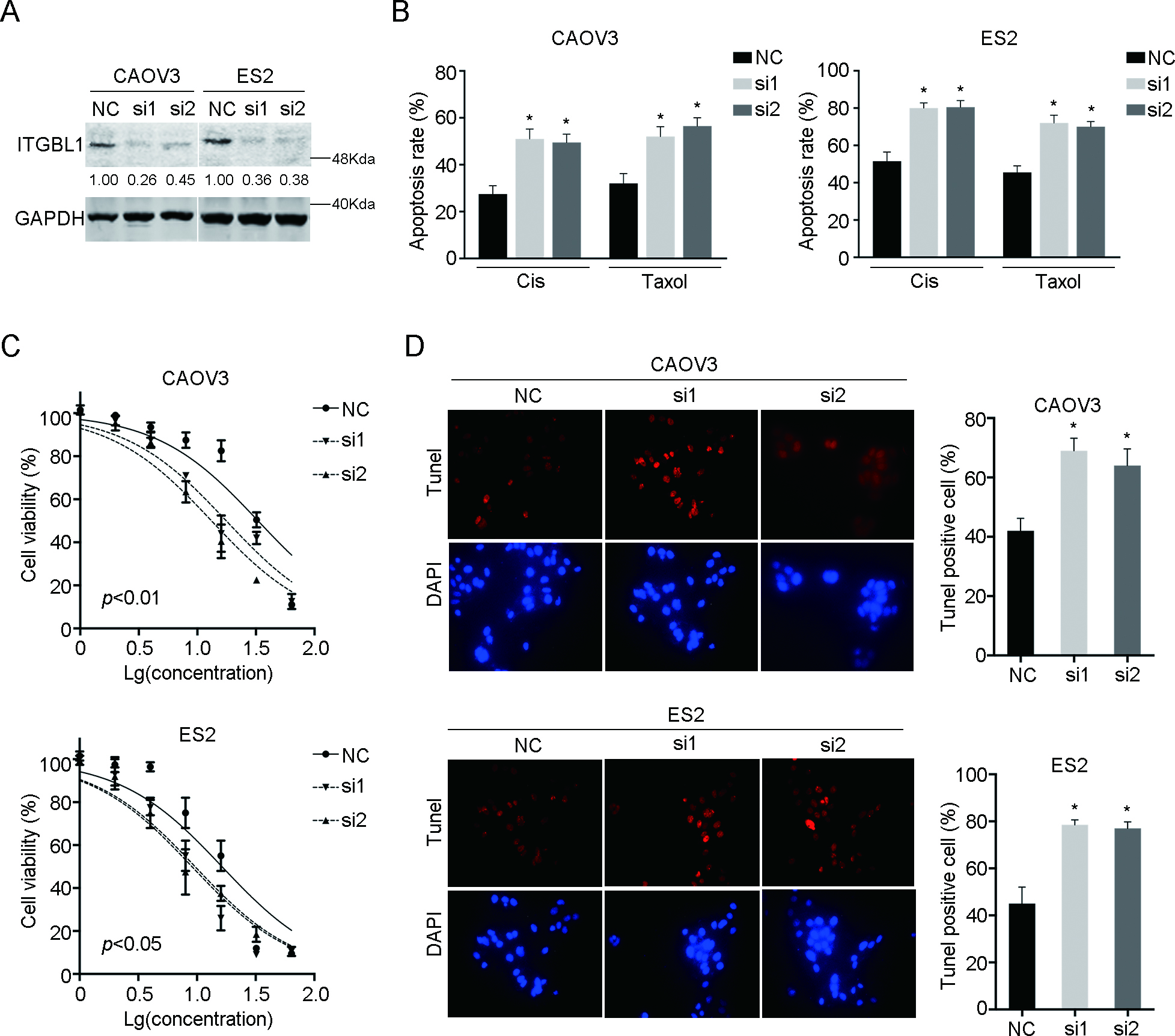
Association between ITGBL1 expression and clinicopathologic parameters of ovarian cancer
ITGBL1 overexpressing cells or knocking down cells with control cells were plated onto 96-well plates in the concentration of 1500 cells/well. Cells were treated with gradient concentration (1, 2, 4, 8, 16, 32, 64
Cell apoptosis assay
Indicated cells were plated onto 6-well plates and then treated with 5
Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay
Cells were plated onto cover glass and treated with 5
In vivo tumor growth assay
1
Statistical analysis
Statistical analysis and graphical representations were performed using SPSS 21.0 (IBM, Chicago, IL, USA) and GraphPad Prism 5 (San Diego, CA, USA). ITGBL1 mRNA in tumour and adjacent non tumour tissues were compared using paired
Cox regression model for overall survival of ovarian cancer
Cox regression model for overall survival of ovarian cancer
Overexpression of ITGBL1 confer chemoresistance. A: The overexpression of ITGBL1 was examined in SKOV3 and OV90 cells. B: Cell apoptosis was examined by FACS after chemodrugs treatment. C: The viability of SKOV3 and OV90 overexpressing ITGBL1 cells and control cells after gradient Cis treatment. The IC50 value was calculated. D: The TUNEL assay of SKOV3 and OV90 overexpressing ITGBL1 cells and control cells after Cis treatment. E: Cell apoptosis was examined by FACS after chemodrugs treatment in the presence of gradient recombinant ITGBL1 (rITGBL1). F: The TUNEL assay of SKOV3 and OV90 treated with gradient recombinant ITGBL1 in the presence of Cis. 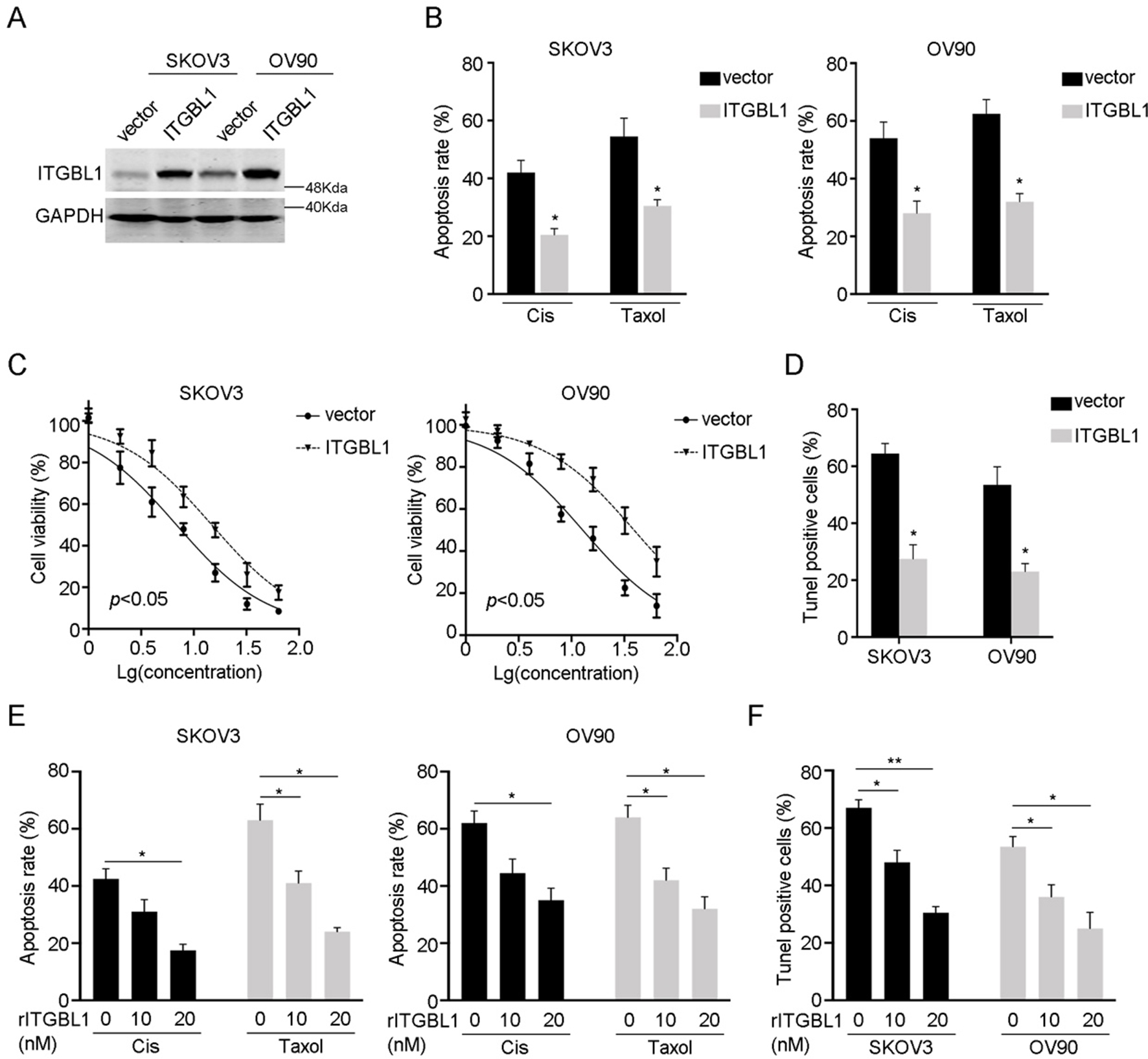
Upregulation of ITGBL1 is associated with poor prognosis of ovarian cancer
To investigate the expression of ITGBL1, we performed IHC in 210 cases of ovarian cancer tissues and adjacent non-cancer tissues and found that ITGBL1 was predominant upregulated in cancer tissues (Fig. 1A). We also detected the mRNA and protein levels of ITGBL1 in ovarian cancer cell lines (Fig. 1B and C). Then, we determined the correlation between ITGBL1 expression and clinicopathological features in ovarian cancer. Among the 210 cases, the high expression of ITGBL1 was significantly correlated with the lymph node invasion and FIGO stages (Table 1). Whereas no significant association was observed between ITGBL1 expression and other features such as age, or histotype, or grade, or tumor size or CA125 levels.
To demonstrate the prognostic value of ITGBL1 in ovarian cancer, Kaplan-Meier plot with log-rank test in 210 cases was performed and we noticed that high expression of ITGBL1 indicated shorter overall survival (Fig. 1D). Furthermore, multivariate analysis indicated that ITGBL1, lymph node invasion and FIGO stage could be the independent prognostic factors of ovarian cancer (Table 2).
Knocking down of ITGBL1 enhances Cisplatin treatment in vivo. A: Four different groups of tumors were shown (shcon, shcon 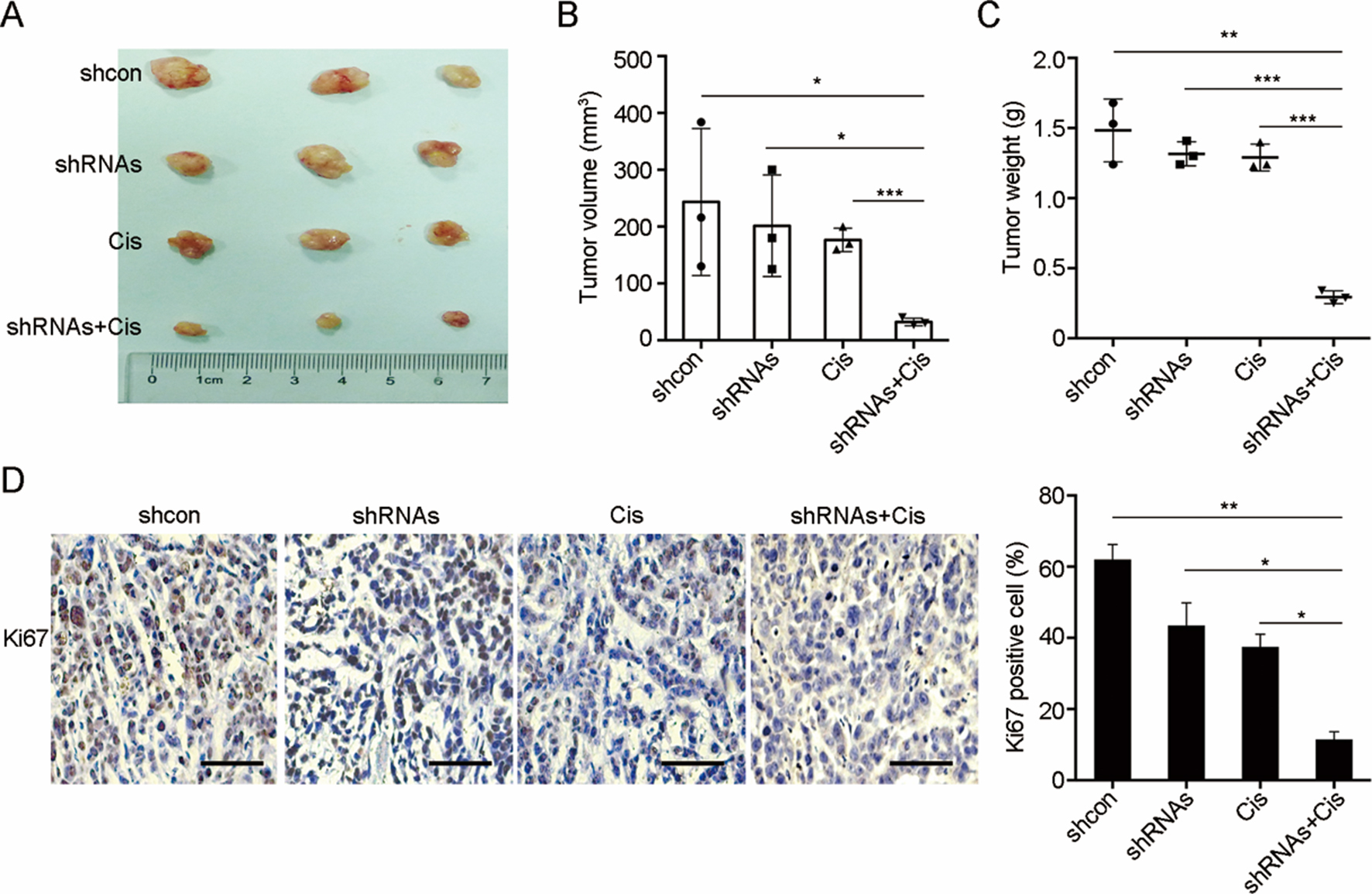
We employed two siRNAs to knock down the expression of ITGBL1 in CAOV3 and ES2 (Fig. 2A). To detect the role of ITGBL1 in ovarian cancer chemoresistance, we examined the effects of two first-line drugs, Cisplatin (Cis) and paclitaxel (Taxol), on ITGBL1 knocking down cells. We found that ITGBL1 knocking down significantly increased chemodrugs induced apoptosis as evaluated by flow cytometry in CAOV3 and ES2 cells (Fig. 2B). Then, we treated cells with Cis in ITGBL1 knockdown and control cells with a gradient of concentrations to evaluate cell viability. As shown in Fig. 2C, compared with control cells, ITGBL1 knocking down significantly decreased the viability of ovarian cancer cells and increased the chemo-sensitivity (details were shown in Table 3). Furthermore, we performed TUNEL assay after Cis treatment in CAOV3 and ES2 ITGBL1 knocking down cells and control cells and noticed that ITGBL1 knocking down enhanced the percentage of TUNEL positive cells (Fig. 2D). Taken together, the results indicated that downregulation of ITGBL1 promoted chemodrugs induced apoptosis.
Overexpression of ITGBL1 confer cells cisplatin and taxol resistance
Next, we overexpressed ITGBL1 in SKOV3 and OV90 cells which had a relative low endogenous expression of ITGBL1 (Fig. 3A). Of note, we treated cells with Cis and Taxol and found that ITGB1L overexpression significantly decreased the apoptosis rate (Fig. 3B). The IC50 values of ITGBL1 overexpression cells were also higher than that of control cells (Fig. 3C and Table 3), demonstrated by the viability assay. In parallel, TUNEL assay indicated similar results as well (Fig. 3D).
IC50 values of Cisplatin
IC50 values of Cisplatin
ITGBL1 promotes chemoresistance via activating PI3K/Akt signaling. (A) Detection of PI3K/Akt signaling activation and downstream targets after ITGBL1 knocking down or overexpression. (B) Knocking down of ITGBL1 suppressed p-AKT expression while rITGBL1 restored PI3K/Akt activation. (C) LY294002 inhibited rITGBL1 induced PI3K/Akt activation. (D, E) LY294002 abrogated rITGBL1 induced chemoresistance in SKOV3 (D) and OV90 (E) cells treated with Cis or Taxol. 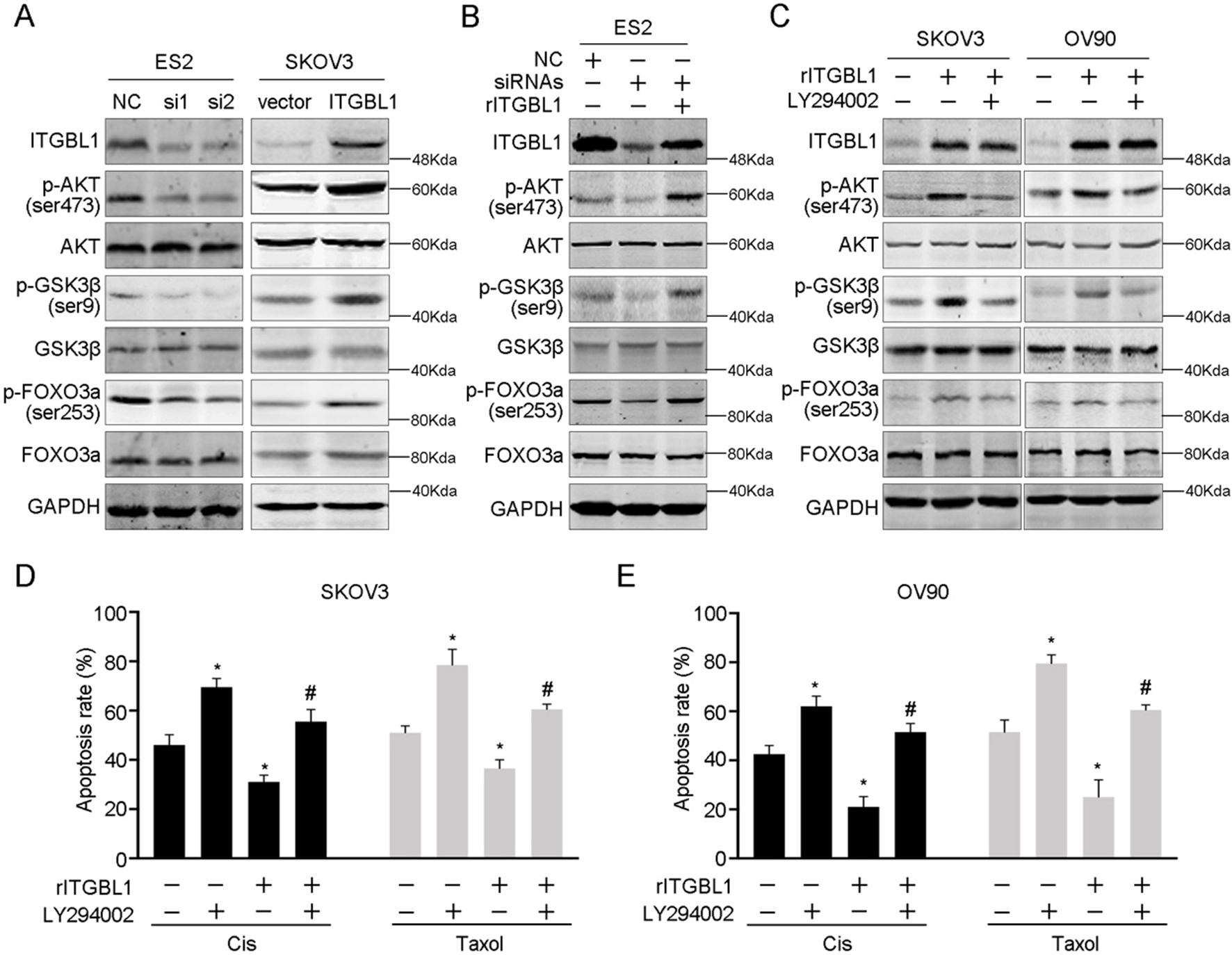
As ITGBL1 could be secreted, we performed apoptosis assay using recombinant ITGBL1 (rITGBL1) and chemodrugs. When treated with Cis or Taxol, the cell apoptosis rate was decreased along with the increased rITGBL1 concentration (Fig. 3E). The results of TUNEL assay were consistent with ITGBL1 overexpression (Fig. 3F). Taken together, the data indicated that ITGBL1 increased ovarian cancer cells chemoresistance.
We further examined the tumor growth and chemo-resistance in vivo. ES2 cells with or without knocking down of ITGBL1 were s.c. injected into nude mice. After 2 weeks, we randomized separated mice and half of them in each group were treated with Cis. After 4 weeks, mice were sacrificed and we found that knocking down of ITGBL1 or treated with Cis only, suppressed tumor growth. But compared with control group, it was not statistically significant. However, combination of knocking down of ITGBL1 and Cis, the tumors were significantly suppressed (Fig. 4A and B). Also, the tumor weight had the same trend (Fig. 4C). Meanwhile, we performed IHC of Ki67, and indicated that knocking down of ITGB1L plus Cis treatment significantly abrogated tumor growth in vivo (Fig. 4D).
ITGBL1 induces chemoresistance via upregulating PI3K/Akt signaling
To explore the mechanism of ITGBL1 induced chemoresistance, we detected the PI3K/Akt signaling which had pivotal role on tumor proliferation, chemoresistance and metastasis. Downregulation of ITGBL1 significantly decreased the activation of PI3K/ Akt evidenced by the suppression of p-AKT and the downstream factors p-GSK3
To further examine whether PI3K/Akt signaling plays a role in chemoresistance induced by ITGBL1, we performed the apoptosis assay. Using LY294002, we noticed that the apoptosis was decreased after rITGBL1 treatment in the presence of Cis or Taxol, while restored by inhibition of PI3K/Akt signaling in SKOV3 and OV90 cells (Fig. 5D and E). Taken together, the data indicated that PI3K/Akt signaling was involved in ITGBL1 induced chemoresistance.
Discussion
Although ITGBL1 has been implied to be highly expressed in ovarian cancer and regulate cancer cell migration and adhesion, this is the first study to examine the role of ITGBL1 protein in ovarian cancer prognosis and chemoresistance [21]. Ovarian cancer is one of the most fatal gynecological malignancies and the chemoresistance is frequently occurred [22, 23]. A majority of patients relapses within 2–3 years, and becomes resistant to chemotherapy [24]. Therefore, our study suggested that the secreted protein, ITGBL1 might contribute to the malignancy, and was identified as an independent prognostic factor for ovarian cancer.
ITGBL1 has been reported to upregulated in a variety types of cancers, such as colorectal cancer, breast cancer and esophageal cancer [25, 26]. However, Gan et al. reported that ITGBL1 was epigenetic downregulated by miR-576-5p and inhibited Wnt signal in non-small cell lung cancer, suggesting its heterogeneous roles [27]. In addition, ITGBL1 was observed as predictor gene for cirrhosis risk in chronic hepatitis B patients [28]. In current study, we performed immunohistochemistry in 210 paired ovarian cancer samples and adjacent non-cancer tissues, together with detecting protein and mRNA expression of ITGBL1 in fresh tissues. We found that ITGBL1 expression was significantly increased in ovarian cancer. Higher expression of ITGBL1 was associated with advanced FIGO stage and lymph node invasion of patients. Importantly, ITGBL1 expression was identified as an independent prognostic factor for ovarian cancer. Given the fact that ITGBL1 could be secreted, further investigation will be focused on detecting ITGBL1 in serum as a biomarker for predicting ovarian cancer patients’ chemotherapy response, recurrence and prognosis.
Previous studies have shown that ITGBL1 was involved in epithelial-mesenchymal transition (EMT), migration and metastasis. By integrating Oncomine, GEO databases, Gene Set Enrichment Analysis of multiple independent datasets revealed that gastric cancer specimens with high expression of ITGBL1 had elevated expression of EMT genes [12]. Also, Sun et al. indicated that ITGBL1 promoted cell migration and adhesion through the Wnt/PCP and FAK/SRC signaling pathways [16]. Li et al. found that RUNX2 could transcriptional enhance ITGBL1 expression, subsequently activate TGF-
In conclusion, our results for the first time, indicated that ITGBL1 played an essential role in chemoresistance in ovarian cancer. Upregulation of ITGBL1 activated PI3K/Akt signaling, thereby contributing to malignancy and poor prognosis. Our findings suggested that ITGBL1 was a potential biomarker for chemotherapy and provide new clinical target for ovarian cancer.
