Abstract
MicroRNAs (miRNAs; miR) have been proven to act as both oncogenes and tumor suppressors. However, the mechanism of action of miR429 in melanoma cells remains to be elusive. The present study aims to explain the functional role and mechanism of miR429 in the pathogenesis of melanoma. In our study, we have demonstrated that has-miRNA429 (miR429) is a tumor suppressor in melanoma cells. Luciferase reporter assays demonstrated that the overexpression of miR429 reduced the transcriptional activity of AKT serine/threonine kinase 1 (AKT1). Furthermore, the results showed that the mRNA and protein expression levels of AKT1 were downregulated in the melanoma cell lines when miR429 was overexpressed according to qRT-PCR and western bolt, indicating MicroRNA-429 may directly target AKT1 in melanoma. In vivo, overexpression miR-429 could obviously enhance the inhibition effect of tumor size and weight in the nude mice. Taken together, our findings suggest that novel miR429-regulated pathway may serve as new insights into melanoma oncogenesis and metastasis.
Keywords
Introduction
Malignant melanoma is an aggressive tumor with increasing incidence and poor prognosis in the metastatic stage [1]. Although many genetic and epigenetic changes are found to be correlated with melanoma, the carcinogenesis remains poorly understood [2].
MicroRNAs (miRNAs) are endogenous small ncRNA of 19–22 nucleotides that regulate gene expressions. They regulate protein-coding gene expressions at the post-transcriptional level through translational inhibition or mRNA degradation [3]. It is wildly accepted that miRNAs are distributed in some cancers [4, 5, 6], and dysregulate key processes of tumor development and progression, including proliferation, differentiation, cell cycle, apoptosis, migration and invasion [7, 8].
miR429, a member of the miR-200 family, has been recently identified to be an oncogene or tumorsuppressor gene in diverse cancers, including ovarian cancer [9], cervical cancer [10], cholangiocarcinoma [11], non-small cell lung cancer [12], gastric cancer [13], hepatocellular carcinoma [14]. Wu et al. [15] have found that miR429 in RCC contributed to cell migration, cell survival and invasion via the downregulation of Sp1. Chen et al. [16] have also demonstrated that miR429 can regulate RCC cell function by targeting VEGF and reduce the 3’UTR luciferase action of VEGF. However, the role and mechanisms of miR-429 in melanoma cells have not been illustrated clearly. In the present study, we identified AKT1 as a target of miR-429 and showed the possibility of miR-429/AKT1-mediated signal pathway as a new target for melanoma treatment.
Primer sequences used in the present study
Primer sequences used in the present study
3’UTR, 3’ untranslated region; WT, wild-type; MUT, mutant.
Cell culture
A-375 cells and 293T were purchased from the American Type Culture Collection (Manassas, VA, USA). Cells were cultured in DMEM medium (Invitrogen Life Technologies, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (FBS; Shanghai ExCell Biology, Inc., Shanghai, China), 100 U/mL penicillin, and 100
Cell transfection
The miR-429 mimics, miR-429 inhibitor, negative control miRNA mimics (mimics NC) and negative control miRNA inhibitor (inhibitor NC) were chemically synthesized by Shanghai GenePharma Co., Ltd. (Shanghai, China).
Some sequences are listed in Table 1. Transient transfection of miR-429 mimics, inhibitor and/or negative control were conducted using Lipofectamine 2000 reagent (Invitrogen) according to the manufacturer’s protocol.
RTqPCR
The total RNA was extracted from cells using TRIzol
The luciferase reporter assay
The luciferase reporter assay is used to verify whether AKT1 is a direct target of miR-429. Briefly, the 3’-untranslated region (3’-UTR region) of AKT1 with the potential binding sites or mutant sites of miR-429 were generated, followed by fusion into the luciferase reporter vector pGL3-Promoter Vector (Promega, Madison, WI, USA, named as AKT1-WT or AKT1-MUT). The amplification was conducted using primers that listed in Table 1. 293T cells were seeded (10000 cells/well) in a 96 well plate and were co-transfected with miR-429 mimic/miR-negative control, miR-429 inhibitor/inhibitor control and WT/MUT-AKT1-3’-UTR vector. Luciferase activity was measured by using a Luciferase Reporter Assay kit (Promega) in accordance with the manufacturer’s guidance at 48 h after transfection.
Protein extraction and western blot analysis
Western blot was performed according to manufacturer’s guidance. Briefly, cells were homogenized and total protein were extracted using RIPA buffer, then the extraction of the proteins was detected by Western blot. Proteins were detected with antibodies. The primary antibodies included AKT1 (1:1000; cat; Abcam, Hong Kong, China) and GAPDH (1:1000; cat; Santa Cruz, CA, USA). Goat antirabbit IgG-HRP used as the second antibodies were purchased from Beyotime. GAPDH was used as an internal control.
In vivo tumor xenograft studies
Six BALB/c-nu mice (6 weeks, 16–18 g), were provided by Shanghai Slac Laboratory Animal Ltd (Shanghai, China). Mice were adapted for 3 days before any experimental procedures. A-375 cells were injected into the underarms of each mouse. Mice received twice intratumoral injection of miR-429 miRNA agomir or agomir negative control each week when the tumor size reached approximately 5 mm
Statistical analysis
Statistical significance was analyzed by the
Results
miR-429 down regulated the expression of AKT1 in the A-375 cell lines
The mRNA and protein analyses were performed to confirm whether the expression of miR-429 affected the expression of AKT1 at the transcriptional or translational levels. Compared to the control group, the cells transfected with the miR-429 mimics suppressed mRNA expression of AKT1 (Fig. 1A), which was consistent with the results from the luciferase report assay. The overexpression of miR-429 decreased AKT1 protein expression dramatically in A-375 cell lines (Fig. 1B).
A. A-375 cells transfected with miR-429 mimic exhibited decreased mRNA expression levels of AKT1 compared with those transfected with the negative control (NC). B. Overexpression of miR-429 resulted in a decrease in the protein expression levels of AKT1 in the A-375 cell lines. The results represent three independent experiments. 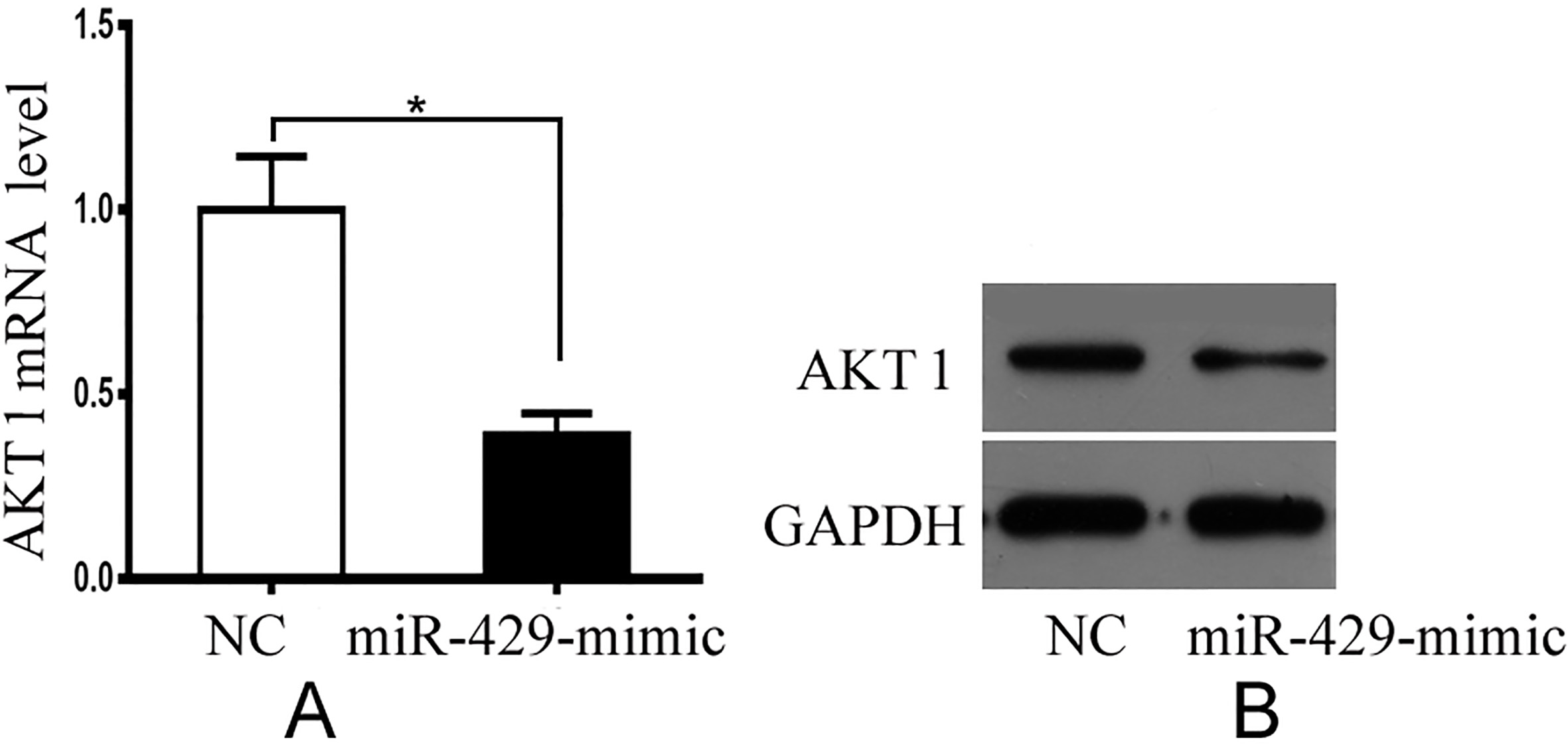
Prediction of the miRNA target was performed using computational algorithms, according to the base-pairing rules between the miRNA and mRNA target sites, location of binding sequences within the target’ 3’UTR, and conservation of target binding sequences within associated genomes. In the present study, gene was predicted using the microRNA (
AKT1 is the target gene of miR-429. A. Putative duplex formation between miR-429 and the AKT1 3’UTR. B. Luciferase reporter constructs containing the 3’UTR of AKT1, together with the miR-429 mimic or negative control; inhibitor or inhibitor NC, were transfected into the 293T cells. After 48 h, luciferase activity was detected. The relative luciferase activity of the reporter, containing the WT 3’UTR was significantly suppressed when miR429 was cotransfected into the 293T cells compared with negative control; containing the WT 3’UTR was significantly promoted when inhibitor was cotransfected into the 293T cells compared with inhibitor NC. C. By contrast, the luciferase activity of the MUT reporter was unaffected. All luciferase assays were repeated three times; UTR, untranslated region; WT, wildtype; MUT, mutant. 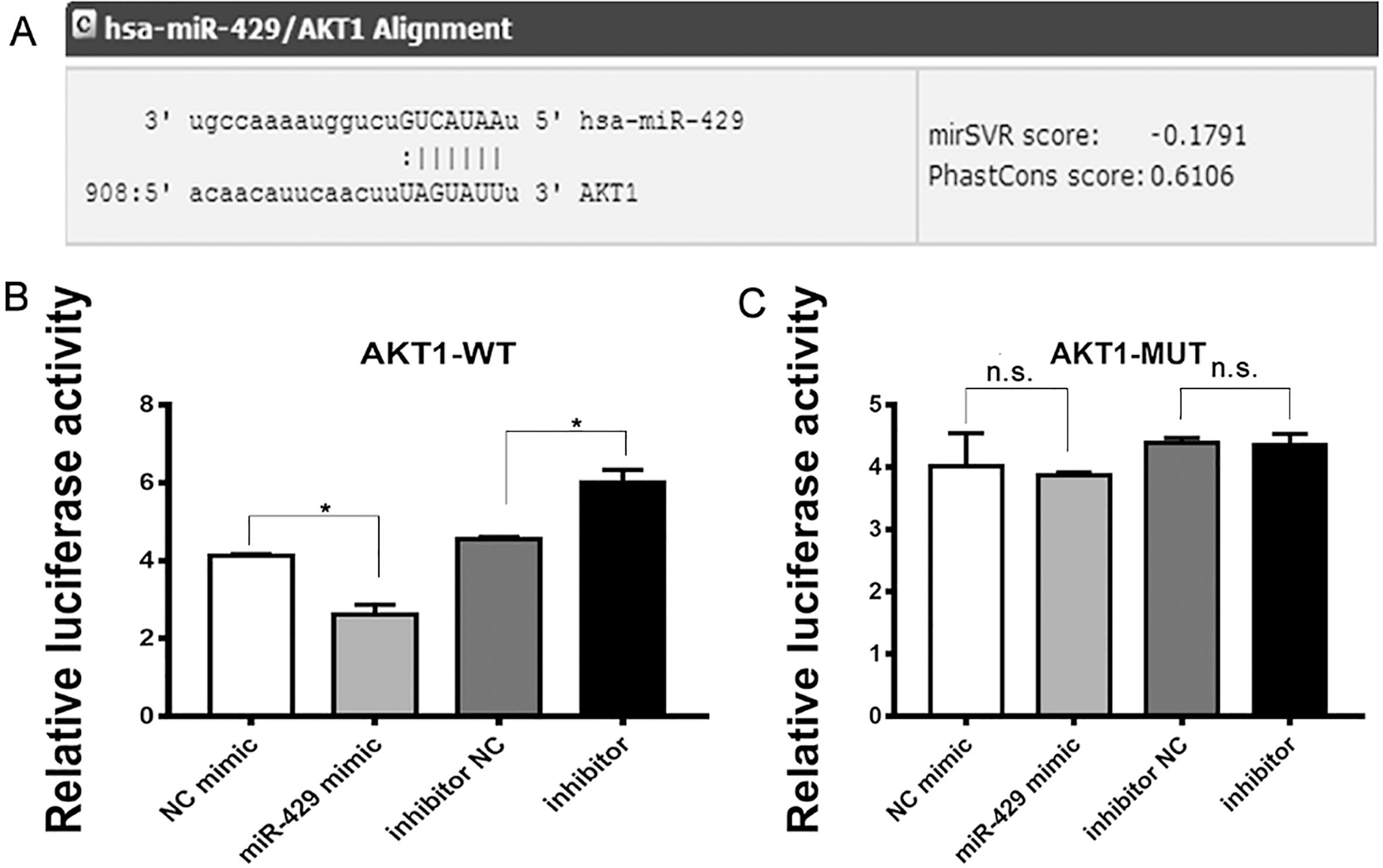
To perform luciferase reporter assay for validating whether miR-429 physically interacts with AKT1 mRNA, we constructed two vectors by cloning WT and MUT 3’UTR into pGL3-Promoter Vector. Luciferase reporter constructs containing the 3’UTR of AKT1, together with the miR-429 mimic or negative control; inhibitor or inhibitor NC, were transfected into the 293T cells. After 48 h, luciferase activity was detected. The relative luciferase activity of the reporter, containing the WT 3’UTR was significantly suppressed when miR429 was cotransfected into the 293T cells compared with negative control; containing the WT 3’UTR was significantly promoted when inhibitor was cotransfected into the 293T cells compared with inhibitor NC (Fig. 2B). By contrast, the luciferase activity of the MUT reporter was unaffected (Fig. 2C). All luciferase assays were repeated three times.
To confirm whether overexpression of miR-429 could inhibit the growth of tumor, a nude mouse xenograft model was established in this study. As a result, miR-429 inhibited the growth effect of tumor effectively. The size and weight in the nude mice with miR-429 treatment were much lower than these in control mice (Fig. 3A–C).
Tumor volumes at the indicated dates B and images as well as tumor weight A at day 28 C in the nude mice treated with intratumoral injection of miR-429 agomir or its negative control (NC agomir) for four weeks. The average values are present in the bar graphs (means 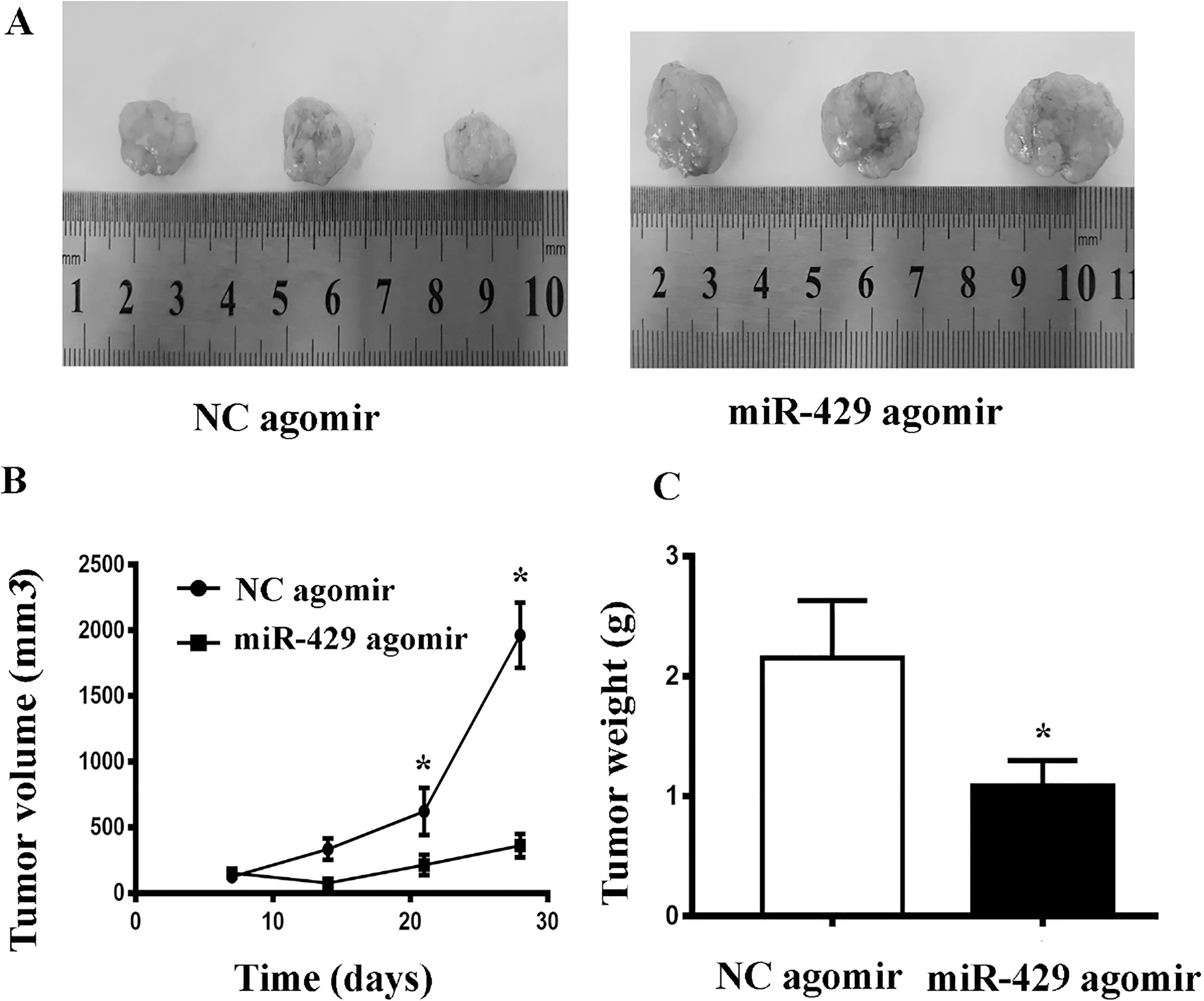
Melanoma is a highly malignant tumor. Radical surgical removal of the tumor is the one of its main therapies. However, the recurrence rate is still high even after surgery. Therefore, it is urgent to search for alternative treatment for melanoma.
The role of miRNAs has been well studied both in cancer and normal tissues (17–20). MiRNAs are considered as important biomarkers or therapeutic targets for cancers [21]. However, the role of miR-429 it has been found to be controversial. Machackova et al. reported that miR-429 acted as a tumor suppressor through inhibiting loss of E-cadherin in renal cancer cells [22]. miR-429 was also found to be capable of inhibit migration, invasion and proliferation of renal cancer cells via targeting directly Sp1 [15]. Furthermore, miR-429 was related to resistance of chemoradiotherapy of pancreatic cancer through regulation of PDCD4 [23]. In 2018, Xue et al. demonstrated that miR-429 was significantly much lower in HCC tissues and cells compared with normal tissues, meanwhile elevated level of miR-429 could inhibit the metastasis ability in HCC cells through targeting RAB23 [14]. Conversely, Tang et al. reported that MiR-429 enhanced HCC cell invasion and migration through targeting the PTEN/PI3K/AKT/
In the present study, we demonstrated that miR-429 act as a tumor suppressor in melanoma cells. Several signal pathways are involved in miR429 regulated tumor development, such as PDCD4 [23], VEGF [16], and cmyc [26]. It is the first time to demonstrate that AKT1 demonstrated was a target in mediating the anti-tumoral function of miR429.
The phosphoinositide 3kinase (PI3K)/RAC seine/ threonineprotein kinase (AKT)/mechanistic target of rapamycin (mTOR) signaling pathway is frequently dysregulated in several cancers [27, 28]. AKT plays a key role in the PI3K/AKT/mTOR signaling pathway, and it is closely associated with a wide variety of cancer pathophysiology, including EMT, growth, migration metabolism, and angiogenesis [29, 30, 31]. AKT consists of three isoforms (AKT1, AKT2, and AKT3), and the different isoforms act as different roles in cancer pathophysiology. AKT1 can suppress breast cancer cell invasion and migration [32, 33] but accelerated the onset of tumorigenesis [34]. Therefore, knockdown of AKT1 gene inhibited the onset of tumorigenesis [35].
To explore potential effect of miR-429 in melanoma tumorigenesis, four computational algorithms were used to predict the targets of miR-429, which identified AKT1 as a novel target gene. The binding sites of miR-429 on the AKT1 3’UTR were verified using a luciferase reporter assay. In addition, cancer cells transfected with miR-429 mimics showed reduced expression of AKT1 at both mRNA and protein level. The in vitro experiment show that overexpression of miR-429 could obviously enhance the inhibition effect of tumor size and weight in the nude mice. All these findings indicate that AKT1 is a direct target gene of miR-429 in melanoma. Therefore, miR429/AKT1 axis can be a novel marker and therapeutic target in melanoma treatment.
In summary, the present study demonstrates that miR429 acts as a tumor suppressor in melanoma via the regulation of AKT1. This newly underlying association between miR429 and AKT1 may provide a new therapeutic strategy for melanoma.
Footnotes
Acknowledgments
We are indebted to the coworkers, whose names were not included in the author list, but who contributed to this work.
