Abstract
INTRODUCTION:
The present study evaluated the effects of the long non-coding RNA (lncRNA) gastric carcinoma high-expressed transcript (GHET1) in cervical carcinoma development.
METHODS:
The expression levels of GHET1 and PTEN were measured using in situ hybridisation, immunohistochemistry (IHC) and quantitative reverse transcription PCR assay to investigate their correlations. In an in vitro study, the effects of GHET1 knockdown on the biological activities of SiHa and HeLa cells were evaluated by MTT, flow cytometry, transwell and wound-healing assays and relative protein expression was measured using western blotting. In an in vivo experiment, cell apoptosis and relative protein expression were measured in nude mice using TUNEL and IHC assays, respectively.
RESULTS:
The expression levels of lncRNA GHET1 and PTEN protein differed significantly between cancer and adjacent normal tissues (
CONCLUSION:
GHET1 knockdown suppressed cervical carcinoma development via the PTEN/PI3 K/AKT signalling pathway.
Introduction
Cervical cancer is the second most serious malignancy in women that poses a significant threat to health [1]. However, its pathogenesis remains unclear. Understanding the pathogenesis of cervical cancer is crucial to its treatment. Long non-coding RNAs (lncRNAs) are a class of RNA with a transcription length of
The present study measured the expression ofGHET1 in cervical cancer and adjacent normal tissues and evaluated the effects of GHET1 knockdown in SiHa and HeLa cervical cancer cell lines as well as the effects of lncRNA GHET1 in vivo.
Materials and methods
Clinical data
A total of 36 pairs of cervical cancer and adjacent normal tissues were collected from patients diagnosed and treated for cervical cancer at the Second Hospital of Jilin University. Adjacent normal tissues were taken
In situ hybridisation
The in situ hybridisation (ISH) probe used for detecting GHET1-labelled digoxin was designed and synthesised by Sanon Biotech Co., Ltd. (Shanghai, China). Tissue slices were processed using an Enhanced Sensitive ISH Detection Kit (POD) (Boster, Wuhan, China) according to the manufacturer’s protocol. Tissue chip de-waxing was performed at 37
Immunohistochemistry
Immunohistochemistry (IHC) was performed as follows. After de-waxing, samples were hydrated and rinsed with PBS, pH 7.4, three times for 5 min each. Peroxidase blocking solution (50
RT-PCR assay
Cervical cancer and adjacent tissues were added to TRIzol reagent before being cut and milled separately. Total cellular RNA was extracted using the PureLink RNA Mini Kit. Genomic contamination was removed using DNase I during extraction. Reverse transcription of total RNA was performed using a reverse transcription kit to generate cDNA. GHET1 and PTEN expression levels were measured using SYBR Green real-time fluorescent quantitative PCR. GAPDH was stably expressed among the different tissues (data not shown) and used as a reference. The primer sequences were as follows: GHET1, R: 5
Cell culture and transfection
HeLa and SiHa cells were seeded at a density of 1
CCK-8 cell proliferation assay
Cell proliferation rates were measured in the different groups by CCK-8 assay. After transfection, cells were seeded in 96-well plates at a density of 2
Flow cytometryanalysis of cell apoptosis and cell cycle
Measurement of cell apoptosis
Cells from different groups were washed twice in pre-cooled PBS and then adjusted to a concentration of 1
Measurement of cell cycle distribution
Cells from the different groups were washed twice in pre-cooled PBS before being fixed in 70% ice-cold ethanol for 4 h at 12
Transwell assay
Transwell chambers were placed into 24-well plates. The upper surface of the basement membrane of the transwell chamber was covered with Matrigel packets, dried at 4
Wound-healing assay
HeLa and SiHa cells were seeded in six-well plates. Cells were transfected when they reached 80% confluence. After 6 h, the medium was replaced, and a scratch was made in the cell layer using a 200-
Western blot assay
A total of 5
Grouping and treatment in the in vivo study
Balb/c nude mice (male, 4–6 weeks age) were purchased from Nanjing Medicine University. HeLa cells, at a concentration of 5
TUNEL assay
Tumour tissues were fixed in 4% paraformaldehyde, followed by conventional dehydration, embedding, slicing and de-waxing in water, and then washed using PBS. After protease K digestion at 37
Statistical analysis
All data were analysed using SPSS 19.0 software. The measured data were shown as mean
Results
Clinical data and analysis
ISH and RT-PCR analyses revealed that GHET1 expression was significantly up-regulated in cervical cancer tissue compared with that in adjacent normal tissue (
Clinical data and analysis. (A) Expression of lncRNA GHET1 in cervical cancer and normal tissues measured by ISH (200 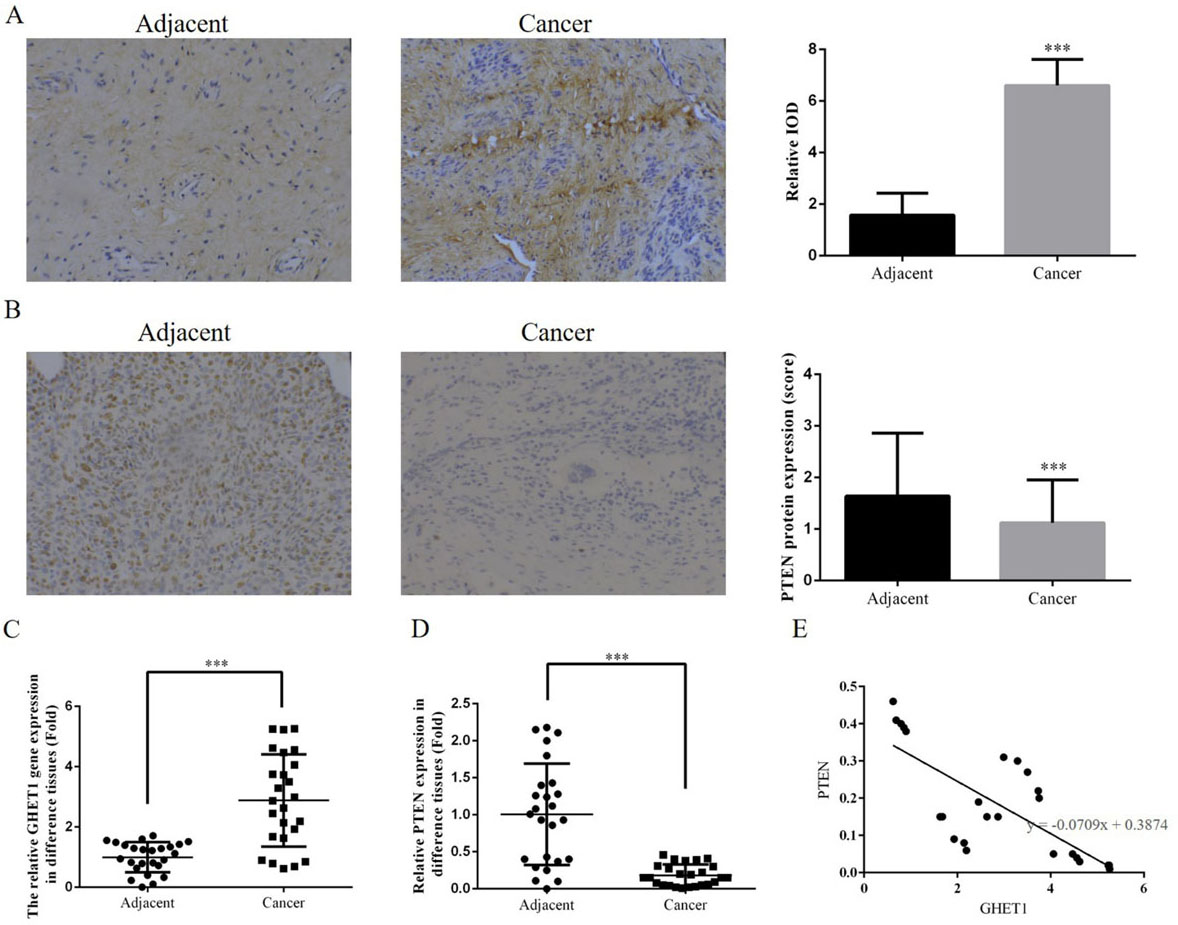
To evaluate the effects of GHET1 knockdown in cervical cancer cells, CCK-8 assay was used to measure the proliferation rates of HeLa and SiHa cells transfected with either si-GHET1 or non-specific siRNA (Fig. 2A and B). Proliferation of HeLa and SiHa cells was significantly reduced in the si-GHET1 groups compared with that in the NC groups (
Cell proliferation rates in the knockdown and control groups measured by CCK-8 assay. HeLa cell (A) and SiHa cell (B) proliferation rates in the two groups. *** 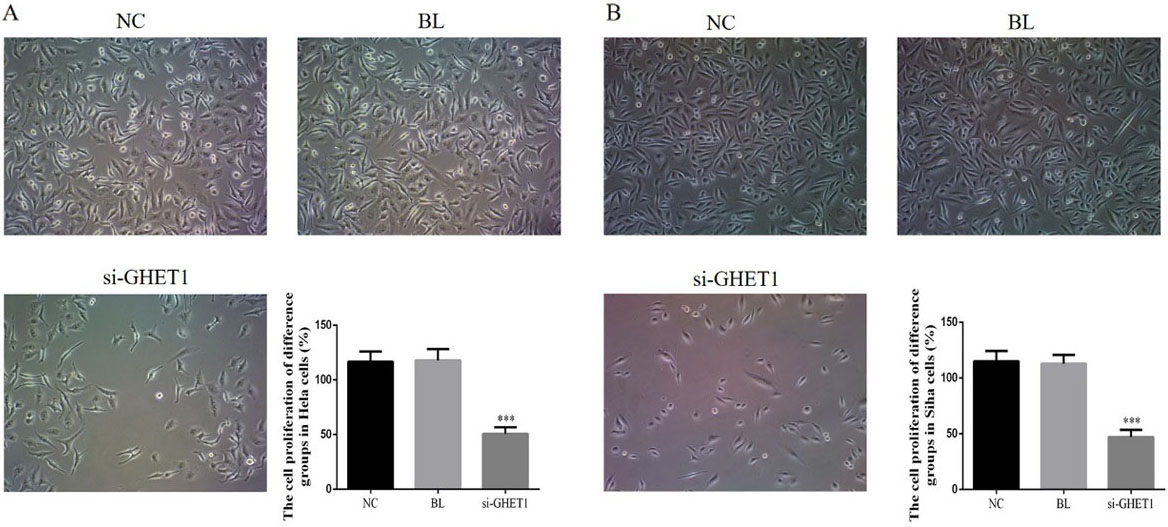
Cell apoptosis rate in the knockdown and control groups measured by flow cytometry. HeLa cell (A) and SiHa cell (B) apoptosis rates in the two groups. *** 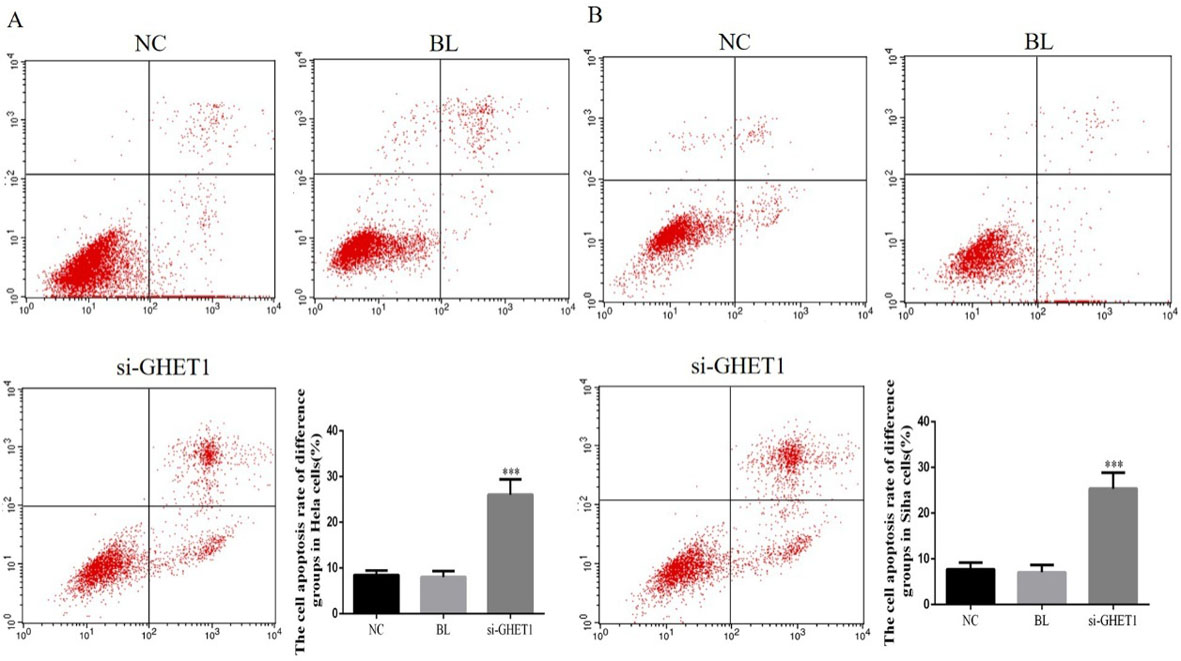
Cell cycle in the knockdown and control groups measured by flow cytometry. HeLa cell cycle (A) and SiHa cell cycle in the two groups. *** 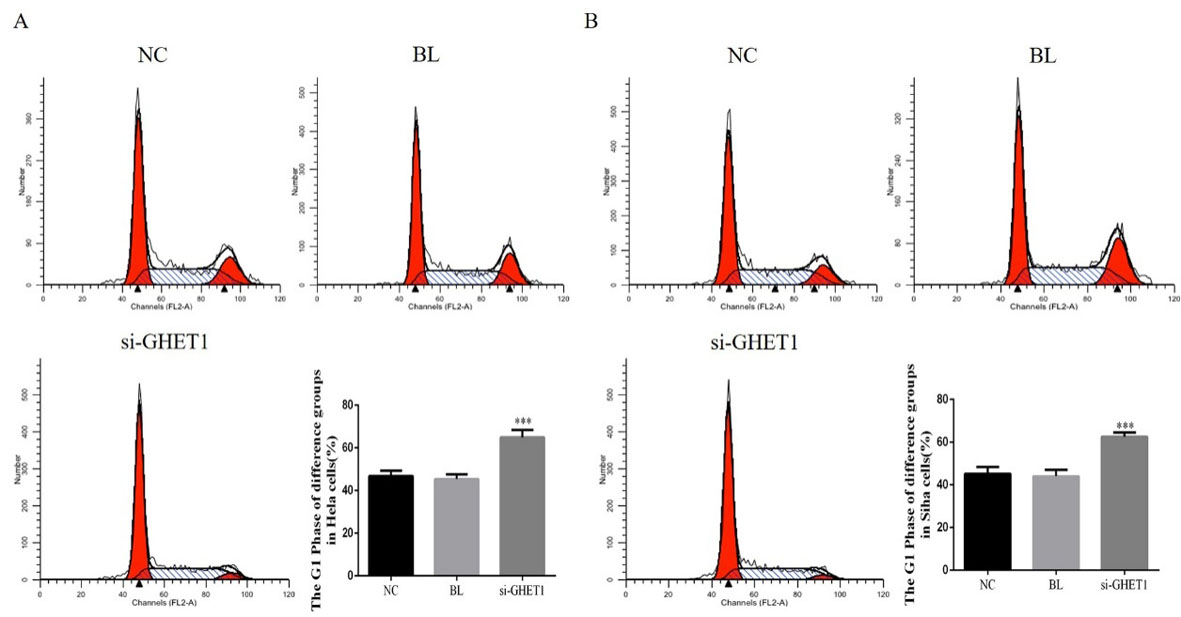
Cervical cancer cell invasion in the knockdown and control groups measured by transwell assay. HeLa cell (A) and SiHa cell (B) invasion in the two groups. *** 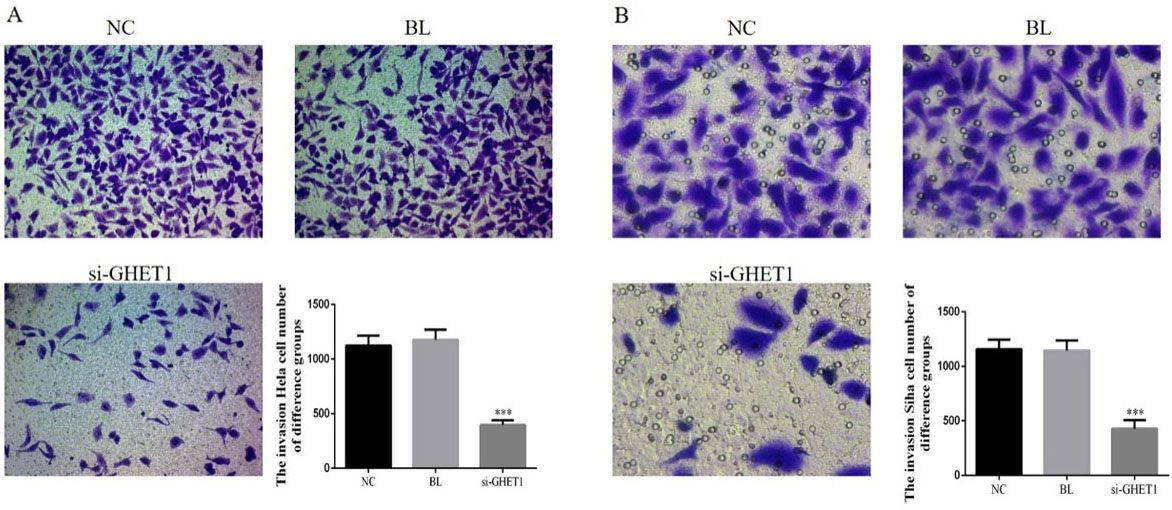
Wound-healing rate in the knockdown and control groups in HeLa and SiHa cells measured by wound-healing assay. HeLa cell (A) and SiHa cell (B) wound-healing rate in the two groups. *** 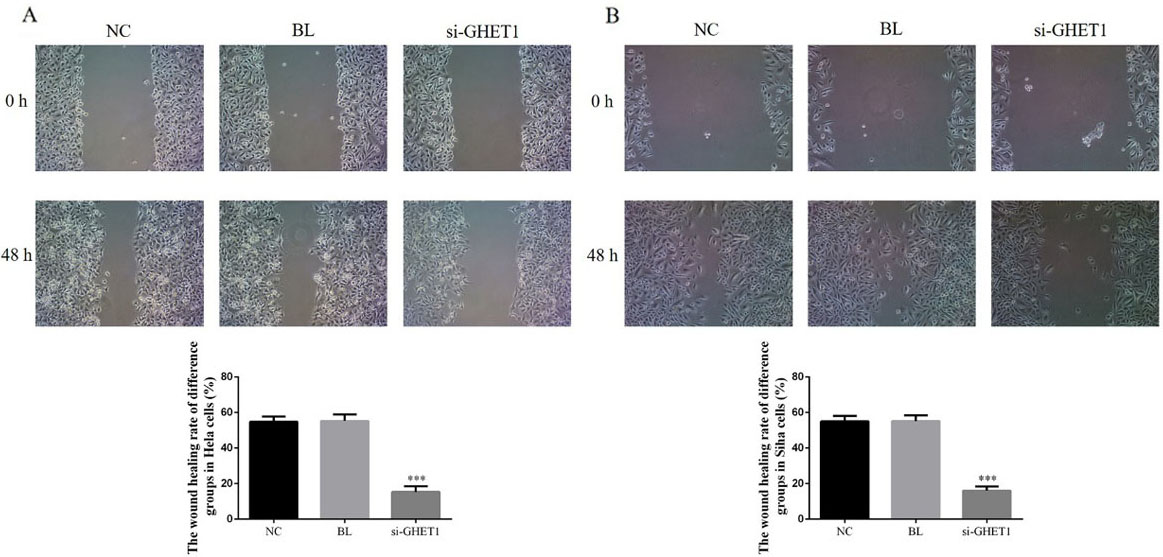
Flow cytometry analysis showed that the apoptosis rates of HeLa and SiHa cells were significantly higher in the GHET-siRNA groups compared with those in the NC groups (
Analysis of the G
phase
To investigate the role of GHET1 in cervical cancer development, we assessed the cell cycle status among the different groups using flow cytometry (Fig. 4A and B). The G
Relative protein expression in HeLa and SiHa cells measured by western blotting. Relative protein expression in the knockdown and control groups in HeLa cells (A) and SiHa cells (B). *** 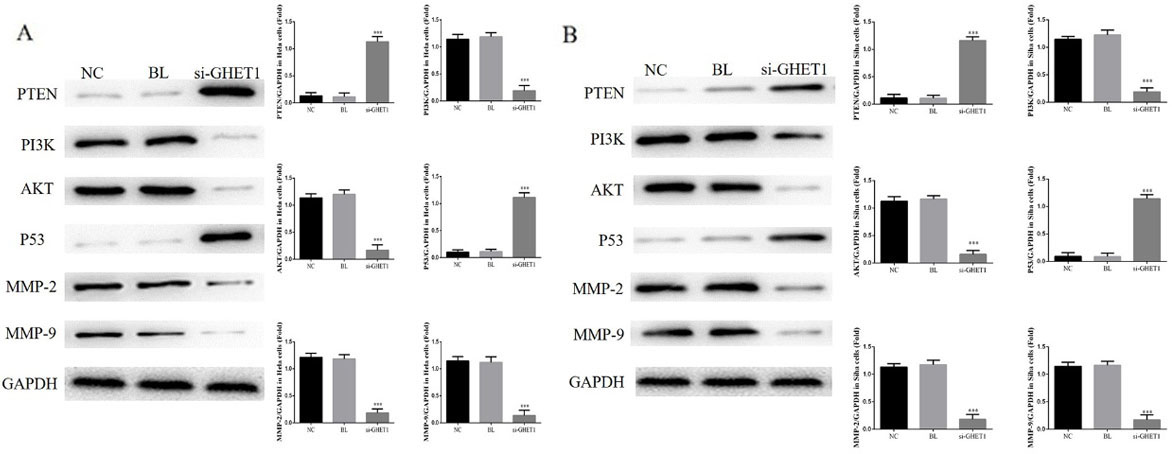
Transwell assay analysis showed that the number of invasive HeLa and SiHa cells was significantly lower in the si-GHET1 groups compared with that in the NC groups (
Migration ability of cervical cancer cell lines
Wound-healing assays revealed that the wound-healing rate was significantly suppressed in the si-GHET1 groups compared with that in the NC groups of HeLa and SiHa cells (
Relative protein expression
To further elucidate the mechanisms of GHET1 in cervical cancer cell lines, we measured relative protein expression by western blot assay. Compared with the NC groups, the si-GHET1 groups showed significant stimulation of PTEN and P53 protein expression (
Tumour volumes and weights, and cell apoptosis. (A) Tumour volumes and weights in the knockdown and control groups. *** 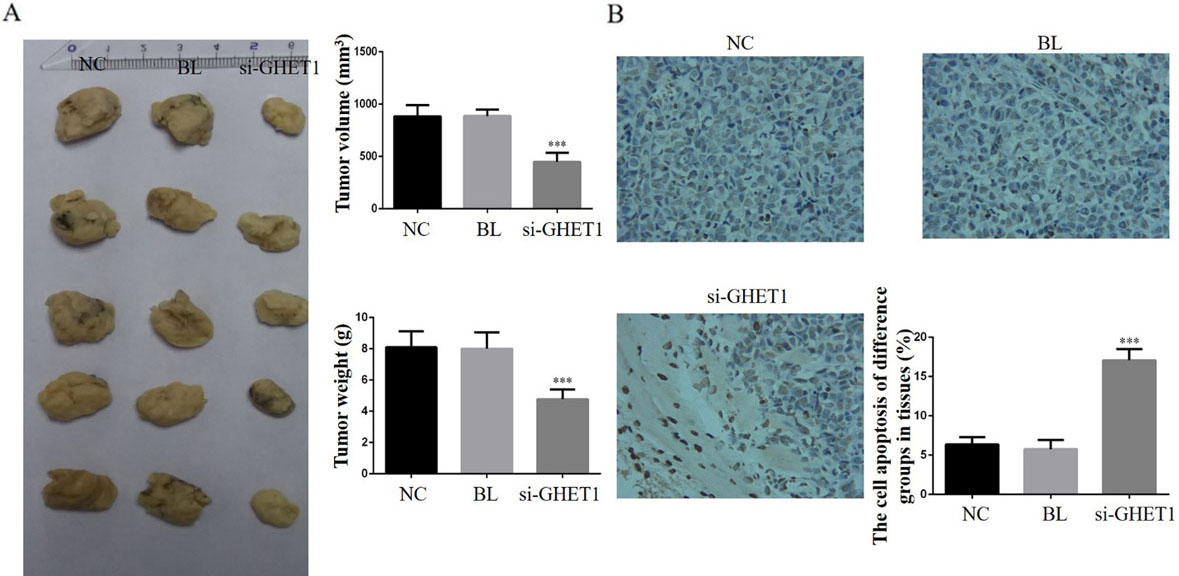
Tissue PTEN and PI3 K protein expression in the knockdown and control groups. PTEN (A) and PI3 K (B) protein expression in the different groups and tissues (200 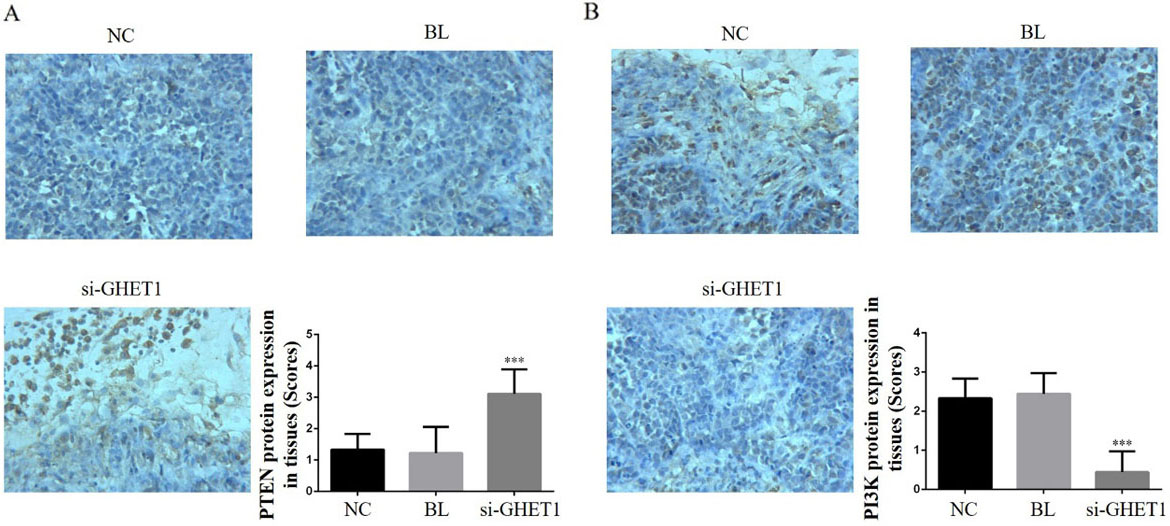
AKT and P53 protein expression in the knockdown and control groups in cervical cancer and normal tissues. AKT (A) and P53 (B) protein expression in the different groups and tissues (200 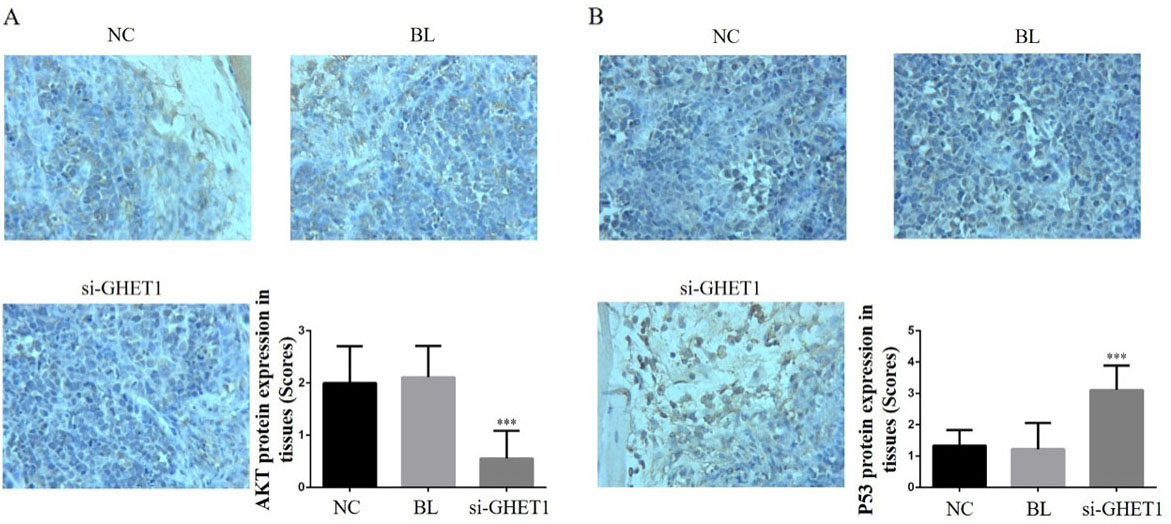
MMP-2 and MMP-9 protein expression in the knockdown and control groups in cervical cancer and normal tissues. MMP-2 (A) and MMP-9 (B) protein expression in the different groups and tissues (200 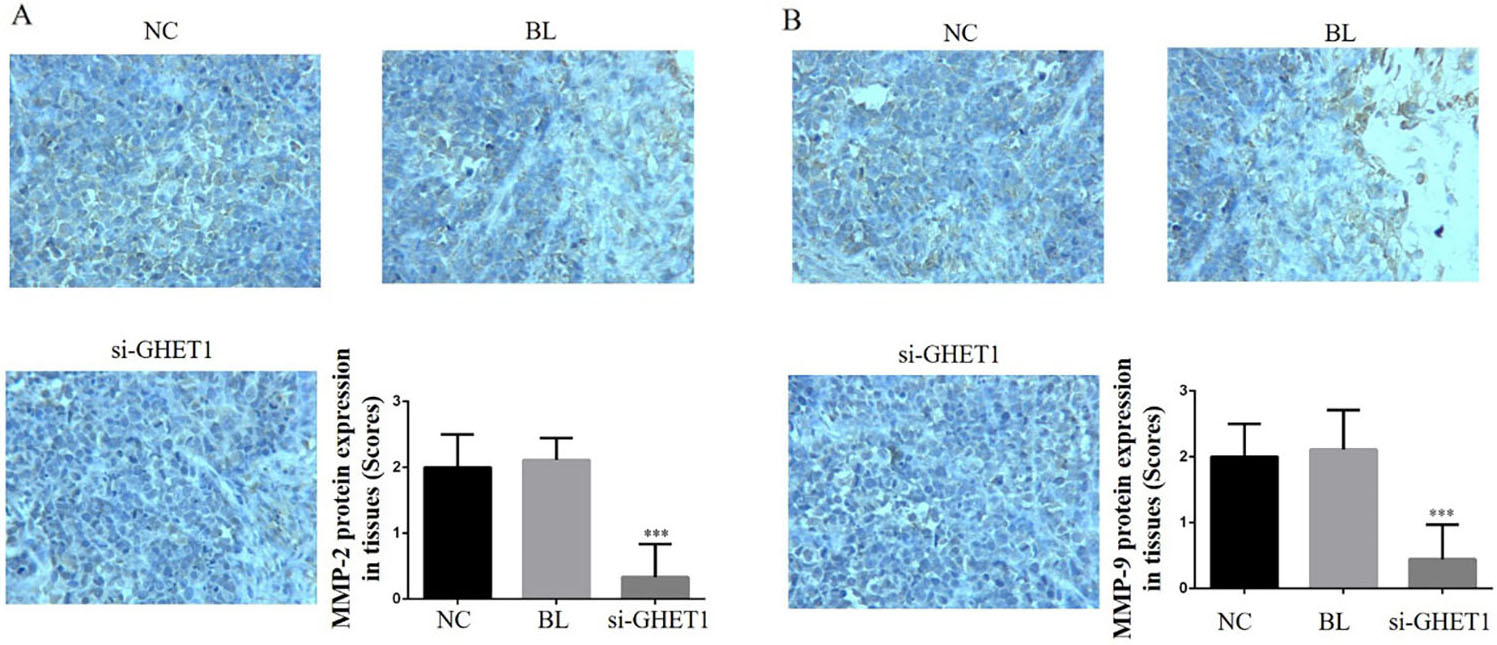
Tumour volumes and weights were significantly suppressed in the si-GHET1 groups compared with those in the NC groups (
Relative protein expression in the tumour tissues
IHC analysis showed that the relative protein expression of PTEN, PI3 K, AKT, P53, MMP-2 and MMP-9 differed significantly between the si-GHET1 and NC groups (
Discussion
lncRNAs are important non-coding RNAs that cannot be translated into protein. Although many studies have shown that lncRNAs play key roles in the development and progression of tumours [13], their precise mechanisms of action remain unclear. In particular, the clinical significance and use of lncRNAs as biomarkers for tumour resection and prognosis have not yet been definitely established. Some studies have shown that lncRNAs are closely correlated with cervical cancer [14, 15, 16, 17]. The functions of lncRNAs vary, and they may act as either tumour suppressor genes or oncogenes according to different tissues, indicating individualtissue-specific expression of lncRNAs. Since lncRNAs may have multiple target genes, regulation of the downstream target genes (with different functions) and their final role in tumour development may differ among organs and individuals.
In recent years, the lncRNA GHET1 has been shown to play a new role in cancer [18, 19]. However, the correlation between GHET1 and cervical cancer remains unclear. The present study evaluated the expression of lncRNA GHET1 in cervical cancer and adjacent normal tissue as well as the anti-tumour effects and mechanisms of GHET1 knockdown in HeLa and SiHa cervical cancer cell lines. We found that GHET1 knockdown suppressed biological activities such as proliferation, invasion and migration of the cervical cancer cell lines and improved apoptosis via regulation of the cell cycle.
We also measured relative protein expression. PTEN is the first tumour suppressor gene of the protein and lipid phosphatase family that blocks the signal transduction pathway mediated by cytokines, thereby inhibiting growth, invasion and metastasis of tumour cells and promoting tumour cell apoptosis [20, 21]. PTEN gene and protein expression varies betweenhaematopoietic malignancies, such as leukaemia, lymphoma, myeloma and other cancer cell lines. Although low expression, deletion, mutations and rearrangement of PTEN are rare, low expression is associated with poor prognosis. Inactivation of the PTEN gene occurs predominantly because of indirect mechanisms such as transcriptional silencing and post-transcriptional modification and methylation [22, 23, 24, 25]. Our clinical data showed that PTEN protein expression was significantly down-regulated in cervical cancer tissue compared with that in adjacent normal tissue and was negatively correlated with GHET1 expression. In the in vitro experiments, PTEN protein expression increased in response to GHET1 knockdown. On the basis of these results, we inferred that GHET1 could be a tumour factor in cervical carcinoma as knockdown of GHET1 suppressed cervical carcinoma with a concurrent increase in PTEN expression levels both in vitro and in vivo.
PI3 K/AKT is an important cell signal transduction pathway that is closely correlated with cell biological activities such as proliferation, apoptosis, invasion and migration [26, 27, 28, 29]. Our results implied that GHET1 knockdown may have exerted its anti-tumour effects via the PTEN/PI3 K/AKT signalling pathway. P53 is a tumour suppressor gene with a wide range of roles, including regulation of tumour formation, growth, development and progression, as well as regulation of tumour stem cell division and self-renewal and inhibition of tumour recurrence [30, 31, 32]. Under normal circumstances, p53 protein acts as a tumour suppressor and regulates all aspects of cell life activities [33]. In response to DNA damage, the DNA damage stress response is induced, p53 protein triggers cell cycle arrest, and complete DNA repair or cell apoptosis is induced to prevent the damaged DNA from continuing to expand and transcribe [34]. The present study revealed that GHET1 knockdown led to significant suppression of PI3 K/AKT pathway activity and induction of PTEN, as well as enhanced P53 protein expression in vitro and vivo. From our results, we inferred that the suppressed proliferation and enhanced apoptosis observed in the cervical cell lines were mediated by the PTEN/PI3K/AKT/P53 pathway and stagnation of the cell cycle in the G
MMPs comprise a large family of proteins that require calcium, zinc and metal ions as cofactors. They can degrade almost all protein components in the extracellular matrix as well as the basement membrane to promote tumour cell invasion and play a key role in tumour invasion and metastasis [35, 36]. MMP-2 and MMP-9 were shown to be involved in angiogenesis and basement membrane degradation, which are directly related to tumour metastasis and prognosis [37, 38]. In the present study, the invasion and migration abilities of the HeLa and SiHa cell groups were significantly suppressed in the si-GHET1 groups. Furthermore, MMP-2 and MMP-9 protein expression was significantly reduced in vitro and in vivo. Therefore, we hypothesised that down-regulation of GHET1 could inhibit cervical cancer cell invasion and migration via regulation of MMP-2 and MMP-9 in vitro and in vivo. In the cervical development, lncRNA GHET1 play a key role to promote cervical cancer occurrence.
Conclusion
GHET1 knockdown showed anti-tumour effects by suppressing cell proliferation, invasion and migration and by improving cell apoptosis via regulation of the PTEN/PI3 K/AKT/P53 pathway and MMP-2 and MMP-9.
Footnotes
Acknowledgment
This study was supported by the Natural Science Foundation of Liaoning Province, China (Grant no. 20170540570), and was a project of the Liaoning Clinical Research Center for Colorectal Cancer (Grant no. 2015225005), which was sponsored by the Liaoning BaiQianWan Talents Programme (2017, no. C13).
