Abstract
BACKGROUND:
MicroRNAs (miRNAs) are emerging as promising molecules in the diagnosis, prognosis and treatment of urological tumours. Recently, our group performed two independent studies highlighting that miR-210-3p may be a useful biomarker not only for diagnosis but also for post-surgery clear cell Renal Cell Carcinoma (ccRCC) management.
OBJECTIVE:
The aim of this study is to further explore the effectiveness of miRNA as non-invasive biomarker for clinical outcomes and ccRCC response to the treatment.
METHODS:
We analyzed miR-210-3p levels in neoplastic and healthy tissue and in urine specimens collected at surgery and during follow-up of 21 ccRCC patients by RTqPCR.
RESULTS:
Firstly, we confirmed that the expression of miR-210-3p was upregulated in tumor tissues and in urine samples of analyzed cohort. Of note is that miR-210-3p expression was significantly reduced in urine samples from disease-free patients during follow-up (from 3 to 12 months) compared to the baseline levels observed at the time of surgery. In a small subgroup of patients presenting metastatic progression (such as bone, intestinal or lung metastasis), the urine levels of miR-210-3p correlated with responsiveness to the therapy.
CONCLUSIONS:
This pilot study highlights the relevance of secreted miR-210-3p as powerful non-invasive prognostic and predictive biomarker for the evaluation of clinical outcomes and treatment response during ccRCC follow up.
Background
Renal cell carcinoma (RCC) represents the tenth most common cancer worldwide [1, 2]. Probability of occurrence is particularly high in men with excessive body weight. Apart from that smoking, hypertension, environmental pollutants, as well as genetic predisposition (e.g. mutation in VHL gene in von-Hippel-Lindau disease), are described as the main risk factors associated to this type of cancer. According to the 2016 WHO classification, RCC can be divided into three major clinical subtypes: the chromophobe RCC (chRCC), the papillary RCC (pRCC) and clear cell RCC (ccRCC). The latest represents the most frequent and aggressive phenotype, with a 30% of cases that developes distant metastases. ccRCC is also the most common cause of death among all kidney cancer patiens [1, 2]. To date, no useful markers are available in clinical practice not only for early diagnosis but also for better stratification and management of high-risk patients [3]. Identifying novel low-expensive and non-invasive tools for a more accurate definition of prognosis and response to therapy would improve patient clinical outcomes and quality of life therefore this is one of the major challenges in oncology research.
Among these novel tools, microRNAs (miRNAs) are emerging as one of the most relevant. MiRNAs are a class of small non-coding RNA of which expression is tissue-specific and highly regulated according to the cell line and its developmental stage [4]. Studies indicate that miRNAs are heterogenously expressed in cancer cells, in which they form unique expression patterns or signatures [5, 6]. Of note, specific miRNA-expression signatures are identified in correlation with tumor classification and are also emerging as predictive markers of metastasis [7, 8]. These small non-coding RNAs, to exercise their function, are assembled in the functional RNA induced silencing complexes (RISCs), where a member of Argonaute (Ago) family of proteins, Ago1-4, provides an unique platform for target recognition and gene silencing [9, 10, 11]. The miRNAs loaded in the RISC complex usually function as negative regulators of gene expression, mainly by targeting the 3’ untranslated region (UTR) of their target mRNAs [12].
MiRNAs and their protein complexes modulate a variety of developmental and physio-pathological processes by fine-tuning translation of specific target mRNAs [13].
MiRNAs are secreted by cells as free molecules and are bounded in a ribonucleoprotein complexes or in vesicles. Discovery of circulating miRNAs in biological fluids as blood plasma, serum and urine suggests the possibility of using this class of non-coding RNAs as non-invasive biomarkers for an early cancer diagnosis. Potentially, they could also serve as prognostic factors and predictors of response to the therapy [14, 15].
These miRNA secretion pathways are dysregulated in cancer, making miRNAs attractive candidate mole-cules for liquid biopsies in several types of cancer [16] and in particular in urologic tumors [17, 18]. Of note, several studies demonstrated that circulating miRNAs can be introduced to the cell by endocytosis or membrane fusion and affect recipient cell gene expression [19].
Recently, our group performed a retrospective cohort study of tissue samples obtained from patients subjected to partial or radical nephrectomy for ccRCC and evaluated the expression of miR-21-5p, miR-210-3p, miR185-5p and miR-221-3p either in FFPE neoplastic tissue and in paired normal parenchyma [20]. miR-21-5p and miR-210-3p resulted significantly up-regulated in ccRCC vs matched normal tissues.
To further support the potential clinical usefulness of these miRNAs in ccRCC management we started a prospective study [21] where we evidenced that miR-21-5p, miR-210-3p and miR-221-3p were up-regulated in fresh frozen ccRCC tissues vs matched healty tissues. Moreover, by using two independent cohorts of patients we evidenced that miR-210-3p was significantly up-regulated in urine specimens collected from ccRCC patients at the time of surgery compared to samples from healthy donors [21].
In this study, to further assess the robustness of miR-210-3p as a diagnostic and prognostic biomarker, we analyzed its levels in tissues and urine specimens collected at surgery and during follow-up visits (from 3 to 12 months) from 21 ccRCC disease-free patients and in a small subgroup of patients presenting metastatic progression.
Methods
Patients and urine samples collection
This study includes a cohort of ccRCC patients (totally 21 patients) who underwent surgical resection between September 2016 and March 2017 (as described in Table 1) at Regina Elena National Cancer Institute of Rome. All subjects gave their informed consent for inclusion before they participated in the study. The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the Ethics Committee of Regina Elena National Cancer Institute (Project identification code: CI IRE 19.11.2012 and 431-13.6.2017).
Clinical characteristics of patients with ccRCC
Clinical characteristics of patients with ccRCC
F: Female; M: Male; R: right; L: left: Bone: Bone matastasis; Intes: Intestinal metastasis; Lung: lung metastasis.
Neoplastic and healthy kidney tissues samples from 13 patients were included. Additional 8 neoplastic samples were considered. Urine specimens were collected at the time of surgery from all the 21 patients and during follow-up when available (from 3 to 12 months, as reported in Table 1). Urine samples were frozen within 30 minutes from collection and stored at
Fresh Frozen samples were homogenized by gentle dissociator (Miltenyi Biotec) in 700
As normalizer we chose the small nucleolar RNA SNORD68 (miScript Primer Assay Hs-SNORD68 # MS00033712) for its low variability expression between fresh tissue samples. To quantify miRNA expr-ession in RT-qPCR, we used a standard curve method; standard curve was performed with serial dilutions of a reference cDNA, obtained from RNA extracted from a tumor sample, to quantify miR-210-3p and SNORD68. Ratio of miR-210-3p over SNORD68 was then used to present the relative expression data.
Total RNA from 200
A Spike-in Control (#219610) (Qiagen, Chatsworth, CA, USA), a synthetic miRNA, C. elegans miR-39 miRNA mimic, to monitor RNA recovery and reverse transcription efficiency. 1.6
All reactions were performed in triplicates. microRNA expression was evaluated on urine samples from the ccRCC cohort and compared to urine samples from healthy donors.
Statistical analysis
All statistical analyses were performed using the GraphPad Prism 6.0 software (GraphPad Software, Inc., USA). The
Results
By using a cohort of fresh frozen renal tissues obtained from 21 ccRCC patients subjected to surgical resection we evaluated the expression level of miR-210-3p. The clinical and demographic details of ccRCC patients and tumor specimens included in this study are summarized in Table 1.
In particular, thirteen patients were males (62%) and 8 patients were females (38%) with a median age of 63 years old (range 43–83) and a median Serum Creatinin concentration of 1.1 mg/dl (range 0.66–1.36). All the cases presented a clear cell histotype of RCC at the histological examination and according to ISUP grade classification, 10 cases (47.6%) were G2 grade, 10 cases (47.6%) were G3 grade and only 1 case (4.7%) was G4 grade. Information about smoking habit, as a main risk factor, was available for 15 patients; among these, 3 patients (20%) were cigarette smokers, 5 patients (33.3%) have a history of smoking and 7 patients (46.6%) were non-smokers (Table 1). The surgery procedures performed as choice treatment for these patients were: Robotic Radical Right Nephrectomy in 1 cases (4.7%), Robotic Partial Right or Left Nephrectomy in 14 cases (66.6%), Open Radical Left Nephrectomy in only 1 case (4.7%) and Laparoscopic Right or Left Nephrectomy in 5 cases (23.8%). All the patients included in the study did not receive any neo-adjuvant therapy before surgery.
Urine specimens were collected at the time of surgery from all the 21 patients and subsequently during follow-up (from 3 to 12 months, as reported in Table 1). Urine samples were also collected from 16 healthy donors with characteristics comparable to the ccRCC patients included in the study (median age: 59.6; males: 50% and females: 50%).
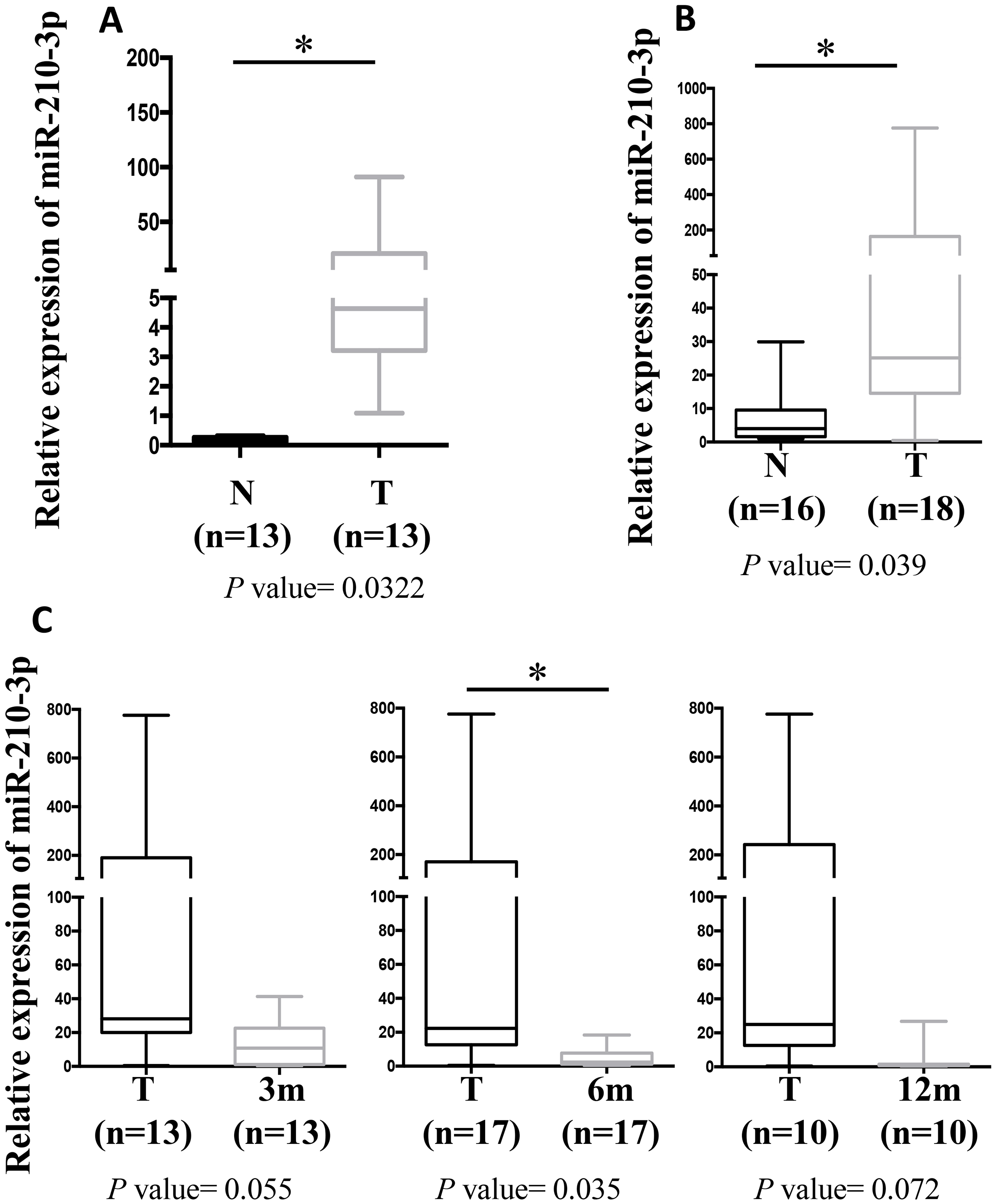
miR-210-3p levels in tissue and urine specimens collected at surgery and during follow-up of ccRCC patients. (A) Box plot showing the modulation of miR-210-3p in a cohort of 21 fresh frozen tissues from ccRCC patients. A total of 13 matched ccRCC tumor (T) and adjacent healthy tissue (N) samples, and additional 8 tumor samples were analyzed by RT-qPCR. The expression value of miR-210-3p was normalized over the SNORD68 expression. 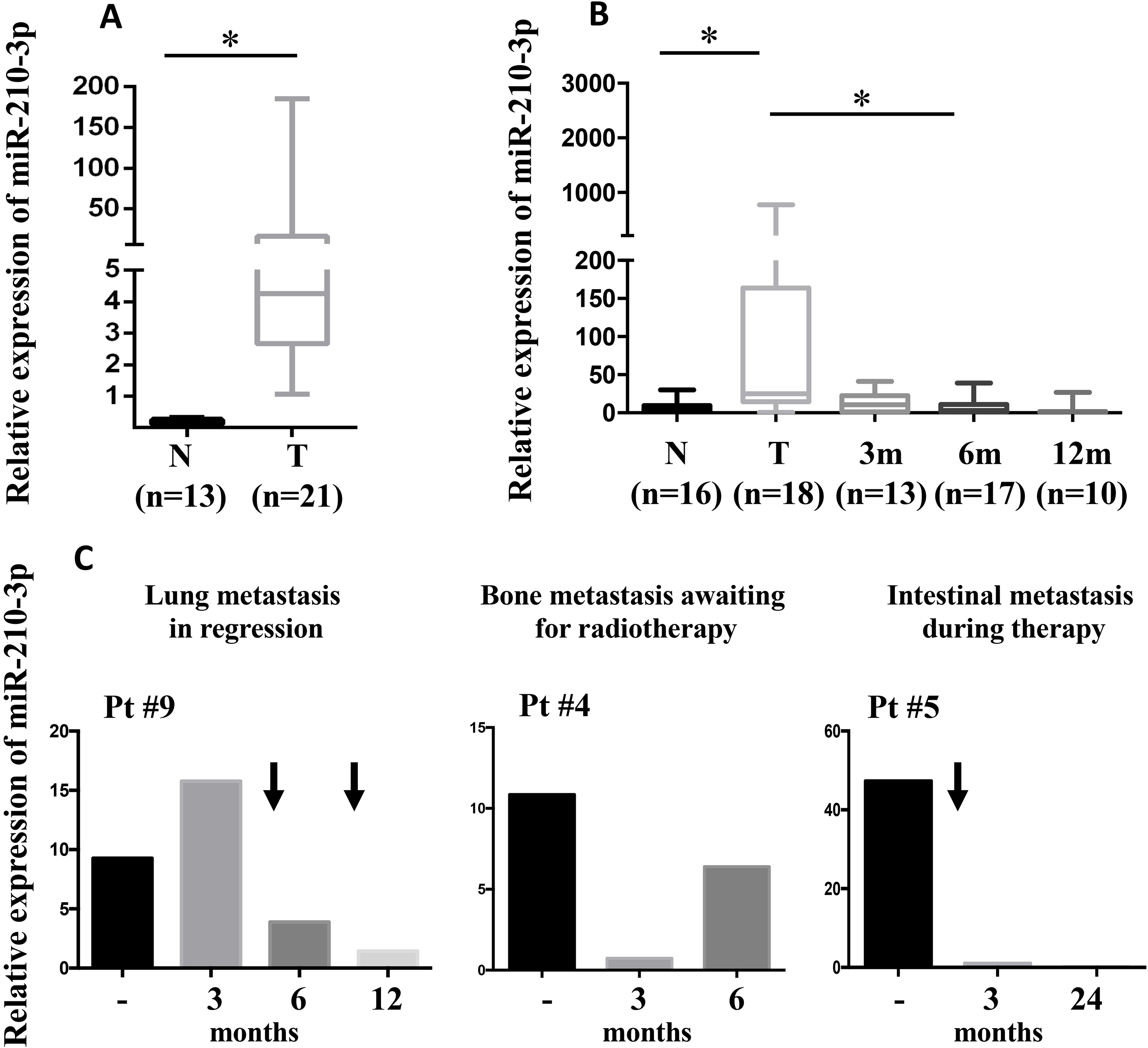
Three patients developed metastasis (to the lungs, bones or intestines). The patient with bilateral lungs metastasis was treated with Sunitinib (Sutent
In agreement with the previous studies [20, 21], we evidenced that miR-210-3p is up-regulated in ccRCC vs adjacent healthy tissue (Fig. 1A and Supplementary Fig. 1A).
To further evaluate the relevance of miR-210-3p as novel low-expensive and non-invasive clinical biomar-ker for ccRCC, we analyzed miR-210-3p expression in 21 urine specimens collected at the time of surgery and during follow-up (1–3, 6 and 12 months of time from surgery) from the same cohort of ccRCC patients (18 disease-free and 3 in metastatic progression); urine specimens from healthy donors with similar characteristics were analyzed as well.
Of note, miR-210-3p was significantly upregulated in urine specimens collected from ccRCC patients at the time of surgery, compared to healthy donor samples (Fig. 1B and and Supplementary Fig. 1B). Importantly, miR-210-3p urinary level was significantly reduced during follow-up in disease-free ccRCC patients of the same cohort, for which follow-up urine samples were available (Fig. 1B and and Supplementary Fig. 1C).
Of note, in a small subgroup of patients presenting metastatic progression to the lungs, bones or intestines, the urine levels of miR-210-3p were down regulated after the treatment (pts #9 and #5) or after surgical resection (pt #4) in comparison to their basaline levels (time of surgert). Patient #4 who was waiting for radiotherapy presented a recovery of high miR-210-3p urine levels (Fig. 1C). However, when a small increase of the omental nodule was evidenced after 24 months from surgery in patient #5 (with intestinal metastasis), the expression levels of miR-210-3p did not correlate with the relapse of the disease.
In this study, we evidenced that at the time of diagnosis the miR-210-3p is up-regulated not only in ccRCC vs matched healthy tissues but also in urine specimens from ccRCC patients compared to healthy donors.
The up-regulation of miR-210-3p in ccRCC tissue was also evidenced by several studies described in the literature [22, 23, 24, 25]. Our study supports and confirms the relevance of this miRNA as molecular diagnostic biomarker for ccRCC evaluation and classification. Moreover, different studies evidenced also the correlation of miR-210-3p expression and different clinical features. In particular, a significant inverse correlation between miR-210 expression and survival in ccRCC tumours was found by Neal et al. [26]. Interestingly, Samaan et al. evidenced that patients with a higher level of miR-210 expression have significantly shorter overall survival than patients with lower miR-210 expression and that miR-210 expression is independent from pathologic stage and tumor size [27]. However, surprisingly, McCormick et al. [22] evidenced the association of miR-210 with good clinico-pathological parameters in ccRCC in terms of a better prognosis and lower grade/stage of tumours.
Our study suggests that the urine level of miR-210-3p was significantly reduced during follow-up samples highlighting this onco-miRNA as a potential biomarker useful not only for diagnosis but also to assess the complete resection and disease-free survival in ccRCC patients. Moreover, in metastatic patients miR-210-3p might represent a useful tool for the evaluation of the treatment response during ccRCC follow-up. Interestingly, these results further support the relevance of miRNAs as potential prognostic tool for urological tumours [15, 17] and the detection of the urine levels of miR-210-3p as biofluid marker for ccRCC management [18, 21].
Dysregulation of miR-210 expression have great relevance in ccRCC pathogenesis. Indeed, this miRNA is not only an hypoximiR, but is also involved in multiple molecular pathways related to different metabolic processes, including autophagy and mitochondrial respiration, linking hypoxia to metabolic reprograming [28]. In this scenario, it is emerging that ccRCC is characterized by reprogramming of metabolism. In particular, the metabolic flux through glycolysis is partitioned [29], and mitochondrial bioenergetics and oxidative phosphorylation are impaired [30]. To date, a several number of metabolic pathways are under investigation, to identify potential therapeutic targets and relevant tools for diagnostic and prognostic biomarkers of ccRCC, including for example glutathione metabolism [31]. Moreover, ccRCC is a tumor highly vascularized and the pathways of VHL/hypoxia – VEGFR in this type of neoplasms are well studied and targeted by actual molecular treatments with monoclonal antibodies such as bevacizumab, or tyrosine kinase inhibitors such as sunitinib or sorafenib [32].
In this context, the characterization of miRNAs contribution to ccRCC establishment and progression could be relevant for the improvement of the clinical management of this tumor.
Conclusions
In conclusion, this study highlights miR-210-3p, which is altered in ccRCC tissues and urine specimens, as non-invasive biomarker for ccRCC management but also suggests this miRNA as an emerging biomarker in the monitoring of tumor progression. Further investigation including larger cohorts of patients will allow to establish the associations existing between miR-210-3p levels and insurgence of metastasis in ccRCC patients.
Footnotes
Acknowledgments
Contribution of Sapienza University of Rome was greatly appreciated.
List of abbreviations
Supplementary data
(A) Box plot showing the modulation of miR-210-3p in fresh frozen tissues from 13 matched samples of the cohort reported in Fig. 1A.