Abstract
BACKGROUND:
MicroRNAs (miRNAs), a group of non-coding post-transcriptional regulators of gene expression, are dysregulated in clear cell renal cell carcinoma (ccRCC) and play an important role in carcinogenesis. Our prior work identified a subset of miRNAs in pT1 ccRCC tumors associated with progression to metastatic disease.
OBJECTIVE:
To investigate the impact of two of these dysregulated miRNA, miR-15a-5p and -26a-5p, in an effort to elucidate the mechanisms underpinning aggressive forms of stage I ccRCC.
METHODS:
The ccRCC cell line 786-O was transfected with pre-miRs-15a-5p and -26a-5p to rescue expression. Cell proliferation was measured via MT Cell Viability Assay. O-GlcNAc-transferase (OGT), a known protein in ccRCC proliferation, was identified by bioinformatics analysis as a target of both miRNA and validated via luciferase reporter assay to confirm binding of each miR to the 3
RESULTS:
Luciferase assay confirmed specificity of miR-15a-5p and -26a-5p for the OGT UTR. Western blot analysis for OGT showed reduced expression following co-transfection of both miRNAs compared to negative control or individual transfection. Co-transfection of these miRNAs greatly reduced proliferation when compared to negative control or the individual transfections.
CONCLUSION:
Our results indicate that the dysregulation of miR-15a-5p and -26a-5p contribute cooperatively to the proliferation of ccRCC through their regulation of OGT. These results give insight into the pathogenesis of aggressive early stage ccRCC and suggest potential therapeutic targets for future research.
Keywords
Introduction
Renal cell carcinoma (RCC) is a heterogeneous group of cancer variants that arise from the renal tubular epithelium [1]. RCC can be subdivided based on pathology (histologic subtypes), genetic, or behavioral characteristics [2]. Of these subgroups, clear cell RCC (ccRCC) is both the most common, representing 75–80% of all RCC, and most overrepresented in metastatic RCC, at about 90% of cases [3]. Traditionally, pathologic stage has been the strongest prognostic indicator for ccRCC progression [4], however recent studies have begun to look at additional features, not traditionally included in staging rubrics, with the goal of improving both prognostic accuracy and tailoring treatment algorithms. As a result of the rise of active surveillance of small tumors during the past decade [5], interest in alternative methods of classifying these tumors to inform clinical management has also risen. One area of particular interest involves pathologic stage I (pT1, N0, Mx) ccRCC. These tumors are associated with favorable patient outcomes; however, the rise of wide-spread cross sectional imaging has created a disproportionate increase in incidence of pT1 RCC compared to other tumor stages [6]. Despite the excellent prognosis for these tumors, with disease specific 5-year survival cited at 81–92%, a small minority of patients will progress to metastatic disease [7]. A prior study from our laboratory addressed this issue by investigating the use of microRNAs (miRNAs) as biomarkers to identify tumors that progress to metastasis [7].
miRNAs are a class of small (
Prior studies have established the prognostic value of miR-15a-5p in a wide variety of cancers, however its role in RCC is in dispute [12]. Specifically, miR-15a-5p has been demonstrated to suppress tumor proliferation in a wide variety of cancer types [13, 14, 15], with downregulation leading to unchecked proliferation. In studies comparing its expression in ccRCC tumors compared to corresponding adjacent normal tissue, reports are conflicting [16, 17].
Numerous studies have linked a downregulation of miR-26a-5p with poor patient prognosis in RCC. Expression levels of miR-26a-5p were lower in RCC tumors of patients who developed tumor relapse [11]. Decreased miR-26a-5p expression was also correlated with aggressiveness in ccRCC tumors [10]. As is seen with miR-15a-5p, miR-26a-5p has also been reported to suppress tumor proliferation in various tumor types, including osteosarcoma and breast cancer [18, 19].
The influence of a particular miRNA at the cellular level is determined by the specific targets and pathways through which they interact. An individual miRNA can interact with the transcripts of multiple potential protein targets, and each protein transcript can be affected by many different miRNAs binding at unique sites [20]. Additionally, multiple miRNA may act on a single target resulting in a cooperative effect, greatly amplifying the impact of each individual miRNA on the target [20]. The role a specific miRNA plays in a disease state can be elucidated by first using algorithmic methods to identify potential protein targets [21] and then using experimental methods to verify those targets [20]. This was the approach we took to examine the impact of miR-15a-5p and miR-26a-5p in ccRCC.
One such target predicted to interact with both of these miRNAs is O-GlcNAc-transferase (OGT) [22, 23, 24], which is an enzyme that catalyzes the addition of an O-Linked
The aim of this study is to identify the potential influence of miR-15a-5p and miR-26a-5p on the proliferation of ccRCC cells, in addition to their potential interaction with OGT. We investigated the link and potential cooperation between these miRNA and OGT in order to better understand the roles of miR-15a-5p and miR-26a-5p in the development of metastatic potential in small stage I ccRCC tumors.
Materials and methods
Cell culture
The human ccRCC line 786-O (ATCC
Prediction of miRNA targets
Target search for predicted protein targets was conducted using a combination of miRcode (
Verification of protein targets in ccRCC
Protein and gene transcript expression levels in ccRCC tumors were investigated using the Human Protein Atlas Pathology database (
Cell transfection of pre-miR constructs
Cells were seeded into CELLSTAR six-well dishes (Greiner, Kremsmunster, Austria) at a density of 2
Luciferase assay
A vector was prepared containing the human OGT 3
Western blot analysis
Cell lysates were prepared from six-well dishes displaying 70–80% confluency, 48 hours after transfection. Cells were lysed in 100
Cell proliferation assay
Cells were seeded into 35 mm dishes (Corning, Corning, NY, USA) at a density of 1.56
Statistical analysis
A one-way repeated measures ANOVA was conducted with post hoc Fisher’s least significant difference tests using GraphPad Prism version 8.0.0 for Mac (GraphPad Software, San Diego, CA, USA,
Results
OGT is overexpressed in ccRCC
OGT expression levels in stage I ccRCC were analyzed using data from the Human Protein Atlas database [27] v19.3 (
High OGT expression in stage I ccRCC tumors is associated with poor patient outcomes. OGT expression and survival data for stage I ccRCC patients available from the Human Protein Atlas v. 19.3 (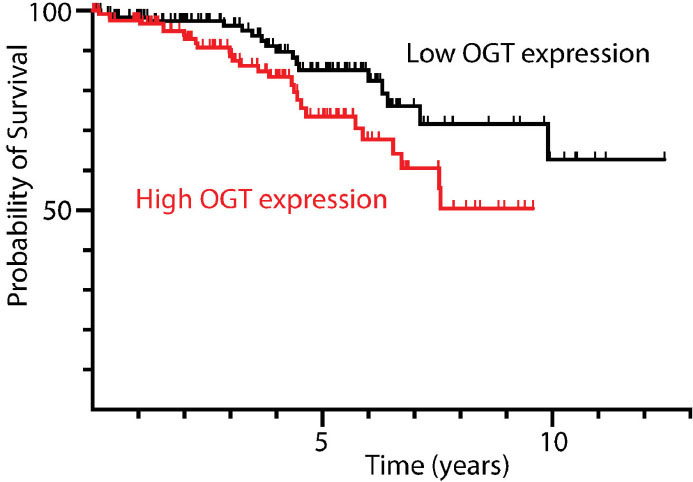
Using the miRNA binding algorithms miRcode [22], miRmap [23], and PicTar [24] we found that miRs-15a-5p and -26a-5p both have high predicted interactions with OGT with good inter-algorithm agreement. The conserved binding sites identified were the 3
Effect of miR-15a-5p and miR-26a-5p transfection on OGT expression and cancer phenotype. (A) Predicted interaction between the human 3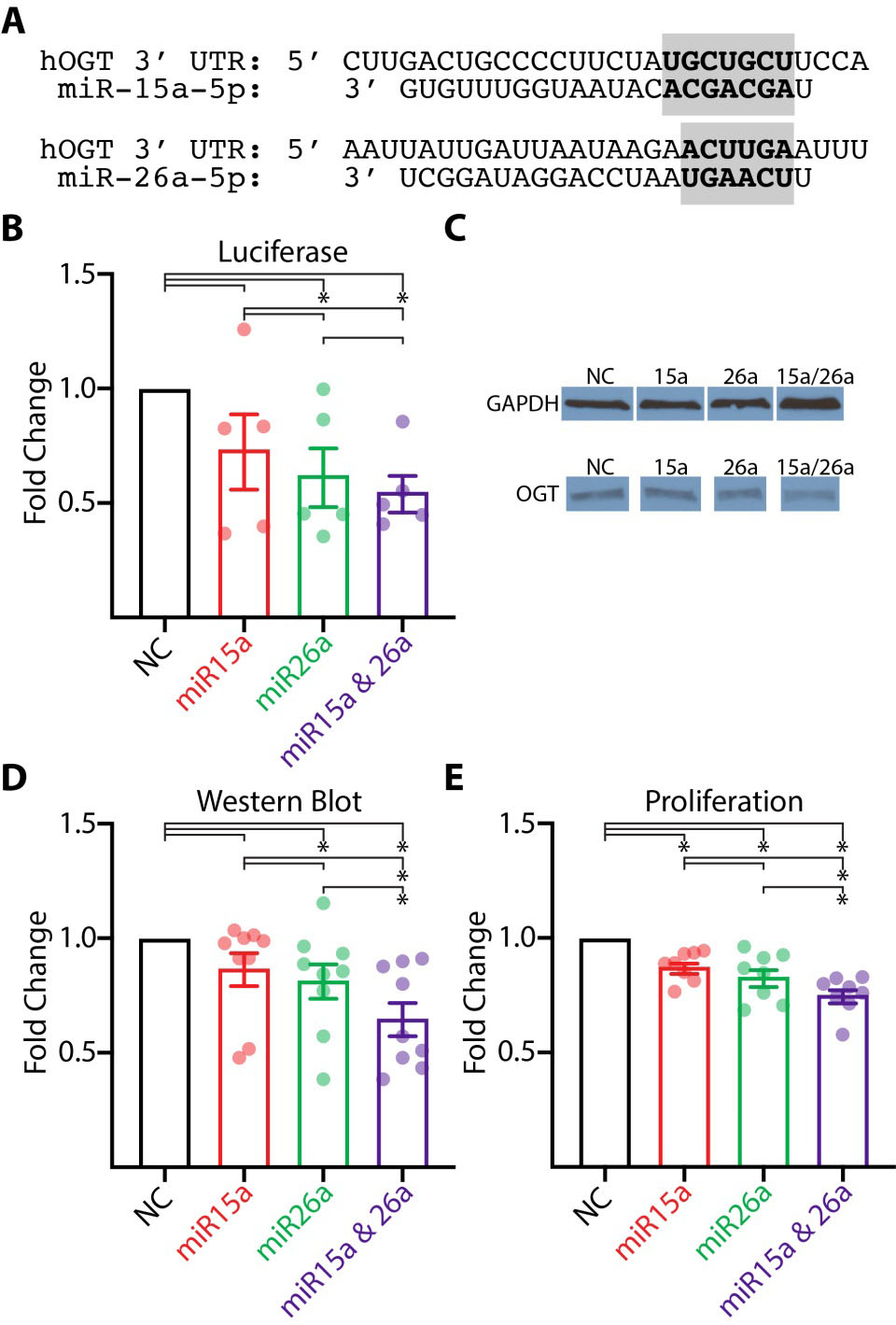
Luciferase, Western blot, and Proliferation statistics. Mean ratios and
miRs-15a-5p and -26a-5p have previously been shown to inhibit cellular proliferation in other cancer types [13, 15, 18]. We performed a cell proliferation assay to assess the impact of these miRNA in ccRCC. Both miRs-15a-5p and -26a-5p significantly inhibited cell proliferation when transfected into 786-O cells compared to negative control (Fig. 2E). Additionally, in cells transfected with both miRs-15a-5p and -26a-5p proliferation was significantly less than cells transfected with either miRNA alone (Fig. 2E). Average proliferation ratios are detailed in Table 1.
Discussion
In order to improve prognostic accuracy for ccRCC patients, many studies have focused on identifying biomarker signatures that correlate with patient outcomes. These investigations cover an array of cellular processes including, but not limited to, cellular proliferation, angiogenesis, chemoresistance, metabolism, and mitochondrial dysfunction [33, 34]. In addition, these biomolecules can be measured in a variety of matrices. For example: Papale et al. found the urinary concentration of RKIP and p-RKIP to be prognostic of patient outcomes in ccRCC patients [35].
Evidence is mounting suggesting that dysregulation of miRNAs can be predictive of patient outcomes as well, playing an important role in cancer pathophysiology. These translational regulators have been linked to altered protein expression of both oncogenic and tumor suppressor proteins. Unsurprisingly, alterations in miRNA expression have also been linked to patient outcomes, presumably through alterations of key cancer related protein expression. Our previous study identified miRs-15a-5p and -26a-5p, among others, as dysregulated miRNA biomarkers in small stage I ccRCC tumors with an aggressive phenotype. These were also shown to have prognostic significance [7]. While each of these miRNA can potentially bind to and silence hundreds of different mRNA transcripts, here we have gained further insight into the tumor suppressor functionality of miR-15a-5p and miR-26a-5p in ccRCC. This study investigated the impact of these miRNAs, individually and in combination, on cell proliferation and on OGT in ccRCC.
We demonstrate a cooperative relationship between miR-15a-5p and miR-26a-5p with respect to proliferation in 786-O cells. Despite the same amount of miRNA added, cells transfected with both pre-miR-15a-5p and pre-miR-26a-5p exhibited a significantly reduced proliferation rate compared to cells transfected with a negative control pre-miR or either miRNA individually. Through luciferase and western blot analysis, we show that both of these miRNA, when transfected together bind to and reduce the expression of OGT in 786-O cells. The same is true of miR-26a-5p when transfected alone. While transfection with miR-15a-5p was unable to significantly decrease OGT expression in these two assays, co-transfection of both miRNA produced an amplified and significant reduction in expression. We hypothesize that the decreased expression of these miRNA in ccRCC leads to an increase in OGT expression, which then contributes to tumor progression. This is supported by OGT upregulation correlating with poorer patient outcomes, even as early as stage I, as seen in the TCGA dataset presented by the Human Protein Atlas. Additionally, OGT protein and mRNA levels were higher in ccRCC tissues compared to that of adjacent normal tissue [28].
Studies on miR-15a-5p and miR-26a-5p have found both miRNA to be downregulated in ccRCC in addition to other cancer types: although previous reports for miR-15a-5p in ccRCC are conflicting [7, 12, 16, 17]. Within the broader miR-15a/16 cluster, miR-15a is characterized by reduced expression in various cancers and plays an important role in tumor progression [37]. In addition to reduced expression, miR-15a-5p has been linked to suppressed proliferation in numerous cancers, such as nasopharyngeal, hepatocellular, and osteosarcoma [13, 14, 15]. Specifically in ccRCC, miR-15a-5p expression has been shown to be reduced in tumor tissue compared to its adjacent normal tissue [16], and our previous study found it to be decreased in aggressive stage I ccRCC [7] as compared to stage I tumors that did not progress to metastatic disease. While the great body of evidence suggests that miR-15a-5p acts as a tumor suppressor, a recent study by Mytsyk et al. found a correlation between the upregulation of this miRNA and poor patient outcomes [12] in ccRCC. While our findings support the former studies, the role of miR-15a-5p in ccRCC is unclear, and we present our findings in anticipation that these results will help to guide future studies in an effort to fully elucidate its oncogenic effects.
As with miR-15a-5p, miR-26a-5p is also downregulated in various cancer types, such as osteosarcoma and breast [18, 19], and its expression level is inversely related to cellular proliferation [18, 19, 38]. Kurozumi et al. identified a tumor suppressive function of miR-26a-5p through its interaction with the proteins LOXL2 and PLOD2 [39]. The mechanism behind the downregulation of miR-26a-5p has been investigated in other cancer types. In prostate cancer, the Myc oncogene binds directly to the miR-26a-5p promoter and negatively regulates expression [40]. As Myc is both activated and upregulated in ccRCC [41], the same mechanism may explain the reduced expression of miR-26a-5p in ccRCC as well. While not as strongly repressed by Myc as miR-26a-5p, there is evidence to suggest that miR-15a-5p expression is also directly reduced by Myc in B cell lymphoma [42]. Future studies are needed to verify these findings in ccRCC, however Myc may be responsible for the expression levels seen in both of our miRNA’s of interest.
Of the abundant potential protein targets from which to choose for each of these miRNA, we chose to focus on OGT, which in addition to its own importance in cancer biology is targeted by both miR-15a-5p and miR-26a-5p [22, 23, 24]. Patient outcomes are significantly worse in ccRCC patients with high OGT expression [27]. OGT, an enzyme that catalyzes the addition of an O-GlcNAc sugar to a target protein [25], acts on a wide array of different protein targets, allowing the protein to have a far-reaching impact on numerous cellular processes [43]. In both prostate and breast cancers, a reduction in cellular O-GlcNAc levels led to G1 cell cycle arrest, inhibiting cellular growth and division [43]. In RCC cells, Wang et al. demonstrated the suppression of cellular proliferation upon OGT knockdown [28]. In addition to its impact on proliferation, OGT has been shown to affect EMT, angiogenesis, and epigenetics [43]. However, while much is understood of the impact of OGT on the cell, the regulation of OGT is not as clear, especially in ccRCC. To our knowledge, no previous study has investigated the regulation of OGT in ccRCC. In breast cancer cells, Sodi et al. demonstrated that the mTOR/Myc axis regulates OGT expression [44]. Because the stability and activity of Myc is dependent on O-GlcNAclyation, there may be a feed-forward regulation mechanism between Myc and OGT [44]. As Myc itself is upregulated in ccRCC [41] and is known to additionally repress the expression of miR-26a-5p and miR-15a-5p in some cancer types, it is possible that these four players may be converging to promote metastatic potential in ccRCC. Because both miR-15a-5p and miR-26a-5p are downregulated and function to attenuate OGT expression, it is plausible that the dysregulation in OGT is a direct result of the dysregulation in these miRNA.
It is important to note that while we present the conservative binding sites predicted by three search engines, there are other potential target sites that were predicted for each miRNA. One limitation of our study was that we did not investigate the validity of the different predicted sites. We present the sites that had the highest prediction score and were the only sites predicted by all three informatics tools. Investigating these potential alternative sites in future work to learn more about why these two miRNA have a cooperative effect with respect to OGT may be worthwhile. Another potential limitation is the use of only one cell line. Evaluation of the influence of miRs-15-5p and -26a-5p on OGT in other ccRCC cell lines in future study may add to the evidence presented here.
In conclusion, the downregulation of miR-15a-5p and miR-26a-5p in aggressive ccRCC tumors is likely at least partially responsible for the upregulation of OGT in these same tumors. We show that these miRNA modulate both cellular proliferation and OGT expression directly in a cooperative manner.
Footnotes
Acknowledgments
R.K. Mellon Family Foundation support to the Lahey Hospital Medical Center Division of Urology.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-200553.
