Abstract
BACKGROUND:
Pancreatic ductal adenocarcinoma is a devastating malignancy with an extremely poor prognosis. Although the most widely used biomarker for pancreatic cancer is carbohydrate antigen CA19-9, it is elevated mainly in the late stage of pancreatic cancer. Some serum natural antibodies against carbohydrates have been shown to be possible diagnostic markers for cancer.
OBJECTIVE:
This study was conducted to determine whether the level of natural antibodies against carbohydrates fluctuates in pancreatic ductal adenocarcinoma.
METHODS:
Serum from pancreatic cancer subjects (
RESULTS:
The level of serum anti-3’-sialyllactose antibodies in pancreatic cancer subjects was significantly lower than that in healthy controls. In contrast, the amounts of serum antibodies against other sialyl glycans were comparable between the two groups. Concentration of serum anti-3’-sialyllactose IgG provided excellent AUC of 0.86, with sensitivity 82%, specificity 81%, and accuracy 82%. The combination of serum anti-3’-sialyllactose IgG with CA19-9 improved the sensitivity of pancreatic cancer detection at an early stage.
CONCLUSIONS:
Natural antibodies against 3’-sialyllactose constitute a promising biomarker for pancreatic cancer detection. The measurement of serum anti-3’-sialyllactose antibodies could play a supportive role in diagnostics and complement the performance of CA19-9 for the early detection of pancreatic ductal adenocarcinoma.
Introduction
Pancreatic ductal adenocarcinoma, which accounts for more than 90% of all pancreatic tumors, is a devastating malignancy with an extremely poor prognosis. Because of the asymptomatic nature of its early stage, and coupled with inadequate methods for early detection, most of the patients present with locally advanced and inoperable disease at the time of diagnosis [1]. Therefore, there is an urgent need to develop accurate markers of pre-invasive pancreatic neoplasms to facilitate prediction of cancer risk and to aid in diagnosis of pancreatic cancer at an early stage [2]. The most widely used serum biomarker for pancreatic cancer is carbohydrate antigen 19-9 (CA19-9), sialyl Lewis
Specific antibodies are commonly monitored for the diagnosis of diseases such as HIV, malaria, autoimmune diseases, and cancers. Immune system success is largely dependent on the presence of several components and well-equipped repertoire of antibodies that are the first line of defense against pathogens and eliminate dysfunctional or malignant cells [9]. Natural antibodies are present in the serum of individuals in the absence of known antigenic stimulation. Human natural antibodies against Gal
In contrast, some natural antibodies are supposed to play a role in surveillance on appearance of aberrant cancer antigens, including carbohydrates [11]. A hexa-saccharide, Globo H, is highly expressed on a various type of cancers, especially breast, prostate, and lung cancers. Wang et al. presented that the amounts of serum antibodies against Globo H from breast cancer patients were significantly higher than those of healthy subjects [12]. CA125 is a high molecular weight, heavily glycosylated transmembrane mucin 16, one of the major carriers of CA19-9 and SLX, and is overexpressed in 80% of ovarian cancer. A previous study showed that the serum CA125 level is increased in ovarian cancer; however, the contents of some anti-sialyl glycan antibodies in serum are significantly decreased in ovarian cancer compared with healthy controls [13].
Since the repertoire of glycan expressed on cell surface undergoes structural alterations with the onset of cancer, the corresponding natural antibody levels can be changed [14]. Although previous studies identified some natural antibodies as diagnostic markers for cancer such as breast, ovarian, and prostate cancers, the involvement of natural antibodies in the detection of pancreatic cancer remains unclear. The current study was conducted to determine whether the level of natural antibodies against pancreatic cancer-associated sialyl oligosaccharides fluctuates. In addition, we examined whether serum anti-3’-sialyllactose antibodies as a biomarker could increase the diagnostic capacity of CA19-9 to detect the early stage of pancreatic cancer.
Materials and methods
Serum
Human sera from healthy (
Demographic data of the subjects
Demographic data of the subjects
Human serum IgG from healthy subjects was purchased from Wako Pure Chemical Industries (Osaka, Japan). Biotinylated 3’-sialyllactose conjugated with polyacrylamide (PAA) was obtained from GlycoTech (Gaithersburg, MD, USA). To prepare human anti-3’-sialyllactose IgG, biotinylated 3’-sialyllactose-PAA and streptavidin-tagged agarose (Thermo Scientific, Waltham, MA, USA) in PBS (-) were incubated for 12 h at 4
Enzyme-linked immunosorbent assay (ELISA)
The wells of 96-well plates (MaxiSorp, Nunc, Denmark) were coated with 10
Glycan array
Glycan polymer (1
Statistical analysis
All statistical calculations were carried out using Prism and InStat software (GraphPad software, La Jolla, CA, USA). The diagnostic ability of serum marker to distinguish between healthy control and pancreatic cancer was determined through cutoffs, derived through analysis of the Area Under the Curve (AUC) using Receiver Operating Characteristic (ROC) curve analysis. Optimal cutoff values for biomarkers were identified using Youden’s J statistic. Linear discriminant analysis was applied to the data using R software (Ver. 3.5.2) with packages MASS. Cross-validation was estimated using a limited number of data sample by the leave-one-out-cross-validation (LOOCV) method. Values are expressed as mean
Diagnostic performance of serum anti-3’-sialyllactose IgG for pancreatic cancer. (A) Both the unbound and bound fractions prepared using a 3’-sialyllactose affinity column were applied to two arrays of 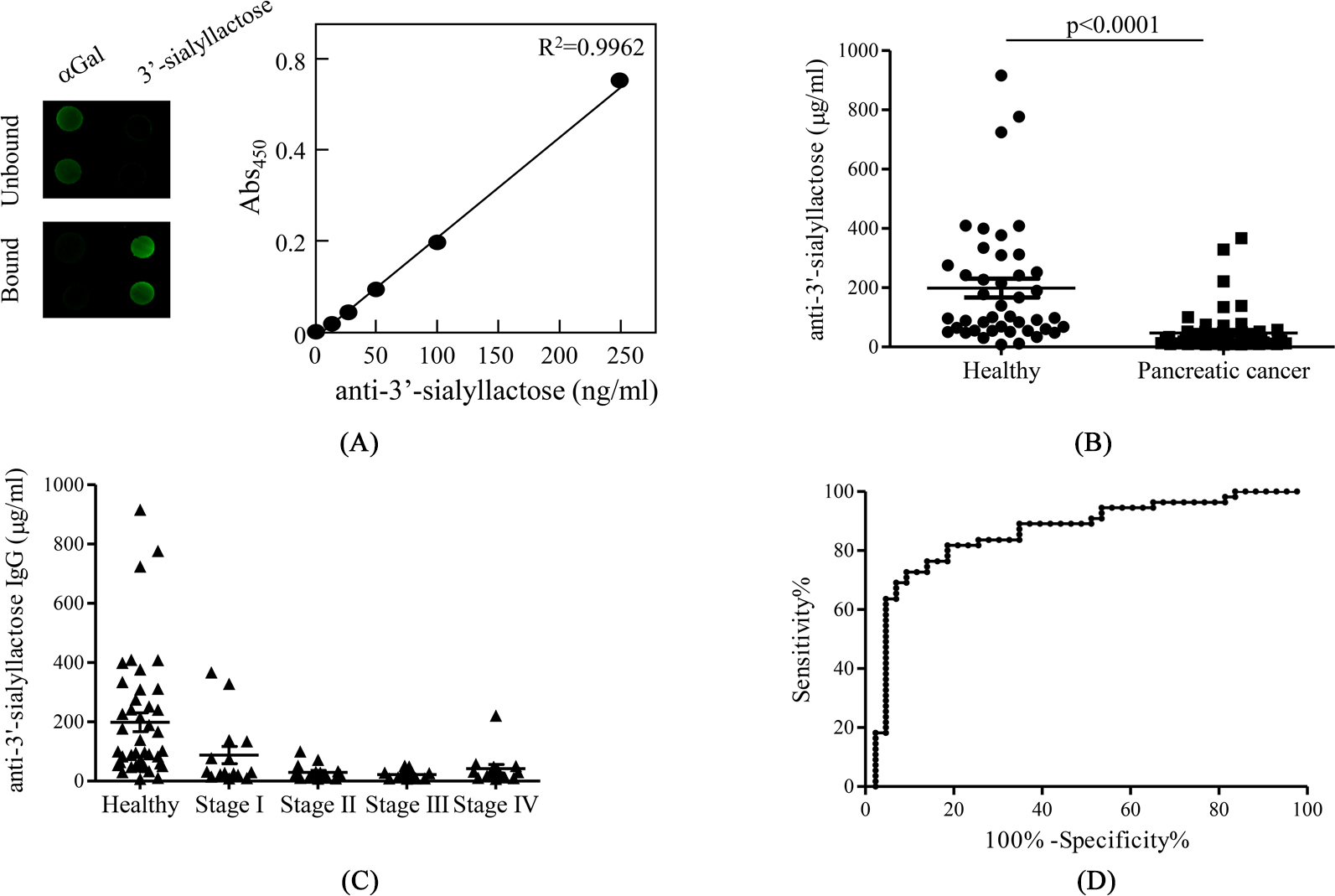
Combined diagnosis of pancreatic cancer at an early stage by serum anti-3’-sialyllactose IgG and CA19-9. (A) The concentration of serum CA19-9 from 43 healthy and 55 pancreatic cancer subjects was determined by commercially available ELISA. (B) Distribution of serum CA19-9 from subjects with different stages of pancreatic cancer. Scatter plots of the level of CA19-9 from healthy (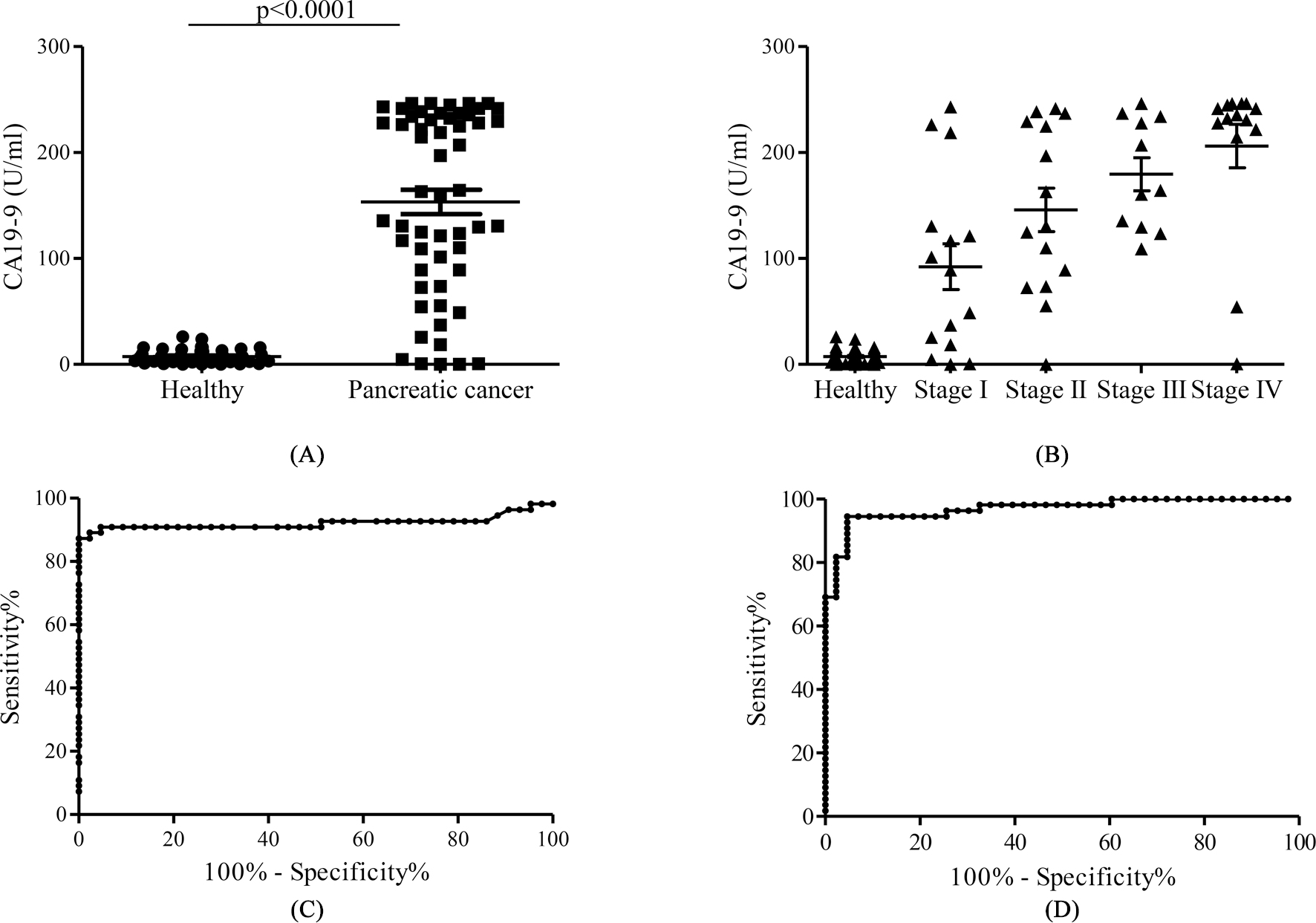
Natural antibodies against pancreatic cancer-associated sialyl glycans
Glycan arrays have emerged as powerful tools for evaluating the interactions between carbohydrates and lectin, whole cells, viruses, or natural antibodies. Using glycan microarrays, previous studies have shown that numerous natural antibodies against oligosaccharides are included in human serum and IgG [15, 16]. To investigate whether the level of natural antibodies against pancreatic cancer-associated sialyl glycans fluctuates, semi-quantitative analysis of anti-sialyl glycan IgG in serum from healthy and pancreatic cancer subjects was performed by ELISA (Supplementary Fig. 1). The concentrations of HRP-conjugated goat anti-human IgG as a detecting antibody were determined necessary to achieve greater linearity from 1:800 to 1:200 dilution of serum (data not shown). Semi-quantitative analysis showed that the content of serum anti-3’-sialyllactose IgG in pancreatic cancer subjects was significantly lower than that in healthy controls, whereas the levels of antibodies against CA19-9, DUPAN-2, SLX, and STN were comparable between the healthy and pancreatic cancer groups(Supplementary Fig. 2).
Diagnostic potential of natural antibodies against 3’-sialyllactose
Previous studies have suggested that human antibodies that bind to 3’-sialyllactose were included in purified IgG [16]. To quantify natural antibodies against 3’-sialyllactose, we first isolated human anti-3’-sialyllactose IgG as a standard using 3’-sialyllactose affinity columns. After purification by affinity, both the unbound and bound fractions were applied to two arrays of
Combined diagnosis for pancreatic cancer at an early stage
The most widely used biomarker for pancreatic cancer is CA19-9; however, CA19-9 is elevated mainly in the late-stage of pancreatic cancer [17]. The results described above led us to raise the possibility that an integrated approach (a combination of serum anti-3’-sialyllactose IgG and CA19-9 tests) would improve the detection of early-stage pancreatic cancer. Serum CA19-9 values from sera of the same individuals as for the anti-3’-sialyllactose IgG test were calculated using the commercially available ELISA kit. Consistent with previous studies, the concentration of serum CA19-9 in pancreatic cancer subjects was significantly elevated compared with healthy controls (Fig. 2A). In addition, the stage-dependent increases in the CA19-9 content were observed (Fig. 2B). The CA19-9 value exhibited an AUC of 0.92 (95% confidence interval 0.86–0.99,
We next explored the possibility that combining anti-3’-sialyllactose IgG and CA19-9 tests would improve diagnostic performance for the early stage of pancreatic cancer. When all data from healthy and pancreatic cancer subjects were plotted against each other, the values were not totally correlated(Supplementary Fig. 4). Similarly, data from pancreatic cancer subjects at stage I were plotted against anti-3’-sialyllactose IgG and CA19-9, and each cutoff value was applied. As shown in Supplementary Figs 5 and 6 out of 15 subjects were diagnosed as having cancer by both tests (solid circle), and 3 or 5 were determined as the presence of cancer by only anti-3’-sialyllactose IgG or CA19-9 test, respectively (open circle). However, 1 out of 15 pancreatic cancer subjects at stage I was diagnosed as a non-cancerous subject by both tests (closed square). In addition, linear discriminant analysis produced the following predictive equations for pancreatic cancer: Composite biomarker
Discussion
Pancreatic cancer is one of the most aggressive and lethal malignancies worldwide, with a very poor prognosis and a five-year survival rate of less than 8%. This dismal outcome is largely due to delayed diagnosis, early distant metastasis, and resistance to conventional chemotherapies [18]. CA19-9 is a serum marker measured in the clinical management of pancreatic cancer; however, CA19-9 is not overexpressed in the early stage of pancreatic cancer [17]. In addition, CA19-9 expression is not elevated in patients who lack the Lewis antigen, even if they have a large pancreatic tumor [19]. Accordingly, the guidelines of the American Society of Clinical Oncology do not recommend the measurement of CA19-9 as a screening test for gastrointestinal cancers, particularly pancreatic cancer [20]. On the other hand, aberrant expression of
In the present study, the content of natural antibodies against the sialyl glycans were measured and found that the level of serum anti-3’-sialyllactose IgG in pancreatic cancer subjects was markedly reduced compared with healthy controls (Fig. 1B). In addition, we demonstrated that the concentration of serum anti-3’-sialyllactose IgG serves as a novel diagnostic marker for pancreatic cancer (Fig. 1D). The CA19-9 test exhibited high sensitivity; however, the cancer of 4 out of 15 pancreatic cancer subjects at stage I failed to be detected in this study. To more sensitively detect the early stage of pancreatic cancer, we examined a combination assay based on the determination of anti-3’-sialyllactose IgG and CA19-9. As expected, the combination assay enabled detection of stage I pancreatic cancer with high sensitivity (93.3%), and the anti-3’-sialyllactose IgG test detected 3 out of 4 subjects with clinical stage I pancreatic cancer in which no increase in CA19-9 was observed (Fig. 2D). In addition, 1 out of 15 pancreatic cancer subjects at stage II was predicated to be non-cancerous in the CA19-9 test, but properly diagnosed as a pancreatic cancer subject in the 3’-sialyllactose IgG test (data not shown). Thus, these results indicated that the combination of anti-3’-sialyllactose IgG with CA19-9 test increases the sensitivity of pancreatic cancer detection at an early stage.
Serum carbohydrates have been studied extensively as potential cancer biomarkers; however, many are restricted to the tumor site and levels that are often proportional to the size of tumor, which is not ideal for early detection. In contrast, a small tumor can potentially give rise to large changes in antibody levels, and these antibodies can be easily detected in biofluids such as serum [14]. We have previously proposed novel methods that facilitate the discovery of carbohydrate-binding molecules as disease markers using glycan arrays [21]. Since previous studies have shown that numerous natural antibodies against oligosaccharides are included in human serum, we determined the levels of serum natural antibodies against 50 species of glycans in addition to sialyl glycans used in this study using glycan arrays, and concluded that the concentration of serum anti-3’-sialyllactose antibodies can robustly distinguish pancreatic cancer subjects from healthy subjects (data not shown). In addition, the diagnostic capacity of anti-3’-sialyllactose IgG to detect pancreatic cancer was superior to that of IgM class (data not shown). Since serum CA19-9 is also elevated in benign diseases of the pancreas and in other malignancies of gastrointestinal tract, we preliminarily evaluated the specificity of anti-3’-sialyllactose antibodies using sera from several cancers and chronic pancreatitis, and obtained prospective results on the clinical availability as a pancreatic cancer marker (data not shown). Collectively, natural antibodies as a serum biomarker for pancreatic cancer are presented here for the first time. In addition, the measurement of serum anti-3’-sialyllactose IgG could play a supportive role in diagnostics, complementing the defects in CA19-9 determination. To apply current evidence to the diagnosis for pancreatic cancer, the findings from large-scale clinical trials of interventions would be the most relevant.
Natural antibodies are supposed to play a role in surveillance on appearance of aberrant cancer antigen [14].
In conclusion, we determined the amounts of natural antibodies that exist against numerous oligosaccharides and demonstrated that serum anti-3’-sialyllactose IgG is a promising biomarker for pancreatic cancer detection. In addition, we observed that the level of serum anti-3’-sialyllactose IgG could improve the diagnostic performance of CA19-9 for the early stage of pancreatic cancer. Future clinical trials will validate the possibility of serum anti-3’-sialyllactose IgG as a promising biomarker for the early detection of pancreatic cancer.
Footnotes
Conflict of interest
K. Higashi, H. Nagahori and K. Saito are employees of Sumitomo Chemical Co. Ltd. Osaka Ohtani University and Sumitomo Chemical Co. Ltd. applied for a patent to use serum anti-3’-sialyllactose antibodies as a biomarker for pancreatic cancer (WO2018–143336).
Supplementary data
The supplementary files are available to download from
