Abstract
We previously demonstrated that the carbohydrate 3′-sialyllactose is overexpressed in cancer stem-like cells such as metastatic pancreatic and poorly differentiated gastric cancer cells, and undifferentiated human embryonic stem cells. In this study, we investigated the possibility of 3′-sialyllactose as a target for theranostics in cancers using a recombinant mouse monoclonal antibody r3B1E2 that binds to 3′-sialyllactose. Immunohistochemistry analysis confirmed an elevated expression of 3′-sialyllactose in tumors of pancreas, stomach, and testis, while no expression of 3′-sialyllactose was observed in corresponding normal controls. In addition, a stage-independent expression of 3′-sialyllactose was observed, especially in pancreatic ductal adenocarcinoma (PDAC). The level of serum 3′-sialyllactose in PDAC subjects was significantly higher than that in healthy controls, providing excellent AUC of 0.88. We next explored the therapeutic potential of r3B1E2 for PDAC
Introduction
Pancreatic ductal adenocarcinoma (PDAC), which accounts for more than 90% of all pancreatic tumors, is a devastating malignancy with an extremely poor prognosis. Because of the asymptomatic nature of its early stage, and coupled with inadequate methods for early detection, most of the patients present with locally advanced and inoperable disease at the time of diagnosis. 1 Therefore, there is an urgent need to develop accurate markers of pre-invasive pancreatic neoplasms to facilitate prediction of cancer risk and to aid in diagnosis of pancreatic cancer at an early stage. 2 We recently determined the amounts of natural antibodies that exist against numerous oligosaccharides and demonstrated that serum anti-3′-sialyllactose immunoglobulin G (IgG) is a promising biomarker for detection of PDAC. 3 In addition, we demonstrated that the level of serum anti-3′-sialyllactose IgG could improve the diagnostic performance of CA19-9, carbohydrate sialyl Lewisa, for the early stage of PDAC. 3
CA19-9 was first identified using a mouse monoclonal antibody raised against human colorectal adenocarcinoma cell line and has been used as a serum marker in the clinical management of pancreatic cancer. 4 However, Sawada et al. discovered a fully human monoclonal antibody, 5B1, that was generated from blood lymphocytes from breast cancer patients immunized with sialyl Lewisa vaccine, and showed that injections of 5B1 significantly improved survival in a mouse xenograft model using sialyl Lewisa-bearing tumor cells. 5 The results from recent clinical trials of radiolabeled 5B1 have also suggested sialyl Lewisa has potential as a theranostic target in PDAC. 6
Cancer stem cells are the malignant counterparts of normal adult tissue stem cells, and they are thought to be responsible for cancer initiation, progression, metastasis, recurrence, and drug resistance. 7 Lacoste et al. 8 showed that highly tumorigenic hepatocellular carcinoma cell line exhibits cancer stem cell–like properties. Since several cell-surface embryonic stem cell (ESC) markers have been reported to overlap with those of cancer stem cells, we previously generated a panel of monoclonal antibodies against undifferentiated human ESCs, and found that the carbohydrate 3′-sialyllactose is overexpressed in cancer stem–like cells such as metastatic or poorly differentiated cancer cells and undifferentiated human ESCs. 9 In this study, we investigated the possibility of 3′-sialyllactose as a target for theranostics in cancers using a recombinant mouse monoclonal antibody, r3B1E2 (IgG2a, κ), that binds to 3′-sialyllactose.
Materials and methods
Cell cultures and reagents
Human pancreatic cancer cell lines were obtained from the Japanese Collection of Research Bioresources Cell Bank (Osaka, Japan), and maintained in accordance with the supplier’s protocol. Human sera from healthy (
Gene analysis and recombinant monoclonal antibody
The immunoglobulin M (IgM) heavy and light chain gene products of hybridoma 3B1E2 were cloned into a pGEM-T Easy vector (Promega), and nucleotide sequences were analyzed. The variable regions of heavy and light chain nucleotides were inserted into the mammalian expression plasmid pUC19-CAG-HC (for mouse IgG2a) and pUC19-CAG-LC (for mouse κ), respectively. A recombinant monoclonal antibody, r3B1E2, was prepared from culture supernatant of HEK293 cells transfected with equal amounts of paired heavy and light chain plasmids, as previously described. 10
Enzyme-linked immunosorbent assay
The wells of 96-well plates (MaxiSorp, Nunc, Roskilde, Denmark) were coated with 10 μg/mL of r3B1E2 at 4°C, and the plates were left overnight. The sera were diluted 100- to 400-fold. The wells were blocked with 5% bovine serum albumin (BSA), the diluted sera were added to the wells, the plates were incubated for 1 h at room temperature, and then incubated with horseradish peroxidase (HRP)-conjugated 3B1E2 for 1 h at room temperature. After washing, peroxidase substrate and stop solution were added to the wells, and then the optical density (OD) at 450 nm was measured. The final OD450 was determined by subtracting the negative control OD450 reading from subject sample OD450 reading.
Immunohistochemistry
The high-density multiple organ tumor and normal tissue microarray, containing 20 tumor types and normal corresponding controls, was immunostained with r3B1E2, as previously described. 11 Briefly, sections were treated with 0.3% hydrogen peroxide, and antigen activation by microwave was performed. After blocking, sections were incubated with 10 µg/mL of r3B1E2 or control mouse IgG at room temperature for 2 h, treated with Histofine Simple Stain MAX PO (Nichirei Biosciences, Tokyo, Japan) at room temperature for 30 min, and then visualized with 3,3′-diaminobenzidine and hematoxylin.
Immunofluorescence
Pancreatic cancer cells were first fixed with 4% paraformaldehyde, incubated with 10 µg/mL of primary antibody at room temperature for 1 h, and then visualized using a 1:1000 dilution of secondary antibody conjugated with Alexa Fluor (Molecular Probes, Eugene, OR, USA) at room temperature for 1 h.
Western blot
Whole cell extracts or culture media were resolved by 7.5% sodium dodecyl sulfate (SDS)/polyacrylamide gel electrophoresis (PAGE) at constant voltage (150 V) for 1 h, transferred to a polyvinylidene difluoride (PVDF) membrane at constant current (250 mA) for 1 h, blocked with 5% BSA for 30 min at room temperature, incubated with 1 µg/mL of r3B1E2 at room temperature for 1 h, incubated with a 1:1000 dilution of anti-mouse IgG conjugated with HRP at room temperature for 1 h, and then the proteins of interest on immunoblots were detected using an enhanced chemiluminescence detection system.
Complement-dependent cytotoxicity assay
3′-Sialyllactose-positive (KP-3L) and -negative (BxPC3) cell lines were used. Target cells (2 × 104) were treated with r3B1E2 or control IgG for 30 min followed by incubation with 25% rabbit complement for another 4 h at 37°C, and then lactate dehydrogenase (LDH) activity in the culture supernatant was measured using a cytotoxicity detection kit. In addition, the number of viable cells was calculated using CellTiter-Glo Luminescent Cell Viability Assay. Positive control samples that had received 5% Triton X-100 were used to determine maximal cytotoxicity, and samples receiving complement alone served as baseline. The complement-dependent cytotoxicity (CDC) activity (%) determined by LDH release was calculated according to the following formula
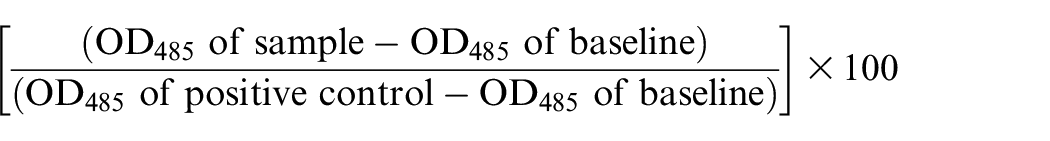
The CDC activity (%) determined by viable cell number was calculated according to the following formula

Statistical analysis
The diagnostic ability of serum marker to distinguish between healthy control and PDAC was determined through cutoffs, derived through analysis of the area under the curve (AUC) using receiver operating characteristic (ROC) curve analysis. Optimal cutoff values for biomarkers were identified using Youden’s J statistic. Values are expressed as mean ± standard deviation (SD). Student’s
Results
Genetic analyses of 3B1E2
We previously generated a panel of mouse monoclonal antibodies against undifferentiated hESCs, and showed that one of the obtained monoclonal antibodies, MAb7, recognized pancreatic and gastric cancer cells. 9 In addition, MAb7 antigen expression levels were increased in metastatic and poorly differentiated cancer cell lines. 9 Analyses of MAb7 antigen indicated that carbohydrate 3′-sialyllactose was critical for MAb7 binding in cancer cells. 9 However, a mouse monoclonal antibody, 3B1E2 (IgM, κ), from another hybridoma reacted with the whole cell lysate and a culture medium prepared from human pancreatic cancer cells, and also binds to 3′-sialyllactose (Supplementary Figure 1). To further characterize 3B1E2, we isolated the antibody gene products and identified the amino acid sequences of variable regions of a heavy (Figure 1(a)) and a light chain (Figure 1(b)). The variable domains of each chain contain three hypervariable loops, named complementarity-determining regions (CDRs), and the length and composition of the CDR sequences are highly variable, especially in CDR3. 12 As shown in Figure 1(a) and (b), 3B1E2 antibody heavy and light chains contained 13 and 9 amino acids in CDR3 loops, respectively.

Amino acid sequence of heavy and light chain variable regions of 3B1E2. (a, b) The complementarity-determining regions (CDRs) of heavy (a) and light (b) chain variable regions of 3B1E2 were determined based on the computer program IMGT/V-QUEST. Amino acid sequences of CDRs are underlined. (c, d) Comparison of amino acid sequences of CDRs in heavy (c) and light (d) chains. The program IMGT/V-QUEST was used to compare the sequences with its reference directory that contains the mouse germline immunoglobulin genes, and the junctions were identified by IMGT/Junction analysis. The identical amino acids of 3B1E2 to germline immunoglobulin are indicated by asterisks.
The software program IMGT/V-QUEST was used to compare the sequences with its reference directory that contains the mouse germline immunoglobulin genes, and the junctions were identified by IMGT/Junction analysis. In the case of the heavy chain, we found that 3B1E2 was derived from the germline IGHV4-1*01; however, the CDR3 amino acid sequence of 3B1E2 exhibited the lowest homology among CDRs, with IGHV4-1*01 sharing 23% identity (Figure 1(c)). In contrast, the amino acid sequences of CDRs between 3B1E2 and MAb7 were totally different (Figure 1(c)). However, the results of IMGT/V-QUEST analysis suggested that the light chain of 3B1E2 was from IGKV16-104*01 with the identical amino acid sequences of CDR1 and CDR2 (Figure 1(d)). By contrast, the amino acid sequences of 3B1E2 in CDRs were entirely distinct from those of MAb7 (Figure 1(d)).
Diagnostic potential of r3B1E2 for PDAC by immunohistochemical examination
To explore the application of 3B1E2 for cancer diagnosis and therapy, we prepared r3B1E2 (IgG2a, κ) using variable regions of heavy and light chain nucleotides of 3B1E2 in a mammalian expression system. As expected, r3B1E2 reacted with human pancreatic and gastric cancer cells and also bound to 3′-sialyllactose, suggesting the conserved antigen recognition (data not shown). Immunohistochemical examination using the high-density multiple tissue microarray was performed to define the presence of 3′-sialyllactose in tumors. To this end, the sections were stained with r3B1E2 or control IgG. As shown in Figure 2(a), a remarkable expression of 3′-sialyllactose was observed in tumors of pancreas (adenocarcinoma) and stomach (adenocarcinoma). Consistent with our previous finding, 3′-sialyllactose was also expressed in embryonal carcinoma (Figure 2(a)). In contrast, no expression of 3′-sialyllactose was detected in normal corresponding controls under the same experimental conditions (data not shown). Specimens stained with control IgG were omitted, and the frequency of 3′-sialyllactose expression in each type of cancer was calculated. As shown in Figure 2(b), 3′-sialyllactose was overexpressed with high frequency in pancreatic adenocarcinoma. Interestingly, a stage-independent expression of 3′-sialyllactose in tumors was shown (Figure 2(b)). However, no significant expression of 3′-sialyllactose was observed in tumors of breast, liver, prostate, colon, head and neck, oral cavity, kidney, uterus, soft tissue, esophagus, lung, brain, bladder, lymph node, or cervix, suggesting a cancer type–specific expression of 3′-sialyllactose (data not shown).

Cancer type–specific expression of 3′-sialyllactose. (a) Representative immunostained tumor sections of pancreas, stomach, and testis by control IgG or r3B1E2 are shown. Areas of the immunostained sections of upper panel (
Diagnostic ability of r3B1E2 for PDAC by serological test
The results described above led us to test the possibility of 3′-sialyllactose as a serum biomarker in PDAC. For this purpose, we constructed a sandwich enzyme-linked immunosorbent assay (ELISA) system using r3B1E2 as a capture antibody and HRP-labeled 3B1E2 as a detection antibody. As shown in Figure 3(a), the optical density at 450 nm (OD450) exhibited high linearity from 400-fold to 100-fold diluted serum. Under these experimental conditions, the content of 3′-sialyllactose in serum from 13 healthy and 55 PDAC subjects was determined. As shown in Figure 3(b), the level of serum 3′-sialyllactose in PDAC patients was significantly higher than that in healthy controls. In addition, a stage-dependent increase of serum 3′-sialyllactose content was also observed (Figure 3(c)). The discriminatory capacity of serum levels of 3′-sialyllactose to detect PDAC was profiled using ROC curve analysis. As shown in Figure 3(d), the concentration of serum 3B1E2 antigen provided an excellent AUC of 0.88 (95% confidence interval 0.79–0.98,

Diagnostic performance of serum 3′-sialyllactose for PDAC. (a) The serum concentration–dependent increase of 3′-sialyllactose content was analyzed by ELISA. (b) The contents of serum 3′-sialyllactose from 13 healthy and 55 PDAC subjects were determined by ELISA. (c) Distribution of serum 3′-sialyllactose from subjects with different stages of PDAC is shown. Scatter plots of the level of serum 3′-sialyllactose from healthy individuals (
Therapeutic potential of r3B1E2 for PDAC in vitro
According to the results of a previous study that demonstrated the potential of sialyl Lewisa as a target for immune attack, 5 we examined whether r3B1E2 exerts the cytotoxicity against the 3′-sialyllactose-expressing pancreatic cancer cells. To find pancreatic cancer cells that express 3′-sialyllactose, we screened the commercially available pancreatic cancer cell lines. As shown in Figure 4(a), the expression of 3′-sialyllactose was observed only in human PDAC cell line KP-3 and its liver-metastatic counterparts (KP-3L) by western blotting. Interestingly, there was no detectable expression of 3′-sialyllactose in BxPC3 cells that bear sialyl Lewisa (Figure 4(a)). In addition, strong staining of 3′-sialyllactose on plasma membrane of KP-3L cells was observed by immunofluorescence, whereas 3′-sialyllactose expression was not observed in BxPC3 cells (Figure 4(b)).

CDC activity of r3B1E2 against 3′-sialyllactose-bearing human PDAC cells. (a) 50 µg of lysates from human pancreatic cancer cells were immunoblotted with r3B1E2. (b) KP-3L and BxPC3 cells were immunostained with r3B1E2 (
To evaluate the functional properties of r3B1E2, we measured the cytotoxic activity, determined by LDH release or viable cell number, of r3B1E2 against KP-3L and BxPC3 cells in the presence of rabbit complement. As shown in Figure 4(c), exposure of KP-3L cells to r3B1E2 significantly exhibited the cytotoxicity determined by LDH release (
Discussion
Theranostics describes a close connection between diagnosis and consequent therapy. The principle of theranostics has acquired greater importance in personalized medicine in recent years, particularly in oncology, where advanced tumors can be treated effectively with low side effects. Molecular alterations in malignant disease result in the expression or upregulation of various targets, and thus monoclonal antibodies that bind to these targets can be used for imaging and treatment of cancers. 6 For cancer diagnosis, monoclonal antibodies labeled with a radioisotope, fluorescent dye, or photoacoustic probe have the potential to advance medical care. In addition, radioisotope-, cytotoxic agent-, or near infrared probe-conjugated monoclonal antibodies are of great interest for therapy of cancers.
It is well known that altered glycosylation is a hallmark of malignant transformation. In pancreatic cancer, a few markers targeting aberrantly glycosylated proteins have been exploited for theranostic applications. 13 The roles of glycans in cancer have been highlighted by the fact that alterations in glycosylation regulate the development and progression of cancer, serving as important biomarkers and providing a set of specific targets for therapeutic intervention. 14 The carbohydrate sialyl Lewisa is a well-established tumor marker for pancreatic cancer. Although sialyl Lewisa-carrying proteins are secreted into blood, it is still an interesting target structure for molecular imaging and therapy, as the concentration is significantly higher in tumor tissue than in serum, and monoclonal antibodies against sialyl Lewisa can therefore accumulate in the tumor tissue. 15 Recent developments in nuclear theranostics targeting sialyl Lewisa have already entered the clinical study phase. In this study, we showed a significant increase of 3′-sialyllactose in pancreatic tumor tissues and sera of PDAC patients. In addition, a monoclonal antibody that binds 3′-sialyllactose exhibited a cytotoxic activity only against 3′-sialyllactose-expressing cells. Further in vivo studies are necessary to confirm the beneficial effects of monoclonal antibodies against 3′-sialyllactose for theranostics in PDAC.
Resistance to chemotherapy is a major clinical problem in cancer medicine, and its attenuation is crucial for reduction of disease relapse and progression. The current knowledge of mechanisms that promote or enable drug resistance, such as drug inactivation, drug target alteration, drug efflux, DNA damage repair, cell death inhibition, and the epithelial–mesenchymal transition, as well as how inherent tumor cell heterogeneity plays a role in drug resistance. 16 A defining hallmark of tumor phenotypes is uncontrolled cell proliferation, while fermentative glycolysis has long been considered as one of the major metabolic pathways that allows energy production and provides intermediates for the anabolic growth of cancer cells. 17 Gemcitabine has been used as a first-line treatment alone for pancreatic cancer; however, a frequent occurrence of gemcitabine resistance has been observed. There are various molecular mechanisms that lead to chemoresistance, and in a general view, many of those have been linked to a stemness-associated survival phenotype. 18 Cancer stem cells have been characterized in terms of their self-renewal capacity, pluripotency, resistance to chemotherapy, and their ability to metastasize to distant organs. Thus, cancer stem cells are considered as the major chemoresistance fraction in tumor cells, which survive different types of treatment and give rise to tumor recurrence and finally progression toward metastasis. 19 In this study, we showed an enhanced expression of 3′-sialyllactose in tumors of embryonal carcinoma (Figure 2(a)), consistent with our previous finding that 3′-sialyllactose marks undifferentiated human ESCs. 9 In addition, we previously described an elevated expression of 3′-sialyllactose in cancer stem–like cells compared with their counterparts, and suggested that carbohydrate 3′-sialyllactose plays a pivotal role in cancer metastasis. 9 To assess the correlation between 3′-sialyllactose expression level and gemcitabine resistance, we performed cell viability assays using several pancreatic cancer cell lines. Interestingly, the resistance to gemcitabine was dependent on the expression level of 3′-sialyllactose (Figure 4(a) and Supplementary Figure 2). Since the frequent expressions of 3′-sialyllactose in tumor tissues at an early stage of cancers were evident (Figure 2(b)), these results raise the possibility of 3′-sialyllactose as a biomarker for the prediction and diagnosis of drug resistance and prognosis. The precise mechanisms by which the aberrant expression of 3′-sialyllactose might exhibit stemness behaviors in cancers continue to be topics for investigation and still remain to be elucidated.
Understanding the molecular basis of antigen binding and recognition by antibodies helps to facilitate the production of better and more potent antibodies for diagnosis, therapy, and various other applications.20,21 The variable domains of antibody heavy and light chains of antibodies contain three hypervariable loops, named CDRs, and together six CDR loops form the antigen binding site. CDR3 of the heavy chain often forms the most significant contact with antigen, is longer than the other CDRs, and usually plays a prominent role in antigen binding. 22 Although both 3B1E2 and MAb7 bind to 3′-sialyllactose and KP-3L cells, the significant differences in amino acid sequence and length of CDRs, especially in CDR3 of the heavy chain, were revealed (Figure 1(c) and (d)). The results from immunohistochemistry using the multiple tissue microarray implied that 3B1E2 antigen differs from that of MAb7 (data not shown). We previously identified MAb7 antigen as 3′-sialyllactose on deleted in malignant brain tumors 1 (DMBT 1), 9 suggesting that 3B1E2 might recognize 3′-sialyllactose on another carrier protein. Identification of a 3′-sialyllactose-carrying protein would enable us to improve diagnostic capacity of 3B1E2 for PDAC.
In summary, we have demonstrated that the expression of 3′-sialyllactose is increased in pancreatic tumor tissues and sera of PDAC patients. Treatment of 3′-sialyllactose-bearing cells with a monoclonal antibody against 3′-sialyllactose exerts the cytotoxicity. These findings raise the possibility of 3′-sialyllactose as a novel target for theranostics in PDAC.
Supplemental Material
revised_figure_Higashi_et_al – Supplemental material for Carbohydrate 3′-sialyllactose as a novel target for theranostics in pancreatic ductal adenocarcinoma
Supplemental material, revised_figure_Higashi_et_al for Carbohydrate 3′-sialyllactose as a novel target for theranostics in pancreatic ductal adenocarcinoma by Kiyoshi Higashi, Keiko Maeda, Kaori Miyata, Saori Yoshimura, Keita Yamada, Daijiro Konno, Taro Tachibana and Koichi Saito in Tumor Biology
Footnotes
Author contributions
K.H., T.T., and K.S. conceived and supervised the study. K.H., K.M., and D.K. designed experiments and analyzed data. K.H., K.M., and S.Y. performed experiments. K.H. wrote the manuscript. K.M., K.Y., D.K., T.T., and K.S. provided editorial input to the manuscript. All authors approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: K.H., K.M., K.M., and K.S. are the employees of Sumitomo Chemical Co., Ltd., which has applied for a patent for monoclonal antibody 3B1E2. S.Y., D.K., and T.T. are the employees of Cell Engineering Corporation.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This collaborative study between Sumitomo Chemical Co., Ltd. and Cell Engineering Corporation was performed, in part, with support of a grant from Sumitomo Chemical Co., Ltd.
Informed consent
All commercially available samples used were collected after obtainment of patients’ written, informed consent.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
