Abstract
Growing evidence have revealed the serum exosomal miRNAs emerged as biomarkers for various cancer types, including colorectal cancer (CRC). Here, we sought to explore the potential clinical significance of serum exosomal miR-150-5p in CRC. A total of 133 CRC patients and 60 healthy volunteers as control group were recruited in this study. Exosomes were isolated from the serum of all the participants. The total RNA was isolated from the exosomes and the serum exosomal miR-150-5p levels were measured by quantitative reverse transcription-polymerase chain reaction. The findings showed that the serum exosomal miR-150-5p levels were significantly reduced in CRC cases compared with those in the control group. Serum exosomal miR-150-5p levels in post-operative blood samples were greatly upregulated one month after surgical treatment. In addition, decreased serum exosomal miR-150-5p expression was closely correlated with poorly differentiation, positive lymph node metastasis and advanced TNM stage. Moreover, receiver operating characteristic (ROC) curve analysis showed serum exosomal miR-150-5p level had good performance to identify CRC cases from healthy volunteers, and a combination of serum exosomal miR-150-5p and carcinoembryonic antigen (CEA) could improve the diagnostic accuracy with an increased the area under the ROC curve (AUC) value. Furthermore, the survival time of patients with higher serum exosomal miR-150-5p expression was significantly longer than those with lower expression. Serum exosomal miR-150-5p was confirmed as an independent prognostic indicator in CRC. Mechanistically, ZEB1 was identified as a direct downstream target of miR-150-5p. Collectively, serum exosomal miR-150-5p might be a novel noninvasive biomarker for CRC diagnosis and prognosis.
Introduction
Colorectal cancer (CRC), one of the most frequent fatal cancers in the gastrointestinal tract, is the fourth leading cause of cancer-related deaths around the world [1, 2]. In China, about 300,000 new CRC cases are reported and 150,000 patients die from this malignancy annually [3]. Although the advances in the diagnosis and treatment of CRC have been made over the past years, the 5-year overall survival (OS) rate of CRC still remains unsatisfied, especially in patients exhibiting metastasis and local relapse [4, 5, 6]. Thus, to improve the prognosis and the survival rate of CRC, novel and effective diagnostic and prognostic biomarkers are urgently required.
MicroRNAs (miRNAs) are a class of small (19–25-nt) non-coding RNA molecules and function as a class of negative regulators by binding to target mRNAs at their 3’-untranslated region, resulting in mRNA degradation or translation suppression [7, 8]. Emerging evidence have shown that miRNAs can function as oncogenes or tumor suppressors, and their deregulation involves in the initiation and progression of various types of cancer, including CRC [9, 10].
Exosomes are small membranous vesicles (30–100 nm) and found naturally in different body fluids (plasma, urine, saliva). Previous reports have showed that most of serum miRNAs are enriched and stably detected in exosomes [11, 12]. Several exosomal miRNAs such as miR-217 [13], miR-203 [14], miR-17-92a and miR-19a [15] are regarded as non-invasive biomarkers for CRC detection and predicting the prognosis.
MiR-150-5p was located on chromosome 19q13 and aberrantly expressed in many tumor cancer types, suggesting that miR-150-5p might be closely involved in carcinogenesis. For CRC, miR-150-5p was previously implicated to exert a tumor suppressor function. For instance, Chen et al. found that miR-150-5p expression was low in both CRC tissues and cell lines, and downregulation of miR-150-5p was correlated with aggressive clinical parameters. In addition, in vitro and in vivo evidence showed that ectopic expression of miR-150-5p greatly attenuated the oncogenic activities of cancer cells by targeting vascular epithelial growth factor A (VEGFA) [16]. Likewise, reduced miR-150-5p levels were found in CRC tissues and cell lines, and its expression was inversely associated with zinc finger antisense 1 (ZFAS1) expression [17]. Wang and colleagues revealed the miR-150-5p directly targeted MUC4, leading to a decreased cell migration, invasion and metastasis [18]. However, the potential clinical significance of serum exosomal miR-150-5p in CRC is unknown.
Materials and methods
Ethic statements
In this study, all clinical and laboratory protocols were approved by the Ethic Committees of Aviation General Hospital. The written informed consents were signed by all the patients, and all specimens were handled and made anonymous according to the ethical and legal standards.
Patients and samples collection
A total of 133 cases with pathologically confirmed CRC and 60 healthy volunteers were enrolled in our study. The patients underwent any chemotherapy or radiotherapy before blood sampling were excluded. Patient characteristics with respect to sex, age, tumor location, local invasion, carcinoembryonic antigen (CEA), differentiation, lymph node metastasis (LNM) and tumor-node-metastasis (TNM) stage were presented in Table 1.
Correlation between serum exosomal miR-150-5p levels and clinical parameters
Correlation between serum exosomal miR-150-5p levels and clinical parameters
NS
Briefly, 5 mL of peripheral blood was withdrawn from each participant before any treatment. Of all CRC patients, 91 cases received surgical resection, and the blood samples were collected from them 30 days after their surgery. All the samples were centrifuged for 10 min at 1500 rpm and for 2 min at 12000 rpm within 12 hours, then the supernatant was stored at
Exosomes were extracted and purified using ExoQuick exosome precipitation solution (System Biosciences, CA, USA) following the manufacturer’s protocol. Exosomal RNA was isolated from exosome pellets using the miRNeasy Serum/Serum Kit (Qiagen, Hilden, Germany). The RNA sample concentration was quantified spectrophotometrically using the NanoDrop ND-1000 system (NanoDrop, Wilmington, DE, USA). Reverse transcription was performed to synthesize cDNA using TaqMan MicroRNA Reverse Transcription Kit (Thermo Fisher Scientific, Waltham, MA, USA). After the synthesis of cDNA, qRT-PCR was performed in triplicate on LightCycler 480 Real-time PCR system (Roche Applied Science, Penzberg, Germany) using Taqman Universal PCR Master Mix (Thermo Fisher Scientific). Before RNA extraction, 100 fmol/mL of synthetic Caenorhabditis elegans miR (cel-miR-39) was added to an equal volume of serum and used as the control. The relative level of serum exosome miR-150-5p was calculated by the comparative 2
CEA detection
CEA as a minimally invasive biomarker has been previously used in CRC diagnosis. The expression levels of serum CEA in patients with CRC were measured by available commercial Elecsys CEA kit (Roche Diagnostics, Basel, Switzerland) using the modular analytics E170 (Roche). The assays were performed according to the manufacturer’s instructions and all samples were run in duplicate. Abnormal serum CEA expression was defined as level higher than 5.0 ng/mL
Cell culture and luciferase reporter assay
The human colorectal cancer cell lines HCT116 and HT-29 were cultured in Dulbecco’s modified Eagle medium supplemented with 10% fetal bovine serum, penicillin (100 U/mL) and streptomycin (100
Statistical analysis
The expression levels of serum exosomal miR-150-5p were compared using the Mann-Whitney U test between groups. Comparisons between categorical data were evaluated with the Chi-square test. The diagnostic accuracy was assessed by receiver operating characteristic (ROC) curve analysis, and the area under the ROC curve (AUC) was computed. Overall survival (OS) and disease-free survival (DFS) curves were constructed using the Kaplan-Meier method with a log-rank test. Cox proportional-hazards regression model was used to determine univariate and multivariate odds ratio for OS. OS was defined as the time between the date of diagnosis and the date of death. DFS was defined as the time between the date of surgery and the date of recurrence. Statistical analysis was performed using GraphPad Prism version 6.0 (GraphPad Software, San Diego, CA, USA).
Results
CRC patients exhibited lower serum exosomal miR-150-5p levels
QRT-PCR was used to detect the relative levels of serum exosomal miR-150-5p in all CRC patients and normal controls, and the difference in serum exosomal miR-150-5p expression between CRC patients and healthy volunteers was shown in Fig. 1. Compared to the control group, the expression levels of serum exosomal miR-150-5p were remarkably reduced in CRC patients (
Serum exosomal miR-150-5p levels were significantly lower in CRC patients than those in the control group.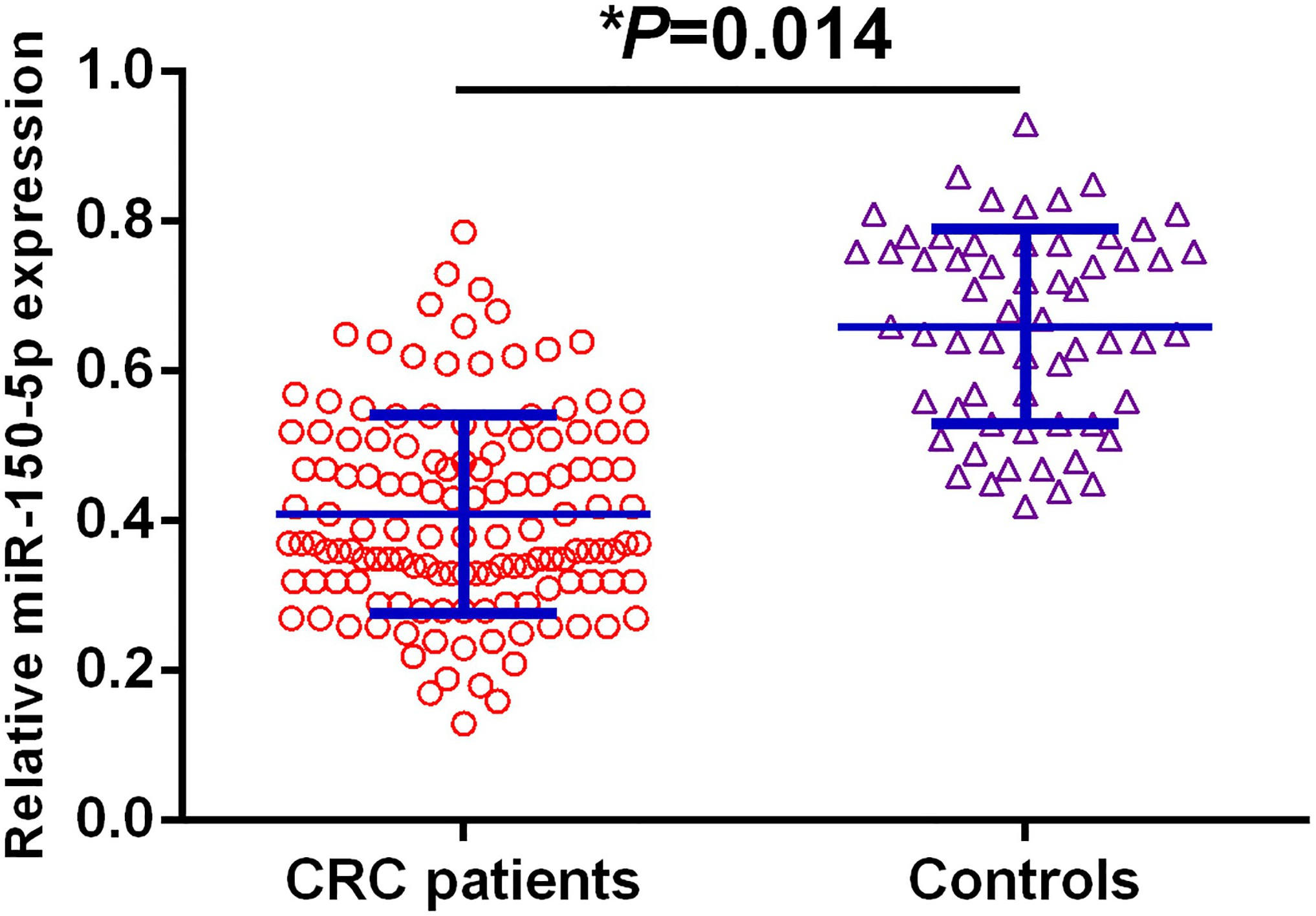
Compared to the levels of serum exosomal miR-150-5p in pre-operative blood samples, a significant increase in serum exosomal miR-150-5p was observed in post-operative samples.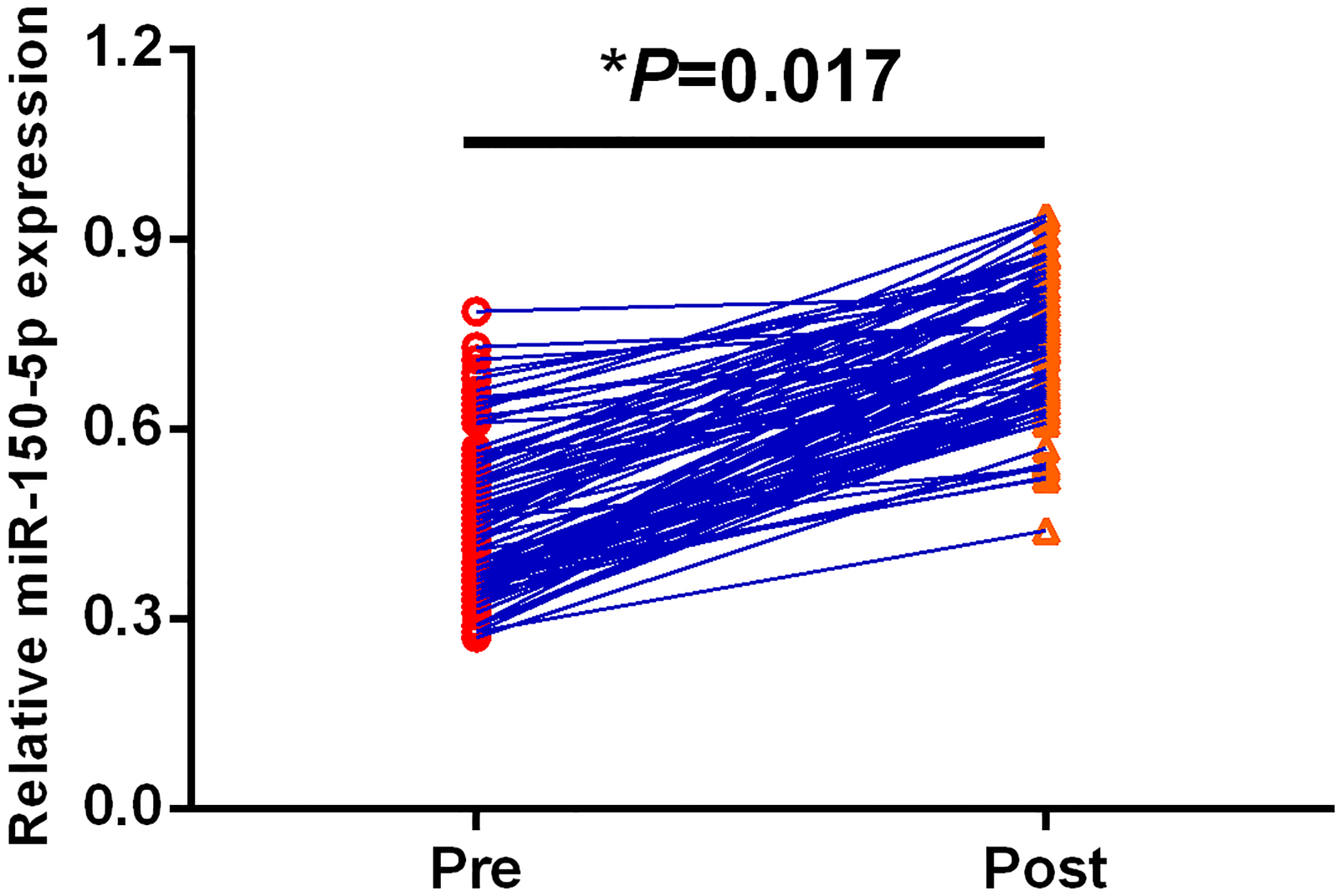
A. ROC curve analysis of serum exosomal miR-150-5p in all CRC patients. B. ROC curve analysis of CEA in all CRC patients. C. ROC curve analysis of serum exosomal miR-150-5p and CEA in s all CRC patients.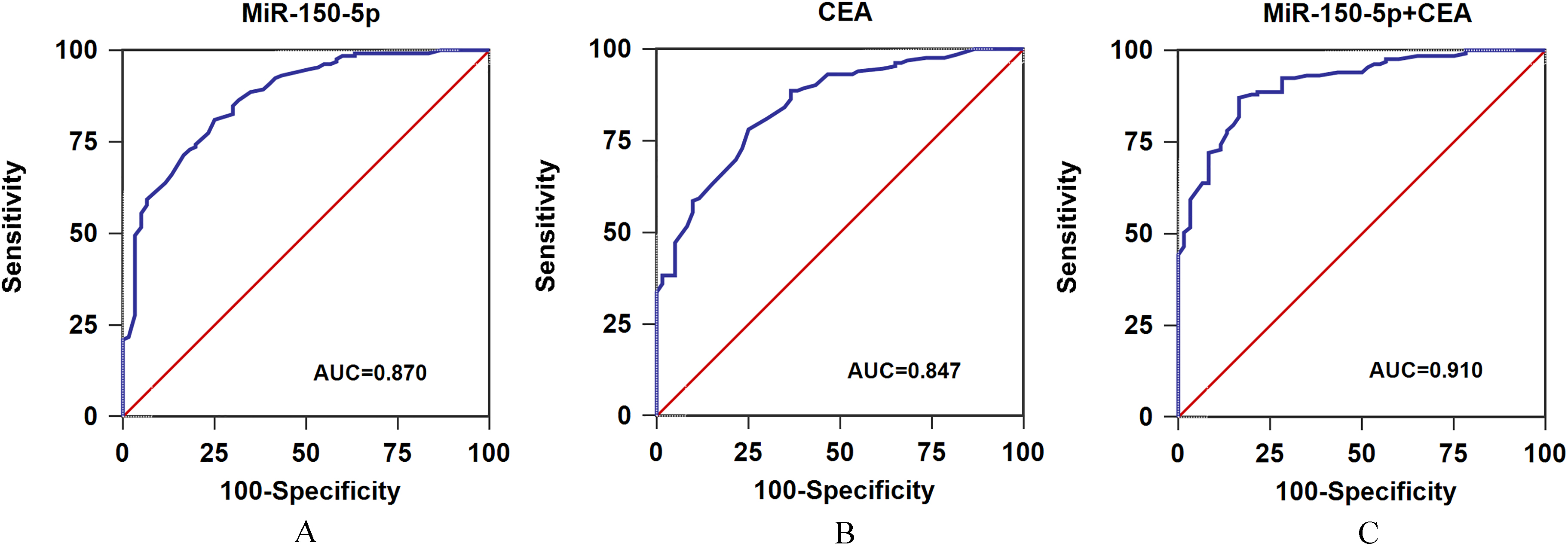
Next, we detected the serum exosomal miR-150-5p levels between ninety-one paired pre-operative samples and post-operative samples. Serum exosomal miR-150-5p levels in post-operative blood samples were statistically significantly higher than those in pre-operative samples (
The median value of serum exosomal miR-150-5p expression in CRC patients was used as the cutoff point. All 133 CRC cases were classified into high serum exosomal miR-150-5p expression group (
Serum exosomal miR-150-5p was a potential biomarker for the diagnosis of CRC
The ROC curve analysis was performed to compare the diagnostic accuracy of serum exosome miR-150-5p and traditional biomarker CEA. As shown in Fig. 3A, serum exosomal miR-150-5p expression was robust in differentiating CRC subjects from healthy volunteers with an AUC value of 0.870, and the specificity and sensitivity were 76.1% and 81.0%, respectively. Figure 3B revealed CEA had an AUC value of 0.847, the diagnostic specificity and sensitivity of CEA were 75.0% and 78.2%, respectively. The data indicated serum exosomal miR-150-5p had better performance to identify CRC patients from controls than CEA. Next, the combination of serum exosomal miR-150-5p and CEA could yield increased diagnostic power of discrimination with an AUC of 0.910, sensitivity of 87.2% and specificity of 83.3% (Fig. 3C). The data showed the combined serum exosomal miR-150-5p and CEA improved the diagnostic accuracy of either alone.
Correlation between serum exosomal miR-150-5p and the OS/DFS of CRC patients
Kaplan-Meier analysis demonstrated that CRC cases with low serum exosomal miR-150-5p levels had significantly worse OS (
In univariate analysis, differentiation (OR
Univariate and multivariate analysis of prognostic parameters for OS of CRC patients
Univariate and multivariate analysis of prognostic parameters for OS of CRC patients
CI
A. Kaplan-Meier survival analysis of OS according to serum exosomal miR-150-5p levels in all CRC patients. B. Kaplan-Meier survival analysis of DFS according to serum exosomal miR-150-5p levels in all CRC patients. C. Kaplan-Meier survival analysis of OS according to differentiation in all CRC patients. D. Kaplan-Meier survival analysis of OS according to lymph node metastasis in all CRC patients.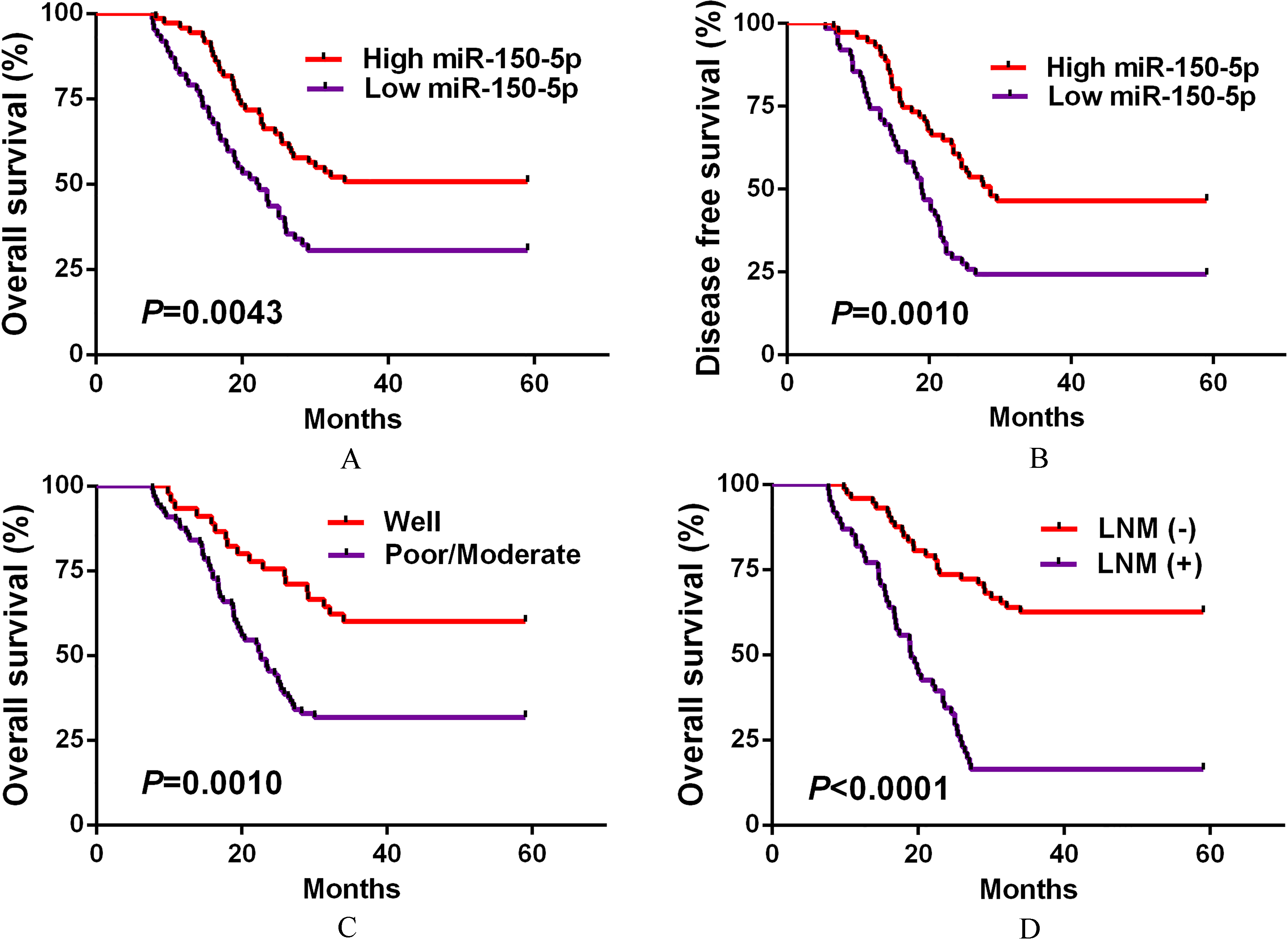
To explore the potential molecular mechanisms accounting for the tumor suppressive role of miR-150-5p in CRC, we first screened the downstream targets of miR-150-5p using miRwalk2.0 and TargetScan7.2. Our results showed that there were 356 and 544 downstream targets of miR-150-5p in miRwalk2.0 and TargetScan7.2 respectively (Fig. 5A). Figure 5B revealed the commonly detected downstream targets. The luciferase reporter assay showed that cotransfection of miR-150-5p mimic together with the wild-type ZEB1 reporter caused significant downregulation of the luciferase reporter activity. However, cotransfection of miR-150-5p mimic together with the mutant-type ZEB1 reporter did not affect the reporter activity (Fig. 5C).
ZEB1 was a direct downstream target of miR-150-5p.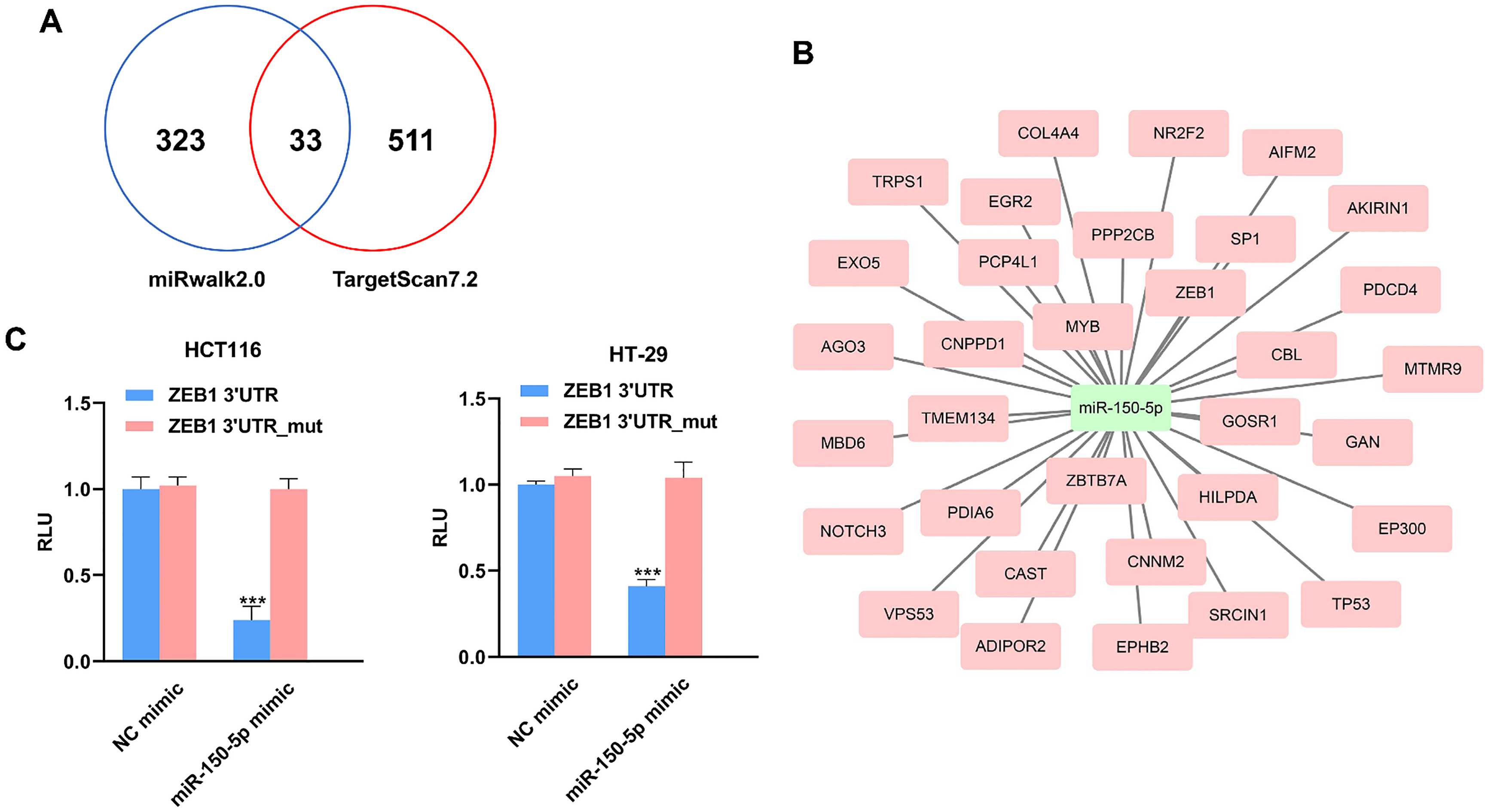
MiRNA dysregulation has been found to be correlated with the initiation and progression of CRC, here we aimed to explore the potential value of serum exosomal miR-150-5p as a potential biomarker for CRC. To the best of our knowledge, this was the first report demonstrating that serum exosomal miR-150-5p affected the prognosis of patients with CRC. In this study, we found serum exosomal miR-150-5p levels were significantly downregulated in CRC patients than those in the controls. In addition, serum exosomal miR-150-5p in post-operative blood samples showed marked upregulation after surgical treatment. Low serum exosomal miR-150-5p expression was strongly associated with poor differentiation, the presence of lymph node metastasis and advanced TNM stage in CRC patients. Moreover, serum exosomal miR-150-5p expression could well differentiate CRC patients from normal control, and the AUC, specificity and diagnostic accuracy of combined serum exosomal miR-150-5p and CEA were superior to those of either alone. Furthermore, patients in low serum exosomal miR-150-5p expression group had significantly worse OS and DFS compared to those in high expression group. Serum exosomal miR-150-5p was identified to be an independent poor prognostic marker in CRC. We have proposed a potential mechanism accounting for the downregulation of serum exosomal miR-150-5p in patients with CRC. The biosynthesis of exosomes was significantly upregulated in cancer [19]. Thus, the tumor cells not only produce and release many more exosomes compared to the normal cells, but also the contents of the cancer cell-derived exosomes are dramatically different from those from normal cells [20]. Maintaining the normal expression level of miR-150-5p might be important for the cellular homeostasis. Under the normal circumstances, miR-150-5p is packaged into the normal cell-derived exosomes and then secreted into the circulation system. However, much less miR-150-5p was produced in the exosomes from tumor cells. For the cancer patients, most of the circulating exosomes are derived from the tumor cells. Therefore, when we compared the level of miR-150-5p in serum exosomes between CRC patients and healthy controls, it was reasonable to observe that the relative serum exosomal miR-150-5p level was significantly lower in CRC patients than in healthy controls.
Mechanistically, ZEB1 was identified as a direct downstream target of miR-150-5p. The reason that we chose ZEB1 for the luciferase reporter assay is as follows: Firstly, epithelial-mesenchymal transition (EMT) is a process which plays a critical role in the tumor initiation, invasion, metastasis and therapeutic resistance [21]. The ZEB family of transcription factors consists of two members, ZEB1 and ZEB2 [22]. ZEB1 is well known EMT activator which promotes the expression of mesenchymal genes and suppresses the expression of epithelial genes. Also, ZEB1 actively regulates many important biological processes including, but not limited to, vertebrate embryonic development, differentiation and DNA damage responses [23]. ZEB1 has been demonstrated to a crucial regulator in the tumorigenesis and progression of CRC. For instance, Ester Sanchez showed that ZEB1 promoted the migration and invasion capacity of CRC cells by regulating the expression levels of uPA and PAI-1 [24]. Loss of basement membrane is important for malignancy metastasis. ZEB1 served as an indispensable transcriptional repressor for inhibiting the biosynthesis of basement membrane components in CRC [25]. Secondly, currently the association between miR-150-5p and ZEB1 in CRC remains poor known. Due to the crucial role of ZEB1 in CRC carcinogenesis, elucidating their potential correlation is important for understanding the tumor suppressive role of miR-150-5p in CRC. Future studies are warranted to validate other predicted downstream targets of miR-150-5p in CRC cells.
Consistent with our findings, various studies have demonstrated that miR-150-5p functions as a tumor suppressor gene in CRC. For instance, circulating miR-150-5p levels were significantly lower in CRC samples than in non-malignant samples [26]. The expression level of miR-150-5p was also reduced in the CRC tissues compared to the adjacent normal tissues. In addition, low miR-150-5p expression was associated with unfavorable survival and therapeutic responses [27]. Ectopic expression of miR-150-5p suppressed the proliferation, migration and invasion capacity of CRC cells in vitro and tumor growth in vivo, while downregulation of miR-150-5p led to opposite findings. Mechanistically, c-Myb was identified to be a direct downstream target of miR-150-5p [28]. Similarly, the expression level of miR-150-5p was also found to be downregulated in CRC cell lines. Overexpression of miR-150-5p inhibited the malignant behaviors of cancer cells by suppressing the transcription factor 4 (TCF4) [29]. Li et al. showed that a negative correlation was found between miR-150-5p and iASPP expression in CRC tissues. Enforced expression of miR-150-5p decreased the viability, induced cell cycle arrest and apoptosis as well as suppressed the migration and invasion of CRC cells. Overexpression of iASPP partially restored the oncogenic behaviors of cancer cells, indicating that iASPP was a functional downstream target of miR-150-5p [30]. Glioma-associated oncogene homolog 1 (Gli1) and High mobility group protein A2 (HMGA2) were also identified to downstream targets of miR-150-5p in CRC cells [31, 32].
In addition to CRC, miR-150-5p was found to inhibit the carcinogenesis in many types of cancer. In epithelial ovarian cancer (EOC), miR-150-5p expression was markedly decreased in EOC tissue samples, miR-150-5p upregulation suppressed ECO cell proliferation while miR-150-5p inhibition enhanced invasion ability [33]. An underexpression of miR-150-5p was found in primary and metastatic hepatocellular carcinoma (HCC) tissues, and knockdown of miR-150-5p dramatically accelerated the tumorigenesis of HCC through regulating the matrix metalloproteinase 14 (MMP14) [34]. In juvenile myelomonocytic leukemia (JMML), Leoncini and colleagues demonstrated miR-150-5p was decreased in JMML patients and in a JMML murine model, and miR-150-5p overexpression remarkably restrained cancer cell proliferation [35]. In osteosarcoma, miR-150-5p was identified as the downstream target of myocardial infarction associated transcript (MIAT). In vitro evidence showed MIAT upregulation increased cancer cell proliferation and invasion, and ectopic miR-150-5p expression inhibited the tumorigenic effects of MIAT in cancer cells [36]. Suetsugu et al. showed miR-150-5p levels were greatly reduced in both tissue samples and cell lines of lung squamous cell carcinoma (LUSQ), miR-150-5p upregulation inhibited the LUSQ cell migration and invasion [37]. Wu and colleagues reported miR-150-5p downregulation more frequently occurred in sera, bile and tissues of cholangiocarcinoma (CCA), and miR-150-5p overexpression strongly suppressed CCA cell proliferation, migration and invasion [38]. In glioma, Sakr et al. showed miR-150-5p levels were greatly reduced in cancerous tissues in comparison with normal tissues, and transfection of miR-150-5p mimic inhibited the proliferation, invasion and migration of the cells in vitro by directly silencing MT1-MMP (membrane-type 1 matrix metalloproteinase) expression [39]. Moreover, an inverse correlation between SPOCK1 and both the strands of pre-miR-150 (miR-150-5p and miR-150-3p) was identified in head and neck squamous cell carcinoma (HNSCC) and esophageal squamous cell carcinoma (ESCC). Koshizuka et al. demonstrated high SPOCK1 expression was observed in HNSCC clinical samples and significantly promoted cell invasiveness, indicating miR-150-5p/miR-150-3p exhibited antitumor effect in HNSCC cells [40]. Similarly, Osako et al. revealed ectopic miR-150-5p/miR-150-3p expression or silencing of SPOCK1 expression markedly decreased ESCC cell viability and invasion [41].
Interestingly, Yan and colleagues reported high miR-150-5p expression significantly stimulated cell proliferation, viability, induced cell apoptosis and promoted epithelial mesenchymal transition process in papillary thyroid cancer (PTC) cell lines by positively regulating BRAF
In conclusion, our findings demonstrated that serum exosomal miR-150-5p levels were significantly lower in CRC. Low serum exosomal miR-150-5p expression was associated with aggressive clinical parameters and poor prognosis. Therefore, serum exosomal miR-150-5p showed great potential as a biomarker for the diagnosis and prognosis in CRC.
Footnotes
Conflict of interest
We deny any conflict of interest.
