Abstract
BACKGROUND:
Non-small cell lung cancer (NSCLC) is one of the most widespread cancer with increasing morbidity and mortality. FAS-associated protein with death domain (FADD) is considered as an essential instrument in cell death, whereas Bcl-XS promotes apoptosis through inhibiting the activity of Bcl-2 and Bcl-XL.
OBJECTIVE AND METHODS:
We detected the expression of FADD and Bcl-XS in resected NSCLC tissues by immunohistochemistry, and investigated their association with clinicopathological characteristics and prognostic significance of NSCLC patients.
RESULTS:
Bcl-XS expression was significantly increased in well and moderate differentiated lung SCC (
CONCLUSION:
Elevated expression of FADD was identified as independent poor prognostic factor for patients with surgically resected lung ADC, however, increased expression of Bcl-XS was an independent good prognostic biomarker for patients with surgically resected lung SCC.
Introduction
Lung cancer is one of the most common cancers with increasing morbidity and mortality that threatens human health. According to the estimation of the American Cancer Society, 83550 men and 70500 women died from lung cancer in 2018, and new diagnosed cases would come up to 121680 and 112350, in males and females respectively, which would rank the first place among all kinds of new malignant tumors in America [1]. Also, more than one-third of all newly diagnosed lung cancers worldwide occurred in China,and it was estimated for 4.3 million new cases and 2.9 million cancer-related death in 2018 [2].
Lung cancer is usually classified into non-small cell lung cancer (NSCLC) and small cell lung cancer (SCLC), which accounted for approximately 80% and 20% respectively for total lung cancer in 2015. Like most human cancers, the occurrence and development of NSCLC is considered as a multiple process and involves numerous cancer hallmarks, one of which is to resist induced apoptosis [3].
Apoptosis is programmed cell death and serves as a natural barrier to cancer development. However, deregulation of apoptotic signaling is universal in many cancer cells, which leads to uncontrolled proliferation and tumor development/growth [4]. Various molecules and signaling pathways involved in this complicated phenomenon include FAS-associated protein with death domain (FADD) and Bcl-XS.
FADD is considered an essential molecule in cell death activation, as it can interact with FAS, tumornecrosis factor receptor 1 (TNF-R1), death receptors (DRs) and other molecules, leading to apoptosis [5].
Bcl-XS belongs to Bcl-2 family, and this family of proteins regulate apoptosis by controlling the permeability of the mitochondrial outer membrane. Among four members, while Bcl-2 and Bcl-XL prevent cells from entering apoptosis, on the other hand Bax and Bcl-XS induce cell death. Bcl-XS can inhibit the activity of Bcl-2 and Bcl-XL, and thus promote apoptosis [6, 7].
It has been demonstrated that high expression of FADD is associated with shorter survival times in patients with head and neck cancer, breast cancer, nasopharyngeal carcinoma (NPC) and NSCLC [8, 9, 10, 11], whereas the overexpression of Bcl-XS is reported to result in strong induction of apoptosis in melanoma cell lines [12]. In Jurkat cells, Bcl-2 inhibits Fas-induced apoptosis by preventing the formation of the death-inducing signaling complex, which is composed of Fas, FADD/MORT1 and caspase 8 [13].
Reviewing published literatures, we haven’t found reports about whether the expression of FADD and Bcl-XS is associated with clinicopathological characteristics for patients with surgically resected NSCLC. Meanwhile, the relationship between the expression of FADD and Bcl-XS in NSCLC is unclear. In this study, we detected the expression of FADD and Bcl-XS proteins in NSCLC tissue arrays including the surgically resected NSCLC tissues and non-cancerous lung control tissues by immunohistochemistry (IHC). Wealso investigated the relationship between expression of FADD and Bcl-XS and clinicopathological characteristics and their potential prognostic significance in patients with surgically resected NSCLC.
Materials and methods
Ethics statement
All samples were obtained with informed consentand all protocols, specimen usage, and data retrieval were approved by the Ethics Review Committee of the Second Xiangya Hospital of Central South University (Scientific and Research Ethics Committee, No. S039/2011). Written informed consent was obtained from all patients, also the written informed consent was obtained from the next of kin, caretakers, or guardians on the behalf of the minors/children participants involved in our study.
Patient cohorts
In this study we included 446 cases patients with age at diagnosis ranging from 36 to 73 years (median, 55.1 years) of paraffin-embedded NSCLC from the primary NSCLC patients which include 227 cases of lung ADC and 219 cases of lung SCC. We also selected 89 cases of non-cancerous lung control specimen from independent patients with non-cancerous pulmonary diseases. All 535 tissue specimens were collected from the Department of Pathology, Second Xiangya Hospital of Central South University (Changsha, China) between January 2007 and December 2016.
The cohort included NSCLC, all of which had undergone clinical surgery at the Department of Thoracic Surgery at the Second Xiangya Hospital of Central South University. All patients with NSCLC had been assessed and staged routinely and accepted definitive surgical resection of the lung and systematic mediastinal lymph node dissection. No patients had been previously treated with neither radiotherapy nor chemotherapy before operation.
Patients had a confirmed histological diagnosis of NSCLC according to WHO [14] histological classification of the lung cancer, and the staging classification was based on the 7th edition of the AJCC/UICC TNM staging system of lung cancer (2009) [15]. Clinicopathological characteristics of patients with surgically resected NSCLC and non-cancerous control lung tissues are shown on Table 1.
Clinicopathological characteristics of patients with surgically resected NSCLC and non-cancerous control lung tissues
Clinicopathological characteristics of patients with surgically resected NSCLC and non-cancerous control lung tissues
Abbreviations: NSCLC: non-small cell lung cancer; SCC: squamous cell carcinoma; ADC: adenocarcinoma; LN: lymph node; LNM, lymph node metastasis.
Complete clinical record and followed-up data are available for all patients. The non-cancerous lung tissues were obtained from patients with pulmonary bulla or tuberculosis, and asmall amount of lungtissues were obtained from trauma patients. In this study, we used the tissue microarrays (TMA) technology to construct the high-throughput NSCLC TMAs according to rules previously described [16].
The IHC staining for FADD and Bcl-XS proteins in TMAs was carried out using ready-to-use Envision TM
To eliminate nonspecific staining, the slides were incubated with appropriate preimmune serum for 30 minutes at room temperature, and then with specific first antibody in 4
All slides were counterstained with hematoxylin. Positive control slides were included in every experiment in addition to the internal positive control. The specificity of the antibody was determined with matched IgG isotype antibody as a negative control.
Positive staining of FADD protein mainly located in both the cytoplasm and nuclei of cancer cells. Positive expression of Bcl-XS protein was found in the cytoplasm and cell membrane of cancer cells. A semiquantitative evaluation of FADD and Bcl-XS expression was independently reviewed by two pathologists (J.F. and S.F), who were blind to the clinicopathological data under light microscopy with 200
Expression of FADD and Bcl-XS proteins in lung SCC cells, lung ADC cells and non-cancerous control lung tissues were detected by IHC using specific antibody. Strong positive staining of FADD was found in cytoplasm and nuclei of lung SCC, lung ADC cells, and negative control showed no staining in lung SCC cells (Fig. 1A–C). Strong positive staining of Bcl-XS was indicated in cytoplasm and membrane of lung SCC, lung ADC cells, and negative control showed no staining in lung ADC cells (Fig. 1D–F). Positive staining of FADD was showed in cytoplasm and nuclei of the on-cancerous control lung tissues (Fig. 1G), positive staining of Bcl-XS was found in cytoplasm and membrane of the on-cancerous control lung tissues (Fig. 1H). Negative control showed no staining in the on-cancerous control lung tissues (Fig. 1I) (IHC, 200 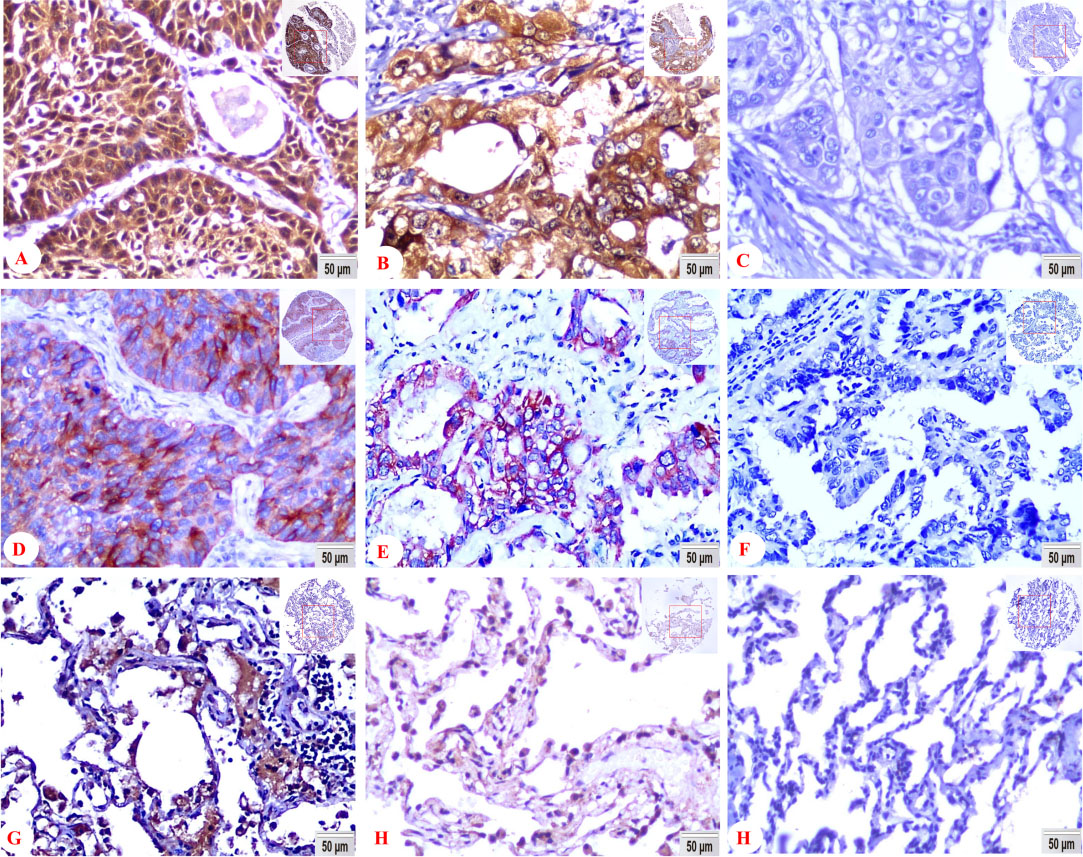
All statistical analysis were performed by SPSS version 22.0 (SPSS, Chicago, IL). The clinicopathologic characteristics and prognostic factors of NSCLC patients were compared between the two cohorts using Chi-square test forcategorical variables. The Spearman’s rank correlation coefficient was used to assess the association between FADD and Bcl-XS in the NSCLC. Kaplan-Meier analysis was performed to obtain overall survival curves, and statistical significance was assessed using the log-rank test. Overall survival times were defined as the time from the treatment initiation (diagnosis) to the date of death or date of last follow-up.
To identify whether overexpression of FADD and Bcl-XS was the independent prognostic factors for overall survival in NSCLC, multivariate analysis using the Cox proportional hazard regression model was performed. All
Results
Expression of FADD and Bcl-XS proteins was ignificantly higher in NSCLC
On the basis of our previous work [20], we constructed NSCLC TMA containing 227 cases of lung ADC, 219 cases of lung SCC and 89 cases of non-cancerous control lung tissues. Positive expression and cellular localization of FADD and Bcl-XS proteins in NSCLC and the non-cancerous control lung tissues were detected by IHC.
Positive expression of FADD protein was located in the cytoplasm and nuclei. Bcl-XS positive staining was found in the cytoplasm and cell membrane (Fig. 1). The matched IgG isotype antibody showed no positive staining of Bcl-XS as a negative control.
The expression rate of FADD protein was 33.9% (77/227), 37.9% (83/219) and 2.2% (2/89) in lung ADC, lung SCC and non-cancerous control lung tissues respectively. In terms of Bcl-XS, the percentage of elevated expression was 48.0% (109/227), 51.6% (113/219) and 13.5% (12/89) in lung ADC, lung SCC and non-cancerous control lung tissues respectively. There was significantly higher positive percentages of FADD and Bcl-XS expression in the lung SCC and lung ADC than that in the non-cancerous control lung tissues (
Expression of FADD and Bcl-XS proteins in NSCLC and non-cancerous control lung tissues (Non-CLT) was compared by Chi-square test. Column charts show that there were significant differences between the groups which were statistically evaluated by Chi-square test. Positive expression of FADD and Bcl-XS proteins was evidently higher in the lung SCC and lung ADC compared to the Non-CLT (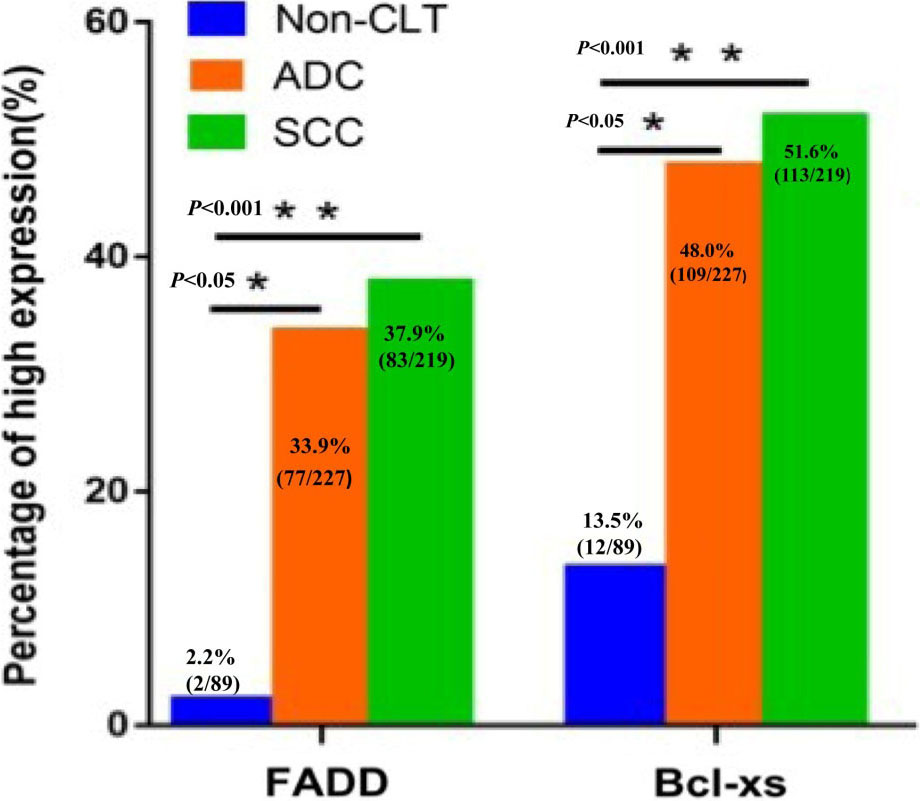
Moreover, we analyzed the pairwise association between expression of FADD and Bcl-XS proteins in 227 cases of lung ADC and 219 cases of lung SCC, we found positively association between expression of FADD and Bcl-XS proteins in lung ADC and lung SCC (
Pairwise association between expression of FADD and Bcl-XS proteins in lung SCC and lung ADC
Abbreviations: NSCLC: non-small cell lung cancer; SCC: squamous cell carcinoma; ADC: adenocarcinoma. NOTE. Spearman’s rank correlation coefficient (r) analysis; * Correlation is significant at the
We further investigated the associations between the overexpression of FADD and Bcl-XS proteins and clinicalpathologic characteristics of NSCLC including age, gender, histological types, clinical stages, lymph node metastasis (LNM) and pathological grades in Chi-square test (Table 3).
Association between expression of FADD and BCL-XS proteins and clinicopathological characteristics of NSCLC (
446)
Association between expression of FADD and BCL-XS proteins and clinicopathological characteristics of NSCLC (
Abbreviations: NSCLC: non-small cell lung cancer; LNM, lymph node metastasis;
Compared to poor differentiated lung SCC, the elevated expression of Bcl-XS protein was significantly higher in well and moderate differentiated lung SCC (
To further examine the impact of FADD and Bcl-XS overexpressionon the overall survival rates (OS) of NSCLC patients, we applied the Kaplan-Meier analysis to plot the survival curve of lung ADC and lung SCC patients, and statistical significance was assessed using the log-rank test. The Kaplan-Meier survival curve was shown for patients with lung ADC and lung SCC according to the extent of expression of FADD and Bcl-XS proteins (Fig. 3).
Univariate survival analysis (log-rank test) showed that the OS rates were significantly lower fo rlung ADC patients with high expression of FADD protein (
Kaplan-Meier overall survival curves of NSCLC patients with expression of FADD and Bcl-XS proteins and different clinicopathological characteristics were statistically evaluated by the log-rank test. The overall survival (OS) rates was significantly lower for lung ADC patients with high expression of FADD protein (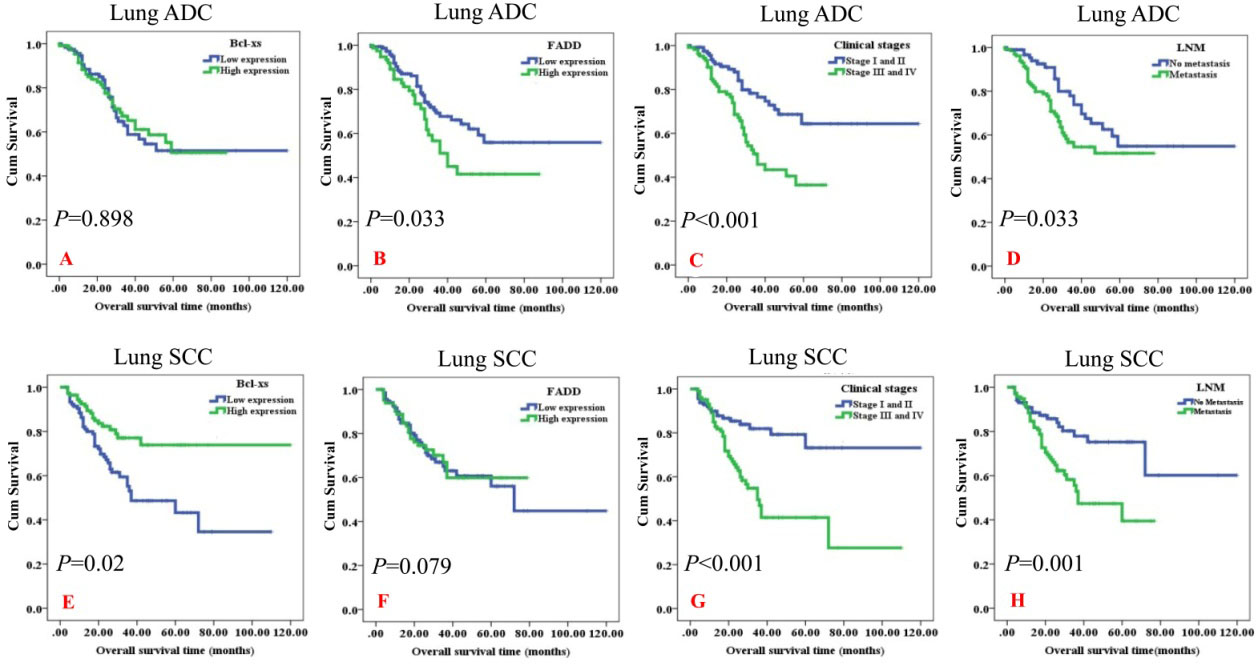
We also analyzed the survival rates of patients with NSCLC according to the conventional prognostic parameters including pathological grades, clinical stage and lymph node metastasis by univariate survival analysis (log-rank test). The results showed that the patients with advanced-stages (clinical stage III and IV) had lower OS rates than those with early-stages (clinical stage I and II) both in lung ADC (
In addition, lung SCC patients with poor pathological grade had significantly lower OS rates than these patients with well and moderately differentiated lung SCC (
Multivariate Cox proportional hazard regression analysis was also carried out to further investigate whether the overexpression of FADD and Bcl-XS proteins were the independent prognostic factors for lung ADC and lung SCC patients, respectively (Table 4).
Summary of multivariate of Cox proportional regression for overall survival in patients with surgically resected lung SCC and lung ADC
Abbreviations: LNM, lymph node metastasis; SCC, squamous cell carcinoma; ADC, adenocarcinoma; CI, confidence interval. Note: multivariate analysis of Cox regression, *
In multivariate analysis of the characteristics of patients with lung ADC or lung SCC, which include gender, age, pathological grades, clinical stages, LNM status, treatment strategy, overexpression of FADD and Bcl-XS proteins, elevated expression of FADD were identified as independent poorer prognostic factors for patients with lung ADC (
There were no prognostic effects detected in lung ADC including gender, age, pathological grades, LNM status, treatment strategy and Bcl-XS expression (all
NSCLC is one of the most widespread cancer with increasing morbidity and mortality, and it is typically fatal. Although comprehensive treatment dramatically improved the prognosis of NSCLC patients, overall survival is still poor. Compared to advanced clinical stages, 5-year survival rates of patients with early stages is significantly higher [21]. Exactly as various kinds of malignant tumors, the occurrence and development of NSCLC is undergoing multiple steps, and involved in diversified kinds of biological and pathological process, one of which is resistant to cell apoptosis.
FADD protein is an adaptor molecule that interacts with many cell surface receptors and mediates cell apoptotic signals. The FADD protein is located mainly in cytoplasm and nucleus of most cells, and its nuclear localization relies on strong nuclear localization and nuclear export signals (NLS and NES, respectively) that reside in the death-effector domain (DED) of the protein [22]. FADD dependent signaling pathway can activate apoptosis via FADD death domain, which allows it to recruit caspase-8, and thereby result in cell apoptosis [23], and it was later implicated the FADD is involved in nonapoptotic cellular processes such as the necroptosome and the inflammasome [24]. High expression of FADD protein can be detected in some kinds of malignant tumors, and overexpression of FADD is confirmed to associate with poor prognosis. It was reported by Choi et al. that FADD expression was associated with T stage in invasive ductal carcinoma of the breast, and combined gene expression scores of TMEM16A, FADD, and PPFIA1 can show association with disease-free survival [11]. Chen et al. also observed that increased FADD mRNA and protein are significantly associated with poor survivalin lung ADC and their further study confirmed that levels of p-FADD might be used as a prognostic biomarker for predicting survival of lung cancer patients [25]. Moreover, FADD expression is associated with a higher incidence of lymph node metastasis in squamous cell carcinoma of the head and neck [26]. Another research reveal that FADD gene copy number and protein expression can be considered potential prognostic markers and is closely associated with lymph node metastasis in patients with oral cavity squamous cell carcinomas in Taiwan [19]. Our previous study also have demonstrated that positive percentage of FADD expression in NPC was significantly higher than that in the noncancerous nasopharyngeal control tissues, and FADD expression was significantly correlated inversely with overall survival rates in NPC patients [10]. Although these studies indicte that overexpression of FADD have inverse effect for patients prognosis, Wang HB etal. reported that patients with early clinical stages have higher expression of FADD than that of advance stages in glioblastoma [27]. In our present study, we found that FADD expression is significant higher in NSCLC compared with non-cancerous lung tissue, however, no significant difference is observed between FADD expression and prognosis of NSCLC patients. On the other hand, Cimino et al. reported tumor samples released significantly more FADD than non-tumor ones (fully in accord with our results), nevertheless, their results showed that extracellular FADD could be a poor prognostic markerin NSCLC patients [9]. One of the reasons this inconsistency result might be due to apply different methods for the detection. We collected resected NSCLC samples and made tissue microarray, using immunohistochemistry to detect the expression of FADD and analyzing the relationship between FADD expression and clinicalpathological characteristics, whereas Cimino et al. were detecting released FADD via ELISA. Additionally, we found an interesting report by Yin et al. that stable overexpression of extrinsic FADD gene can significantly ameliorate apoptosis-inducing effects of 5-fluorouracil on colorectal adenocarcinoma cells [28]. Although their study is only focused on cell lines, we can assume that it might have some relationship between FADD expression and 5-fluorouracil efficacy in NSCLC patients, and we will do some deeper researches in that aspect in the future.
Bcl-2 family can regulate cell apoptosis in double ways, on the one hand, some family members such as Bax, Bad and Bcl-XS can promote cell apoptosis, and on the other hand, others can inhibit apoptosis including Bcl-2, Bcl-XL and BID. It is believed that the proteins of the BCL-2 family are playing roles through controlling mitochondrial outer membrane permeabilization [29, 30]. It is reported that Bcl-XScan be induced in spontaneous and drug-induced apoptosis in chronic lymphocytic leukemia cells [31]. Besides, Yamaguchi et al. has reported that Bcl-XS negative patients with acute myeloid leukemia will have shorter overall survival than those with positive Bcl-XS expression [32]. Moreover, it has been confirmed that the overexpression of Bcl-XS in MCF-7 cells induces a marked increase in chemosensitivity to VP-16 or Taxol [33]. These evidences indicate that Bcl-XS protein might play analogously protective roles. To our knowledge, there has been no report discussing the relationship between Bcl-XS and the prognosis of patients with NSCLC via immunohistochemistry. Our results show that the overexpression of Bcl-XS could act as an independent favorable prognostic biomarker for lung squamous cell cancer that have undergone radical resection and standardized prophylactic chemoradiotherapy, which might partly make compliment for Bcl-XS roles.
FADD/caspase-8/Bid/cytochrome Caxis is a crucial linker between death receptors and mitochondria, where they play a role in ROS generation and apoptosis [34]. FADD/Mort1 was initially reported as a pro-apoptotic adaptor molecule that recruits the initiator caspases 8 and 10 to promote formation of the death-inducing signal complex (DISC) and mediates receptor induced apoptosis [35]. Apoptosis is regulated by the action of the Bcl-2 family of proteins, which includes pro-apoptotic members such as Bcl-xS and Bax [36]. The apoptotic pathway is well characterized; however, a signaling linker between the FADD and Bcl-XS has not been clearly elucidated. Our results showed that overexpression of FADD protein was associated positively with expression of Bcl-XS in lung ADC and lung SCC Additional studies are planned to confirm these initial findings.
We have to admit that there are still some flaws deserved to improve. On the one hand, we have taken notice that some researchers study EGFR mutation and Bcl family or FADD protein simultaneously [37, 38], and more comprehensive follow-up statistics could be better. On the other hand, we found slightly positive correlation between FADD proteins and Bcl-XS in lung ADC patients, more detailed and deeper work should be done in the future.
In summary, there was significantly elevated expression of FADD and Bcl-XS proteins in the lung SCC and lung ADC. Overexpression of FADD positively associated with Bcl-XS expression in the NSCLC. Elevated expression of FADD was identified as independent poor prognostic factors for patients with surgically resected lung ADC, likewise high expression of Bcl-XS was identified as a good prognostic factor for patients with surgically resected lung SCC.
Footnotes
Funding
This work was supported by the National Natural Science Foundations of China (No: 81773218, 81472773, 81703009 and 81272566).
