Abstract
BACKGROUND:
Although risk stratification of mantle cell lymphoma (MCL) is most frequently performed using the simplified MCL International Prognostic Index (sMIPI), the identification of host-related factors and tumor microenvironment, including absolute monocyte counts (AMC) and peripheral blood T lymphocyte subsets, especially absolute natural killer cell counts (ANKC) has been suggested to be critical in the prediction of prognosis and the guidance of treatment.
OBJECTIVE:
This study was aimed at investigating whether peripheral blood ANKC and AMC at diagnosis had an impact on MCL prognosis.
METHODS:
A total of 92 newly diagnosed MCL patients was enrolled in this retrospective study. Flow cytometric analysis was conducted on fresh peripheral blood samples with a FACSCalibur flow cytometer (BD Biosciences, San Jose, CA, USA).
RESULTS:
The median follow-up was 42 months (range, 2–144 months) and the median overall survival (OS) of all cases was 45 months. High AMC (
CONCLUSIONS:
Low ANKC (
Introduction
Mantle cell lymphoma (MCL), a distinct subtype of B cell non-Hodgkin lymphoma (NHL), represents 3%–6% of all non-Hodgkin lymphoma. In addition to classical histology, in more than 95% cases, MCL patients have a cytogenetic feature of chromosomal translocation t (11; 14) (q13; q32), resulting in the overexpression of cyclin D1 and deregulation of cell cycle. Generally, patients with MCL experience a median overall survival (OS) of 3–5 years [1]. The currently used intensive treatment protocols including anti-CD20 antibody, high-dose cytarabine and autologous stem cell transplantation increased the response rate and remission duration but have failed to improve long-term OS so far. The simplified MCL International Prognostic Index (sMIPI), incorporating Eastern Cooperative Oncology Group (ECOG) performance status, age, leukocyte count and lactic dehydrogenase (LDH) was formulated in clinical routine to identify patient risk groups with different courses of disease [2]. Nonetheless, as sMIPI was derived from patients with advanced stage disease in the pre-rituximab era, adoption of the index has been confined since it has been shown that it inadequately separates low and intermediate risk group patients. To improve outcome and minimize treatment-related complications, additional parameters including Ki-67, TP53, serum
Tumor microenvironment and host immunity are significantly associated with outcome in patients with MCL. Natural killer (NK) cells, as important components of the innate immune response, play crucial roles in eliminating viruses, regulating dendritic cells and killing malignant cells [6]. Kim et al. revealed that low NK cell counts at diagnosis are associated with poor clinical outcomes in non-germinal center (non-GC) diffuse large B-cell lymphoma (DLBCL) patients treated with R-CHOP therapy [7]. As reported by Shafer et al., low NK cell counts in peripheral blood are associated with inferior OS in patients with follicular lymphoma (FL) [8]. However, no research to date has focused on the predictive role of absolute natural killer cell counts (ANKC) in the progression of MCL.
The present study aimed to evaluate the prognostic relevance of absolute monocyte counts (AMC) and ANKC and examine whether ANKC could be served as an independent prognostic parameter for progression free survival (PFS) and OS.
Materials and methods
Patients
One hundred and thirteen consecutive subjects histologically newly diagnosed MCL patients and detailed T/NK cell subsets at admission between April 2006 and November 2017 were enrolled in this retrospective study from the First Affiliated Hospital of Nanjing Medical University, Jiangsu Province Hospital (Nanjing, China). This study was approved by the Institutional Review Boards of our hospital. In consideration of the confounding effects of different medications, we eliminated 21 patients who took palliative care or abandon treatment or disrupt treatment for economic and other causes. Diagnosis in accordance with World Health Organization (WHO) classification 2008 should be dependent on an operative specimen, preferably a lymph node biopsy. Additionally, detection of t (11; 14) (q13; q32) and overexpression of cyclin D1 must be sufficient [9]. Laboratory measurements consisting of complete blood count (CBC), blood chemistry, imageological examination such as computed tomography (CT) and positron emission tomography (PET) of whole body as well as bone marrow aspiration and biopsy were obtained within 24 h after first admission. Baseline clinical characteristics were entirely available, including gender, age, bone marrow involvement (BMI), Ann Arbor stage, ECOG score, B symptoms, leukocyte count, serum lactic dehydrogenase (LDH),
Peripheral ANKC and AMC at diagnosis
Flow cytometric analysis was conducted on fresh peripheral blood samples with a FACSCalibur flow cytometer (BD Biosciences, San Jose, CA, USA). All murine anti-human mAbs, offered by BD Biosciences (San Jose, CA, USA), including anti-CD3-FITC, anti-CD4-PE, anti-CD8-APC, anti-CD16-PE, anti-CD19-APC and anti-CD56-APC were used to stain peripheral lymphocytes. T/NK cell subsets were counted for all patients at diagnosis. ANKC refers to CD3-CD16+ and/or CD56+ lymphocytes. Combined with the simultaneous white blood cell (WBC), the counts of NK cell subset were calculated from the percentages collected from peripheral blood flow cytometry (PBFCM). The AMCs were obtained from routine complete blood counts.
Treatment decision at diagnosis
Patients younger than 60 years old received a hyper-CVAD regimen (cyclophosphamide, doxorubicin, vincristine and dexamethasone) alternated with the MA regimen (high-dose methotrexate and cytarabine). However, patients
Statistical analysis
For the entire cohort, the OS and the PFS were the end points for the prognostic analyses. OS was defined as the time between diagnosis and death by any cause or last follow-up. PFS was calculated as the time from the initial diagnosis to the time of progression, relapse from response, death due to any cause or last follow-up. All statistical analyses were performed using SPSS for Windows (version 21.0; IBM Corporation, Armonk, NY, USA) and Graphpad Prism 6. The best cut-off values were determined by x-tile according to OS. The distribution of OS and PFS was estimated using the Kaplan-Meier method and statistical significance was assessed using the two-sided log-rank test. The Cox proportional hazards models was established for the estimation of hazard radio and its confidence interval in both univariable and multivariable analysis. Univariable analysis was performed with each potential prognostic factor. Multivariable proportional hazards regression was performed with statistically significant factors from the univariable analysis (factors with
Baseline clinical characteristics of 92 patients with mantle cell lymphoma
Baseline clinical characteristics of 92 patients with mantle cell lymphoma
ECOG, Eastern Cooperative Oncology Group; BMI, bone marrow involvement; sMIPI, simplified mantle cell lymphoma International Prognostic Index; LDH, serum lactate dehydrogenase; NUV, normal upper value; WBC, white blood cells;
Correlations between AMC and ANKC in patients with mantle cell lymphoma and patient characteristics
Progression-free survival and overall survival of 92 patients with mantle cell lymphoma according to absolute monocyte counts (AMC) and absolute natural killer cell counts (ANKC) at time of diagnosis, by Kaplan-Meier estimation.
Patient characteristics
The baseline clinical characteristics of 92 enrolled patients with MCL at admission were presented in Table 1. Of the 92 patients whose age ranged from 28 to 78 years, approximately 55% of them were over 60 years of age and 69 (75.0%) were male, which showed a striking male predominance. The majority of patients (93.5%) had advanced disease (Ann Arbor stage III–IV) and 59.8% had BMI. Elevated LDH was shown in 23 (25.0%) patients. B symptoms were found in 40 (43.5%) patients. Forty-seven (51.6%) patients had decreased ALB prior to treatment, while 67 (77.9%) patients had
Baseline patient characteristics in relation to AMC and ANKC
The baseline characteristics according to AMC and ANKC are described in Table 2. A statistically evident dominance was identified that high AMC was more likely in men (
Univariable Cox regression analysis of the main factors for PFS and OS
Univariable Cox regression analysis of the main factors for PFS and OS
Multivariable Cox regression analysis of the main factors for PFS and OS
In the current study, high AMC, low ANKC and other relevant variables were subjected to Cox regression analyses for further research of their prognostic significance. In patients with low AMC, the median PFS and OS were 29 months (range, 2–83 months) and 53 months (range, 2–109 months) respectively, which were significantly better compared to patients with high AMC, in whom the median PFS was 17 months (range, 1–56 months) and median OS was 18 months (range, 4–56 months) (
Subgroup survival analysis of sMIPI grades (A), age (B) for overall survival (OS). sMIPI, simplified mantle cell lymphoma International Prognostic Index.
Progression-free survival and Overall survival of 57 patients taking rituximab-containing regimens with mantle cell lymphoma according to ANKC at time of diagnosis, by Kaplan-Meier estimation.
Progression-free survival and Overall survival of 92 patients with mantle cell lymphoma according to ANKC to AMC ratio (NKMR) at time of diagnosis, by Kaplan-Meier estimation.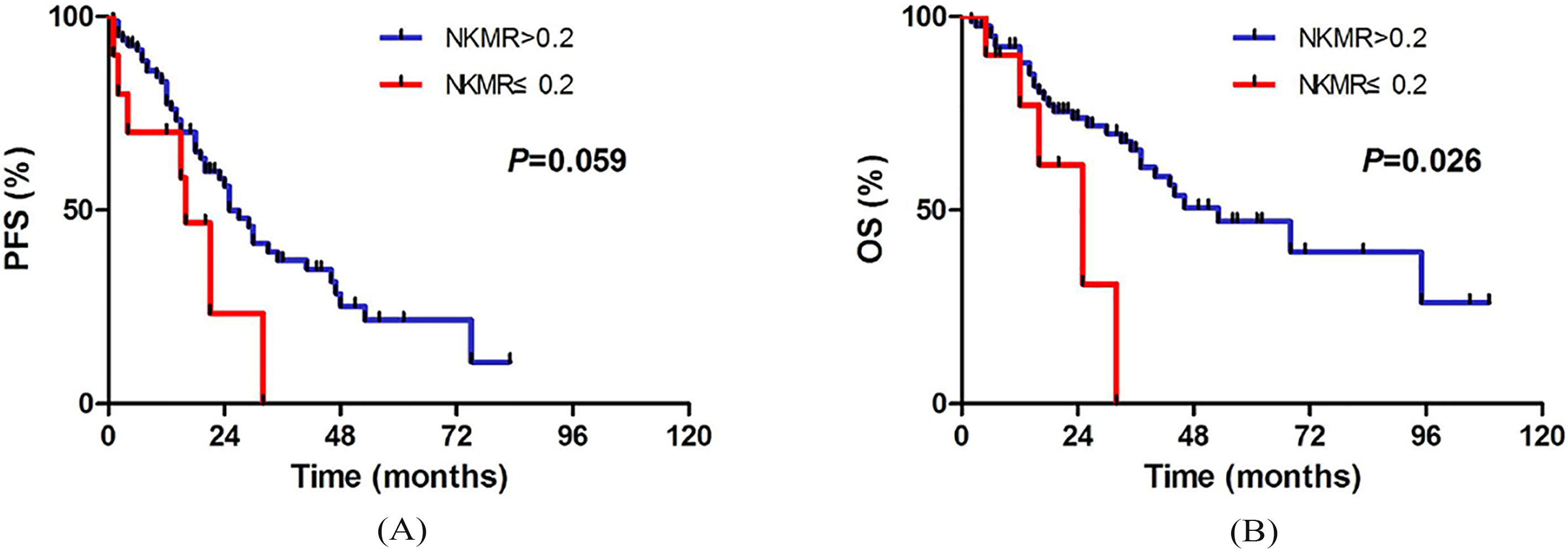
Tables 3 and 4 summarize the univariable and multivariable Cox regression analyses of PFS and OS for potential risk predictors in MCL. In consideration of the limited data, only six cases in our cohort had stage I/II disease, so we did not evaluate the influence of staging. Univariable Cox regression analysis showed that B symptoms (
We investigated the influence of ANKC at diagnosis combined with sMIPI and age for MCL patients. In subgroup analysis of sMIPI score, we separated our 92 MCL patients into two categories: 70 in the low- to intermediate-risk group (sMIPI 0–5) and 22 in the high- risk group (sMIPI 6–11). We conducted a survival analysis for OS based on specific prognostic risk, as is illustrated in Fig. 2. Obviously, low ANKC was a significant risk predictor for patients in low- to intermediate-risk group (
Moreover, as is reflected in Fig. 3, patients who received chemotherapy including rituximab obtained a better OS in high ANKC group compared to low ANKC group (
Discussion
To our knowledge, the host immune system and tumor microenvironment covering lymphoma cells, stromal cells, monocytes/macrophages and immune cells play extremely vital parts in the pathogenesis and progress of lymphoma. As far as monocyte is concerned, von Hohenstaufen et al. defined 0.5
Given the importance of NK cells in the tumor microenvironment, great concern has been focused on allogeneic NK cell therapy in several preclinical or clinical trials targeting breast, ovarian, non-Hodgkin lymphoma and non-small cell lung cancer [21, 22]. Iliopoulou et al. revealed that repetitive infusions of allogeneic, in vitro activated and expanded with interleukin-15 (IL-15)/hydrocortisone (HC) NK cells, in combination with chemotherapy are safe and potentially clinically effective in non-small cell lung cancer [22]. Additionally, Ishikawa et al. demonstrated that NK cell-rich effector cells could be expanded ex vivo from peripheral blood mononuclear cells (PBMCs) in patients with recurrent malignant glioma and that NK cell therapy was safe and partially effective in patients suffering this malignancy [23]. Thus, appropriate NK cell therapy might be applied in MCL in future with more clinical evidence.
In our cohort, the majority of MCL patients took rituximab-containing regimens, which give us a feasible interpretation for the possible connection between low ANKC and poor OS. As previously stated, the OS was better in the high ANKC group among patients took rituximab-containing regimens. Rituximab, is a well-known murine/human chimeric monoclonal antibody (mAb) that binds to CD20 on the surface of human B cells. Substantial evidences have described that rituximab exerts excellent anti-neoplastic effect in malignant B cell lymphomas, involving four signaling pathways: antibody-dependent cell-mediated cytotoxicity (ADCC), complement-dependent cytotoxicity (CDC), direct signaling triggering apoptosis, and increased sensitivity to chemotherapy [24]. Among these pathways, ADCC seems to have overwhelming advantage at anti-tumor activity, by which rituximab recruits NK cells towards malignant B cells via CD16, and the NK cells subsequently kill the malignant cells. Besides, CD16 can activate NK cells locally, producing cytokines which enhance ADCC mediated by other receptors and other cells [25]. Veeramani et al. also confirmed that NK activation occurs within 4 hours of rituximab infusion in subjects with the high-affinity CD16 polymorphism but not those with the low-affinity CD16 polymorphism [26]. Therefore, it makes sense theoretically that higher ANKC and rituximab-induced cytotoxicity are more associated with the preferable OS. These results may remind us many new agents and combinations targeting the activation of NK cells, such as lenalidomide and ibrutinib. Lenalidomide is reported to be an immunomodulatory agent clinically beneficial for patients with relapsed or refractory MCL. Hagner et al. demonstrated that lenalidomide enhanced NK cell-mediated cytotoxicity against MCL cells primarily via increased lytic immunological synapse formation and secretion of granzyme B instead of direct cytotoxic effects against MCL cells [27]. Furthermore, several studies have proved that lenalidomide and ibrutinib play a vital role in augmenting rituximab-dependent NK cell-mediated cytotoxicity [28, 29], which may provide beneficial strategies for MCL patients.
In this retrospective study, our findings suggested that the ANKC value 0.1
In conclusion, we aimed to demonstrate that a low ANKC (
Footnotes
Acknowledgments
This study was supported by National Natural Science Foundation of China (81470328, 81600130, 81770166, 81720108002), Jiangsu Province’s Medical Elite Programme (ZDRCA2016022), Project of National Key Clinical Specialty, National Science and Technology Pillar Program (2014BAI09B12), Jiangsu Provincial Special Program of Medical Science (BL2014086 and BE2017751) and National Science and Technology Major Project (2017ZX09304032).
Conflict of interest
The authors declare no conflict of interest.
