Abstract
BACKGROUND:
Amplified in breast cancer 1 (AIB1) is a candidate oncogene in human breast cancer, which has been identified to be amplified and overexpressed in several types of other human cancers. Abnormalities of AIB1 and its clinical/prognostic significance, however, in upper tract urothelial carcinoma (UTUC) remain unclear.
OBJECTIVE:
To explore what role AIB1 plays in upper tract urothelial carcinoma.
METHODS:
The expression of AIB1 was analyzed using immunohistochemical staining in 133 UTUC patients. Overall, cancer specific and recurrence-free survival rates (OS, CSS, and RFS) were estimated using the Kaplan-Meier method. Multivariable COX regression models containing relevant clinicopathological variables addressed the prediction of postoperative outcome.
RESULTS:
High AIB1 expression was observed to be associated with increased hazard ratios for 5-year CSS (80.6% vs. 55.8%,
CONCLUSIONS:
AIB1 can be used as an independent molecular marker for the prognosis of clinical outcomes of UTUC.
Introduction
Relationship between AIB1 expression and clinicopathological parameters
Relationship between AIB1 expression and clinicopathological parameters
SD: standard deviation.
Urothelial carcinomas are derived from the urothelium and have been classified into two different types, bladder cancer and upper tract urothelial carcinoma (UTUC), according to their tumor location. UTUC is a heterogeneous disease that constitutes approximately 5% of urothelial carcinomas [1]. UTUC incidence has increased in the last 30 years and has been shown to be prevalent in patients who are approximately 70 years old [2]. UTUC has a tendency to relapse and is associated with a generally poor clinical prognosis. Tumors with invasive stage, nodular involvement, metastasis, and residual tumor following surgery are associated with poor clinical outcomes [3]. Therefore, a substantial amount of research regarding UTUC has focused on the discovery of sensitive molecular markers present in UTUC cells that could function as reliable prognostic factors. Several studies have investigated the prognostic value of factors involved in the cell cycle, apoptotic pathways, cell adhesion, metastasis, and microsatellite instability (MSI) [4]. However, the reported specific molecular and/or genetic alterations in UTUC that have prognostic significance remain limited.
The amplified in breast cancer 1 (AIB1) gene, also referred to as SRC-3, p/CIP, RAC3, ACTR, and TRAM-1, is isolated from chromosome 20q and has been identified as a human oncogene [5]. AIB1 has been demonstrated to be frequently amplified and overexpressed in human breast [5], prostate [6], ovarian [7], esophageal [8], gastric [9], and hepatocellular cancers [10]. AIB1 amplification and expression in bladder cancer has been previously studied by our research group, and we have demonstrated that upregulation of AIB1 is associated with poor patient prognosis in patients with bladder cancer [11]. However, significant differences were identified regarding tumor location and behavior between the upper and lower urinary tract [4]. Thus, further research is required regarding the molecular state of the AIB1 gene in UTUC and the prognostic significance of this state. In this study, we examined AIB1 expression in UTUC using immunohistochemistry (IHC), and studied its correlation with clinicopathological features and prognosis. The primary goal of this study was to study whether AIB1 activity possesses prognostic value in UTUC patients.
Study design and patient selection
Surgical specimens from 133 patients who underwent operation for UTUC in our hospital from 2000 to 2016 were used. Of the 133 UTUC patients selected, a total of 83 had invasive UTUC (pT2-4) and the remaining 50 UTUC patients had superficial UTUC (pTa-1). We identified 83 male and 50 female patients in this study. The median patient age was 66 years (mean 64.7
All surgical specimens were processed according to standard pathological procedures and all slides were retrieved and re-reviewed by genitourinary pathologists to confirm the original diagnosis. Tumors were staged according to the American Joint Committee on Cancer-Union Internationale Contre le Cancer TNM classification. Tumor grading was assessed according to the 1998 WHO/International Society of Urologic Pathology consensus classification. The representative paraffin block was selected to freshly cut into 4-
IHC
IHC study was performed using a standard strep- tavidin-biotin-peroxidase complex method [8]. Briefly, 4-
Positive AIB1 expression in UTUC exhibited a primarily nuclear pattern. AIB1 expression levels were assessed using a semiquantitative scoring system and the AIB1 score was calculated using the following formula: staining intensity
Statistical analysis
All analyses were performed using SPSS version 20.0 (IBM Corp., Armonk, NY, USA), GraphPad Prism 5 (GraphPad Software, Inc., La Jolla, CA, USA), and R for Windows, version 3.3.1 (R Foundation for Statistical Computing, Vienna, Austria). The association between AIB1 protein expression and UTUC patient clinicopathological features was assessed by the
Immunohistochemical stainings of AIB1 in upper urinary tract urothelial carcinoma. (a) AIB1 score 0 points (negative staining), (b) Low expression of AIB1 (score 3 points), (c) High expression of AIB1 (score 9 points).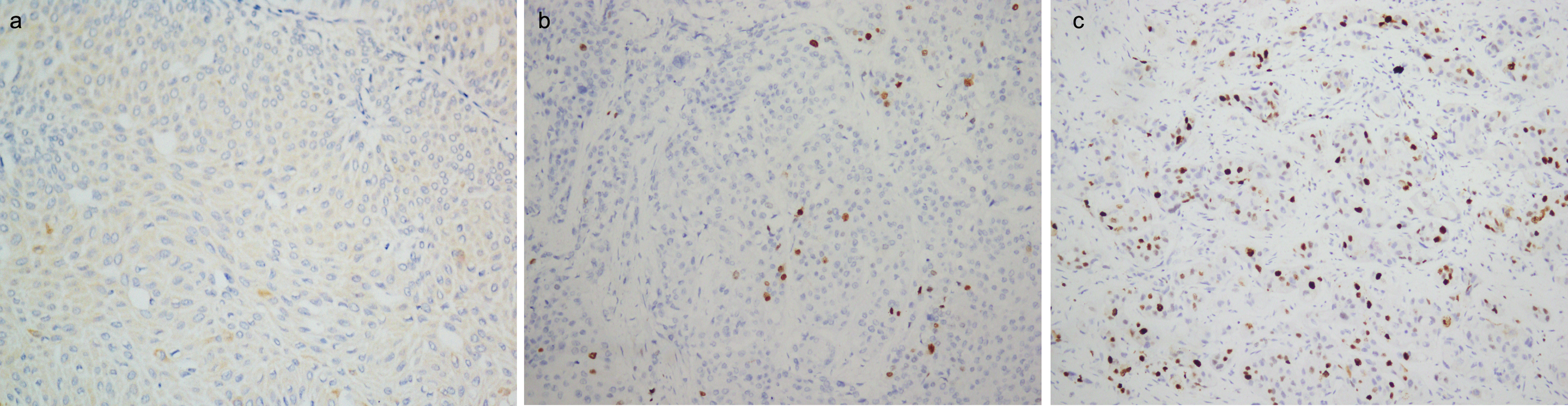
Kaplan-Meier analysis of overall survival, cancer-specific survival and recurrence-free survival of patients following surgery for UTUC according to AIB1 expression (a-c) and according to the prognostic models with AIB1 expression, pT, pN, and hydronephrosis status that are stratified (g-i); ROC curves comparing the predictive accuracy by the combined AIB1, pT, pN, and hydronephrosis status model, the AIB1 alone model, the pT model, and the pN alone model for overall survival (d), cancer-specific (e) and recurrence-free survival (f).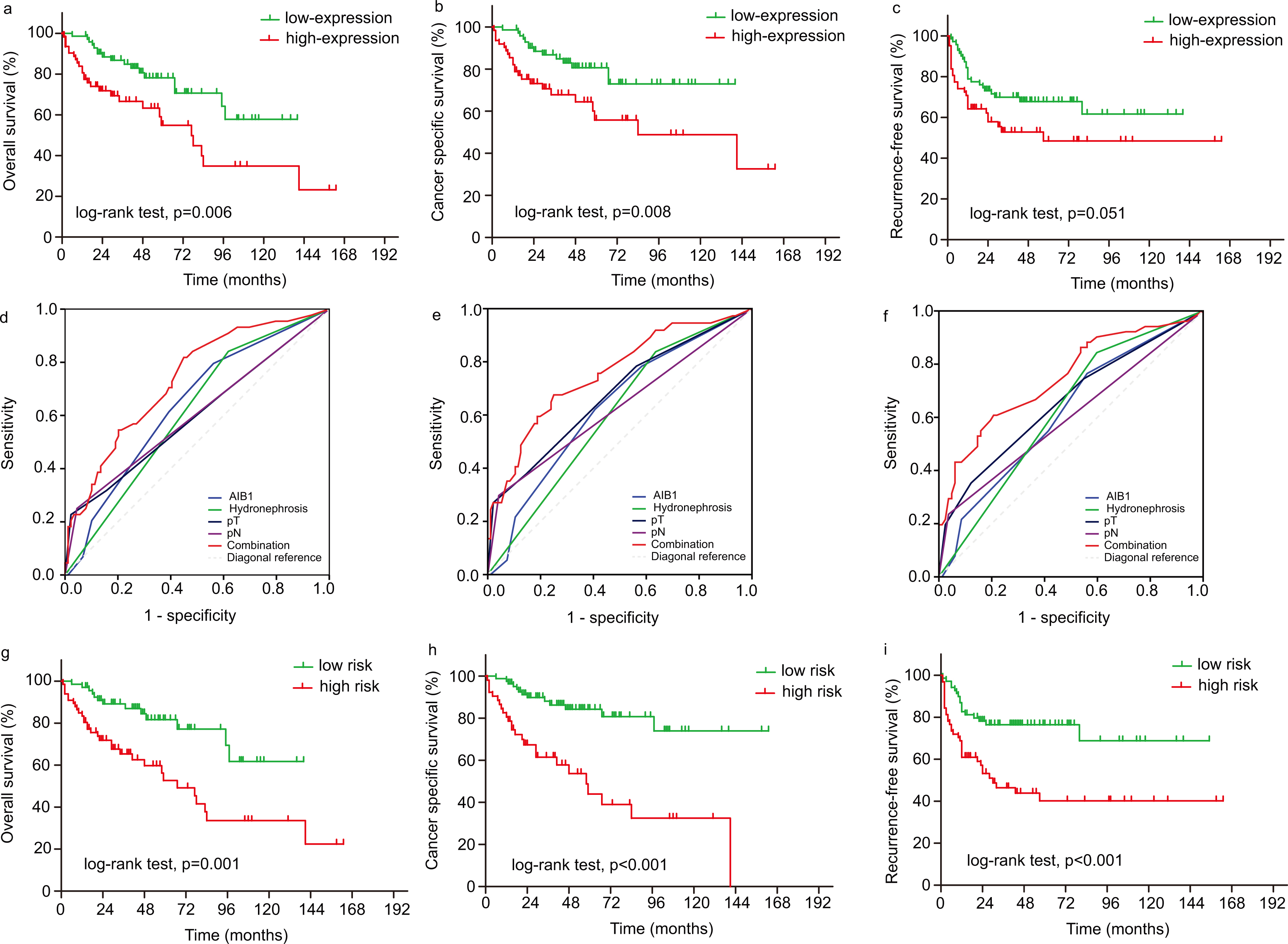
Patient characteristics
All specimens were fixed in 10% formalin and embedded in paraffin. IHC was performed on all slides using anti-AIB1. According to the cut-off value (score 4), patients were divided into low AIB1 (
Univariate and multivariate Cox regression analysis of potential prognostic factors for prediction of overall, cancer specific and recurrence-free survival in patients treated radical nephroureterectomy for upper tract urothelial carcinoma
Univariate and multivariate Cox regression analysis of potential prognostic factors for prediction of overall, cancer specific and recurrence-free survival in patients treated radical nephroureterectomy for upper tract urothelial carcinoma
HR: hazard ratio; 95%CI: 95% confidence interval.
In the low AIB1 expression group, median survival was 46 months (range 6 to 140), whereas in the high AIB1 expression group the median survival was determined to be 25 months (range 1 to 163). Median recurrence-free survival in the low and high AIB1 expression groups was observed to be 41 months (range 2 to 140) and 22 months (range 1 to 163), respectively. Statistically significant differences were observed between the low and high AIB1 expression groups in the 5-year cancer specific survival rate (80.6% vs. 55.8%,
Independent prognostic factors of UTUC: Multivariate cox regression analysis
The proportional hazards assumption was accepted for OS (
The areas under ROC curves (AUC) of the combined AIB1, pT, pN and hydronephrosis status model, the AIB1 alone model, the pT model, and the pN alone model for overall survival, cancer-specific and recurrence-free survival
The areas under ROC curves (AUC) of the combined AIB1, pT, pN and hydronephrosis status model, the AIB1 alone model, the pT model, and the pN alone model for overall survival, cancer-specific and recurrence-free survival
Nomograms to predict overall survival probability (a), cancer-specific survival probability (b), and recurrence-free survival probability (c) at 2, and 5 years for UTUC patients. OS: Overall survival, CSS: Cancer-specific survival, RFS: Recurrence-free survival.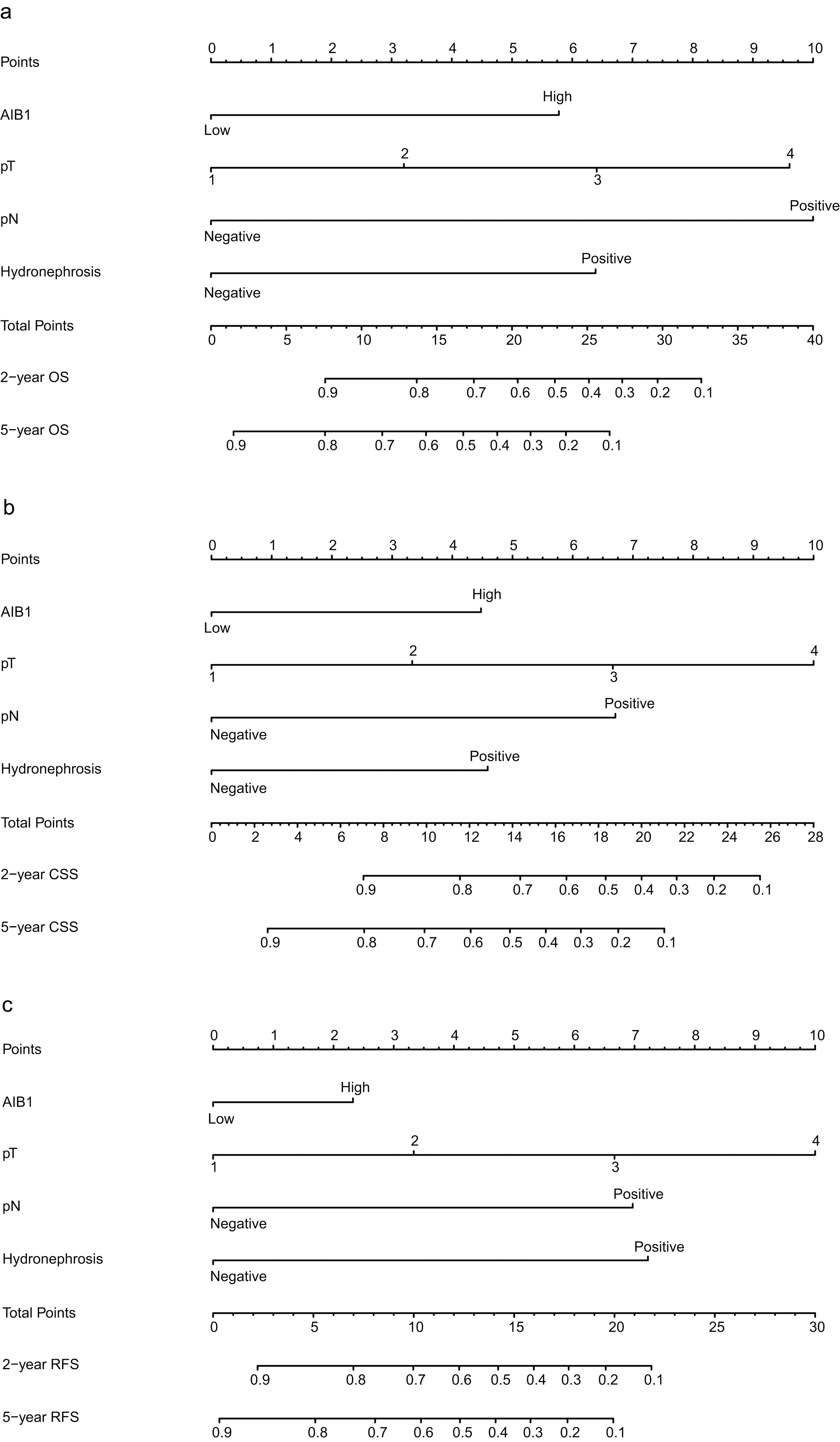
Calibration plots demonstrate virtually ideal predictions for 2, and 5-year overall survival (a), cancer-specific survival (b), and recurrence-free survival (c), respectively.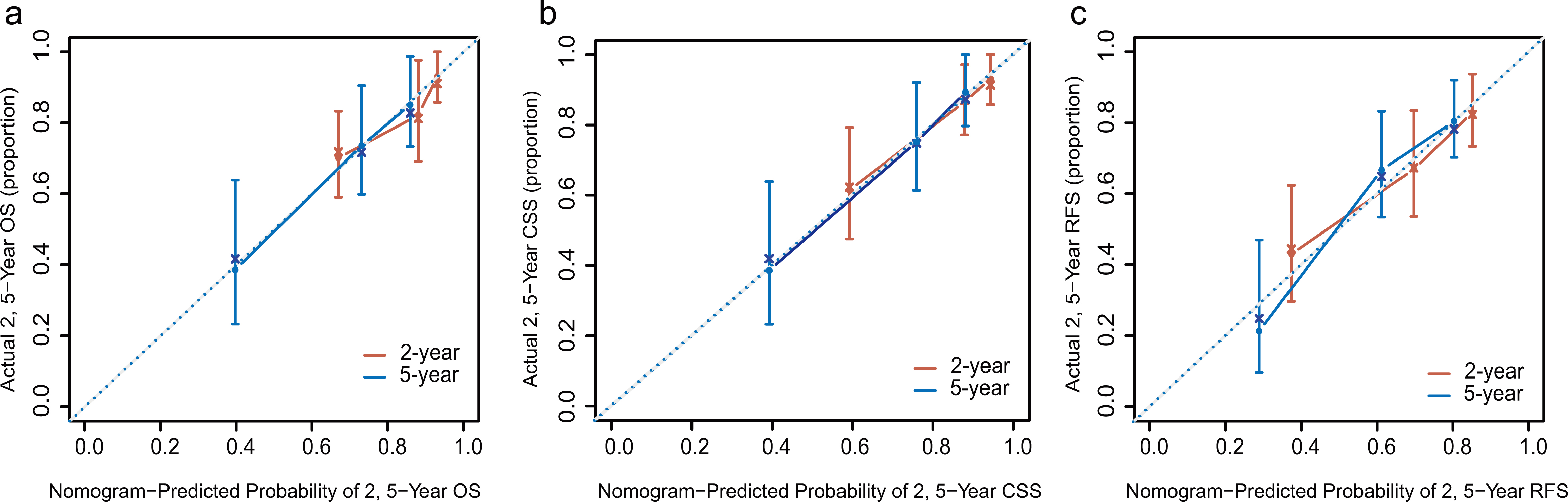
We applied the ROC curve analysis in order to explore the predictive value of AIB1 expression in regards to overall survival, recurrence-free survival, and cancer specific survival. A prognostic model was generated to combine AIB1 with three independent prognostic factors (tumor stage, pN status and hydronephrosis) using Cox proportional hazards regression (the model for overall survival
Survival and recurrence analysis for risk stratification
Based on the median scores of the three prognostic models described above, patients were stratified into low- and high-risk groups. Results from these studies demonstrated that patients in low-risk group exhibited a much better overall survival rate, 5-year cancer specific, and recurrence-free survival rate compared with those patients in the high-risk group (81.7% vs. 52.7%; 84.2% vs. 43.9%; 76.1% vs. 40.2%, respectively, Fig. 2g–i). These results demonstrate that these three prognostic models with AIB1 could practically identify patients with UTUC who had poor clinical outcome and needed further adjuvant chemotherapy.
Construction of the prognostic nomograms
We developed three competing-risk nomograms to predict the probability of overall survival, cancer specific survival, and intravesical recurrence at 2, and 5 years. The models were based on AIB1, hydronephrosis, tumor stage, and pN status. Calibration plots demonstrated that the nomograms compared well with an ideal model (Figs 3 and 4), and the c-index of the nomograms for the predictions of overall survival, cancer specific survival, and recurrence-free survival was 0.723, 0.747, and 0.716, respectively. According to these nomograms, a patient with a T2 UTUC, high AIB1 expression, no hydronephrosis, and no lymphatic metastasis received a total of 8.9 points, corresponding to a greater than 85% overall survival rate for 2 years following surgery. A patient with T3 UTUC, high AIB1 expression, hydronephrosis, and no lymphatic metastasis accumulated 15.9 points with a 63% of 2-year cancer specific survival rate, and about 38% of 5-year cancer specific survival rate. This same patient also accumulated 16.4 points with a 60% risk of tumor recurrence in 2 years following surgery, and a greater than 70% risk for 5-year recurrence.
Discussion
At present, current pTNM staging and histopathological grading systems are established for prognosis prediction in patients with UTUC [12]. However, these parameters may have reached their limits in providing critical information that influences patient prognosis and treatment strategies. Patients who stay at the same clinical stage and/or histological grade often present considerable variability in terms of disease recurrence and survival [13, 14]. Therefore, there exists an urgent need for the development of novel variables that can function to distinguish between patients with an unfavorable prognosis and other patients with a better prognosis. As is known, patients’ survival might be prolonged by more effective adjuvant therapy, if individuals with different prognosis could be identified by molecular biomarkers at the time of surgery [15, 16]. Therefore, further studies are required for the development of appropriate UTUC biomarkers.
The AIB1 gene is located at 20q12 and has been reported to promote proliferation in hormone-dependent cancers and some hormone-independent cancers [5, 8, 9, 17]. Though our previous studies have observed that in bladder cancer, AIB1 overexpression contributes to a shortened survival by promoting bladder cancer cell proliferation through the AKT pathway and acting as coactivator of E2F1 in non-muscle-invasive bladder cancer [11, 18]. However, recent studies have reported different genetic presentations between upper and lower urinary tract urothelial carcinoma [19], and the role of AIB1 in UTUC remains unclear. In order to study the potential oncogenic effects of AIB1 in UTUC, we initially studied AIB1 expression using IHC in a cohort of UTUC tissue samples. The key findings in this study were that AIB1 was much more frequently overexpressed in UTUC and was strongly linked with overall survival, cancer specific survival, and recurrence free survival in patients with UTUC. These findings present a potentially critical role of AIB1 as an underlying biological mechanism related to UTUC recurrence and/or progression. Therefore, AIB1 expression analysis can be applied in order to optimize individual UTUC therapy management.
UTUC in our series presented aggressive behavior, as 63.2% of our patients exhibited a pejorative clinical outcome. Patients with invasive tumors typically have a poor survival rate and have a distinct tendency to relapse, with work suggesting that the tumor stage is a primary prognostic factor [20]. In a study carried out by Ng et al. [21] preoperative clinical features were controlled and it was confirmed that hydronephrosis was independently associated with cancer metastasis and cancer specific survival by preoperative multivariable analysis. Chung et al. [22] graded hydronephrosis as none/mild vs. moderate/severe, and demonstrated degree of hydronephrosis in patients with advanced UTUC was associated with local/systemic recurrence-free survival (HR
The accurate estimation of treatment success, recurrence, and long-term morbidity is essential for both physicians and their patients to make informed medical decisions regarding the management of their disease [14]. To obtain the most accurate and reliable predictions of outcomes, we designed nomograms with AIB1 to predict overall survival, distant or metastatic recurrence, and cancer-specific mortality following surgery, respectively. We observed that our nomograms were well calibrated, as the slopes of the calibration curves were close to 1. With our accurate, well-calibrated predictive models, it is helpful to improve clinicians’ abilities to become familiar with the strengths and weaknesses of the predicted probabilities according to their distribution. These results enrich the present findings from the current literatures [14, 24].
The main limitation of our study centers on the retrospective analysis of data which was collected from a single center. Therefore, the number of cases studied is small. External validation of the current nomograms is required before widespread application. However, we still believe that our postoperative nomograms of tumor recurrence and cancer specific survival are useful for the consideration of the management of postoperative chemotherapy and can provide meaningful counseling for the patients. In addition, lymphovascular invasion, surgical margins and tumor necrosis have been proved significant postoperative factors, [25, 26, 27] and it might be more effective to add these factors to the nomograms. However, in this retrospective study, these pathological factors were not available for all patients.
Conclusion
In the current study, we have identified certain factors that predict poor clinical outcome for UTUC. High AIB1 expression was found to be an important predictor for shortened cancer specific and recurrence-free survival in patients with UTUC. We identified a mixed panel of four markers (AIB1, hydronephrosis, tumor stage, and pN status) that proved to be a powerful predictor of UTUC recurrence. Further prediction models can be used for patient counseling and selecting postoperative systemic therapy.
Footnotes
Acknowledgments
This study was funded by National Key Research and Development Program of China (Grant/Award Number: 2016YFC0902600), Guangdong Provincial Science and Technology Foundation (Grant/Award Number: 2014B020212015, 2017B020227004, 2017A 030313538), National Natural Science Foundation of China (Grant/Award Number: 81372357, 81725016, 81572905, 81602219), Guangdong Medical Science and Technology Research Foundation (Grant/Award Number: A2018040), and the Guangzhou Science and Technology Foundation (201607010238, 2017040 20174).
Conflict of interest
The authors declare that they have no conflict of interest.
