Abstract
Upper tract urothelial carcinoma (UTUC) is a rare malignancy. The standard treatment for localized high-risk disease is radical nephroureterectomy, which confers significant morbidity and is not appropriate for all patients. Patients harboring low-risk, non-invasive disease may be candidates for organ-sparing treatment, which includes endoscopic resection with or without intracavitary drug therapy. Successful administration of intracavitary chemotherapy to the upper tracts is impeded by rapid washout of the agent and short dwell times. This has limited the clinical utility of mitomycin C for treatment of upper tract tumors, despite the successful outcomes observed in low-grade urothelial carcinoma of the bladder. Currently, there is an unmet need for development of a technically feasible and oncologically sound intracavitary therapy for management of low-grade UTUC. UGN-101 (Jelmyto™) is a novel formulation of mitomycin C that uses a unique hydrogel designed to increase urinary dwell time, and thereby efficacy of treatment. Preclinical data demonstrated promising results regarding the safety and feasibility of this agent. Preliminary results of a phase III trial (OLYMPUS study) [ClinicalTrials.gov identifier: NCT02793128] demonstrated the efficacy of UGN-101 as a successful chemo-ablative agent for low-grade upper tract tumors. UGN-101 may represent a pivotal paradigm shift in the treatment of low-grade UTUC. Indeed, the drug has recently been granted approval by the US Food and Drug Administration as the first treatment for low-grade UTUC, which may lead to significant improvements in patient care and a long-awaited decrease in the burden of disease.
Introduction
Upper tract urothelial carcinoma (UTUC) is a rare malignancy, encompassing only 5% of all urologic neoplasms. 1 Treatment of UTUC is dependent on several clinical variables, as staging alone is inaccurate due to the inherent difficulty of obtaining satisfactory upper tract biopsy specimens. Hence, risk-adapted treatment algorithms from current guidelines are driven primarily by stage and grade of disease, although location (pelvis versus ureter), imaging findings, size, cytology, and number of lesions factor into the clinical decision-making process. 2 The current gold-standard therapy for localized, high-risk UTUC is surgical resection in the form of radical nephroureterectomy (RNU) with bladder cuff excision and regional lymphadenectomy. 2 Select patients, such as those with low-risk disease, a solitary renal unit, end-stage renal disease and non-surgical candidates, may be appropriate for endoscopic nephron-sparing surgery (NSS), which includes resection, laser ablation, and intracavitary drug instillation.
The options available for intracavitary drug therapy for the management of UTUC are limited. Bacillus Calmette–Guérin (BCG) and mitomycin C (MMC) have been the most widely investigated, usually as adjuvant therapy with endoscopic ablation or resection. 3 The rarity of UTUC, combined with the inherent technical limitations of upper tract accessibility for drug instillation, have resulted in a limited number of investigations on the topic. Correspondingly, the literature to date is sparse, with a preponderance of retrospective data, small sample sizes, and expert opinions. 3 Consequently, long-term oncologic outcomes remain elusive, although the available data suggest therapeutic equivalency for NSS and RNU for appropriately selected patients.4,5 The paucity of high-level evidence pertaining to this topic is reflected in current guidelines, most of which do not recommend routine use of drug instillation therapy for management of UTUC. 2 To date, there is a clear unmet need for the development of a technically feasible and oncologically sound therapeutic strategy for intracavitary treatment of UTUC.
Recently, UGN-1016–9 (Jelmyto™, formerly MitoGel, UroGen Pharma, Israel) has been approved by the US Food and Drug Administration (FDA) as the first treatment for low-risk UTUC. 10 UGN-101 is a temperature-sensitive, water-soluble gel preparation of MMC composed of a unique combination of polymers.6–9 This hydrogel formulation is liquid at room temperature and converts to a gel at body temperature, thereby conforming to each patient’s distinct pelvicalyceal and ureteric anatomy. Furthermore, dissolution of the formula through continuous urinary bathing results in slow-release, sustained drug delivery. Preliminary clinical trial results are highly favorable, and suggest a durable treatment response for patients with low-risk disease following therapy with UGN-101, which was subsequently confirmed with longer follow up.6–9
Herein, we review preclinical investigations pertaining to the successful development of UGN-101, its implementation into clinical practice and the latest data that support its use as a novel therapy for low-grade UTUC.
Development, safety and feasibility of UGN-101: preclinical data
In contrast to bladder cancer, UTUC has unique anatomic and physiologic considerations that preclude the development and use of effective drug delivery. The major limiting factor to successful intracavitary drug therapy lies in ureteral and pelvic peristalsis, which causes rapid drainage of urine and thus limits exposure of the urothelium to the administered agent. 11 This issue is of particular importance for MMC, as studies have shown a clear correlation between exposure time and efficacious pharmacologic activity.12,13 Hence, the focus of preclinical studies has been to develop a suitable method of intracavitary delivery that achieves appropriate concentration of drug for use in the clinical setting.
Wang et al. described the use of a reverse thermosensitive (RT) polymer plug to augment the dwell time of MMC within the upper tract of a porcine model. 14 The authors deployed the plug agent into the ureter and were successful at maintaining an MMC instillation time of 60 min, which compares with its length of administration in the bladder for successful intravesical therapy. Combination therapy using a thermosensitive hydrogel polymer with MMC was subsequently developed. In a preclinical model, Lifschitz et al. instilled this formulation via retrograde catheters into the renal pelvis of anesthetized swine and determined pharmacokinetic parameters. 15 The authors concluded that dwell time in the upper tract was increased to 4–7 h post instillation without causing significant systemic absorption. Notably, elevated intrarenal pressures recovered quickly, and renal function was not adversely affected by the instillation.
UGN-101 was developed using RTGel as a hydrogel polymer base combined with MMC. 11 The safety and feasibility of this formulation was tested in a preclinical setting using a Yorkshire swine model. 11 In an eloquent three-phase study design, the authors instilled UGN-101 into swine via percutaneous nephrostomy tubes in various settings, including single administration, incrementally increasing doses, and an alternate administration schedule of six twice-weekly instillations. Pharmacokinetic and imaging data was obtained from all animals, and nephrectomy specimens were used for histologic analysis. UGN-101 dwell time was significantly increased compared with MMC control (4–6 h versus minutes, respectively). There was no evidence of renal insufficiency, obstruction, myelosuppression or significant systemic absorption in both the single and serial administration settings. In addition, there were no major concerns identified on histologic analysis. Allergic or anaphylactic reactions were not recorded; however, data from intravesical MMC administration in human subjects suggest that hypersensitivity skin reactions may occur and that systemic toxicity is rare.16,17 In one study using UGN-101 in the upper tracts, only one patient developed pancytopenia. 9 These pharmacologic results were confirmed in an animal study using retrograde instillation of UGN-101 at once-weekly intervals for 6 consecutive weeks. 18
Notable limitations of preclinical trials include the inherent shortcomings of using animal models, and in particular, the absence of a model specific for UTUC. Nonetheless, the promising results of these studies validated the mechanical properties of the agent and generated interest, paving the way for evaluation of this novel agent in the clinical setting. Studies to date evaluating UGN-101 are summarized in Table 1.
Studies evaluating the role of UGN-101 in UTUC.
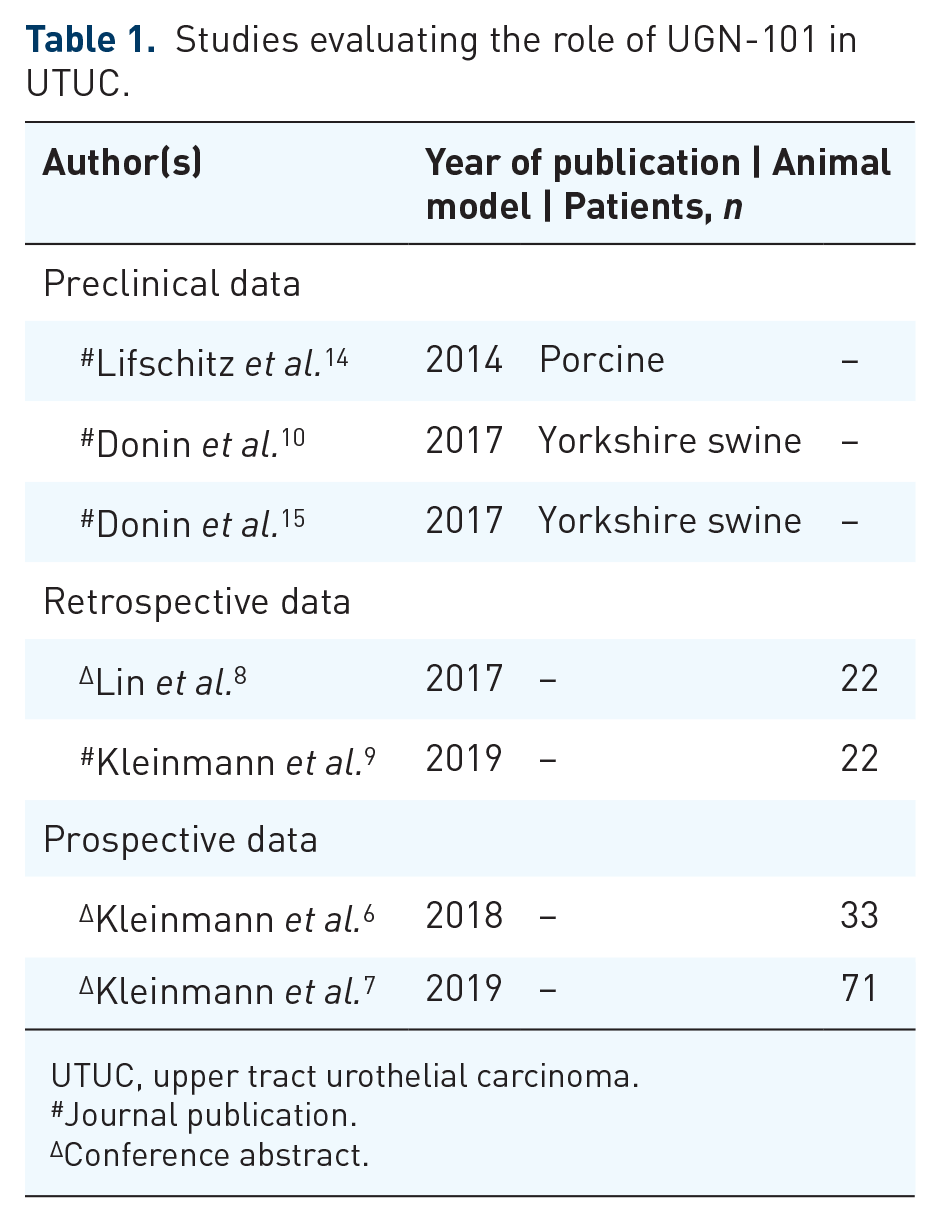
UTUC, upper tract urothelial carcinoma.
Journal publication.
Conference abstract.
Use of MMC and UGN-101 for treatment of UTUC: retrospective series
The role of MMC as a topical chemotherapeutic agent for select cases of UTUC has been investigated in small, retrospective series.19–22 The majority of studies have evaluated MMC in an adjuvant setting, combined with endoscopic tumor ablation or resection. One study investigated the role of an induction and maintenance course of MMC, and reported favorable intermediate- and short-term outcomes. 23 The patient cohorts in these studies are highly selected individuals, mostly with favorable risk disease or those with contraindications to RNU, resulting in an inherent selection bias. The clinical efficacy of MMC remains elusive, with recurrence rates reported to be in the range of 35–68%.19,22 Notably, some series suggest that progression-free survival rates similar to those reported for patients undergoing RNU may be achieved in appropriately selected patients.4,5
Several methods of MMC delivery into the upper tract have been explored, including antegrade administration through a percutaneous route, direct retrograde instillation via a ureteral catheter, and bladder instillation with an indwelling double-J stent, resulting in passive vesico-ureteral reflux. 3 Small sample sizes make direct comparisons between these approaches a challenge; however, it has been suggested that the double-J stent technique confers the least favorable outcomes. 24 One animal study suggested that retrograde instillation via ureteral catheter provided the best retention and coverage of the renal pelvis. 25 As discussed, the major barriers to effective administration of MMC lie in achieving an appropriate dwell time, and assuring drug distribution throughout the entire collecting system. 11 It remains unclear whether the reported outcomes and recurrence rates are reflective of true treatment response, or poor administration technique. Nonetheless, it has been reported that 70% of patients with low and intermediate-risk UTUC undergo RNU, 26 thus highlighting a clear need for the development of successful nephron-sparing treatment options.
The feasibility, efficacy and tolerability of UGN-101 for the treatment of UTUC was evaluated in a multi-institutional retrospective cohort study. 9 A total of 22 patients approved for UGN-101 therapy on a compassionate-use basis from five countries were reviewed. Patients selected for treatment were not candidates for RNU and included those with a solitary kidney, poor surgical candidates, inability to treat tumor with endoscopic ablation therapy, and patients refusing RNU. Most patients (81.8%) were diagnosed with low-grade tumors, and the remainder had high-grade or indeterminate lesions. The treatment regimen consisted of 6-weekly UGN-101 administrations via retrograde ureteral catheter or percutaneous nephrostomy tube. The volume and concentration of the drug were not standardized, but the majority of patients received a dose of 4 mg/cc. Of 16 patients that completed the treatment regimen, eight achieved a complete response (CR), five had a partial response and two patients did not respond. Notably, two patients in the CR group recurred within 12 months of therapy and were treated with endoscopic ablation. Six serious adverse events related to UGN-101 were recorded, which included hydronephrosis, severe inflammation, acute-on-chronic kidney injury, hyperkalemia, and pancytopenia. Limitations of the data include the small sample size, heterogeneity of patient selection and dosing regimens, and short follow-up intervals. Furthermore, given the retrospective nature of the study, results may be prone to reporting and selection bias. Nonetheless, this study is an important proof of concept demonstrating feasibility of chemo-ablation therapy with UGN-101 in UTUC.
Prospective data in support of UGN-101: the OLYMPUS study
OLYMPUS (Optimized deLiverY of Mitomycin for Primary UTUC Study) [ClinicalTrial.gov identifier: NCT02793128] is a phase III, single-arm, open-label study designed to ascertain the safety, efficacy, and tolerability of UGN-101 for treatment of localized UTUC.6,7 A single-arm trial design was used for several reasons. First, the target population is small and therefore randomization would not be feasible. Second, randomization to a comparator arm would not be practical given that there is no standard-of-care alternate treatment available. Lastly, a placebo comparator would be unethical. Hence, the trial was designed as a single-arm phase III with CR as the primary endpoint to demonstrate clinical impact without a direct comparator arm, and adverse event rate as a key co-primary endpoint. Patients with new or recurrent low-grade UTUC located superior to the ureteropelvic junction (i.e. exclusively the pelvis and calyces) measuring between 5 mm and 15 mm in size were eligible for trial enrollment (Table 2). Partial resection was allowed for tumors that were greater than 1.5 cm in size. Exclusion criteria included a diagnosis of high-grade or invasive histology, carcinoma in situ, receipt of BCG therapy or history of invasive urothelial carcinoma within 6 months of trial enrollment, and active treatment with systemic chemotherapy.
BCG, bacillus Calmette–Guérin; CIS, carcinoma in situ; HG, high grade; LG, low grade; UC, urothelial carcinoma; UPJ, ureteropelvic junction; UTUC, upper tract urothelial carcinoma.
The primary objectives of the study were to assess CR and adverse events. CR in the primary objective was defined as no evidence of disease at the primary disease evaluation (PDE), which consisted of ureteroscopy and wash cytology at 4–6 weeks following the last instillation. Secondary endpoints included CR rates at 3, 6, 9 and 12 months, partial response rates, and measurement of MMC plasma levels to evaluate systemic absorption. Treatment consisted of six once-weekly instillations of UGN-101 at a dose of 4 mg MMC per 1 ml of RTGel into the upper tract using retrograde catheters. The dose of 4 mg/ml was chosen, based on previous studies. 9 A maximum volume of 15 ml was based on capacity of renal pelvis and calyces. Patients were also given oral 1.3 g sodium bicarbonate the evening before, morning of and 30 min prior to UGN-101 administration. Patients with a CR at PDE proceeded with monthly maintenance instillations for 12 months or until time of first tumor recurrence, whichever occurred first.
Results from an interim analysis of the OLYMPUS trial were presented at the 2019 American Urological Association Annual Meeting. 7 CR at time of PDE was achieved in 42 patients (59%). Of 41 patients that underwent 6-month surveillance ureteroscopy, 89% demonstrated a durable disease-free response. Mild, moderate, and severe adverse events occurred in 27%, 24% and 34% of patients, respectively. These events included hematuria, urinary tract infection, dysuria, ureteral stenosis, hydronephrosis, systemic symptoms (nausea/vomiting) and flank pain. Two patients experienced a life-threatening event and three patients experienced fatal events; however, these were not attributable to UGN-101 exposure. Final results with longer follow up were just published and consistent with these same outcomes. 9
The OLYMPUS study is the first well-designed, prospective trial to demonstrate a complete and durable chemo-ablative response to intracavitary therapy for low-grade UTUC. Since the unique hydrogel is technically considered a delivery device, the compound is considered a drug–device combination, and indeed, was recently approved by the FDA as the first treatment for low-grade UTUC. 10 The FDA release contained an update on longer term follow up, with 46% of patients maintaining a CR at 12 months. 10 the novel formulation using a hydrogel polymer overcomes previous limitations of intracavitary drug delivery to the upper tract and provides a feasible organ-sparing treatment option, akin to that available for treatment of non-muscle invasive bladder cancer. Second, administration of UGN-101 achieved CR in residual tumors, validating the novel chemo-ablative paradigm. Results from the OLYMPUS trial suggest that UGN-101 administration may have prevented 48% of patients from undergoing invasive surgery. If the trial data remain robust, this could lead to a practice-changing development for low-grade UTUC.
There are several limitations to the existing literature regarding UGN-101. Results from even longer follow-up intervals are required to assess the rates of disease recurrence, progression, treatment failure, and ultimately, requirement for RNU. The ideal trial would be a randomized design, wherein patients with low-grade UTUC are randomized to repeat endoscopic tumor ablation or chemo-ablation with UGN-101 with a primary outcome of progression to RNU. However, the feasibility of such a study design is limited and unlikely to be developed, given the rarity of the disease. Moreover, it is the authors’ experience that lower pole calyx tumors can be very difficult to treat owing to angulation and need for ureteral catheter deflection to administer the compound into the calyx. Lastly, long-term sequelae of severe adverse events, notably ureteral stenosis and hydronephrosis, and their prevention, will require further study.
Conclusion and future directions
To date, intracavitary treatment options for UTUC are limited and lack support from prospective trials. Given its recent FDA approval, UGN-101 is an important addition to the armamentarium of currently available treatment options for low-grade UTUC. The most significant impact may be on patients with contraindications to RNU, such as those with chronic renal insufficiency, bilateral upper tract involvement, hereditary predisposition to recurrent lesions (i.e. hereditary nonpolyposis colorectal cancer) or those with significant medical comorbidities rendering them poor candidates for invasive surgery. The use of UGN-101 in an adjuvant setting to complement minimally invasive endoscopic treatment has not been explored, and further studies to address this role are required. The technique required to administer UGN-101 does require specific training and attention to some important nuances, but once mastered, delivery is simple, utilizes minimal resources (other than fluoroscopy and a cystoscopy suite), and does not require a steep learning curve. For these reasons, we believe it will be adopted into the urologic oncologic community with ease. Furthermore, the indications for use of this unique hydrogel formulation may be expanded to intravesical instillation for the management of non-muscle invasive bladder cancer. An ongoing prospective phase IIb study [ClinicalTrials.gov identifier: NCT03558503] was designed to determine the safety and efficacy of UGN-102, a formulation combining a thermal hydrogel and MMC, for patients with low-grade non-muscle invasive bladder cancer.
In conclusion, UGN-101 is the first-ever FDA-approved treatment for low-grade UTUC and may represent a pivotal paradigm shift with significant implications for improving patient care and decreasing the burden of disease. UGN-101 is likely to become implemented as the new standard of care. Key points are summarized in Table 3.
Key points.

FDA, US Food and Drug Administration; LG, low-grade; MMC, mitomycin C; UTUC, upper tract urothelial carcinoma.
Footnotes
Conflict of interest statement
SFM was a member of the Urogen Scientific Advisory Board (ended January 2019) and is a member of the Taris Bio Scientific Advisory Board. A.K. has no disclosures.
Funding
The authors disclose the following sources of funding for the research, authorship, and/or publication of this article: this work was supported in part by the Monteleone Family Foundation for Research on Kidney and Bladder Cancer, and the Eleanor and Scott Petty Fund for UTUC.

