Abstract
Abnormally expressed microRNAs (miRNAs) contribute widely to human cancer, including oral squamous cell carcinoma (OSCC), by regulating their downstream targets. MiR-223 has been proved to be up-regulated in both gastric cancer and ovarian cancer. However, the effect of miR-223 on OSCC is still unclear. Here, we showed that miR-223 was over-expressed in OSCC tissues using qRT-PCR. Next, we investigated the biological mechanism of miR-223 in OSCC. The results demonstrated that miR-223 facilitated the cell proliferation and migration of OSCC using MTT assay and Transwell assay. Furthermore, we stated that the FBXW7 expression was decreased in OSCC and re-expression of FBXW7 inhibited the proliferation and migration of OSCC. In addition, FBXW7 mimic inversed the promotion effect of miR-223 in regulating of OSCC cells. In short, miR-223 promoted OSCC cell proliferation and migration by downregulating FBXW7, which provided a novel therapeutic strategy for OSCC.
Introduction
Oral cancer is a common malignant tumor appearing in mouth, and oral squamous cell carcinoma (OSCC) accounts for about 90% of them [1]. Previous studies have shown that even after available treatment, over 50% of patients might die within five years due to the related complications, and the prognosis of OSCC remains dismal [2]. Therefore, it is necessary to carry out in-depth studies to reveal the internal mechanism of tumor occurrence and look for possible treatment methods of OSCC.
MicroRNAs (miRNA) are evolutionary conservative non-coding small molecules. They are encoded by the higher eukaryotic genome and regulate their expression by binding to the 3’ untranslatable region (3’UTR) of the target genes [3]. There were increasing evidences that abnormal expression of miRNAs caused a variety of cancers, including oral cancer. For example, the expression level of miR-381-3p was significantly decreased in oral squamous cell carcinoma and it suppressed OSCC cell proliferation [4]. MiR-155 acted as an oncogene in OSCC tissues, regulating cell cycle and apoptosis [5]. MiR-373-3p promoted tongue squamous cell carcinoma metastasis by targeting DKK1 [6]. Previous studies have demonstrated that miR-223 was up-regulated in oral cancer [7, 8], but the specific biological function of miR-223 in OSCC was not well understood.
FBXW7 was first discovered in drosophila as a regulatory protein for cell cycle and was named CDC4, acting as an important tumor suppressor gene found recently. FBXW7 is critical for the regulation of human cell cycle, cell growth and cell differentiation, and its deletion can lead to or accelerate the proliferation of cancer cells and increase poor prognosis [9, 10, 11]. Previously studies have shown that the absence of FBXW7 could lead to poor prognosis for colorectal cancer, esophageal cancer and gastric cancer [12, 13, 14]. Recently, some studies have shown that FBXW7 was a target gene of miRNAs in regulating different cancers. For instance, miR-223 regulated the acute lymphoblastic leukemia by inhibiting of FBXW7 [15]. MiR-27a promoted the growth of lung cancer cells via suppressing FBXW7, suggesting that FBXW7 played an important role in human tumorigenesis as a tumor suppressor [16]. Moreover, FBXW7 acted as a target of miR-23a in regulating of colorectal cancer cells proliferation [17]. Nevertheless, the regulation of miR-223 in OSCC by targeting FBXW7 is not reported ever.
The current study stated that miR-223 was over-expressed in OSCC and could regulate the cell proliferation and migration. Moreover, we first proved that miR-223 directly targeted FBXW7 in regulation of OSCC.
Material and method
Tissue samples
Fifty paired cancerous (tumor center) and corresponding adjacent tongue epithelial tissue samples were obtained from OSCC patients who underwent surgery in the Yantai Yuhuangding Hospital. All patients signed informed consent before sample collection and were untreated before surgery. All samples were stored at
Cell culture
SCC-15 (ATCCCRL-1623), SCC-4 (ATCC CRL-1624) and HSC-3 (BNCC341400) cell lines were purchased from American Type Culture Collection (ATCC). OECM1 cells were generous gifts from the National Defense Medical Center. CGHNC9 cells and normal keratinocyte cells CGHNK2 were obtained from Chang Gung Memorial Hospital. Normal keratinocytes CGHNK2 were cultured and maintained in Keratinocyte Serum Free Medium (K-SFM) (Life Technologies, USA). The oral cancer cell lines were cultured in RPMI-1640 with 10% fetal bovine serum (Gibco, USA), penicillin (100 U/ml) and streptomycin (100
Cell transfection
The miR-223 mimic, miR-223 inhibitor or control mimic, control inhibitor used in this study was purchased from GenePharma (Shanghai, China). OSCC cells were transfected with miRNA mimic or miRNA inhibitor using the Lipofectamine 2000
Western blot assay
Total proteins were extracted from cells by RIPA lysis containing proteinase inhibitors (Beyotime, China) and phenylmethanesulfonyl fluoride (PMSF). The protein concentration was detected by BCA reagent kit (Merck). Total proteins (50
RNA isolation and qRT-PCR
TRIzol reagent (Invitrogen) was used to isolate total RNA from cells. All-in-One
Methyl thiazolyl tetrazolium (MTT)
MTT assay was used to detect cell proliferation. The cells with different transfection were added into 96-well plates and cultured for 0, 24, 36, 48, 96 h, respectively. Then 20 ul MTT medium was added to each well. After incubation at 37
Transwell assay
Cell migration ability was performed using transwell assay. The transwell chamber with 8
Dual luciferase reporter assay
The recombinant pMIR-reportor luciferase vector was used for FBXW7 3’UTR luciferase assays. The wild-type and mut-type miR-223 putative targets on FBXW7 3’UTR were constructed into the pMIR-reporter luciferase vector, OECM1 cells were transfected with control mimic or miR-223 mimic using Lipofectamine 2000. Then, the Dual Luciferase Reporter Assay System (Promega, USA) was used to measure the luciferase activity after transfection for 48 h.
Statistical analysis
All experiments were repeated in triplicate. SPSS v.19.0 software was used to perform statistical analyses and GraphPad Prism 5.02 Software was used to complete graph presentations. Results are represented as the mean
Results
MiR-223 was frequently up-regulated and FBXW7 was down-regulated in OSCC cell lines and tissue specimens
We used real-time PCR to quantify the miR-223 expression level in OSCC cell lines and 50 paired OSCC oral squamous cell carcinoma tissues, as results shown in Fig. 1A and B, miR-223 expression was increased in OSCC cell lines and OSCC tissues compared with the normal ones. We also found that, compared with the normal cells lines and normal tissues, FBXW7 expression levels were down-regulated in OSCC cell lines and OSCC tissues (Fig. 1C and D). Regression analysis showed the inverse correlation between FBXW7 and miR-223 expression level in 50 paired OSCC specimens (Fig. 1F). Furthermore, FBXW7 was correlated with the prognosis of patients (Fig. 1E). In addition, as we saw in Table 1, miR-223 was highly related to stage and tumor size. The results above indicated that miR-223 was correlated with OSCC progression.
Relationship between miR-223 expression and their clinic-patho- logical characteristics of OSCC patients
Relationship between miR-223 expression and their clinic-patho- logical characteristics of OSCC patients
Statistical analyses were performed by the
Increased miR-223 expression and decreased FBXW7 expression in OSCC cell lines and tissues. (A and B) Detection of miR-223 expression in OSCC cell lines and tissue samples by qRT-PCR. (C and D) Detection of FBXW7 mRNA expression in OSCC cell lines and tissue samples by qRT-PCR. (E) The relationship between FBXW7 expression and survival. (F) Regression analysis of negatively correlation of FBXW7 and miR-223 expression in 50 OSCC samples (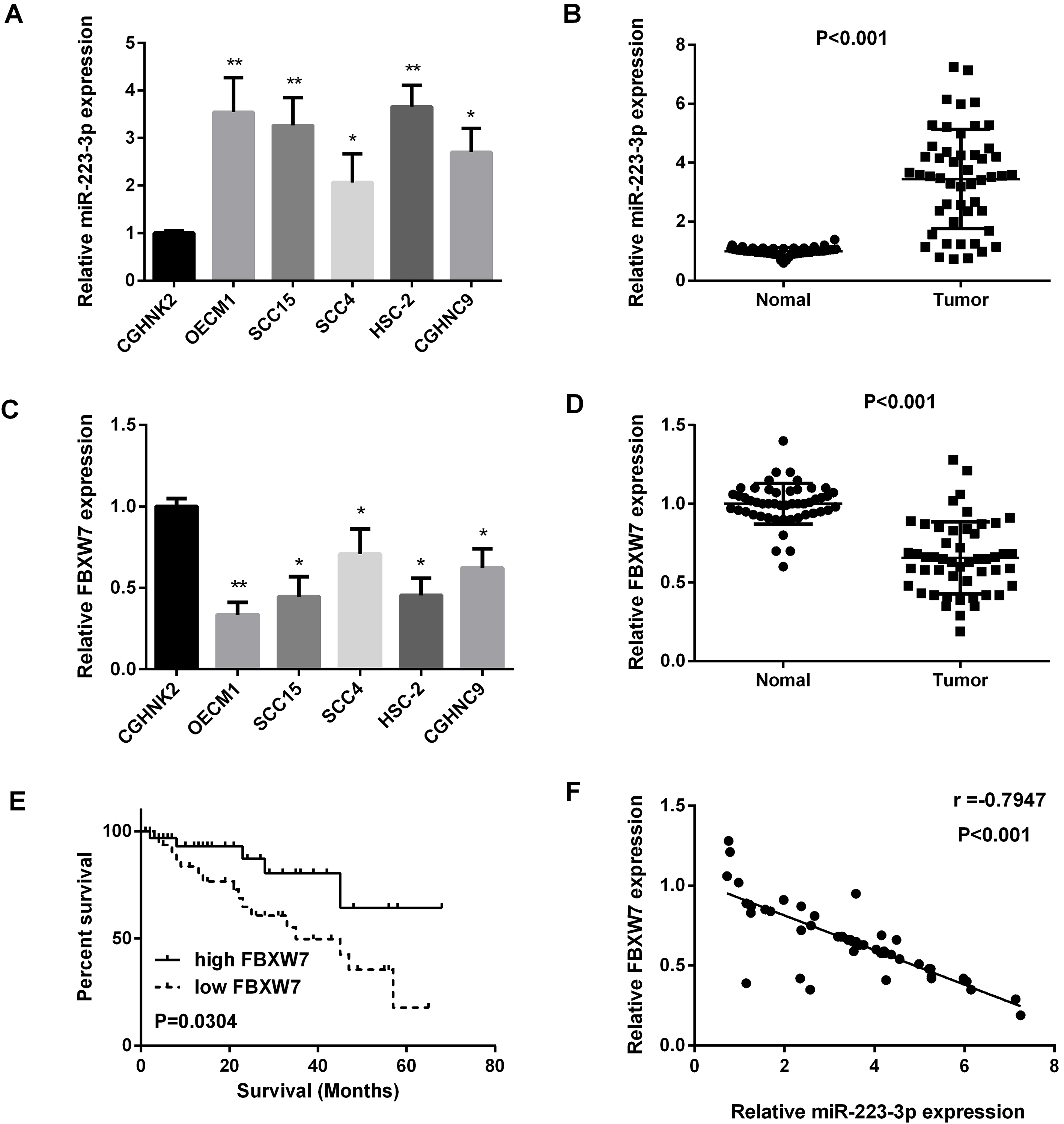
Control mimic/inhibitor and miR-223 mimic/ inhibitor were transfected into OECM1 and SCC15 cells. We used qRT-PCR to examine the miR-223 expression level in OSCC cells with different transfection, as shown in Fig. 2A and B. MTT assay showed that re-expression of miR-223 facilitated cell viability, while inhibiting miR-223 suppressed cell viability in both OECM1 and SCC15 cells (Fig. 2C and D). QRT-PCR results showed that miR-223 mimic increased PCNA expression, while miR-223 inhibitor decreased PCNA expression in both OECM1 and SCC15 cells (Fig. 2E and F). Moreover, Western blot showed that miR-223 mimic suppressed cell apoptosis while miR-223 inhibitor promoted cell apoptosis in both OECM1 and SCC15 cells (Fig. 2G). These results suggested that over-expression of miR-223 promoted OSCC cell proliferation, and inhibited cell apoptosis.
The promotion of miR-223 in the proliferation and apoptosis of OSCC cells. (A and B) Detection of the relative miR-223 mRNA expression in OECM1 and SCC15 cell lines after transfected with control mimic/inhibitor or miR-223 mimic/inhibitor (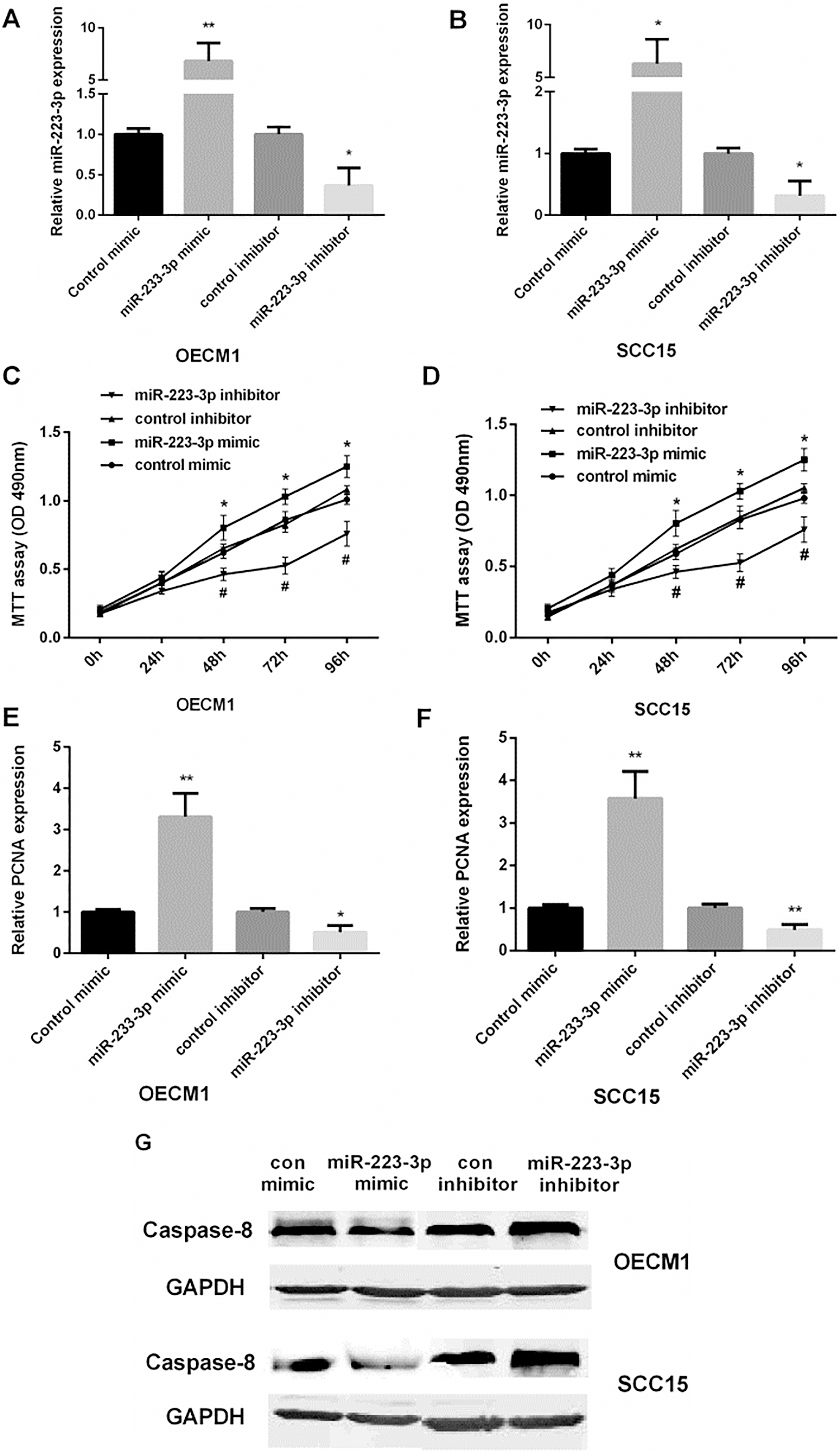
We performed transwell assay to investigate the effect of miR-223 on OSCC cell migration, as shown in Fig. 3A and B, miR-223 mimic enhanced cell migration, while inhibiting miR-223 suppressed cell migration in both OECM1 and SCC15 cells.
The promotion of miR-223 in the migration of OSCC cells. (A and B) Detection of relative cell migration in OECM1 and SCC15 cells lines after transfected with control mimic/control mimic, miR-223 mimic/inhibitor by transwell assay (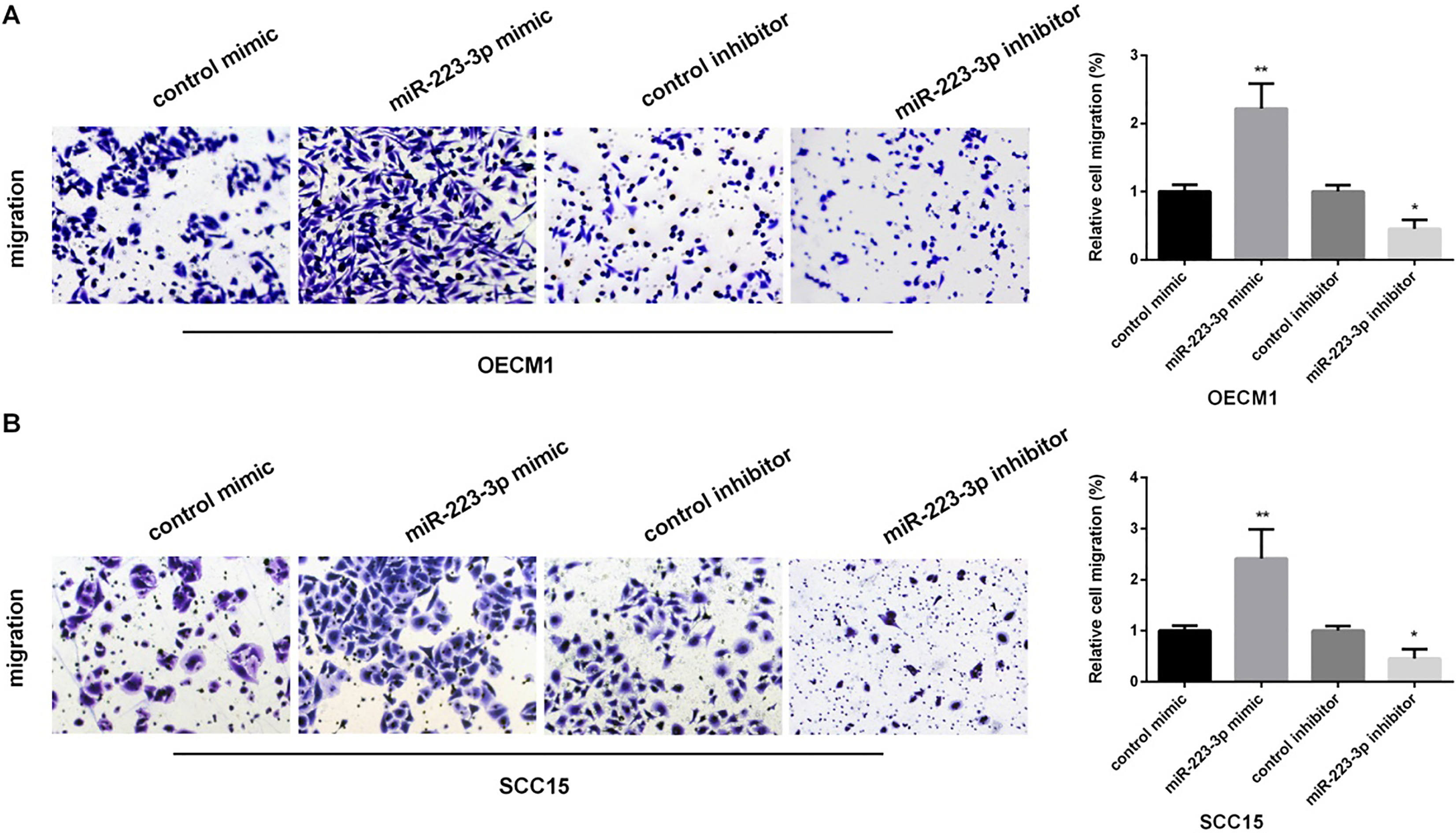
FBXW7 as a target gene of miR-223 in regulating of OSCC cells. (A) The predicted sites of miR-223 in the 3’UTR of FBXW7. Between the 3’-UTR of FBXW7 and the complementary sites for the seed regions in miR-223 generated mutation. (B) Detection of the luciferase activity in OECM1 cells after transfected with miR-223 mimic/inhibitor (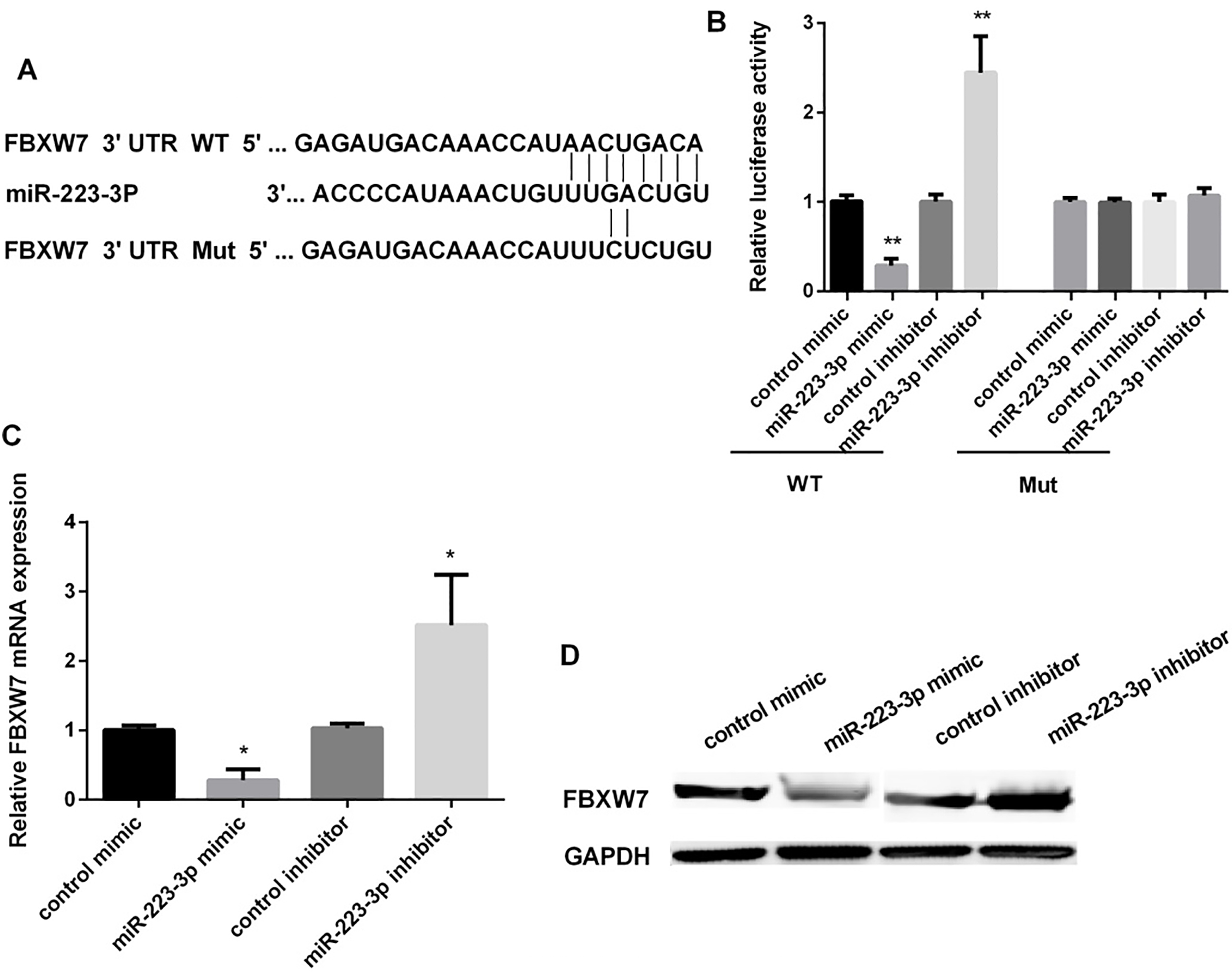
FBXW7 reversed miR-223-mediated OSCC cells proliferation and migration. (A) Detection of the relative FBXW7 mRNA expression in OECM1 cells after overexpression of FBXW7 for 48 h (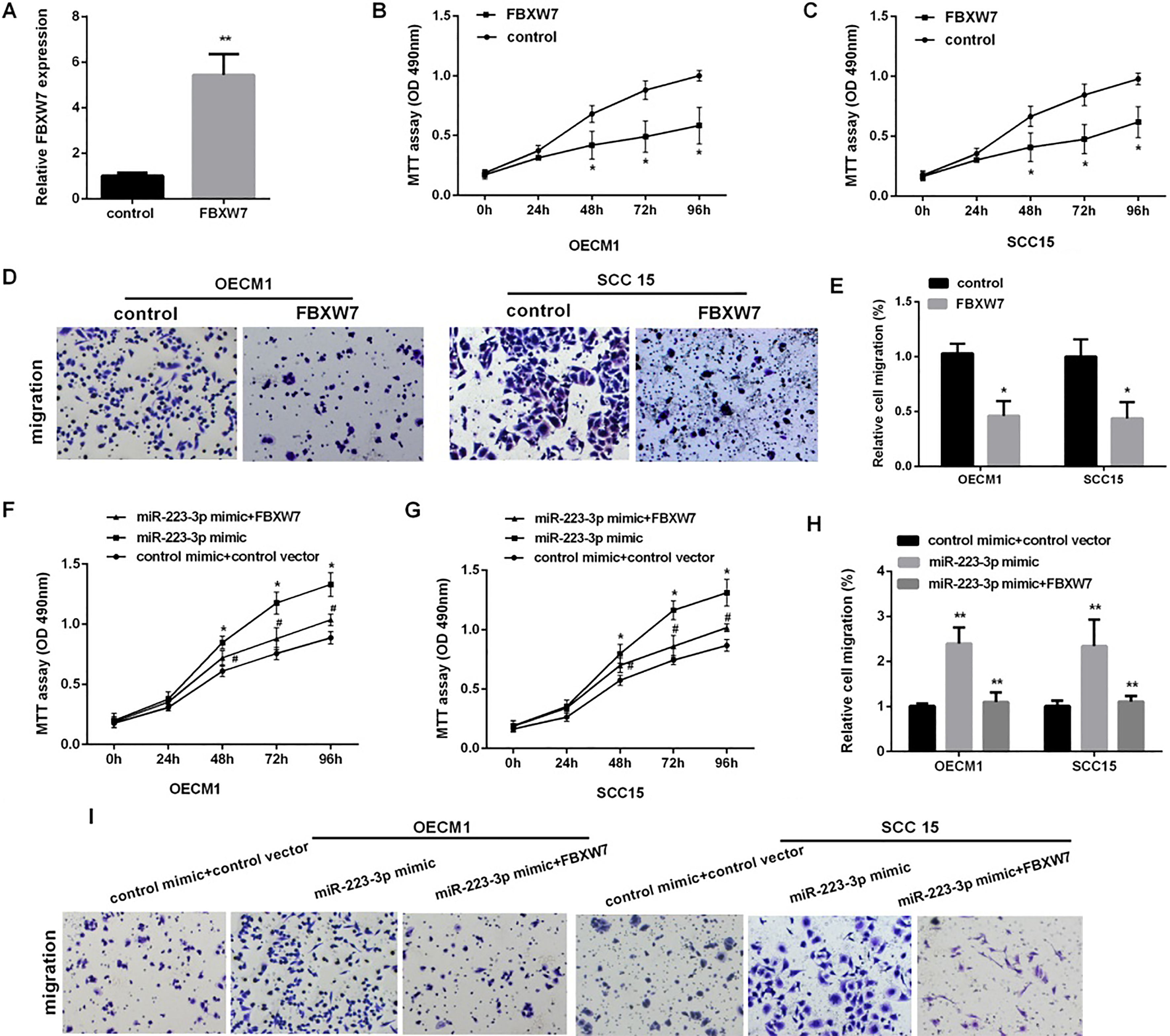
Target Scan Human 7.1 was used to find the potential targets of miR-223, as the Fig. 4A showed, the FBXW7 might act as a target of miR-223. The effect of miR-223 mimic or inhibitor on the expression of FBXW7 in OECM1 cells was detected using luciferase reporter assay. MiR-223 mimic significantly inhibited the expression of FBXW7 (approximately decreased 71.5%), while miR-223 inhibitor promoted FBXW7 expression after OSCC cells transfected with wild-type FBXW7 3’-UTR. However, it has no effect on the expression of FBXW7 when transfected with mutant FBXW7 3’-UTR (Fig. 4B). Furthermore, miR-223 mimic could decrease the mRNA and FBXW7 protein expression. MiR-223 inhibitor increased the FBXW7 expression both in mRNA and protein levels (Fig. 4C and D).
FBXW7 inversed the effect of miR-223 in regulating OSCC progression
To confirm the function of FBXW7 on the proliferation and migration of OSCC, we first over-expressed FBXW7 (Fig. 5A). As the Fig. 5B–D shown, increasing FBXW7 could inhibit the cells proliferation and migration in OECM1 and SCC15. Then, we detected cell proliferation and migration in OSCC co-transfected with FBXW7 vector and miR-223 mimic, the results showed that over-expression of FBXW7 markedly reversed miR-223-medicate promotion of cell proliferation in OECM1 and SCC15 cells (Fig. 5E), we also found that the migration cells number was decreased in OECM1 and SCC15 cells co-transfected with FBXW7 and miR-223 compared with the cells transfected with the miR-223 alone (Fig. 5F).
Discussion
Mounting researches suggest that miRNAs play important roles in progression of OSCC [18, 19]. Previous miRNA microarray analyses showed that miR-223 is down-regulated in OSCC tissues compared to adjacent normal tissues [20]. However, since then, there have no reports on further expression or functional data on miR-223 in OSCC. Here, we aimed to explore the biological function and underling mechanism of miR-223 in OSCC. The results showed that miR-223 expression was elevated in OSCC, whereas, FBXW7 expression was down-regulated. Furthermore, we also provided evidences that miR-223 and FBXW7 mRNA expression were negatively correlated, and FBXW7 could reverse the promotion effect of miR-223 in OSCC cell proliferation and migration.
Emerging studies have shown that miR-223 was abnormally expressed in cancer tissues, suggesting an essential role in tumorigenesis and tumor progression. For instance, miR-223 was up-regulated in gastric cancer, colorectal cancer, non-small cell lung cancer and pancreatic cancer [21, 22, 23, 24]. While, it was down-regulated in cervical cancer [25], prostate cancer [26], nasopharyngeal carcinoma [27] and acute myeloid leukemia [28]. Recent studies have reported that miR-223 expression in oral cancer was up-regulated [7, 8], which was consistent with our study that miR-223 expression was increased in OSCC cells compared with normal cells. In addition, Fang and his colleagues showed that miR-223 could promote ovarian cancer cells proliferation and invasion by targeting MAFB [29]. Also, Ma et al. found that miR-223 mimic could increase the gastric cancer cell proliferation and migration [30]. So we speculated that miR-223 might regulate cell proliferation, migration and invasion as an oncogene in OSCC based on the above researches. Our results showed that miR-223 mimic increased the cell proliferation and migration. Nevertheless, inhibiting miR-223 exerted the inverse effect in both OECM1 and SCC15 cell lines.
To better understand the tumor inhibitory effect of miR-223, bioinformatics analysis was used and FBXW7 was identified as a putative target of miR-223. FBXW7 was further confirmed as a direct target of miR-223 using the luciferase reporter assay. The expression of miR-223 was increased while FGFR2 was down-regulated in OSCC cells. FBXW7 was proved to be a tumor suppressor in regulating tumors progression. A study showed that FBXW7 was identified as a potential target of miR-367 in regulating tumor growth of NSCLC [31]. Also, miR-92a mimic promoted the tumor growth of osteosarcoma via suppressing FBXW7 [32]. Additionally, there was a study stated that FBXW7 level was remarkably reduced in breast cancer and it was a target of miR-223 in regulating the proliferation and migration of breast cancer cells [33]. Otherwise, miR-223 regulated the proliferation and apoptosis of testicular germ cells tumors by FBXW7 [34]. The researches above indicated that FBXW7 mainly function as a tumor suppressor in regulating tumor development, acting as a target gene of miRNA. The results in our study stated that FBXW7 expression level was down-regulated in OSCC and re-expression of FBXW7 inhibited cell proliferation and migration. Moreover, FBXW7 over-expression reversed the promotion effect of miR-223. In clinical tissues, FBXW7 expression was inversely correlated with that of miR-223. All of our data strongly support a role of FBXW7 as a downstream mediator of miR-223-dependent regulation of OSCC.
We summarize all our results and draw a conclusion that miR-223 expression was higher in OSCC and acted as an oncogene in the development of OSCC. We also found the target gene of miR-223 in regulating of OSCC cell proliferation and migration was FBXW7. The newly discovered miR-223/FBXW7 axis provided insight into the mechanisms of oral cancer and provided a potential therapeutic target for OSCC.
Footnotes
Conflict of interest
No conflicts declared.
