Abstract
BACKGROUND:
Long noncoding RNA MIAT expression is related to the development of some diseases. However, the role of MIAT in melanoma was has seldom been studied.
OBJECTIVE:
To investigate the effect of the lncRNA MIAT on melanoma cells.
METHOD:
Microarray was used to analyze the lncRNAs expression in tissue samples. The expression of the lncRNA MIAT was detected by qRT-PCR. A CCK-8 assay was used to assess cell viability, and cell counting was used to analyze cell proliferation. Wound healing and Transwell invasion assays were used to detect the migration and invasion abilities, respectively, of melanoma cells. Western blotting was performed to explore the molecular mechanisms of MIAT in melanoma.
RESULTS:
The lncRNA MIAT was overexpressed in melanoma. The overexpression of MIAT promoted cell proliferation, cell invasion and migration, while the knockdown of MIAT expression got the opposite results. MIAT significantly upregulated the phosphorylation of PI3K and AKT and promoted cMyc and cyclin D1 protein expression.
CONCLUSION:
LncRNA MIAT was a key factor to promote cell invasion, migration and proliferation through the PI3K/AKT signaling pathway. These findings may give us a potential way to treat melanoma.
Introduction
Melanoma is the most aggressive type of skin cancer and the crucial cause of death from skin cancer in America [1]. Furthermore, melanoma accounts for 80% of all deaths among patients with skin cancer but only accounts for 4% of the total number of skin cancer patients [9]. In addition, melanoma has the ability to metastasize to various sites, such as the brain, lungs, liver and eye [10]. The major risk factor for melanoma is ultraviolet (UV) radiation, which is responsible for a characteristic genetic signature in melanoma. The mutational rate in melanoma is the highest mutational rate across all cancer types and is considered the reason that immunotherapy fails. Due to the high metastatic burden in melanoma, patients with advanced disease have a poor prognosis [15]. Therefore, revealing the molecular mechanism of melanoma and investigating new targets for the treatment of melanoma are essential [21].
The data from many studies in recent years have shown that only 1% of genes have the ability to be transcribed into mRNA, which can then be translated into protein, and the remaining 99% of RNAs are named noncoding RNAs (ncRNAs) [7]. Long noncoding RNAs (lncRNAs), whose lengths are
MIAT, also called myocardial infarction associated transcript, which is located at 22q12.1 in humans, was discovered in chronic lymphocytic leukemia [16]. Knocking down the expression of the lncRNA MIAT is considered to exert a tumor suppressor effect and is effective in many cancers, such as gastric cancer [12] and non-small cell lung cancer [22]. Increasing evidence shows that MIAT expression is closely related to tumor formation and the development of other diseases. For example, MIAT expression knockdown inhibits cell metastasis in neuroendocrine prostate cancer [6], and MIAT regulates microvascular dysfunction by functioning as a competing endogenous RNA that sponges miR-18 [14]. However, the role of MIAT in melanoma has seldom been studied.
In the study, we found that the migration of melanoma cells was regulated by the lncRNA MIAT, and as for tumor growth, MIAT also played a vital role. The expression of the lncRNA MIAT in melanoma tissue was significantly higher than that in normal tissue. Furthermore, significant upregulation of the expression of the lncRNA MIAT promoted cell migration, invasion and proliferation. Therefore, these results indicated that the lncRNA MIAT could be a key factor for the treatment of melanoma.
Materials and methods
Microarray
Global gene expression analysis was performed on the skin sections using a NimbleGen array (Roche, Basel, Switzerland). The extracted RNA was reverse transcribed into cDNA using a double-stranded cDNA synthesis kit (invited by Logan) and a color DNA labeling tool from NimbleGen (Roche). Samples were hybridized in a NimbleGen Array 12
Patient tissue samples
There were 20 malignant melanoma specimens with matched adjacent tissue samples from patients who did not receive preoperative chemotherapy or radiotherapy. All experiments in this study were performed after obtaining consent from the patients, Sichuan Academy of Medical Sciences and Sichuan Provincial People’s Hospital (Chengdu, China). Moreover, this study obtained the approval of the ethics committees of the Sichuan Academy of Medical Sciences and Sichuan Provincial People’s Hospital (Chengdu, China).
Cell lines and cell cultures
BeNa Culture Collection (Beijing, China) provided the normal skin cell line HaCaT and the human malignant melanoma cell lines A-375, SK-MEL-28, M21, and A2508. All cells were cultured in DMEM (Life Technologies, Grand Island, NY, USA) that contained 10% fetal bovine serum (FBS).
Quantitative real-time PCR (qRT-PCR)
Trizol reagent (Takara, Dalian, China) was used to extract total RNA from melanoma tissue and cell lines based on the manufacturer’s instructions. Next, M-MLV Reverse Transcriptase (Takara, Dalian, China) was utilized to reserve transcribe the RNAs into double-stranded cDNAs according to the manufacturer’s instructions. Then, a NanoDrop 2000c spectrophotometer was employed to detect the RNA concentrations, GAPDH was considered as the internal control, and the 2
Cell transfection
The plasmids to silence or overexpress MIAT, pCMV-MIAT and pCMV-shMIAT, respectively, and the empty vectors were purchased from GenePharma (Shanghai, China). The following groups were used for the experiments: the Blank group (untreated melanoma cells), NC group (transfected with the empty pCMV vector), pCMV-MIAT group (transfected with pCMV-MIAT) and pCMV-shMIAT group (transfected with pCMV-shMIAT). Twenty-four hours prior to transfection, melanoma cells were seeded into 6-well plates (1
CCK-8 assay
To determine cell viability, a cell counting kit-8 assay (Beyotime, Shanghai, China) was employed. Melanoma cells were placed in 96-well plates and incubated after being transfected with pCMV-MIAT, pCMV-shMIAT or the empty vector plasmid (NC). At different time points (24 h, 48 h and 72 h), each well was supplemented with 10
Cell count
To analyze cell proliferation, melanoma cells were seeded into 6-well plates and transfected with pCMV-MIAT, pCMV-shMIAT or the empty vector plasmid (NC) for 72 h. The cells were trypsinized and the cell numbers were estimated by using a hemocytometer.
Wound healing assay
To analyze cell migration, a wound healing assay was conducted. Transfected melanoma cells were plated into 6-well plates at a density of 5
Western blot analysis
Western blotting was performed using a standard protocol. Protein extracts were electrophoresed on 10% sodium dodecyl sulfate-polyacrylamide gels and transferred onto nitrocellulose membranes. The membranes were blocked at room temperature with 5% powered milk and incubated overnight with an appropriate primary antibody. Secondary antibodies were diluted in TBST with 0.05% powered milk and incubated with the membranes for 1 h. Immunoreactive bands were visualized using a chemiluminescent detection system (Pierce Biotechnology, Rockford, IL, USA).
The MIAT expression levels in melanoma tissues and cell lines. (A) The heat map shows the top 10 lncRNAs whose expression levels were upregulated in the melanoma. (B) The expression levels of MIAT in adjacent normal and tumor tissue samples were detected by qRT-PCR. **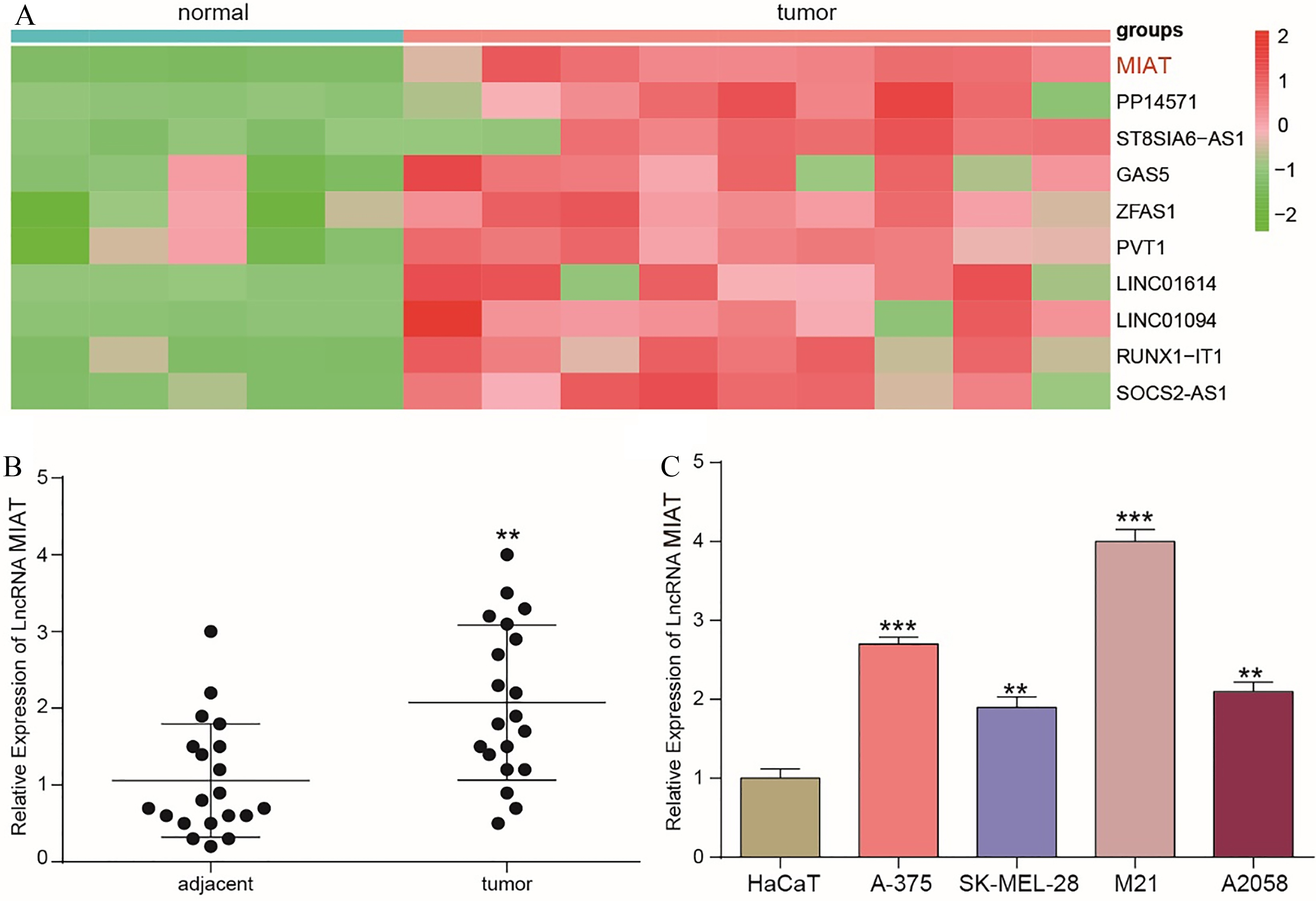
Invasion was investigated with an in vitro Transwell Matrigel invasion assay. Cell invasion was assessed using BD BioCoat Matrigel™ invasion chambers (BD Biosciences, USA) precoated with BD Matrigel matrix. The 24-well artificial basement membrane inserts had 8
Statistical analysis
Data are shown as the mean
MIAT enhanced the cell viability of A375 cells. (A) A375 cells were transfected with NC, pCMV-MIAT or pCMV-shMIAT plasmids or left untreated. The MIAT expression levels in the four groups were examined by qRT-PCR. (B) A CCK-8 proliferation assay detected the cell viability of the four groups at 0 h, 24 h, 48 h and 72 h. (C) Cell counting was performed to evaluate cell proliferation at 72 h. **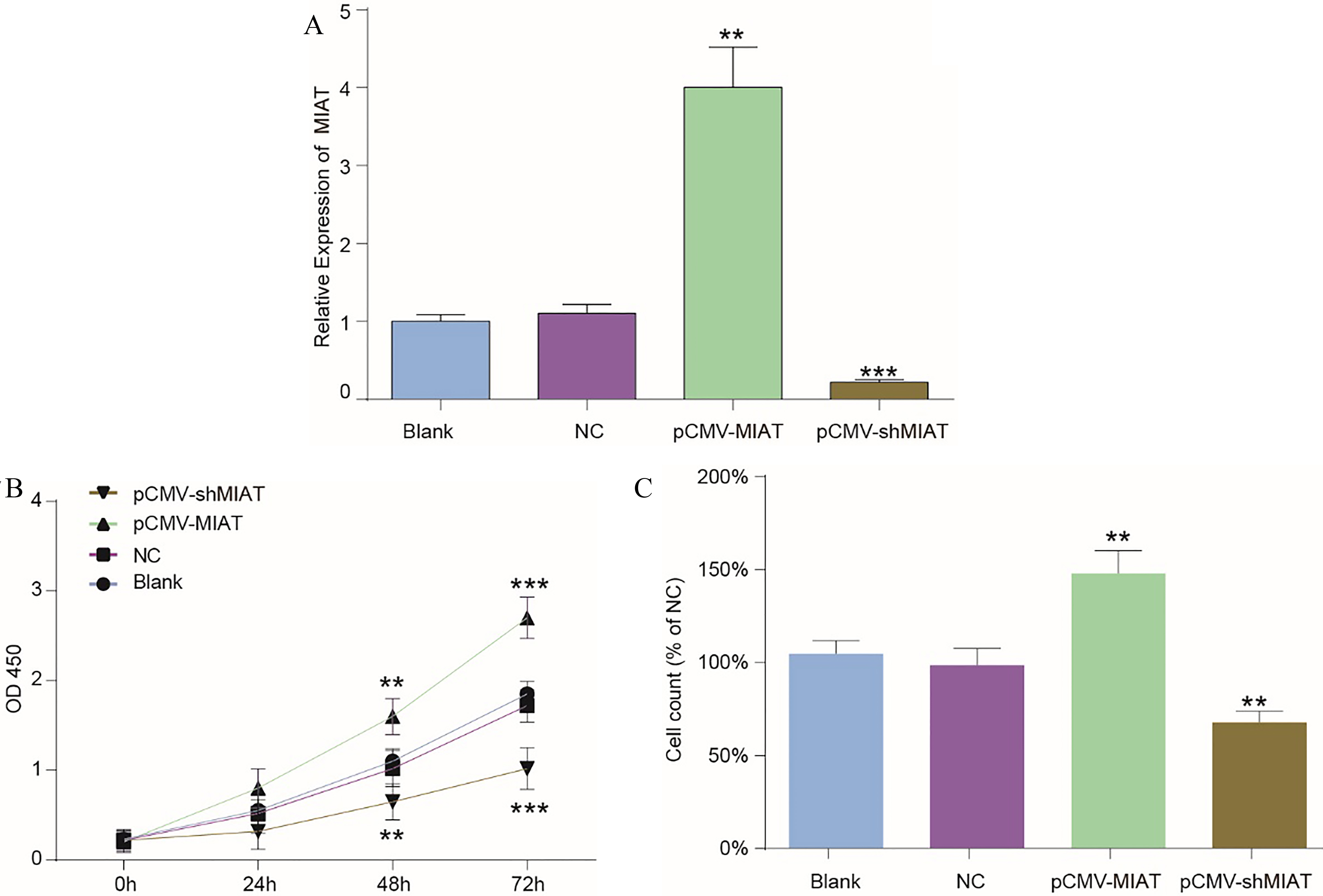
LncRNA MIAT expression was significantly upregulated in melanoma tissues
First, microarray analysis was employed to investigate dysregulated genes. In addition, all differentially expressed genes were defined by
LncRNA MIAT promoted melanoma cell proliferation
To explore the correlation of the expression of the lncRNA MIAT with the proliferative ability of melanoma cells, pCMV-MIAT, pCMV-shMIAT and the empty plasmid were transfected into melanoma cells for further study. pCMV-MIAT obviously promoted MIAT expression, while pCMV-shMIAT visibly downregulated MIAT expression (Fig. 2A). Moreover, the potential biological effects of the lncRNA MIAT on the viability and proliferation of melanoma cells were detected by a CCK-8 assay and the cell counting method, respectively. After the transfection of pcDNA3.1-MIAT into A-375 cells, a CCK8 assay and cell counting were employed. The proliferation and viability of the pcDNA3.1-MIAT group cells were significantly higher than those of the NC group, which means that the lncRNA MIAT promoted the proliferation and enhanced the viability of A-375 cells (Fig. 2B and C).
The cell migration and invasion abilities of A375 cells after altering the expression of MIAT. (A) The representative images from the wound healing assay. (B) The wound healing closure rates of the four groups. (C) The representative images from the Transwell invasion assay using A375 cells with increased or decreased expression of MIAT or NC. (D) The numbers of invading cells were examined. **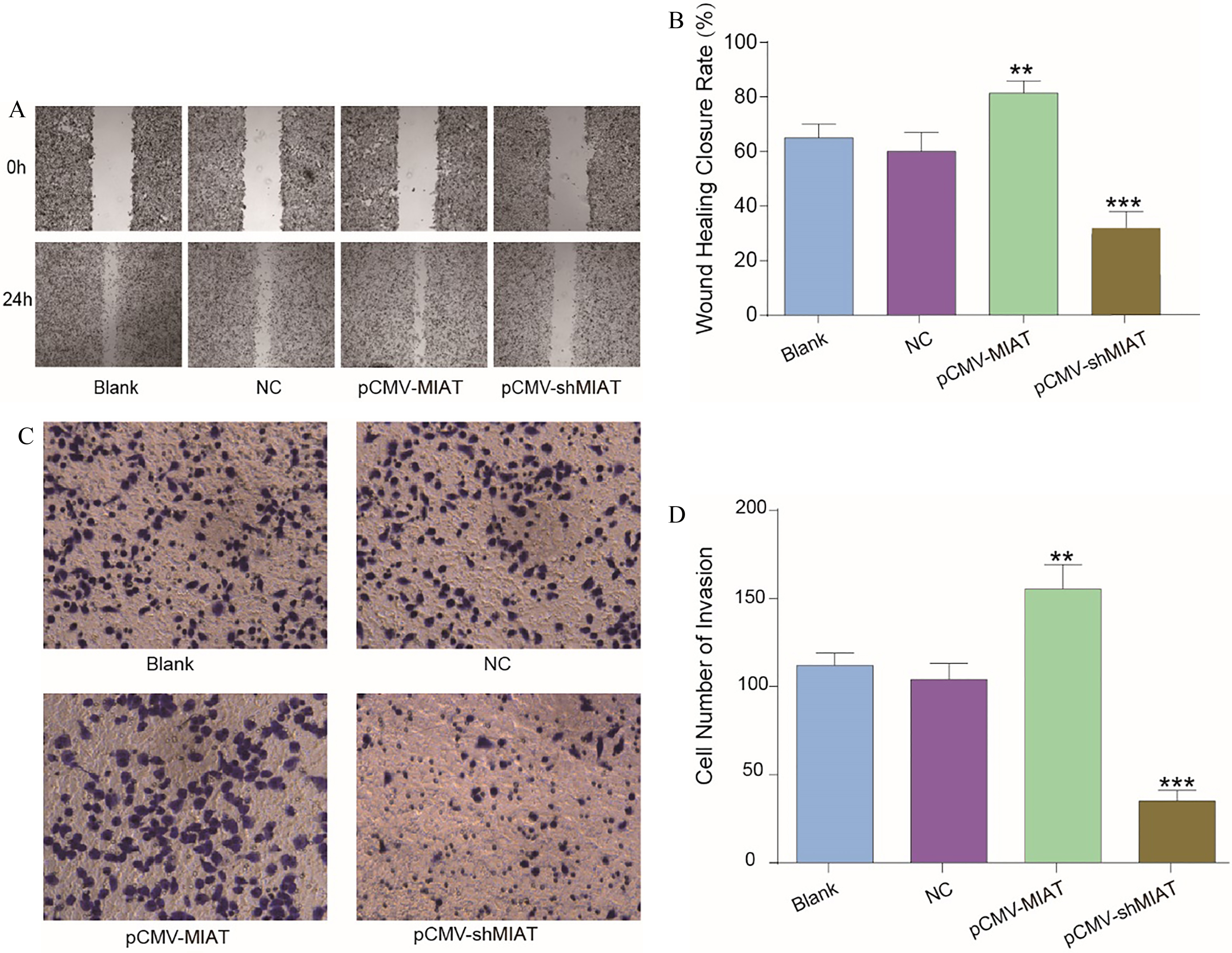
MIAT is involved in altering the physiological state of cells via the PI3K/AKT signaling pathway. (A) The proteins related to the PI3K/AKT pathway were detected by Western blotting. (B and C) The phosphorylation of PI3K and AKT after dysregulating MIAT expression. (D and E) The expression levels of the related proteins c-Myc and Cyclin D1. (F and G) The mRNA expression levels of c-Myc and Cyclin D1. **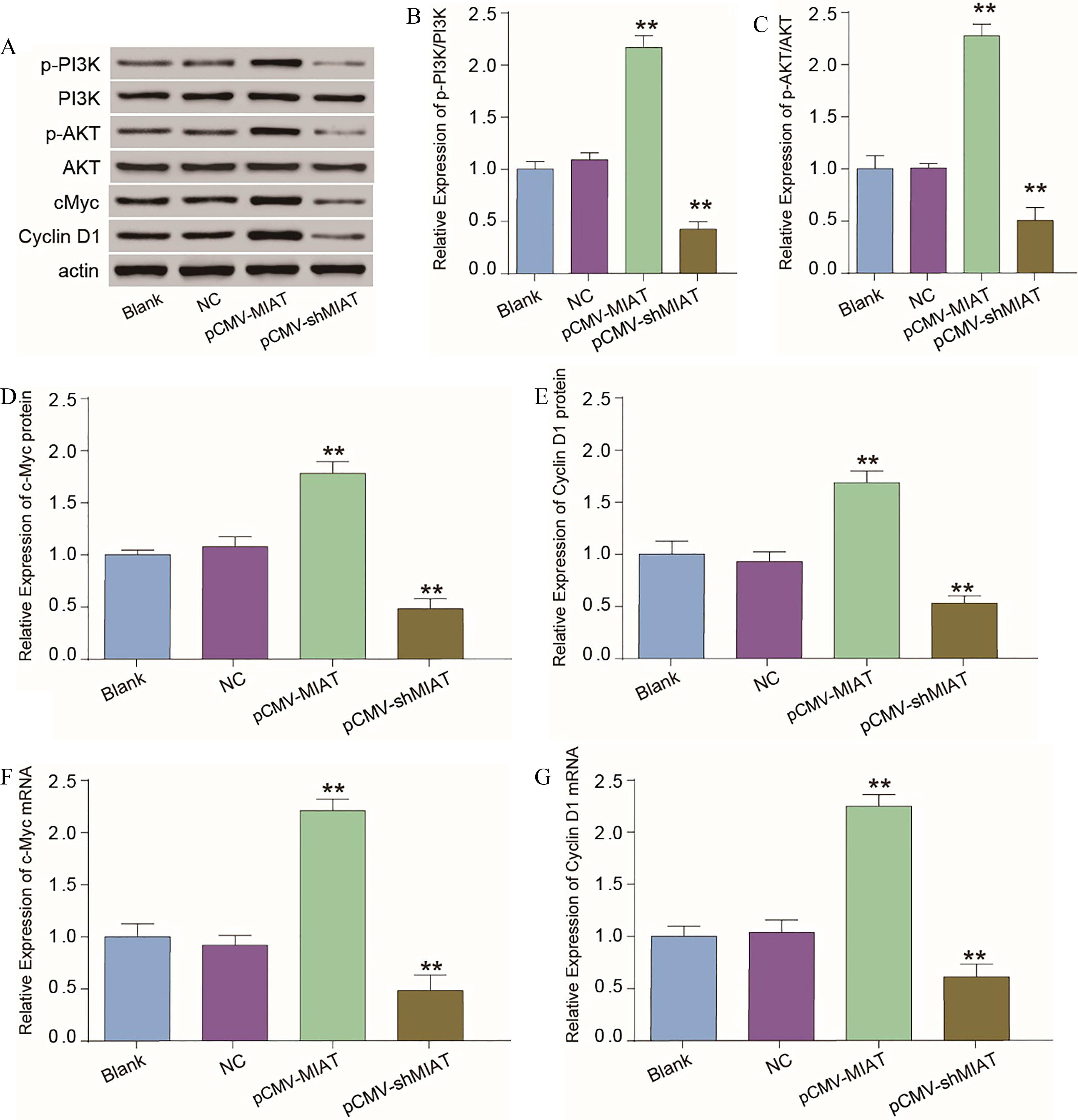
To investigate cell migration and invasion capacities, wound healing assays and Transwell analysis, respectively, were utilized. According to the results, the migratory capacity of MIAT-overexpressing cells was enhanced compared with that of the NC group, whereas the suppression of MIAT expression decreased the migratory capacity of the cells (Fig. 3A and B). In addition, the Transwell invasion assay presented similar results, showing that melanoma cells with upregulated MIAT expression showed enhanced invasive ability and melanoma cells with knocked down MIAT expression exhibited hindered cell invasion compared with the cell invasion of the NC group (Fig. 3C and D). Briefly, MIAT promoted melanoma cell migration and invasion and contributed to the progression of melanoma.
MIAT activated the PI3K/AKT signaling pathway
Subsequently, we further explored whether MIAT affected the progression of melanoma cells through the PI3K/ AKT signaling pathway by determining the phosphorylation statuses of PI3K and AKT as well as the expression levels of cyclin D1 and c-Myc through Western blotting after transfecting control vector, pCMV-MIAT and pCMV-shMIAT into melanoma cells. The overexpression of MIAT significantly upregulated the phosphorylation of PI3K and AKT, and decreasing MIAT expression notably inhibited the phosphorylation of PI3K and AKT (Fig. 4A–C). In addition, there were no differences in the protein levels of PI3K and AKT in the transfected cells (Fig. 4A). The cyclin D1 and cMyc mRNA and protein expression levels were also downregulated by knocking down MIAT expression (Fig. 4D–G). Ultimately, the dysregulation of MIAT expression might influence cell proliferation, migration and invasion by modulating the PI3K/AKT signal pathway.
Discussion
A significant increase in the incidence of melanoma and an increase in the prevalence of risk factors for melanoma mean that melanoma will become a substantial social burden, and this burden is further worsened by the refractoriness of later stage melanoma. Therefore, an in-depth understanding of the molecular mechanism regulating the occurrence and development of melanoma and the development of effective biomarkers for melanoma and targeted treatments are the top priorities [2].
In this study, the significant, high expression of the lncRNA MIAT in melanoma tissue was uncovered. Furthermore, the upregulation of the expression of the lncRNA MIAT significantly increased the invasive and proliferative abilities of melanoma cells. In contrast, the knockdown of the expression of the lncRNA MIAT reversed the effects on cell invasion and proliferation.
The present study suggests that lncRNAs are important regulators in melanoma. The expression of the lncRNA MIAT was found to be upregulated in melanoma tissue and cells in our study. In a previous investigation, the expression levels of the lncRNA HEIH [24] and lncRNA RMEL3 [8] were also upregulated in melanoma. In addition, some studies on the lncRNA MIAT showed that MIAT was involved in many other tumors. For instance, Zhang et al. showed that the expression of MIAT in glioblastoma patients was significantly upregulated [23]. In addition, Crea et al. also reported that MIAT was overexpressed in prostate cancer cell lines and tissue [6]. Therefore, all these reports provide the message that the lncRNA MIAT might play a negative role in melanoma as a carcinogenic factor.
MIAT is an lncRNA that has rarely been studied in melanoma. However, in our research, we demonstrated the influence of MIAT on cell proliferation. Similarly, some studies showed that many lncRNAs exhibit analogous functions in melanoma. For example, the lncRNA HOXD-AS1 significantly increases the proliferative ability of melanoma cells [21]. Mazar et al. showed that the overexpression of SPRY4-IT1 could promote melanoma cell proliferation, and silencing SPRY4-IT1 promoted melanoma cell apoptosis [13]. Zhang et al. showed that the overexpression of MIAT significantly enhanced proliferation in non-small cell lung cancer by sponging miR-150 [22]. These reports mean that MIAT might play a promotive role in the occurrence and progression of melanoma.
In our study, by using wound healing assays and Transwell assays, we found that the overexpression of MIAT increased the invasive and migratory capacities of A-375 cells. Insight into how the invasion and migration of melanoma cells are influenced by the dysregulation of lncRNAs has been obtained with the finding that the lncRNA ILF3-AS1 sponged miR-200b/a/429, resulting in the promotion of melanoma cell invasion and migration [3]. Furthermore, the role of the lncRNA FTH1P3 in facilitating uveal melanoma cell invasion by negatively regulating miR-224-5p has been uncovered [25]. Even in other diseases, for instance, posterior capsule opacification, the ability of the lncRNA MIAT to promote cell migration has been demonstrated [17].
Based on our results, PI3K/AKT was demonstrated to be modulated by the lncRNA MIAT by monitoring the phosphorylation levels of PI3K and AKT. Furthermore, some previous reports have suggested that activation of the PI3K/AKT signaling pathway would promote melanoma cell proliferation, migration and invasion [19]. In addition, Ye et al. also found that inositol polyphosphate 5-phosphatases regulated the PI3K/AKT signaling pathway to exert a suppressive role in human melanoma [20]. Furthermore, silencing MIAT expression inactivated PI3K/ AKT signaling [5], which parallels the results of our experiments.
In summary, MIAT expression was upregulated in melanoma cells and could promote invasion, migration and proliferation through the PI3K/AKT signaling pathway. Therefore, this research may provide a promising strategy for the treatment of melanoma.
Footnotes
Conflict of interest
The authors confirm that they have no conflicts of interest.
