Abstract
OBJECTIVE:
This study aimed to investigate the correlation of long non-coding RNA taurine-upregulated gene 1 (lncRNA TUG1) with clinicopathological characteristics and prognosis in acute myeloid leukemia (AML) patients, as well as its function in cell proliferation and apoptosis.
METHODS:
Two hundred and thirty six de novo AML patients were consecutively enrolled and then underwent conventional induction chemotherapy. Bone marrow samples were obtained from all AML patients and controls. Quantitative polymerase chain reaction assay was performed to detect lncRNA TUG1 expression. KG-1 cells were transfected by TUG1 inhibitor (TUG1 (
RESULTS:
LncRNA TUG1 expression was higher in AML patients compared to controls, and it was positively correlated with white blood cell counts as well as poor risk stratification. Additionally, elevated lncRNA TUG1 expression was observed in non-complete remission (non-CR) patients compared to CR patients, and it was correlated with shorter event-free survival and overall survival in AML patients. In the in vitro experiments, lncRNA TUG1 expression was upregulated in AML cell lines compared to control cells, and cell proliferation ability was reduced, but cell apoptosis rate was promoted in TUG1 (
CONCLUSIONS:
LncRNA TUG1 predicts advanced disease conditions and poor prognosis in AML patients, and its knockout decreases proliferation and increases apoptosis of AML cells.
Keywords
Introduction
Acute myeloid leukemia (AML), a heterogeneous hematopoietic malignancy, is characterized by accumulated clonal myeloid progenitor cells with limited ability to differentiate into mature blood cells including red blood cells, platelets and granulates, which is clinically manifested by anemia, hematostaxis and serious infection [1]. The incidence rate of AML in infants aged below 1 year is 1.5 per 100,000, and it is less in those younger than 25 years, while for the population elder than 25 years, its rate increases with the growth of age and eventually reaches 25 per 100,000 in octogenarians [2]. Although AML has become curable in a limited number of patients during the past 3 decades with the adoption of remissive induction and post-remission consolidation treatments, the prognosis in the vast majority of AML patients remains unsatisfactory, bringing forward a clinical challenge in AML treatment [3]. Therefore, it is essential to explore novel biomarkers to monitor disease progression as well as to improve the prognosis in AML patients.
Long non-coding RNAs (lncRNAs), broadly defined as non-protein coding RNA molecules with lengths over 200 nucleotides, are enormously abundant in mammalian cells with approximately 28000 distinct transcripts [4]. Recent studies have exhibited that lncRNAs are pivotal regulators of genes and are involved in the initiation and development of various cancers including AML [4, 5, 6, 7]. LncRNA taurine upregulated gene 1 (TUG1), a member of lncRNAs firstly identified in mouse retinal cells, has been found to be highly expressed in human malignant tissues and correlates with adverse clinicopathological characteristics as well as poor prognosis in patients with several carcinomas (e.g., bladder cancer and lung adenocarcinoma) [8, 9, 10]. However, only a recently published study reveals that lncRNA TUG1 is upregulated in AML and correlates with worse prognosis in AML patients in a small population [11], while a microarray data published on GEO database with entry No. GSE 96535 (
Thus, the purpose of this study was to investigate the correlation of lncRNA TUG1 with clinicopathological characteristics and prognosis in AML patients with large sample size and to further explore its function in cell proliferation and apoptosis in AML cells.
Materials and methods
Participants
This study consecutively enrolled 236 de novo AML patients at the Department of Hematology, Renmin Hospital Affiliated to Hubei University of Medicine from 2015/1/1 to 2017/12/31. Patients with the following conditions were included: (1) Diagnosed as primary AML according to World Health Organization (WHO) Classification of Tumors of Hematopoietic and Lymphoid Tissues (2008). (2) Age between 18 and 65 years. Patients with the following conditions were excluded from this study: (1) Diagnosed as acute promyelocytic leukemia (APL), central nervous system leukemia (CNSL) or secondary AML; (2) History of other malignant tumors (3) Previous exposure to radiotherapy, chemotherapy, targeted therapy, immunotherapy or allogeneic stem cell transplantation; (4) Severe or uncontrolled heart, renal or liver disease; (5) Pregnancy or breastfeeding women. Meanwhile, 118 age and gender-matched controls who underwent bone marrow biopsy (non-hematologic malignancy patients) were also recruited. The Ethics Committee of Renmin Hospital Affiliated to Hubei University of Medicine had approved this study before its initiation, and all patients provided written informed consents.
Sample acquisition and lncRNA TUG1 detection
Bone marrow sample was obtained by biopsy from AML patients and controls before initiation of any treatments, and lncRNA TUG1 expression in bone marrow was subsequently determined by quantitative polymerase chain reaction (qPCR) assay.
Baseline data collection and assessments
Baseline information of AML Patient was collected including age, gender, white blood cells (WBC), French-American-British (FAB) classification, cytogenetics, molecular genetics and risk stratification. The FAB classification was categorized in line with FAB classification system; the cytogenetics was evaluated according to An International System for Human Cytogenetic Nomenclature (ISCN 2009), and the risk status was classified according to National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines in Oncology of AML (Version 2.2013). In addition, data of controls including age and gender were also documented.
Treatment and outcome evaluation
After the enrollment, AML patients underwent conventional induction chemotherapy regimens including: (1) IA, Idarubicin 8–12 mg/m
Treatment response was evaluated after chemotherapy, and CR was defined as bone marrow (BM) with at least 20% cellularity and BM blasts below 5% at steady state after treatment, without cytological evidence of leukemia, no transfusion requirement, leucocyte count above 1
Cell culture
AML cell lines including KG-1, MOLM-14, Kasumi-6, NB-4, THP-1 and HL-60 were purchased from Cell Biology of the Chinese Academy of Sciences (Shanghai, China) or American Type Culture Collection (ATCC) (Manassas, USA), while normal control cells were obtained by isolating CD34
Transfection and assays
LncRNA TUG1 inhibitor and blank inhibitor plasmids were constructed and transfected into KG-1 cells as TUG1 (
AML patients’ characteristics
AML patients’ characteristics
Data were presented as mean value
LncRNA TUG1 relative expression in AML patients and controls. (A) LncRNA TUG1 expression was higher in AML patients compared to controls. (B) ROC curve showed that lncRNA TUG1 had a good potential to distinguish AML patients from controls. Wilcoxon rank sum test was used to compare lncRNA TUG1 expression in AML patients and controls, and ROC curve was used to evaluate the predictive value of lncRNA TUG1 for AML. 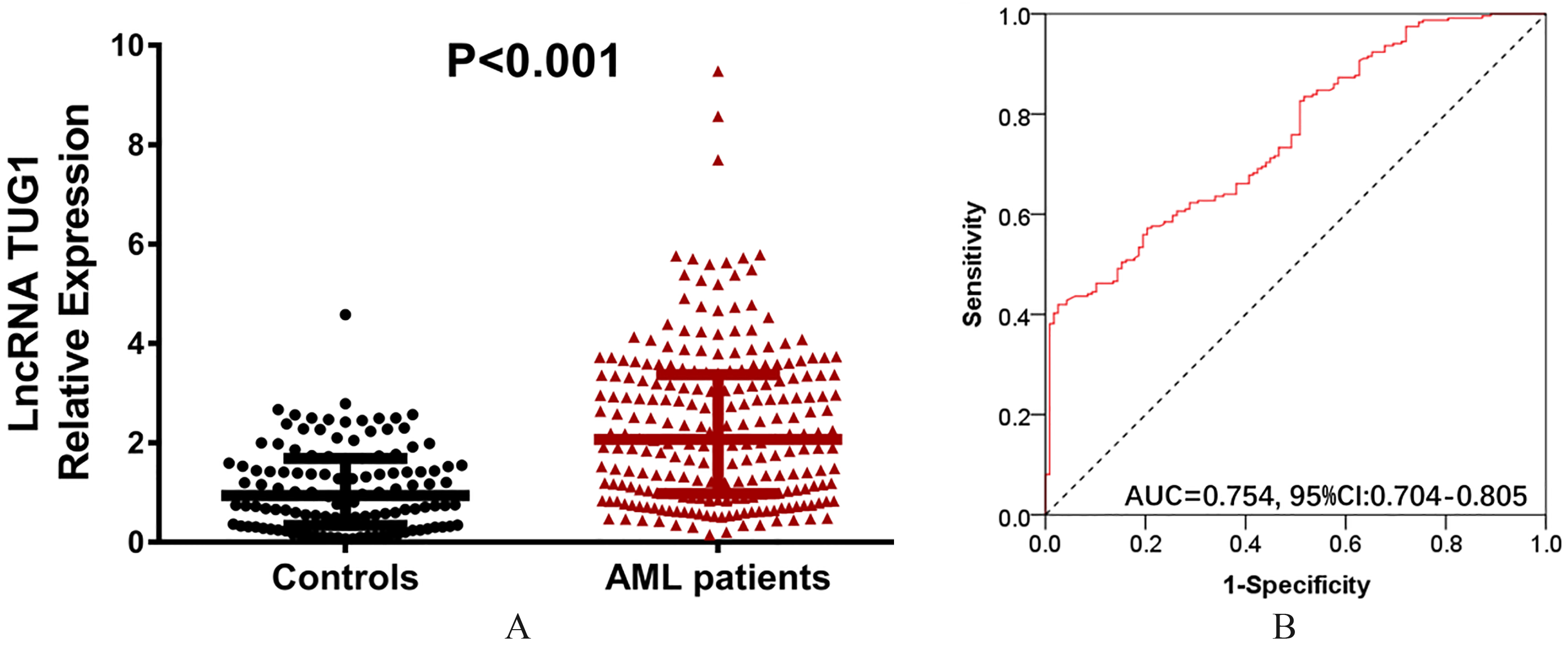
Total RNA was extracted using TRIzol reagent (Invitrogen, USA) and qualified by concentration and purity, as well as agarose gel electrophoresis. Subsequently, RNA was reversely transcribed into cDNA using PrimeScript™ RT reagent kit (TAKARA, Japan): (1) mix 1 ug of RNA with distilled water to constant volume 14 ul, then place at 65
CCK-8 assay
Ten ul of CCK-8 (Dojindo, Japan) and 90 ul of medium were added to each group of KG-1 cells, and following that, the cells were incubated at 37
AV/PI assay
Before being suspended in 100 ul blinding buffer, KG-1 cells were initially digested using pancreatin and washed with phosphate buffer solution (PBS). Subsequently, 2 ul AV (Invitrogen, USA) was added to the cells; then the cells were placed on the ice for 15 mins in darkness. Following that, 1 ul PI (Invitrogen, USA) was added to the cells, and cell apoptosis rate was analyzed using flow cytometry (FCM) (Becton Dickinson, USA).
Western blot assay
KG-1 cells were centrifuged after lysing with RIPA lysis buffer (Thermo Scientific, USA). The total protein concentration was detected by the BCA™ protein assay kit (Pierce, USA). Proteins were separated by electrophoresis on sodium dodecyl sulfate-polyacrylamide (SDS) gel. Following that, proteins were transferred to PVDF membrane (Millipore, USA) and the membrane was then blocked for 1 hour at room temperature with 5% non-fat dried milk in PBST. After incubation with C-Caspase3, Bcl-2 and GAPDH antibodies (Cleaved Caspase 3 Rabbit mAb (CST, USA), Anti-Bcl-2 Rabbit Antibody (Abcam, USA) and GAPDH Rabbit mAb (CST, USA)), the PVDF membrane was finally incubated with the mouse anti-rabbit IgG HRP-conjugated secondary antibody (CST, USA). The specific bands indicating protein abundance were visualized by ECL advanced Western blot analysis detection kit (BD, USA).
Correlation of LncRNA TUG1 relative expression with clinical characteristics of AML patients
Correlation of LncRNA TUG1 relative expression with clinical characteristics of AML patients
Data were presented as median (1/4–3/4 quartiles). Comparison was determined by Wilcoxon rank sum test or Kruskal-Wallis rank sum test.
Analysis of statistics was performed using GraphPad Prism 5.01 software (GraphPad Software Inc, USA) and SPSS 21.0 software (IBM, USA). Data were presented as mean
LncRNA TUG1 expression in non-CR patients and CR patients. Compared to CR patients, lncRNA TUG1 expression was observed to be higher in non-CR patients. Wilcoxon rank sum test was used for the comparison of lncRNA TUG1 expression between non-CR patients and CR patients, and 
Baseline characteristics
The mean ages for AML patients (
Correlation of lncRNA TUG1 expression with EFS and OS in AML patients. (A) Worse EFS was presented in AML patients with lncRNA TUG1 high expression compared to patients with lncRNA TUG1 low expression. (B) Shorter OS was observed in AML patients with lncRNA TUG1 high expression compared to patients with lncRNA TUG1 low expression. K-M curve was used to assess EFS and OS in AML patients, and comparison of median EFS and OS between patients with lncRNA TUG1 high expression and low expression was carried out by log-rank test. 
LncRNA TUG1 expression in AML cell lines and control. LncRNA TUG1 expression was increased in AML cell lines including KG-1, MOLM-14, Kasumi-6, THP-1 and HL-60 compared to control cells, and the most significant increase was presented in KG-1 cells. Comparison between two groups was performed using t-test. NS non-significant, *
LncRNA TUG1 expression in AML patients (2.070 (0.981–3.373)) was increased compared to controls (0.746 (0.343–1.686)) (
Correlation of lncRNA TUG1 expression with clinicopathological characteristics in AML patients
LncRNA TUG1 expression was positively correlated with WBC counts (
LncRNA TUG1 expression in non-CR patients and CR patients
There were 56 non-CR patients and 180 CR patients after induction chemotherapy, and lncRNA TUG1 expression in non-CR patients (2.611 (1.393–3.725)) was higher than that in CR patients (1.946 (0.857–3.315)) (
EFS and OS in AML patients with lncRNA TUG1 high expression and low expression
The median EFS in AML patients with lncRNA TUG1 high expression (9.0 (95% CI: 7.1–10.9) months) was shorter than (
Correlation of lncRNA TUG1 expression with AML cell proliferation and apoptosis. (A) At 24 hours post-transfection in KG-1 cells, lncRNA TUG1 expression was reduced in TUG1 (
In order to further understand the underlying mechanism of lncRNA TUG1 in AML, cells experiments were performed. LncRNA TUG1 expression was increased in AML cell lines including KG-1 (
Function of lncRNA TUG1 in cell proliferation and apoptosis in AML
At 24 hours post-transfection in KG-1 cells, qPCR assay was performed and disclosed that lncRNA TUG1 expression was decreased in TUG1 (
Discussion
In the present study, we observed that: (1) LncRNA TUG1 high expression was correlated with advanced clinicopathological characteristics as well as shorter EFS and OS in AML patients. (2) Knockout of lncRNA TUG1 repressed proliferation and facilitated apoptosis of KG-1 cells in vitro.
LncRNA TUG1 is considered as a novel oncogene, which is highly expressed in various cancers and correlates to advanced disease conditions in cancer patients. In a recent study, lncRNA TUG1 expression is detected to be elevated in endometrial carcinoma tissues compared to adjacent normal tissues [12]. Another study demonstrated that lncRNA TUG1 is overexpressed in intrahepatic cholangiocarcinoma, and it associates with poor clinicopathological characteristics including advanced tumor stage, intrahepatic metastasis, lymph node metastasis and perineural invasion [13]. Also, lncRNA TUG1 is highly expressed in gastric cancer and positively correlates with invasion depth and TNM stage in gastric cancer patients [14]. These previous studies illuminate that lncRNA TUG1 high expression is associated with aggravated disease conditions in patients with different cancers. In addition, several studies have illustrated the cancerogenic function of lncRNAs such as lncRNA H19 and lncRNA zinc finger antisense 1 (ZFAS1) in AML [15, 16]. However, considering that the tumorigenic function of lncRNA TUG1 in hematological malignancies, especially in AML, remains obscure, we aimed to investigate the correlation of lncRNA TUG1 with clinicopathological characteristics and prognosis in AML patients. From our result, lncRNA TUG1 expression was elevated in AML patients compared to controls and was positively correlated with WBC count and poor risk stratification in AML patients. The possible explanation could be that upregulation of lncRNA TUG1 altered corresponding signaling pathways to promote proliferation and suppress apoptosis of AML cells, hence facilitated disease progression and worsened disease conditions in AML patients, which was supported by the following in vitro experiment.
In the meantime, the prognostic value of lncRNA TUG1 has been investigated in researches on cancer, which indicate that lncRNA TUG1 is negatively correlated with prognosis in cancers including muscle-invasive bladder cancer, colorectal cancer and esophageal squamous cell carcinoma [17, 18, 19]. Although one recently published study by Wang et al. illustrates the negative correlation of lncRNA TUG1 with EFS and OS in AML patients, the sample size in that previous study is relatively small, and their patients mainly come from East China [11]. Therefore, in the present study, we enrolled 236 patients, which was relatively larger sample size and from a different region (Middle China) and discovered that lncRNA TUG1 high expression was correlated with worse EFS and shorter OS in AML patients. Our results were in accordance with that of Wang et al. and the possible explanations were: (1) LncRNA TUG1 altered genes and corresponding cellular signaling pathways to promote AML cell growth, facilitating disease progression and increasing disease severity, thereby contributed to poor prognosis in AML patients. (2) LncRNA TUG1 might attenuate drug-sensitivity of cells to alter the chemotherapy outcomes, hence increased the recurrence of disease and led to poor prognosis in AML patients. However, the follow-up duration in this study was relatively short with the median follow-up time being 17 months (range from 1-36 months). Thus, the long-term effect of lncRNA TUG1 on prognosis in AML patients was not investigated, and further study with longer follow-up was needed. In addition, due to lack of non-hematologic malignancy patients underwent bone marrow biopsy, the number of controls (
In order to explore the underlying mechanisms of lncRNA TUG1 in various cancers, in vitro experiments have been extensively performed. For example, lncRNA TUG1 promotes cell proliferation, migration and invasion by inhibiting miR-29c in bladder cancer and enhances cancer cell growth through sponging miR-382 in pancreatic cancer [10, 20]. Also, lncRNA TUG1 increases proliferation and decreases apoptosis in osteosarcoma cells via downregulating miR-132-3p and upregulating expression of SOX4 [9]. These studies imply that lncRNA TUG1 promotes tumorigenesis by initiating cell proliferation and inhibiting cell apoptosis in different cancers, however, its mechanism in leukemias remains unclear. To further understand the function of lncRNA TUG1 in AML, we transfected TUG1 inhibitor mimic and blank mimic plasmids into KG-1 cells and assessed cell proliferation and cell apoptosis rate. Our result revealed that knockout of lncRNA TUG1 suppressed proliferation and accelerated apoptosis of KG-1 cells, suggesting that it was involved in the pathogenesis of AML by regulating cellular proliferation and apoptosis, which was in line with the results from previously stated in vitro experiments and revealed the potential therapeutic application of lncRNA TUG1 in AML.
In summary, lncRNA TUG1 predicts increased disease severity and poor prognosis in AML patients, and its knockout decreases proliferation and increases apoptosis of AML cells.
Footnotes
Conflict of interest
The authors declare that they have no conflict of interest.
