Abstract
Treatment for acute myeloid leukemia (AML) generally consists of a combination of cytarabine and an anthracycline. Although induction therapy leads to complete remission (CR) for most patients, refractoriness to chemotherapy or relapse after initial response is associated with poor outcomes. The 1-year survival rates after first relapse have been reported at 29%, declining to 11% at 5 years. Prognosis is particularly poor among older patients whose higher prevalence of unfavorable cytogenetics and high frequency of comorbidities diminish their ability to tolerate intensive chemotherapy. There is no standard of care for relapsed/refractory (R/R) AML, and no new therapies have shown consistently superior outcomes in this setting in over two decades. Vosaroxin is an anticancer quinolone derivative (AQD) that was evaluated in combination with cytarabine for the treatment of R/R AML in the randomized, double-blind, placebo-controlled, phase III VALOR study (n = 711). Compared with placebo/cytarabine, the vosaroxin/cytarabine regimen demonstrated favorable CR rates and survival in patients ⩾60 years of age, with toxicities similar to other AML regimens. Here we review outcomes of recent studies of commonly used chemotherapy regimens for the treatment of R/R AML and evaluate the results of the VALOR trial in the context of the current treatment landscape.
Introduction
Acute myeloid leukemia (AML) is a heterogeneous hematologic malignancy that arises from uncontrolled clonal expansion of myeloid stem cells in the peripheral blood or bone marrow. 1 AML accounts for 1.2% of all cancers, with an estimated 19,950 new cases and 10,430 deaths in the United States in 2016. 2 It is primarily a disease of older adults, with a median age of ~70 years at diagnosis; patients ⩾65 years of age account for 53–57% of all cases, and those >85 years of age account for 5–10% of all cases. 3 The estimated 5-year survival rate across all patients is 25.9%; 2 among patients >60 years of age, it is <10%. 4
Treatment of AML consists of induction chemotherapy, generally a combination regimen of cytarabine and an anthracycline (e.g. idarubicin or daunorubicin), followed by post-remission consolidation therapy to achieve durable disease control. Strategies for treatment are stratified based on age, with 60 years considered a therapeutic divergence point. 5 For older patients, one must take into consideration the higher prevalence of unfavorable cytogenetics, antecedent myelodysplasia, and multidrug resistance in this population, as well as the higher frequency of comorbidities that may impact the ability to tolerate intensive therapies.5,6 As such, induction strategies for older patients often rely on a patient’s individual characteristics, with or without inclusion of post-remission therapy. 5
Although induction chemotherapy leads to complete remission (CR) in over 60% of patients, overall outcomes, including survival, remain poor for the vast majority. 4 This poor prognosis is attributed to refractoriness of disease to chemotherapy and relapse after achieving CR. Refractory disease has been reported in one-quarter of AML patients, and nearly 50% relapse after initial response. 7 Outcomes in clinical studies have been consistently poor in the relapsed setting, with lower rates of survival associated with increased age, shorter duration of first CR, unfavorable karyotype at diagnosis, and prior allogeneic stem cell transplant. 8 After first relapse, 1-year survival rates have been reported at 29%, declining to a 5-year survival rate of 11%. 9 Increased age is a predictor of poor prognosis in patients with AML, with CR rates declining from 78% of patients ⩽39 years of age to 66% in patients 40–59 years, 45% in patients ⩾60 years, and 35% in patients ⩾70 years of age. 4 For patients ⩾60 years of age, relapse occurs in 80–90% of patients within 5 years of diagnosis.4,5,10 In patients with relapsed disease, overall survival (OS) also declines with age: 1-year survival rates decline from 36% in patients ⩽35 years of age to 30% in patients 36–45 years and 25% in patients >45 years of age; corresponding 5-year survival rates decline from 18% to 11% and 8%, respectively, in the same age cohorts. 9
In the relapsed/refractory (R/R) setting, there is no standard of care. Over the past two decades, no alternative approach has definitively proven to be superior to the cytarabine-anthracycline regimens, and enrollment in a clinical trial is recommended.5,6,11 Chemotherapy alone is not curative in relapsed patients, but in eligible patients, chemotherapy can often be considered as a bridge to allogeneic stem cell transplantation, the only curative option for R/R AML. Approaches to improving remission rates have included altering doses of agents, use of novel combinations of conventional chemotherapies, and use of novel investigational agents.
VALOR is a recently completed phase III clinical trial evaluating vosaroxin, an anticancer quinolone derivative (AQD), in combination with cytarabine in patients with R/R AML. 12 To place the results of VALOR in the context of the current R/R AML treatment landscape, we reviewed outcomes of recent clinical trials evaluating representative chemotherapy regimens for aggressive therapy for R/R AML. 5 These include (1) high-dose cytarabine (HiDAC) with or without an anthracycline; (2) etoposide and cytarabine with or without mitoxantrone; (3) fludarabine, cytarabine, and granulocyte colony-stimulating factor (G-CSF; FLAG); (4) cladribine, cytarabine, and G-CSF (CLAG) with or without mitoxantrone; and (5) clofarabine-containing regimens.
Methods: Literature review
To understand the current landscape for treatment of R/R AML, a literature review was conducted to identify treatment regimens in current practice. We searched in PubMed and Web of Science for phase II or III studies evaluating the efficacy and safety of treatment regimens used in the R/R AML setting. The following search strings were used to identify potential studies: (cytarabine or ‘chemotherapy’ or salvage) AND (relapse* or refractory) AND (AML[title] or ‘acute myeloid leukemia’[title] or ‘acute myeloid leukaemia’[title] or ‘acute myelogenous leukemia’[title]). The search was limited to English language publications on clinical trials from January 2000 onwards. We confirmed our selection of treatment regimens by review of current treatment guidelines and select publication bibliographies and limited our search to studies of ⩾50 patients published after the year 2000 to assess outcomes that are reflective of current practice, including supportive care practices. Data presented reflect only current therapies and do not include the investigational arm of published studies.
Results
Summary of the efficacy and safety of select salvage therapies in patients with R/R AML
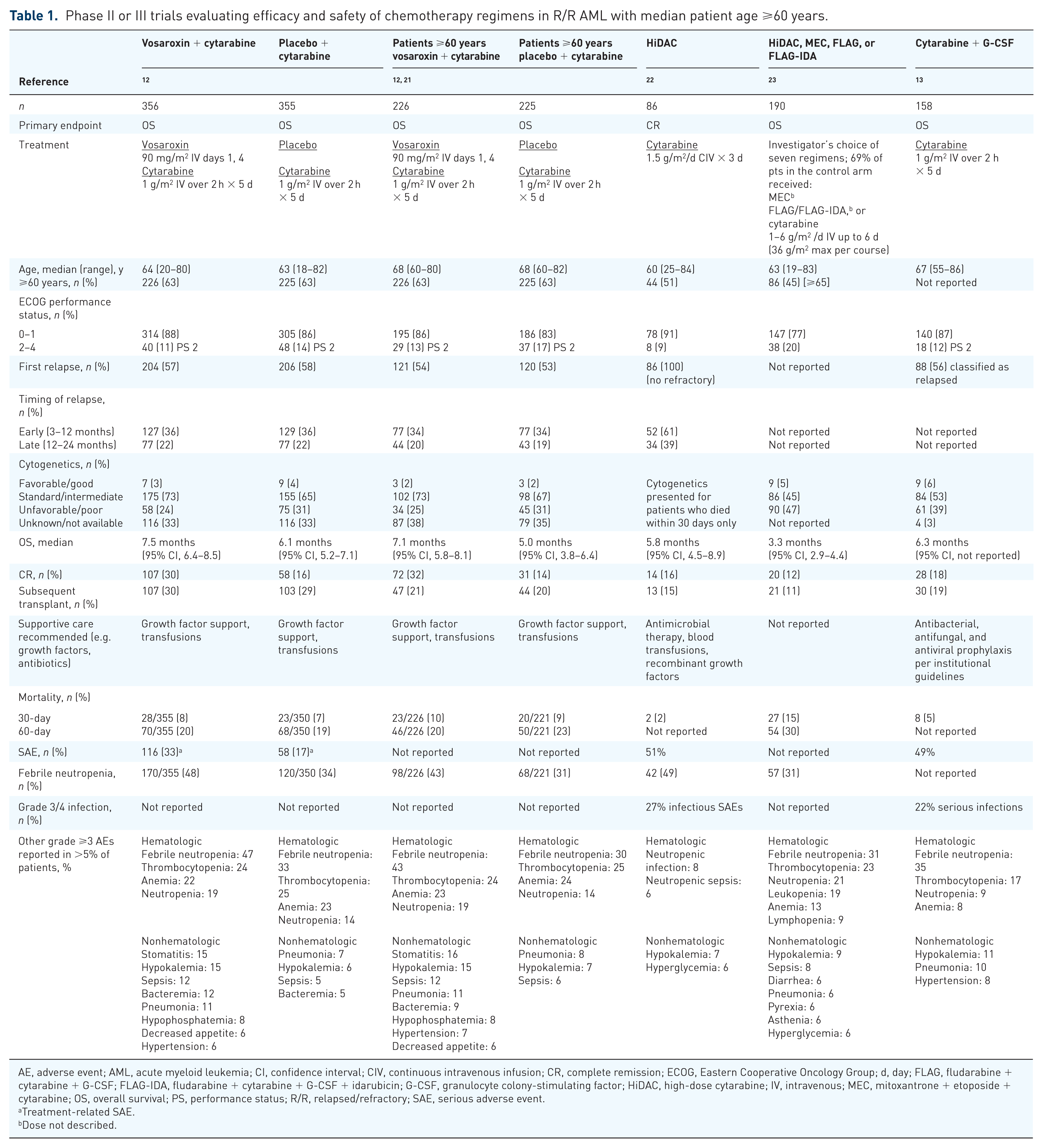
A total of 11 studies were identified, and information on baseline characteristics, treatment, efficacy endpoints (OS and remission rates), and safety was collected for each study. For ease of comparison, studies with a median patient age ⩾60 years are presented in Table 1 and studies with a median patient age <60 are presented in Table 2. The number of patients evaluated in each study ranged from 50 to 190.
Phase II or III trials evaluating efficacy and safety of chemotherapy regimens in R/R AML with median patient age ⩾60 years.
AE, adverse event; AML, acute myeloid leukemia; CI, confidence interval; CIV, continuous intravenous infusion; CR, complete remission; ECOG, Eastern Cooperative Oncology Group; d, day; FLAG, fludarabine + cytarabine + G-CSF; FLAG-IDA, fludarabine + cytarabine + G-CSF + idarubicin; G-CSF, granulocyte colony-stimulating factor; HiDAC, high-dose cytarabine; IV, intravenous; MEC, mitoxantrone + etoposide + cytarabine; OS, overall survival; PS, performance status; R/R, relapsed/refractory; SAE, serious adverse event.
Treatment-related SAE.
Dose not described.
Phase II or III trials evaluating efficacy and safety of chemotherapy regimens in R/R AML with median patient age <60 years.

AE, adverse event; ALT, alanine aminotransferase; AML, acute myeloid leukemia; AST, aspartate aminotransferase; CI, confidence interval; CLAG, cladribine + cytarabine + G-CSF; CR, complete remission; CRp, complete remission with incomplete platelet recovery; CSF, colony-stimulating factor; ECOG, Eastern Cooperative Oncology Group; d, day; FLAG, fludarabine + cytarabine + G-CSF; G-CSF, granulocyte colony-stimulating factor; HiDAC, high-dose cytarabine; IV, intravenous; MEC, mitoxantrone + etoposide + cytarabine; NR, not reached; ORR, overall response rate; OS, overall survival; PS, performance status; R/R, relapsed/refractory; SAE, serious adverse event; SC, subcutaneous; SD, standard deviation; WHO, World Health Organization.
Includes nine patients with myelodysplastic syndrome.
Includes grades 3 and 4 tachycardia, arrhythmias, cardiomyopathy, pericarditis, pericardial effusion, myocardial infarction/ischemia, congestive heart failure, hypertension, and cardiovascular disorder.
Includes grades 3 and 4 hepatic failure, hepatomegaly, jaundice, liver damage, and abnormal liver function tests.
Patient demographics and baseline characteristics
Patient baseline characteristics were variable across studies. The median age varied from 45–67 years (range, 17–84 years); most studies included both younger and older patients (with the exception of the study by Faderl and colleagues, in which patients <60 years of age were excluded). 13 Disease status (refractory versus early relapsed versus late relapsed) also varied across studies, with the proportion of patients in first relapse ranging from 10–100%. In general, most patients had an Eastern Cooperative Oncology Group or World Health Organization performance status of 0–1 at baseline (range, 59–100%), and most had intermediate or unfavorable/poor cytogenetic status.
Dosing
Cytarabine was the backbone of treatment regimens across all studies reviewed. Dosing and schedules for cytarabine were variable, with doses ranging from 1.0–6.0 g/m2 daily for 3–6 days.
Efficacy
Efficacy outcomes were comparable with those reported previously in R/R AML,14,15 with median OS ranging from 3.3–9 months and CR rates from 12–53%. In studies with a median patient age ⩾60 years, median OS was 3.3–6.6 months, and CR rates were 12–35% (Table 2). Across all studies, higher CR rates were generally accompanied by a longer median OS. Rates of subsequent transplantation ranged from 11–23%.
For studies that stratified CR rates by age, CR rates tended to be lower in older patients compared with the overall patient population. In HiDAC-treated patients, 7% of patients >60 years of age achieved CR, compared with 16% in the overall population. In mitoxantrone-etoposide-cytarabine (MEC)-treated patients, 20% of patients ⩾50 years of age achieved CR, compared with 25% in the overall population. In FLAG-treated patients, 9% of patients >60 years of age achieved CR, compared with 46% in the overall population. In CLAG-treated patients, 50% of patients 54–66 years of age achieved CR compared with 58% in the overall population.
Safety
Grade ⩾3 febrile neutropenia was reported in 31–94% of patients, and grade ⩾3 infection rates ranged from 40–91% where reported. Early (30-day) mortality rates ranged from 2–17%; infections or sepsis were the most common causes. Other causes of early mortality included hemorrhage, cardiac failure, and progressive disease.
Findings from the VALOR study
Vosaroxin is an AQD that intercalates DNA and inhibits topoisomerase II. 16 It elicits its anticancer activity by inducing replication-dependent, site-selective DNA damage, G2 arrest, and apoptosis. Vosaroxin is distinct from traditional topoisomerase inhibitors in that it is minimally metabolized, not a substrate of p-glycoprotein, and can induce p53-independent apoptosis.16–18 The rationale for combining vosaroxin with cytarabine in clinical studies was based on preclinical studies demonstrating synergistic antiproliferative activity with the combination. 19 Proof of concept of anticancer activity of the combination regimen was demonstrated in a phase II study in the R/R AML setting. 20
VALOR was a randomized, double-blind, placebo-controlled phase III trial evaluating vosaroxin plus cytarabine versus placebo plus cytarabine in patients with refractory or first-relapsed AML who had received no more than two prior cycles of induction therapy (including one cycle of cytarabine + anthracycline). 12 Vosaroxin was administered intravenously (IV) at 90 mg/m2 over 10 min on days 1 and 4, and 70 mg/m2 in subsequent cycles. Cytarabine was administered at 1 g/m2 IV over 2 h on days 1–5. Patients received induction therapy for 1 or 2 cycles. Patients who had CR or CR with incomplete platelet recovery (CRp) received an additional 1–2 cycles of consolidation therapy.
Primary endpoints were OS and all-cause mortality at 30 and 60 days. The secondary endpoint was CR rate. Other endpoints included combined CR rate, overall remission rate, event-free survival, leukemia-free survival, rate of post-treatment allogeneic stem cell transplant, and safety/tolerability.
A total of 711 patients were randomized (n = 356 to vosaroxin/cytarabine and n = 355 to placebo/cytarabine). Median patient age was 64 years (range, 20–80 years); 63% of patients were ⩾60 years of age (Table 1).
Median OS was 7.5 months in the vosaroxin/cytarabine arm, compared with 6.1 months in the placebo/cytarabine arm (p = 0.06). CR rate was 30.1% in the vosaroxin/cytarabine arm versus 16.3% in the placebo/cytarabine arm (p = 0.0001), and composite CR rate [CR + CRp + CRi (CR with incomplete platelet or neutrophil recovery)] was 37.1% versus 18.6%, respectively (p < 0.0001). The rate of subsequent transplant was 30.1% versus 29.0% in the vosaroxin/cytarabine and placebo/cytarabine arms, respectively.
In a prespecified analysis of patients stratified by age, median OS of patients ⩾60 years of age was 7.1 months in the vosaroxin/cytarabine arm and 5.0 months in the placebo/cytarabine arm (hazard ratio 0.75; 95% confidence interval, 0.62–0.92; p = 0.003). CR rates in patients ⩾60 years of age were similar to those in the overall population: 31.9% in the vosaroxin/cytarabine arm versus 13.8% in the placebo/cytarabine arm (p < 0.0001).
Evaluation of safety showed that the 30-day all-cause mortality rate was within the range observed with other regimens. The 30-day all-cause mortality rate was 7.9% in the vosaroxin/cytarabine arm and 6.6% in the placebo/cytarabine arm; the 60-day rate was 19.7% and 19.4%, respectively. Most deaths due to toxicity were associated with infection. Grade ⩾3 myelosuppression and grade ⩾3 infection were common and had rates comparable with those observed with other regimens. Grade ⩾3 stomatitis occurred in 14% of patients. In comparison, rates of grade ⩾3 stomatitis up to 25% were reported with other regimens.
Expert opinion/perspective
R/R AML remains one of the major challenges in the treatment of AML. More than half of all newly diagnosed patients will eventually fall into the R/R category, and these patients face a dismal outlook. Hematopoietic stem cell transplant provides some hope for long-term survival; however, because pretransplant leukemia burden has been shown to impact transplant outcomes, it is desirable to first obtain a disease response with leukemia-directed therapy. Even in cases where the option of stem cell therapy is not available, response to leukemia-directed therapy is closely related to OS. Therefore, effective second-line treatment is of paramount importance. Historically, regimens for fit patients were based on anthracyclines and cytarabine in an attempt to mimic first-line induction therapy; however, this produced suboptimal response rates and toxicities. Trials in which a third or even fourth agent were added to, or substituted for, an anthracycline or cytarabine created a plethora of protocols without producing significant impact on practice patterns and outcomes. While a substantial number of combinations and agents showed promising results in early clinical trials, almost universally they failed to improve survival in randomized trials.
In that regard, the VALOR trial provides some interesting observations. Caution is necessary when comparing data from the VALOR trial with other trials conducted in similar settings but in smaller, sometimes younger patient populations, or trials looking at different primary endpoints (i.e. asking different questions). For example, the median OS observed in the experimental arm in the VALOR trial seems to fall within the previously reported range; however, when compared with studies in patients with a similar median age, it may be improved. Especially interesting are the results of the predefined subgroup analysis that showed improved survival in patients 60 years of age and older. This group has a particularly dismal outlook and has often been underrepresented in larger clinical trials.
While increased toxicity was observed in the experimental arm compared with the placebo plus cytarabine control arm, mostly due to a higher incidence of neutropenia and neutropenia-related adverse events, 30-day and 60-day mortality remained at a level that compared favorably with rates previously reported for other therapies. One conclusion that may be drawn from review of previous trials is that achievement of an improved CR rate with more aggressive therapy does not necessarily improve OS to any significant extent because of increased toxicity that frequently translates into increased early mortality, particularly in older patient populations. The data obtained from the VALOR trial indicate that it is possible to improve the CR rate in elderly patients without increasing treatment-related mortality, and therefore provides patients with a valuable treatment option.
In conclusion, the VALOR trial, one of the largest randomized trials exploring treatment options for R/R AML to date, is encouraging. In particular, the results in patients ⩾60 years of age, a group targeted in a predefined subgroup analysis, indicate that vosaroxin/cytarabine may be an important new treatment for this high-risk population.
Footnotes
Funding
Medical writing assistance was provided by Janis Leonoudakis of Powered 4 Significance LLC, Annandale, NJ, USA and was funded by Sunesis Pharmaceuticals, South San Francisco, CA, USA.
Conflict of interest statement
Robert Stuart has received research funding from and has served as a consultant to Sunesis Pharmaceuticals. Valeriy Sedov has no relevant conflicts of interest to disclose.
