Abstract
BACKGROUND:
Drug resistance in clinical cancer treatment has become an issue.
OBJECTIVE:
We focus on abnormally expressed lncRNAs in glioma and investigating the function of PVT1.
METHODS:
The paclitaxel-resistant glioma cells SHG-44 RE was obtained through screening the SHG 44 cells that were cultured in medium containing a certain concentration of paclitaxel. Cell survival of SHG 44 RE and SHG 44 cells under the treatment of paclitaxel was detected by MTT assay. The aberrant expressed lncRNAs were screened out with microarray analysis. Further qRT-PCR was utilized to validate the expression of lncRNA PVT1 in the two cells. After manipulating the expression of PVT1, cell viability and apoptosis were measured by MTT and flow cytometry respectively.
RESULTS:
LncRNA PVT1 was overexpressed in glioma cells SHG-44 RE compared with parent SHG-44 cells. Down-regulation of lncRNA PVT1 inhibited the SHG-44 RE cell viability and increased glioma SHG-44 RE cells apoptosis after paclitaxel treatment, suggesting that inhibition of lncRNA PVT1 improved paclitaxel sensibility in human glioma cells.
CONCLUSION:
Down-regulation of PVT1 could enhance chemosensitivity of paclitaxel, induce apoptosis of glioma cells and noteworthy inhibit glioma cells proliferation. Our findings of PVT1 could contribute to attenuate paclitaxel resistance in clinical medicine.
Introduction
Gliomas are one of the most common brain tumors which account for about 70% of main brain tumors in adults [1]. Present therapy containing surgery, chemotherapy, and radiotherapy had little impact on improving the prognosis while only 5% live for more than 5 years and a median overall survival rate between 12–15 months. Therefore, the effective treatment and early diagnosis of glioma is urgently concerned for improving the outcome for patients.
Long noncoding RNAs (lncRNAs), by definition, are a class of transcripts of
Paclitaxel alias yew Taxol, the best natural anti-cancer drugs have been found, is widely used in clinical breast cancer [5], ovarian cancer [6] and lung cancer [7] treatment. Chemoresistance is one of the major obstacles in the treatment of cancer clinically. Most malignancies are sensitive at the beginning of treatment, but develop drug resistance during the process of chemotherapy. The mechanisms of chemotherapy resistance are complex and diverse [8]. P-glycoprotein (Pgp) overexpression is a notable molecular mechanism which involves the process of multidrug resistance (MDR) and the paclitaxel resistance in cancer is also included in.The increase in Pgp expression is to pump out a series of chemotherapy agents that are structurally and functionally different from cancer cells, leading to a decrease in the accumulation of anti-cancer drugs in the cells [9]. With the inspiration of lncRNA NEAT1 regulating ZeB1 expression via mir-194 to conduce to paclitaxel-resistance in ovarian cancer cells [2] and lncRNA SNHG12 conduces to multidrug resistance in non-small cell lung cancer through activating the MAPK/Slug pathway [10].
In this study, We explored the expression of PVT1 in glioma, and investigated the relationship between PVT1 and paclitaxel sensitivity and finally confirmed that downregulating lncRNA PVT1 could attenuate the paclitaxel resistance of glioma cells.
Methods and materials
Cell culture
Glioma cell lines SHG-44 were provided from the Chinese Academy of Stem Cell Bank (Shanghai, China) and cultivated in RPMI-1640 (GIBCO, Grand Island, NY, USA) medium containing 1.5 g/L NaHCO
Generation of a Taxol-resistant cell line
The SHG-44 cells (1
Microarray assay
Microarray analysis was performed by KangChen Bio-tech (Shanghai, China). In short, the labeled cRNAs were hybridized onto the 4
Cell transfection
Si-NC and si-PVT1 were provided from GenePharma (Shanghai, China) commercially. The sequence of PVT1-specific siRNAs (si-PVT1) was 5’-GCUUG GAGGCUGA-GGAGUUTT-3’. Then the si-NC and si-PVT1 were transiently transfected in SHG-44 RE via using Lipofectamine 2000 Reagent (Invitrogen, Carlsbad, CA, USA) following the producer’s instructions. After 48 hours, siRNA was utilized to transfect cells and QRT-PCR was performed to determine the efficiency of transfection.
RNA isolation and quantitative PCR analysis
RNA from glioma cells was totally extracted with TRIzol reagent (Invitrogen, Carlsbad, CA, USA). After quantification by NanoDrop2000 (Thermo Fisher Scientific Inc, Waltham, MA, USA), 200 ng of total RNA was used for reverse transcription with ReverTra Ace qPCR RT Kit (Toyobo, Japan) following the instructions.
According to the contents of internal reference GADPH, which forward and reverse primers were 5’-CCTTCATTGACCTCAACTAC-3’ and 5’-GGAA GGCCATGCCAGTGAGC-3’, three replicate experiments were conducted on each group respectively, and the relative expression amount of PVT1 was calculated by relative quantification (using 2
MTT assays
Cells were dosed in a single cell suspension with 10% fetal bovine serum and cultivated in 96-well plates at 3000 cells per well in a volume of 200
Colony formation assay
For the plate colony formation assay, cells in logarithmic growth phase were resuspended and seeded into 6-well plates which contain DMEM medium with 0.5% agar. After two weeks, colonies were stained with 0.1% crystal violet (Beyotime, Shanghai, China) for 30 minutes and more than 50 cells were counted with a microscopy.
Flow cytometry (FCM) analysis
Glioma cells SHG-44 were cultivated for 48 h after transfection before cell apoptosis analysis. The incubated cells were stained with a FITC Annexin V Apoptosis Detection Kit (BD Biosciences, Bedford, MA, USA), and FACS Calibur flow cytometer (BD, USA) was used to analyze the cell cycle. The experiment was repeated for three times.
Statistical analysis
In this research, all data were expressed as mean
Effect of time and paclitaxel concentration on cell growth inhibition rate. (A) Under the condition of paclitaxel concentration of 2.5 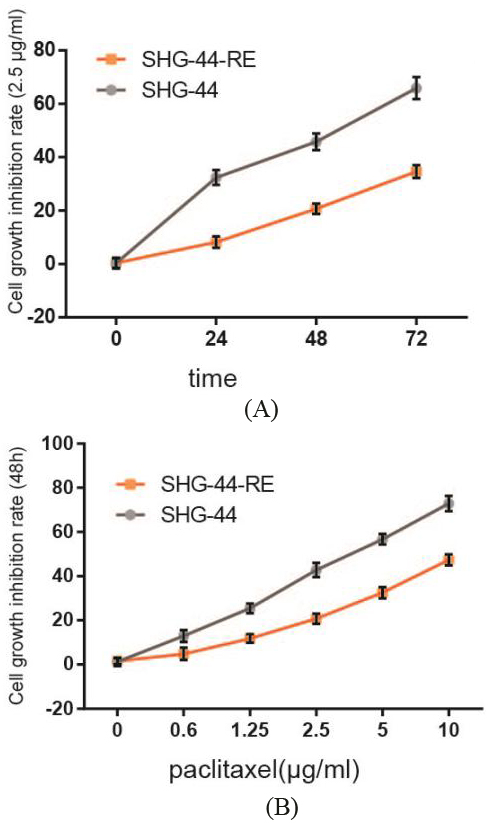
Cluster diagram of lncRNA expression data in glioma was detected by the Agilent Human LncRNA 4 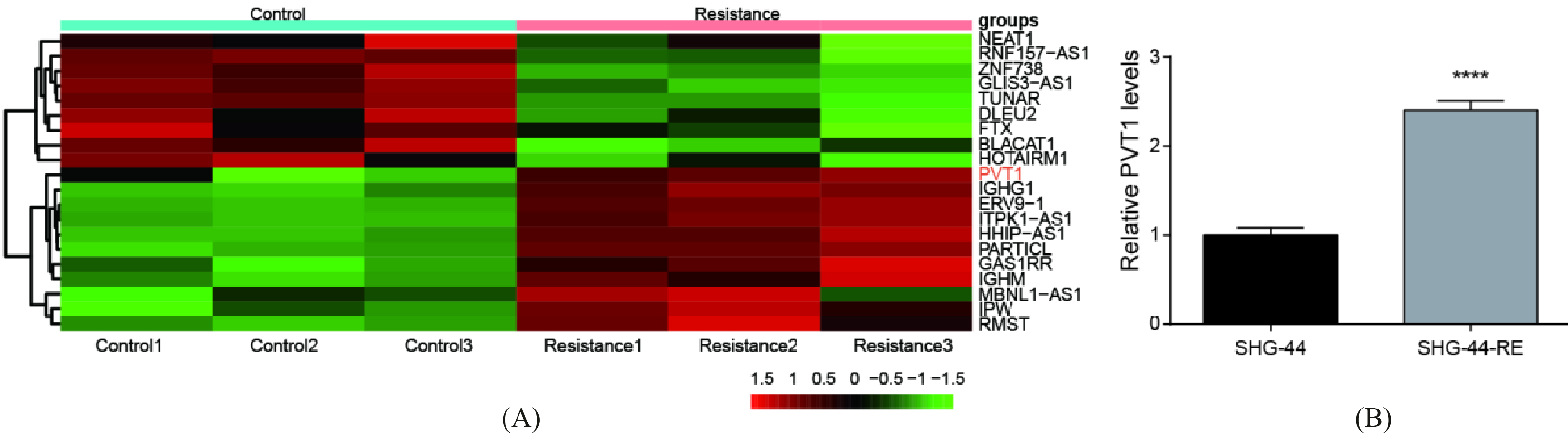
LncRNA PVT1 is upregulated in human glioma cell lines
One of the main obstacles in the clinical therapy of cancers today is chemoresistance. We firstly obtained paclitaxel-resistant cell lines (SHG-44-RE) through screening. Then we detected the effect of different time on the cell growth inhibition between SHG-44-RE and SHG-44 under the condition of paclitaxel concentration of 2.5
In order to investigate the different expression of lncRNAs in paclitaxel-resistance glioma cells, microarray assays were performed. The results showing by heat map revealed that in comparison with the normal cell lines, lncRNA PVT1 seemed to have higher expression in majority of glioma (Fig. 2A). Next, in order to examine the association of PVT1 and paclitaxel resistance in glioma cells, the expression of PVT1 was detected via QRT-PCR. Further development of QRT-PCR was performed to quantify PVT1 in the paclitaxel-resistant glioma cell lines (SHG-44-RE) and the corresponding parental cell lines (SHG-44). The relative PVT1 level was significant high in paclitaxel-resistant glioma cell lines in comparison with the corresponding parental cell lines (Fig. 2B, **
The cell proliferation in vitro. (A) Relative PVT1 levels shown in SHG-44-RE, si-NC and si-PVT1. (B) Under the conditions of 48 hours culture, the effect of different concentrations of paclitaxel on the growth inhibition of cells among SHG-44-RE, si-NC and si-PVT1.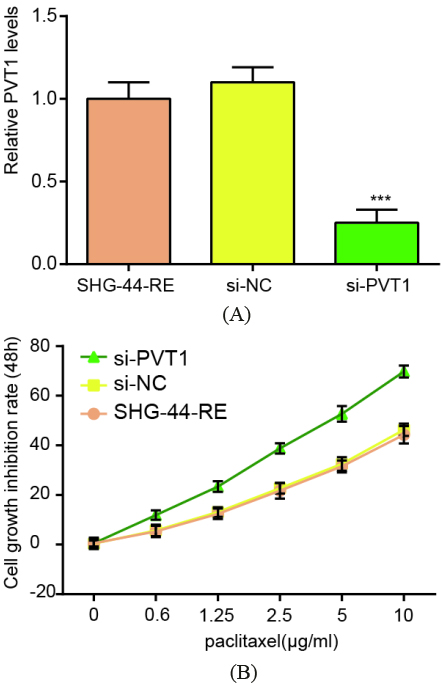
The cell apoptosis in vitro. (A) Apoptosis rates of glioma cell lines were detected with paclitaxel at a concentration of 0 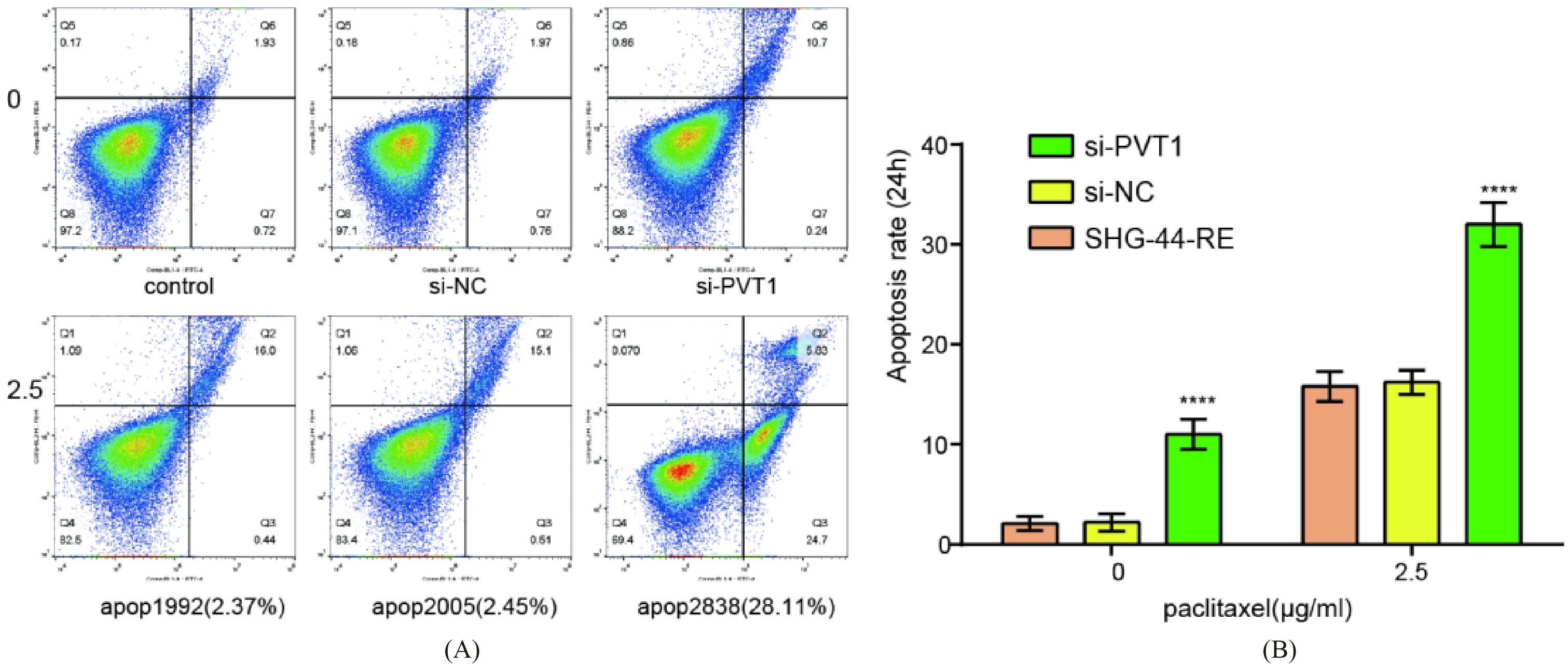
The paclitaxel sensitivity of glioma. (A) and (B) Colony formation assays to determine the influence PVT1 on paclitaxel resistance. The colony formation of three groups (control, si-NC and si-PVT1) treated with paclitaxel at a concentration of 0 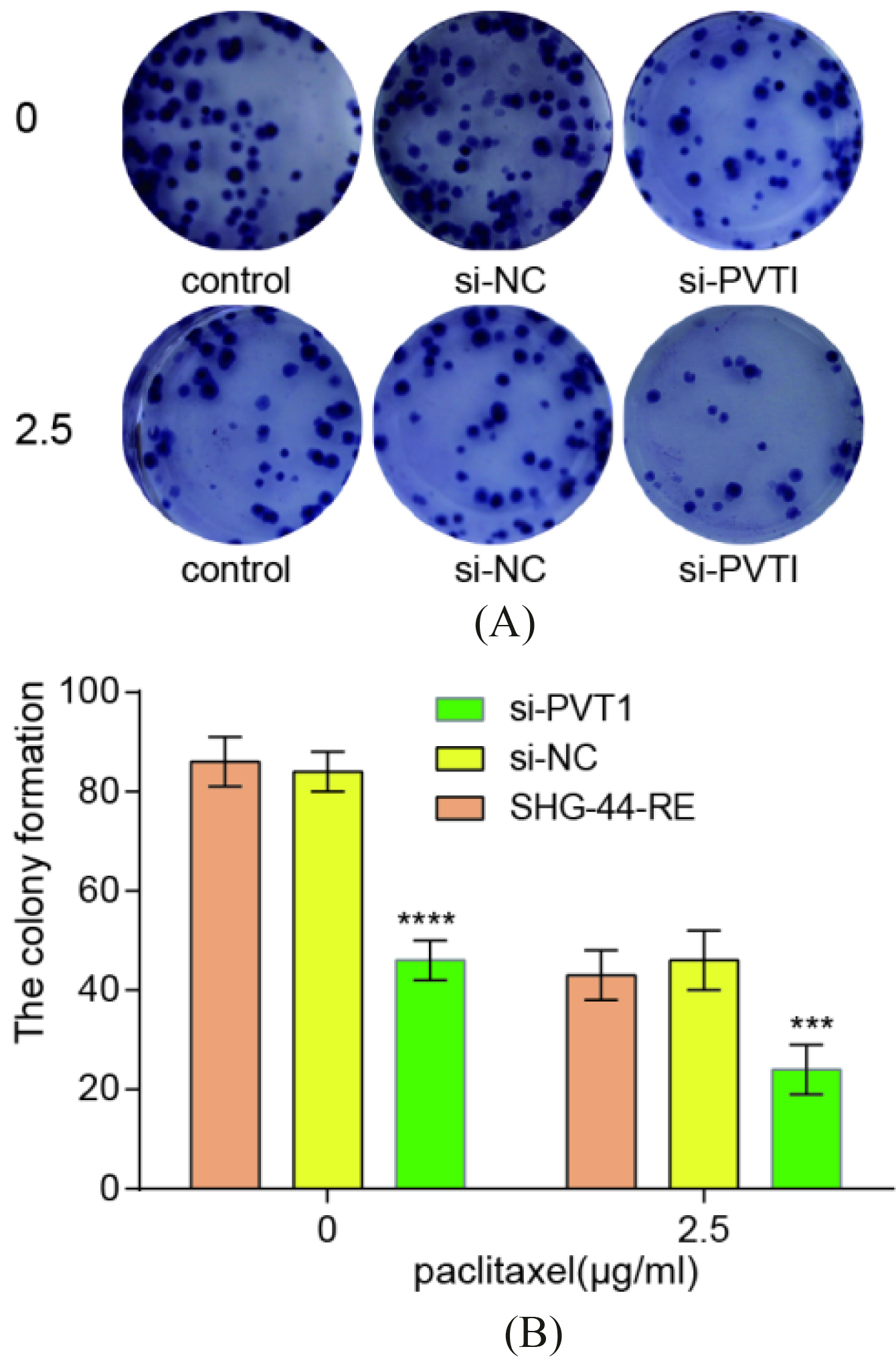
To further examine the function of PVT1 in paclitaxel resistance, an PVT1 downregulation vector was transiently transfected into the SHG-44-RE cells, and a negative control (NC) in our study is the scrambled siRNA. The results indicated that the relative PVT1 level in si-PVT1 was very high compared with control and si-NC groups (Fig. 3A, **
The expression of lncRNA PVT1 affects the cell apoptosis in vitro
To explore the influence of PVT1 on cell apoptosis, flow cytometry analysis was carried out. Data showed that downregulation of PVT1 leaded to increasing apoptosis in si-PVT1 cells compared with the control group and the NC group when treated with or without paclitaxel, suggesting that PVT1 knockdown caused an increase in apoptosis treated with paclitaxel (Fig. 4A and B, **
The down expression of lncRNA PVT1 affects the paclitaxel sensitivity of glioma in vitro
Colony formation assays made it clear that the proliferation of SHG-44-RE cells transfected with si-PVT1 was lower in comparison with si-NC group and control group when treated with or without paclitaxel, and the effect of paclitaxel with higher paclitaxel concentration (2.5
Discussion
Nowadays more and more evidence suggests that most lncRNAs are biologically relevant [11]. It has been found that LncRNAs are usually transcribed in the mammalian genome [12]. Primary structure, secondary structure and expression levels of lncRNAs, as well as alterations of their cognate RNA-binding proteins, generally relate to human diseases, particularly in cancer area [12]. Maladjustment of lncRNA has also been discussed to be conducive to the exploration of the pathogenesis of tumor and provide new therapeutic opportunities for the further treatment of tumor [13]. For example, Pan et al. has proved that chemoresistance is regulated in docetaxel-resistant lung adenocarcinoma cells by lncRNA ROR via epithelial mesenchymal transition pathway [14] and Chen et al. has proved that lncRNA CCAT1 promotes chemoresistance acting as an oncogene in docetaxel-resistant lung adenocarcinoma cells [15]. In addition, many studies revealed that PVT1 has an important influence on drug resistance of some certain tumors, like promoting cell proliferation and indicating a poor prognosis of gastric cancer [16]. However, scarcely any of the studies have found the influence of PVT1 on paclitaxel resistance in glioma cells. In this study, the relative PVT1 level has been evaluated via QT-qPCR and it is confirmed that PVT1 is upregulated in human glioma cell lines. In addition, we provided the probably mechanical contact between lncRNA PVT1 and its antitumor activity in subsequent experiments, demonstrating the functional significance of lncRNA PVT1 in vitro.
To find deeply effects of lncRNA PVT1 in glioma, a series of tests were conducted, with some vital discovery that downregulation of PVT1 restrains cell proliferation and promotes cell apoptosis, whose principle are akin to the following examples. An et al. found that LncRNA NEAT1 is conducive to paclitaxel resistance by regulating ZeB1 expression through sponging miR-194 in ovarian cancer cells [2]. Besides, a potential mechanism of PVT1 that upregulating PVT1 could increase carboplatin-docetaxel-induced activity in ovarian cancer has also been studied by Liu et al. [4]. We used microarray analysis, MTT assay, flow cytometry (FCM) analysis and colony formation assay to attain data and results. All of studies mentioned above contribute to our conclusion.
However, some of the restrictions of this study also ought to be taken into account. For instance, we carried out this study only in vitro, experiment in vivo could also be further considered which might better uncover the mechanism of glioma. What’s more, other lncRNAs involved in the paclitaxel resistance were not investigated and the underlying molecular mechanism of PVT1 could be further discussed.
In summary, with an ever so high expression in glioma cells, lncRNA PVT1 is of great importance in paclitaxel resistance in glioma. This study showed that down-regulation of PVT1 effectively inhibited glioma cell proliferation, improved apoptosis and enhanced chemosensitivity of paclitaxel. Our finding provides a novel insight into the mechanism and the function of PVT1. Data gives proof of PVT1 as a glioma promoter and could be a great target for the therapeutic intervention of glioma in clinical medicine.
Conclusion
Taken all together, these experiment recommended that PVT1 could improve the capacity of proliferation, apoptosis and chemotherapy resistance of paclitaxel in glioma cells.
Footnotes
Acknowledgments
This study was supported by Harbin Science and Technology Talent Innovation Fund Project (No. 2016 RAQXJ173); Scientific Research Project of Heilongjiang Provincial Wei Planning Commission (2016-129).
Conflict of interest
The authors declare that they have no conflict of interest.
