Abstract
OBJECTIVE:
Osteosarcoma is the most common malignant tumor of bone with high recurrent rate. miR-486 was downregulated and acted as a tumor suppressor in plenty of tumors. The purpose of this study was to explore how miR-486 worked in osteosarcoma on cell invasion and EMT.
RESULTS:
miR-486 was low expressed in osteosarcoma while PIM1 was overexpressed, and it had negative correlation between miR-486 and PIM1. miR-486 upregulation or PIM1 downregulation could inhibit osteosarcoma cell invasion and EMT. Meanwhile, miR-486 mediated PIM1 expression through binding to PIM1 mRNA 3’-UTR. PIM1 could reveal partial function of miR-486 on osteosarcoma invasion. In addition, miR-486 low expression or PIM1 overexpression predicted poor prognosis of osteosarcoma patients.
CONCLUSION:
miR-486 regulated osteosarcoma cell invasion and EMT through targeting to PIM1. miR-486 low expression or PIM1 overexpression predicted poor prognosis of osteosarcoma patients. The newly identified miR-486/PIM1 axis provides novel insight into the pathogenesis of osteosarcoma.
Introduction
Osteosarcoma (OS), the most common primary malignant tumor of bone, originates in the mesenchymal tissue [1]. Although combination of surgery and chemotherapy could improve the survival, one third of patients with localized osteosarcoma have recurrent or progressive metastases [2, 3]. Due to high malignant degree, metastasis rate and fatality rate, osteosarcoma is regarded as a difficulty in clinical treatment [4]. Thus, it is essential to discover novel pathogenic factors to help early diagnosis and improve prognosis.
MicroRNAs (miRNAs), small no-ncoding RNAs with 22–28 nucleotides, could repress translation or promote target mRNAs degradation through binding to 3’-UTR at post-transcriptional [5, 6]. miRNAs could cause tumor differentiation, metastasis and other functions [7]. miR-486 acted as a tumor suppressor was downregulated in plenty of tumors, including cervical cancer, esophageal cancer, prostate cancer and oral tongue squamous cell carcinoma [8, 9, 10, 11]. In esophageal cancer, Lang and Zhao discovered that miR-486 acted as tumor suppressor and cell cycle, invasion and colony formation [9]. What’s more, miR-486 inhibited cell proliferation, colony formation, cell cycle and tumor growth in breast cancer [12]. Similar findings were discovered by Ye that miR-486 could repress cell proliferation, migration and invasion in cervical cancer [13]. However, there were little studies explore the function of miR-486 on cell invasion and EMT in osteosarcoma.
Provirus integrating site Moloney murine leukemia virus (PIM) family were highly conserved serine/threonine kinases, which had three main members: PIM1, PIM2 and PIM3 [14, 15]. PIM1 could was upregulated and play important in tumor progression in many kinds of cancers, including triple negative breast cancer, gastric cancer, hepatocellular carcinoma and mesothelioma [16, 17, 18, 19]. Braso-Maristany et al. discovered that PIM1 regulated cell death, tumor growth and chemotherapy response in triple-negative breast cancer [20]. Similar findings were expounded by Herzog et al. found that PIM1 was upregulated in glioblastoma multiforme and mediates tumor cell survival [21]. In addition, Pang and Tian, knockdown of PIM1 could inhibit cell growth and migration in lung cancer [22]. However, in osteosarcoma PIM1 was upregulated and reverse partial function of miR-486 [23]. Therefore, to investigate the role of miR-486 and PIM1 in osteosarcoma, we started this study.
Material and methods
Patients and clinical samples
A collection of osteosarcoma patients were obtained from Qingdao West Coast New Area Central Hospital with 51 osteosarcoma tissues and corresponding paracancerous tissues. All the patents had not been treated before surgery, and the fresh tissue samples were immediately frozen in liquid nitrogen and saved at
Cell lines and culture condition
Two osteosarcoma cell lines (MG63 and SaOS2) and human osteoblastic cell line NHOst were purchased from the American Type Culture Collection (ATCC, Rockville, MD, USA). Both osteosarcoma and osteoblastic cells were cultured in RPMI-1640 medium (Gibco, Carlsbad, CA, USA) with 10% fetal bovine serum (FBS; Gibco) added in and incubated in a 37
RNA isolation and qRT-PCR
TRIzol reagent (Takara Bio, Shiga, Japan) and MIRcute and Separation of miRNAs Kit (Tiangen, Beijing, China) were applied to extract total RNA or miRNAs from samples and cells. Followed, miRNA cDNA Synthesis kit (Abm Canada Inc., Milton, ON, Canada) and RT Master Mix (Abm Canada) were employed to synthesize cDNA or miRNA. In addition, miR-486 or PIM1, which were normalized by U6 small RNA and
Protein extraction and Western blotting
RIPA Lysis Buffer (Beyotime, Shanghai, China) supplemented with phenlymethanesulfonyl fluoride (PMSF) was utilized to assess the protein level of tissues and cells. After quantified the concentration by BCA Reagent Kit (Solarbio, Beijing, China), the proteins were separated by 12% SDS-PAGE through electrophoresis and then transferred onto 0.45
The correlation of miR-486 and PIM1 in osteosarcoma tissues. (A) and (B) miR-486 was low expressed while PIM1 was over expressed in osteosarcoma tissues versus corresponding paracancerous tissues. (C) miR-486 had negative connection with PIM1. (D) miR-486 was low expressed in osteosarcoma cells MG63 and SaOS2 versus osteoblastic cell NHOst.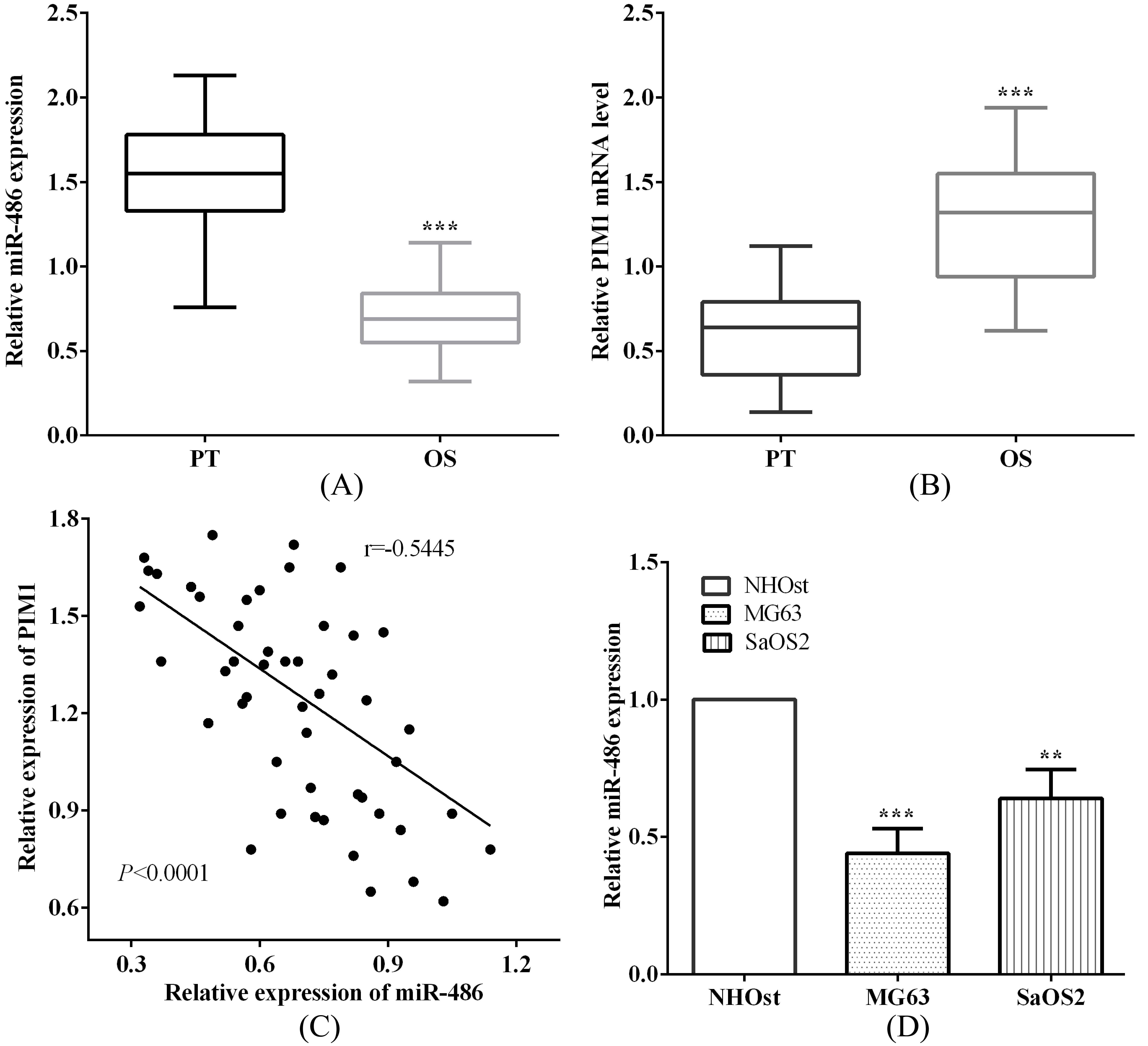
The invasive ability of osteosarcoma cells was evaluated by transwell assay. The transwell were put into 24-well plate, which appeared upper and lower chambers and the upper chamber covered with Matrigel (Clontech, Mountain View, CA, USA). Suspense the cells with medium without FBS and added 200
Transfection
For miRNA analysis, miR-486 mimic and inhibitor (GenePharma, Shanghai, China) were employed to overexpression or knockdown miR-486 in MG63 Cells. For PIM analysis, siRNA-PIM and pcDNA3.1-PIM as well as their negative control were purchased from GenePharma (Shanghai, China) to knockdown or re-express PIM. MG63 cells were seeded into 6-well plate and cultured for overnight at 37
miR-486 suppressed cell invasion and EMT of osteosarcoma cells. (A) We employed miR-486 mimic and inhibitor to over or knockdown miR-486. (B) The invasive ability was reduced after transfected with miR-486 mimic, while promoted when knockdown miR-486. (C) miR-486 overexpression inhibited osteosarcoma cell EMT, while knockdown promoted. (D) It shows the morphologic changes of MG63 cells after transfected with NC/miR-486 mimic/miR-486 inhibitor.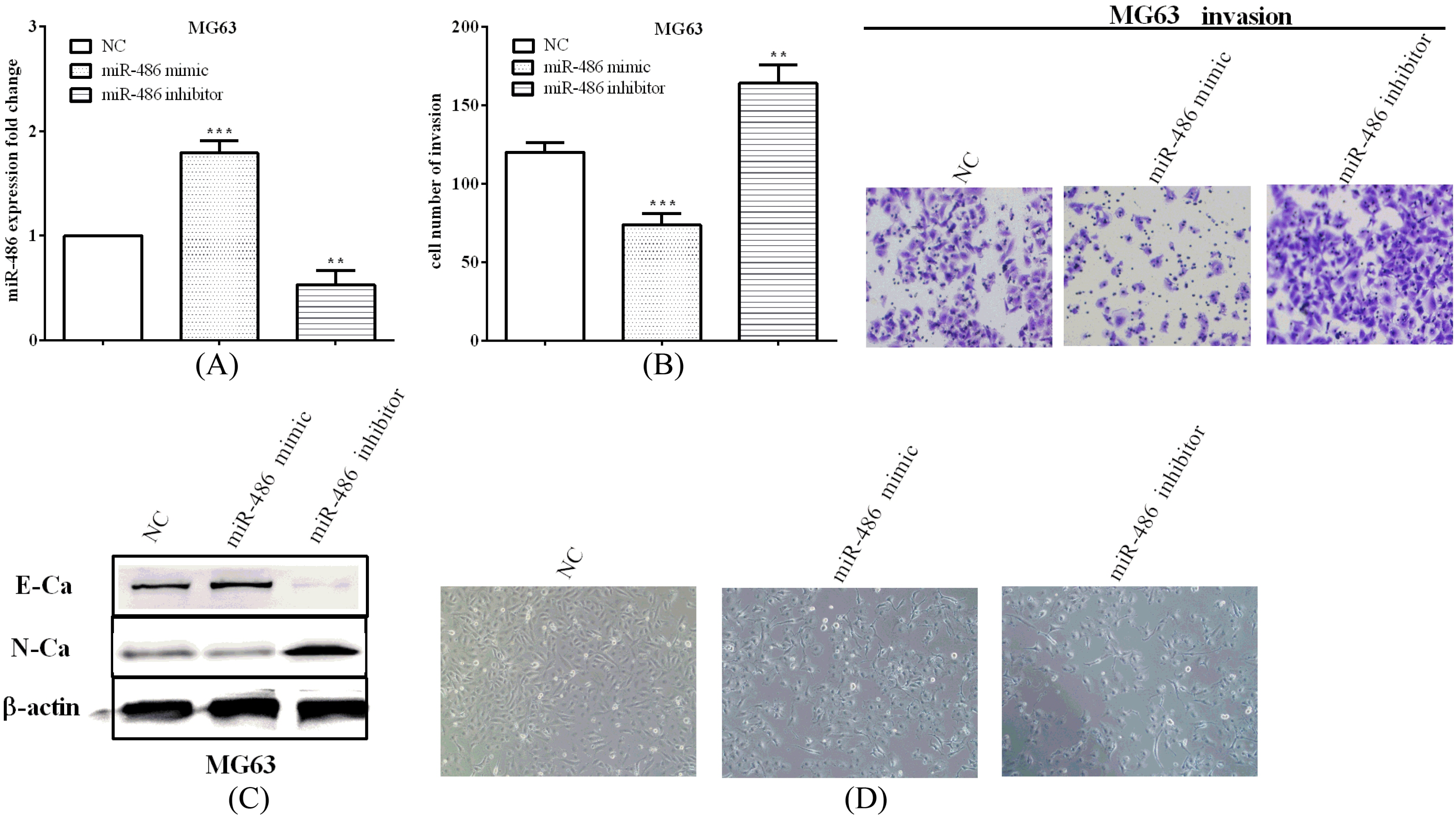
TargetScan predicted that PIM1 was a target gene of miR-486 and the binding sequences were 5’-UCUGAG UGAAAUACUGUACAGGG-3’ on 3’-UTR. We mutated the binding sequences from GUACAGG to CAUGUCC and inserted the nucleotide fragments contain wide type and the mutant sequences into pmirGlo luciferase vector, which named pmirGlo-PIM1-WT (WT) and pmirGlo-PIM1-MUT (MUT) respectively. Whereafter, we co-transfected WT or MUT and miR-486 mimic into MG63 cells using Lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA). The luciferase ability of firefly and Renilla were assayed used dual luciferase reporter assay system (Promega, Shanghai, China) after transfected 48 h, in which Renilla luciferase was the normalization for the firefly luciferase activity.
Statistical analysis
Statistical analysis was performed by SPSS version 16.0 software. The comparisons between two or multiple groups were analyzed by Student’s
Results
The correlation of miR-486 and PIM1 in osteosarcoma tissues
To analyze the relationship of miR-486 and PIM1 in osteosarcoma, we calculated the miR-486 and PIM1 level by qRT-PCR. As expected, miR-486 was low expressed (
PIM1 was upregulated in osteosarcoma cells and knockdown PIM1 inhibited cell invasion and EMT. (A) PIM1 was upregulated in osteosarcoma cells MG63 and SaOS2 versus osteoblastic cell NHOst. (B) siRNA-PIM1 was transfected into MG63 cells to knockdown PIM1. (C) The invasive ability was reduced after transfected siRNA-PIM1. (D) Knockdown PIM1 could inhibit osteosarcoma cell EMT.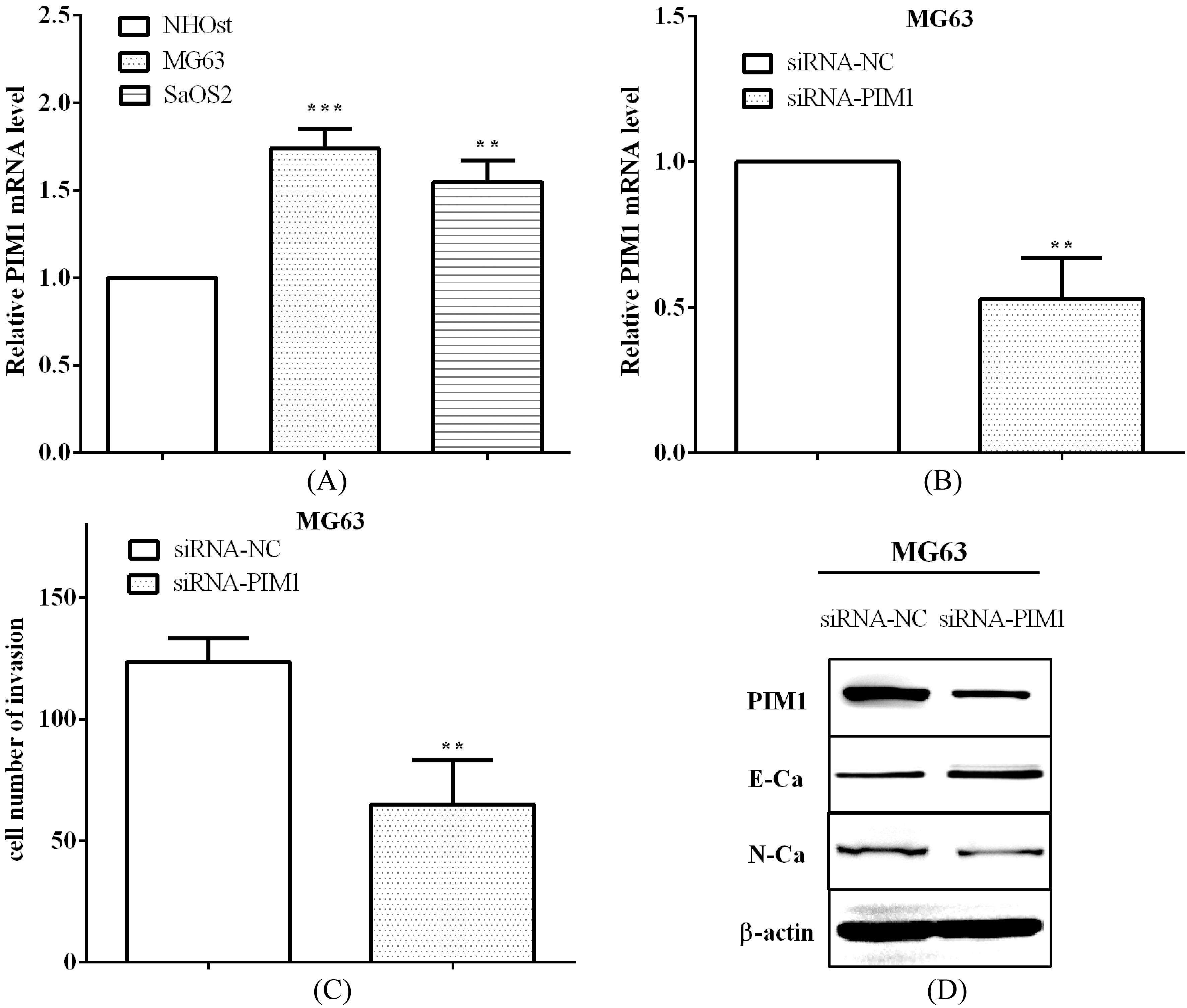
Transwell assay and western blot were performed to evaluate the migration, invasion and EMT abilities in osteosarcoma. We employed miR-486 mimic and inhibitor to over (
PIM1 was upregulated in osteosarcoma cells and knockdown PIM1 inhibited cell invasion and EMT
The expression of PIM1 was also evaluated to be upregulated in osteosarcoma cells MG63 (
miR-486 binding to PIM1 and mediated its expression. (A) TargetScan predicted the binding sequences of miR-386 and PIM1, and mutant sequences of PIM1 3’-UTR. (B) The luciferase activity was reduced in cells with PIM1 3’-UTR wide type, while not in mutant cells. (C) PIM1 mRNA levels were evaluated in MG63 cells, after transfected with miR-486 mimic or miR-486 inhibitor.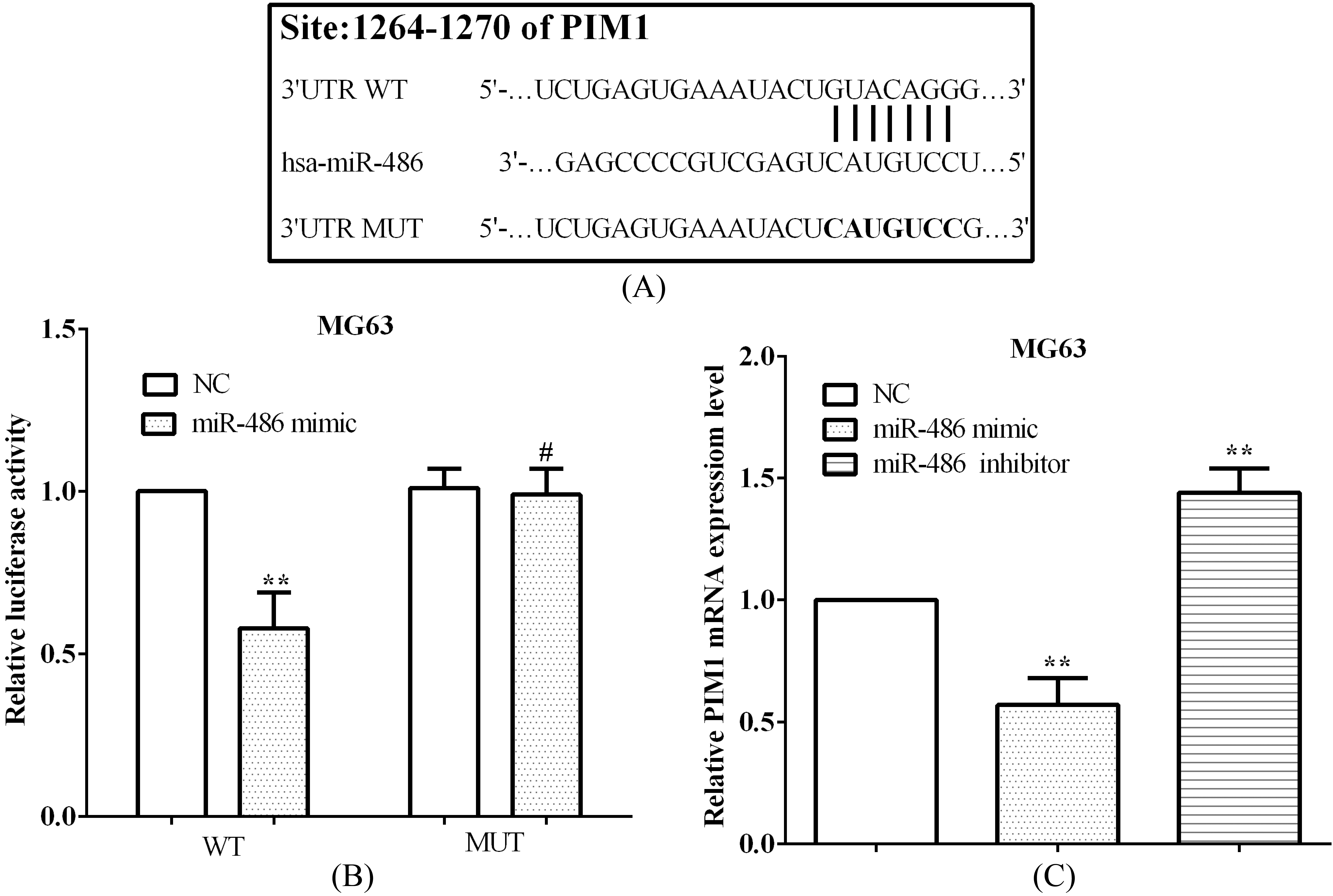
PIM1 could reverse partial roles of miR-486 on cell invasion. (A) PIM1 could re-expressed through overexpressed PIM1, which were inhibited by miR-486 mimic, both in mRNA and protein level. (B) The invasive ability, which suppressed by miR-486 was rescued by re-expressed PIM1 in MG63 cells.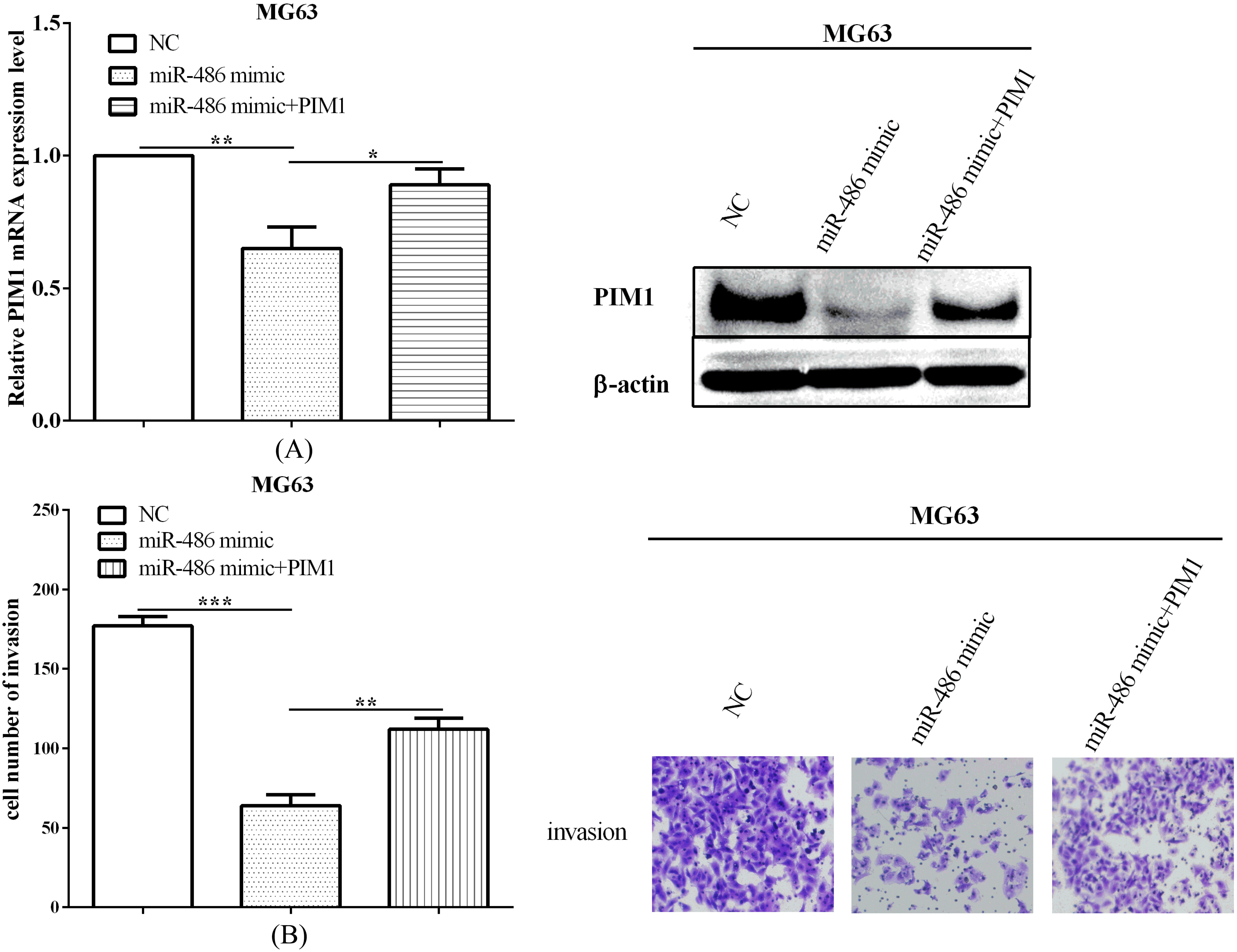
Identification of miR-486 and PIM1 associated with poor survival in osteosarcoma. (A) The OS in miR-486(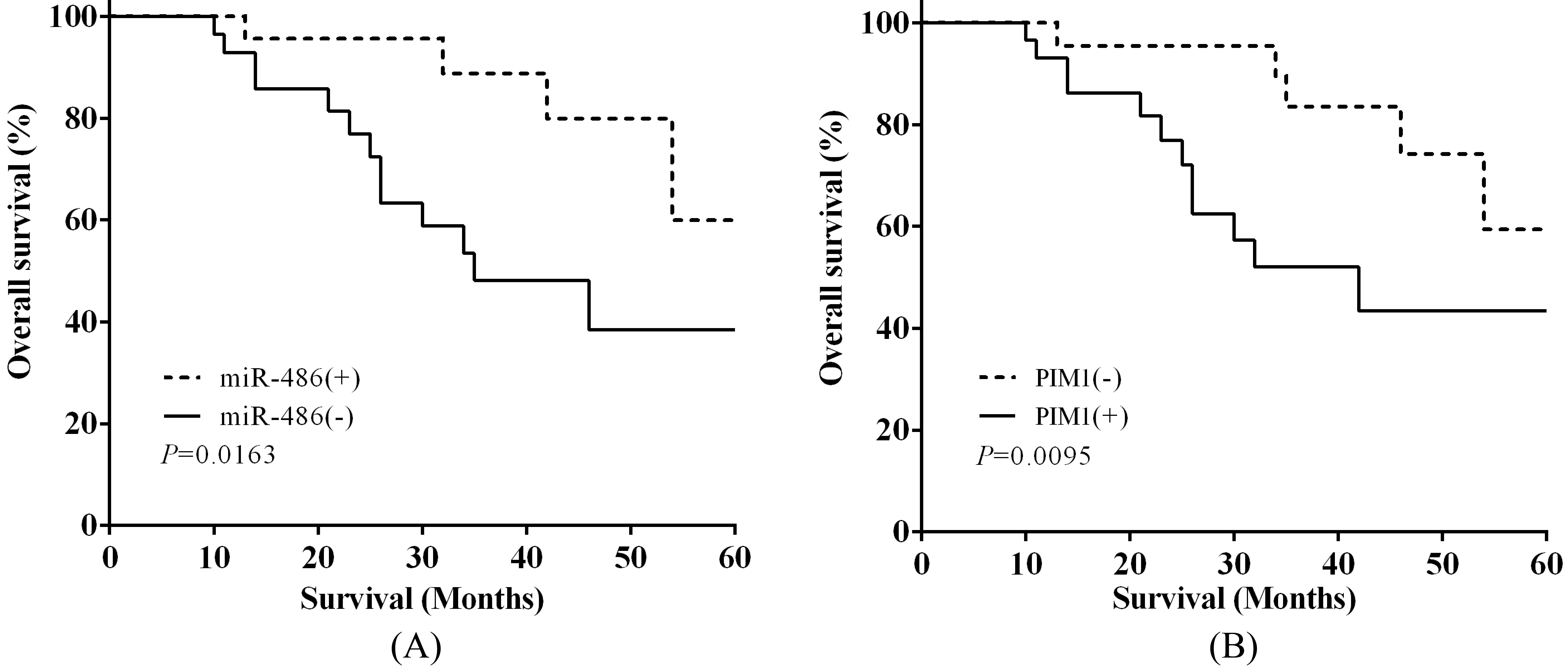
TargetScan was utilized to predict the binding sequences between miR-486 and PIM1, and we discovered that miR-486 binding to PIM1 at 1264-1270 on 3’-UTR of mRNA, which were shown in Fig. 4A. Followed, binding sequences were mutated from GUAC AGG to CAUGUCC to certify miR-486 direct binding to PIM1 (Fig. 4A). Therewith, the wide type (WT) or mutant (MUT) sequences of PIM1 3’-UTR were both inserted into pmirGlo luciferase vector and then calculated the luciferase activity. As expected, the luciferase activity was reduced (
miR-486 expression and clinicopathological features in 51 osteosarcoma
miR-486 expression and clinicopathological features in 51 osteosarcoma
We have evaluated PIM1 was downregulated by miR-486 before, and we next evaluated the roles of PIM1 in miR-486-mediated MG63 cells. Followed, we discovered that PIM1 could re-expressed (
Identification of miR-486 and PIM1 associated with poor survival in osteosarcoma
To identify the connection between miR-486 and the clinicopathological features in osteosarcoma, 51 patients were separated into high and low two groups based on miR-486 expression, gender, age, tumor size, TNM stage, lymph-node metastasis, metastasis and PIM1 respectively. Chi-square test demonstrated that low level of miR-486 had collection with tumor size (
Kaplan-Meier analysis was applied to evaluate the 5-year overall survival (OS) depend on miR-486 and PIM1 expression. The OS in miR-486(
Discussion
Osteosarcoma was the most common primary malignant tumor of bone, which had recurrent or progressive metastases [1, 2, 3]. Thus, it is essential to discover novel pathogenic factors to help early diagnosis and improve prognosis. miRNAs could repress translation or promote target mRNAs degradation through binding to 3’-UTR at post-transcriptional [5, 6]. In esophageal cancer, Lang and Zhao discovered that miR-486 acted as tumor suppressor and cell cycle, invasion and colony formation [9]. Similar findings were discovered by Ye et al. that miR-486 could repress cell proliferation, migration and invasion in cervical cancer [13]. Consistent with all the findings, we discovered that miR-486 was downregulated in osteosarcoma tissues and cells. What’s more, miR-486 low expression could inhibit cell invasion and EMT in osteosarcoma, which consistent with the results of Zhang et al. [24]. Consistent with Zhang et al. that PIM1 was upregulated and was a target of miR-486 in breast cancer [12], we illuminated that miR-486 inhibited cell invasion and EMT through PIM1. In addition, we first proposed that miR-486 downregulation predicted poor prognosis.
PIM1 was a member of PIM family was highly conserved serine/threonine kinases [14, 15]. PIM1 regulated cell death, tumor growth and chemotherapy response in triple-negative breast cancer [20]. Similar findings were expounded by Pang et al. knockdown of PIM1 could inhibit cell growth and migration in lung cancer [22]. Consistent with these findings, we explored that PIM1 was upregulated in osteosarcoma and had negative connection with miR-486. Knockdown PIM1 could suppress cell invasion an EMT in osteosarcoma. In addition, we also discovered that PIM1 overexpression predicted poor prognosis, which consistent with the findings of Herzog et al. that PIM1 was upregulated in glioblastoma multiforme and mediates tumor cell survival [21].
In conclusion, miR-486 was low expressed in osteosarcoma while PIM1 was overexpressed, and it had negative correlation between miR-486 and PIM1. miR-486 upregulation or PIM1 downregulation could inhibit osteosarcoma cell invasion and EMT. Meanwhile, miR-486 mediated the expression of PIM1 through binding to PIM1 mRNA 3’-UTR. PIM1 could reveal partial function of miR-486 on osteosarcoma invasion. In addition, miR-486 low expression or PIM1 overexpression predicted poor prognosis of osteosarcoma patients.
