Abstract
Purpose
There are conflicting opinions on whether miR-486 could be used for cancer diagnosis and prognosis. Therefore, this present study investigated the potential effect of miR-486 on lung cancer diagnosis and prognosis.
Methods
We researched PubMed, Embase, Wanfang and Chinese National Knowledge Infrastructure databases to select relevant publications. Specificity and sensitivity were obtained for the pooled and subgroup diagnostic meta-analysis while the hazard ratio was for prognostic meta-analysis. Publication analyses and sensitivity analyses were conducted to investigate possible sources of heterogeneity.
Results
The overall sensitivity and specificity with 95% confidence intervals were 0.8 (0.8–0.9) and 0.9 (0.9–0.9). Results of subgroup analysis showed that high diagnostic efficacy might be obtained by miR-486 combined with other microRNAs (area under the curve (AUC): 0.9 (0.9–1.0)) to distinguish lung cancer patients from healthy controls (AUC: 1.0 (0.9–1.0)), especially for lung adenocarcinoma (AUC: 1.0 (1.0–1.0)) in the Asian population (AUC: 0.9 (0.9–1.0)). For prognosis prediction of miR-486 in overall non-small cell lung cancer, the overall hazard ratio with 95% confidence interval was 1.15 (0.85–1.54) for high versus low expression of miR-486, which indicated that a high miR-486 level was not related to the high risk of poor outcome. However, for the subgroup of progression-free survival and patients with chemotherapy, the hazard ratio was 0.41 (0.21–0.77), indicating that the higher miR-486 level would decrease the risk of poor progression-free survival for lung cancer patients with chemotherapy.
Conclusion
This study suggested circulating miR-486 combined with other microRNAs could be used as ideal biomarkers in early diagnosis and prognosis prediction for lung cancer, especially for lung adenocarcinoma in the Asian population.
Introduction
microRNA (miRNA) is a set of single-stranded, small, and conserved non-coding RNA with 19–22 nucleotides, which regulates the expression of the target gene by integrating with the 3′-untranslated region of messenger RNA. 1 miRNAs are associated with numerous biological behaviors and tumorigenesis, such as cellular differentiation, proliferation, and cell apoptosis.2,3 Due to the easy access to the miRNAs with high stability from circulating or other specimens, miRNAs tend to be a potential tool for early detection and prognostic prediction for human cancers.
Recently, several articles have reported that abnormal expression of miR-486-5p (miR-486) might be related to different cancers such as breast cancer,4 hepatocellular carcinoma,5 lung cancer6, which would act as both oncogene and tumor suppressor involved in the tumorigenesis and progression of different cancers. The up-regulation of miR-486 can promote the progression of pancreatic cancer,7 gliomas, and chronic myeloid leukemia 8 ; while it is usually down-regulated in lung cancer, 6 hepatocellular carcinoma, 9 osteosarcoma, 10 and breast cancer. 4
A series of studies indicated that miR-486 can participant in different progression processes of lung cancer such as tumor proliferation, metastasis, invasion, and apoptosis,22,23 In addition, whether it is a protective factor16,24 or a risk factor 25 for the prognosis of lung cancer is still controversial. Moreover, no relevant meta-analysis exists to specifically and systematically assess the efficacy of miR-486 expression level on the early diagnosis and prognosis prediction of lung cancer.
Given these numerous observations from different studies, the evaluation of potential efficacy for miR-486 expression in lung cancer detection and prognosis was urgent. Therefore, a systematic review and meta-analysis was conducted to specifically assess the value of miR-486 for the precise detection and exact prognosis prediction in lung cancer.
Materials and methods
Literature search
This present study was conducted according to the Preferred Reporting Items for meta-analyses (PRISMA). Embase, PubMed, Wanfang, and the Chinese National Knowledge Infrastructure (CNKI) databases were enrolled to retrieve all related literature for the value of miR-486 as a potential surveillance tool for precise diagnosis and prognosis prediction in lung cancer. The search strategy was: (miRNA486 or miR-486 or hsa-mir-486 or miRNA-486) and (lung neoplasms or lung cancer or lung tumor or lung carcinoma) updated to June 11, 2021. References of relevant articles were also retrieved for other related publications.
Publication selection
Criteria for publication inclusion and exclusion were drawn up. The inclusion criteria were as follows: (a) Cases were individuals with lung cancer; (b) controls were people with benign pulmonary diseases (BPD) or healthy controls (HC); (c) diagnosis or prognosis value of miR-486 (miR-486-5p) was assessed; and (d) true positive, false positive, false negative, true negative for diagnosis analysis, and hazard ratio (HR) with 95% confidence interval (CI) for prognosis analysis could be obtained from these publications. The exclusion criteria were as follows: (a) Duplicate articles, reviews, meta-analysis, and unrelated articles; (b) studies with the same population or without complete data; and (c) miR-486-3p was the research subject rather than miR-486-5p.
Data collection and methodological quality assessment
Two authors independently collected the necessary data as follows: Author, publication year, country, individual ethnicities, mean/median age, types of cases and controls, sample size, the detection method of miR-486 expression level, miRNA profiling, specimen, outcomes, period of follow-up, and the information for data synthesis including true negative, false negative, false positive, true positive, sensitivity (SEN), specificity (SPE), HR, and 95% CI. The HR was calculated as high versus low miR-486 expression. The Quality Assessment of Diagnostic Accuracy Studies 2 (QUADAS-2) guidelines were as applied to evaluate the methodological quality of these diagnostic articles while the Newcastle–Ottawa Scale were used for records with prognostic efficacy.26,27
Statistical analysis
STATA (STATA-Corp, College Station, TX, USA, version 11.0), meta-disc (version 1.4) and RevMan (version 5.3) were applied for statistical analysis. SEN, SPE, positive likelihood ratio (PLR, PLR = SEN/(1-SEN)), negative likelihood ratio (NLR, NLR = (1-SPE)/SPE), diagnostic odds ratio (DOR, DOR = PLR/NLR), and HR were calculated by bivariate effect regression model. The area under the curve (AUC) of the constructed summary receiver operating characteristic curve was calculated. In addition, heterogeneity among these included studies was evaluated by I2 value and Q test. 28 I2 ≥ 50% or P < 0.05 indicated that significant heterogeneity existed among these enrolled studies, and then a random synthesis analysis would be performed. Otherwise, we would conduct the fixed pooled meta-analysis. In diagnostic analysis, meta-regression and subgroup analyses (based on (a) ethnicity: Caucasian, Asian, and Caucasian/African; (b) specimen: plasma, serum, and other sources; (c) miRNA profiling: miR-486 alone or combined with other biomarkers; (d) control-type: HC or BPD; (e) cancer-type: lung adenocarcinoma, lung squamous cell carcinoma, and non-small cell lung cancer or pooled lung cancer; (f) treatment: surgery or without any treatment; (g) stage: early, late, or overall stage; (h) sample size: ≥100 and <100; (i) cut-off value: <1, ≥1 and <3, ≥3; (j) reference miRNA: single or multiple; and (k) detection method: quantitative real-time polymerase chain reaction (qRT-PCR) or other methods) were conducted to explore heterogeneity sources while an asymmetry test by Deek’s funnel plot was conducted for detection of publication bias. If the P-value was less than 0.01, statistically significant publication bias might exist. 29 In prognostic analyses, we assessed publication bias by Begg's and Egger's tests. Subgroup analysis was also performed by ethnicity (Asian and Caucasian), specimen (serum and tissue), stage (overall and just late), cut-off value (<1 and >1), treatment (surgery, chemotherapy, or without any treatment), and outcome (overall survival (OS), relapse-free survival (RFS), and progression-free survival (PFS)). We also conducted the sensitivity analysis for heterogeneity exploration in diagnostic and prognostic meta-analysis.
Results
Literature search
A total of 388 eligible articles were enrolled based on a literature search (Figure 1), of which 61 were excluded due to duplicate records. Then 64 reviews and 232 unrelated publications were removed, which left 31 articles. Another 10 articles were excluded for incomplete data or with the same population after careful reading. Ultimately, 21 articles15–18,21–25,30–41 with 27 studies were included in the quantitative synthesis from 2010 to 2021. A total of 14 articles15,17–18,21–23,31–35,38–40 with 19 overall studies analyzed the detection efficacy of miR-486 for lung cancer while the other 8 records15–16,24–25,30,36–37,41 with 8 studies explored the value of prognosis prediction of miR-486 in lung cancer. Among these enrolled articles, a study reported by Li et al.15 provided both diagnostic and prognostic value of miR486 for human lung cancer.
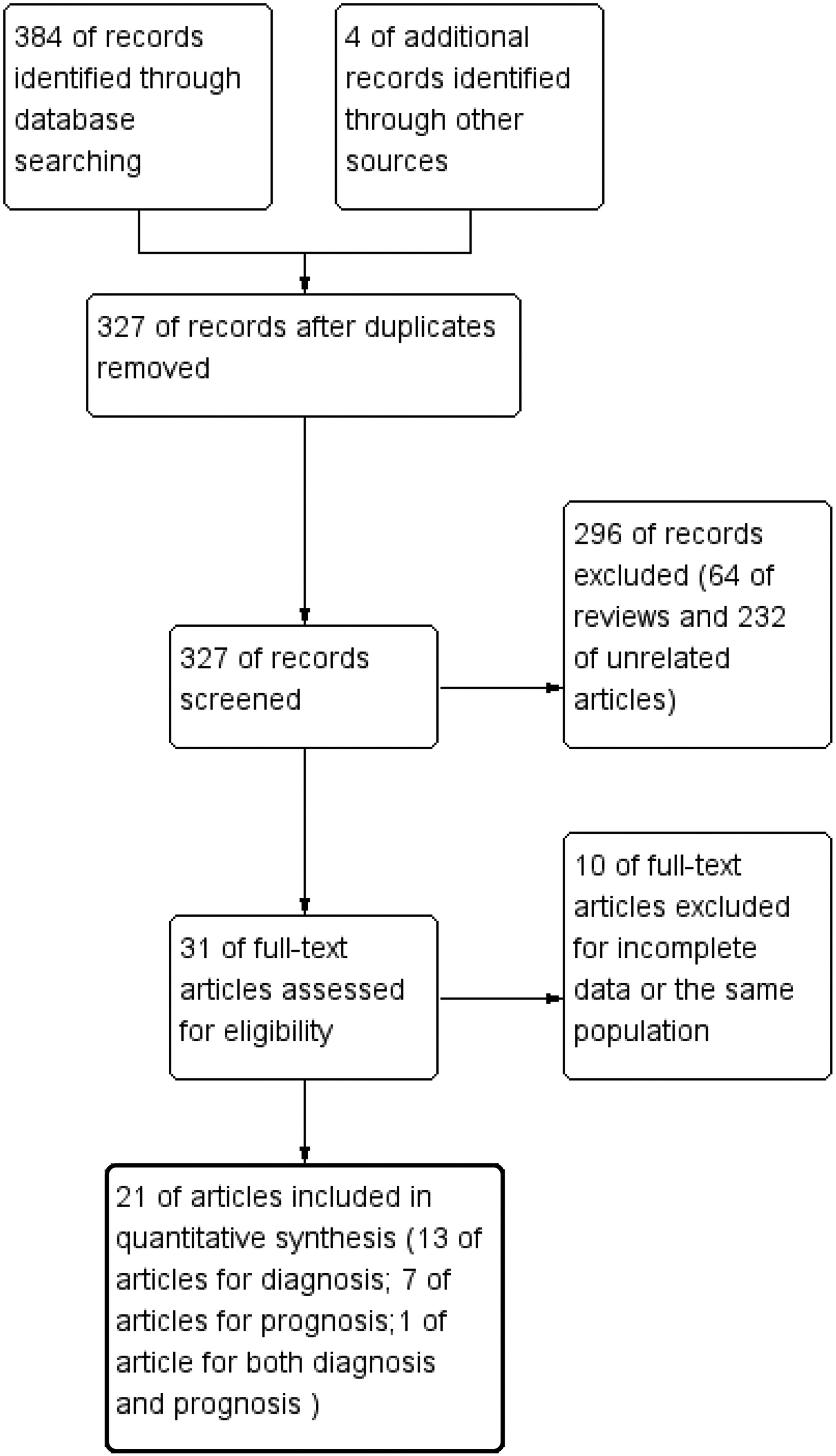
Flow diagram of this meta-analysis for publication screening.
Diagnosis meta-analysis
Research characteristics and methodological quality evaluation
A total of 14 articles with 19 overall studies including 1132 cases and 879 controls were enrolled with the main characteristics displayed in Table 1. The populations enrolled in this study included Asian, Caucasian, and African, with the average age of 63 and 61 years old for the case group and the control group, respectively. The main cancer type was non-small cell lung cancer (NSCLC). For the control population, healthy controls was the more common population compared with individuals with benign pulmonary diseases. Serum and plasma were the most common sources of samples. The qRT-PCR was often applied for detection of the miR-486 expression level, which was usually combined with other miRNAs. In addition, RNU6B, miR-39, and miR-16 were the most common reference miRNAs used as endogenous normalization controls. Methodological quality for these enrolled studies was generally good, which is shown in Figure S1.
The main features of 19 included studies in diagnostic meta-analysis.
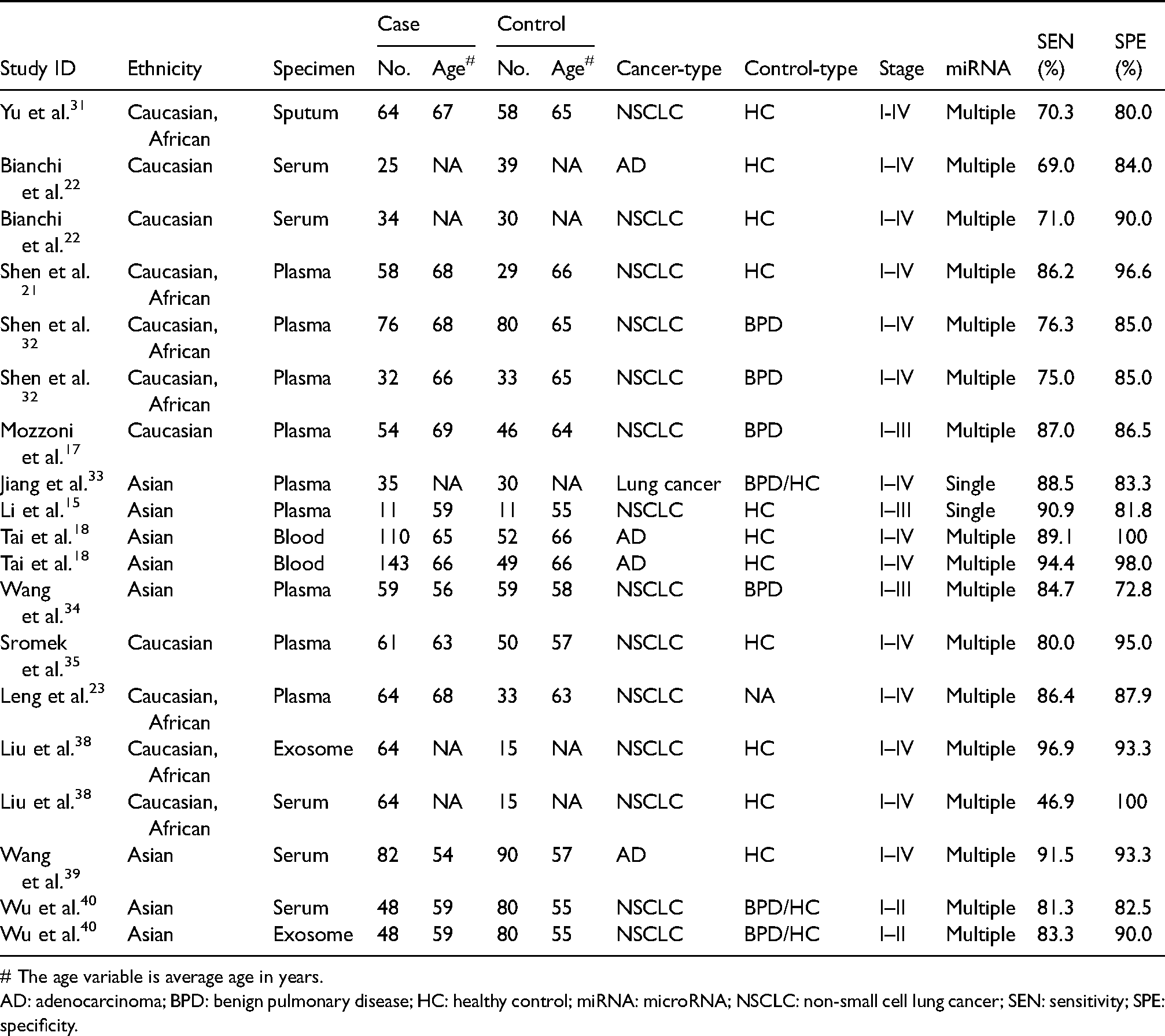
# The age variable is average age in years.
AD: adenocarcinoma; BPD: benign pulmonary disease; HC: healthy control; miRNA: microRNA; NSCLC: non-small cell lung cancer; SEN: sensitivity; SPE: specificity.
Overall diagnostic efficacy
A random-effects model for the pooled SEN and SPE was conducted with I2 of 83.70% (77.2–90.18%) and 61.77% (42.76–80.79%), respectively. Overall SEN and SPE for the 19 studies were 0.8 (0.8–0.9) and 0.9 (0.9–0.9), respectively, to distinguish lung cancer patients from control individuals (Figure S2). The NLR, PLR, and DOR were 0.2 (0.1–0.3), 7.9 (5.6–11.2), and 43 (24–77), respectively; while the AUC was 0.9 (0.9–1.0) (Figure 2(a)).
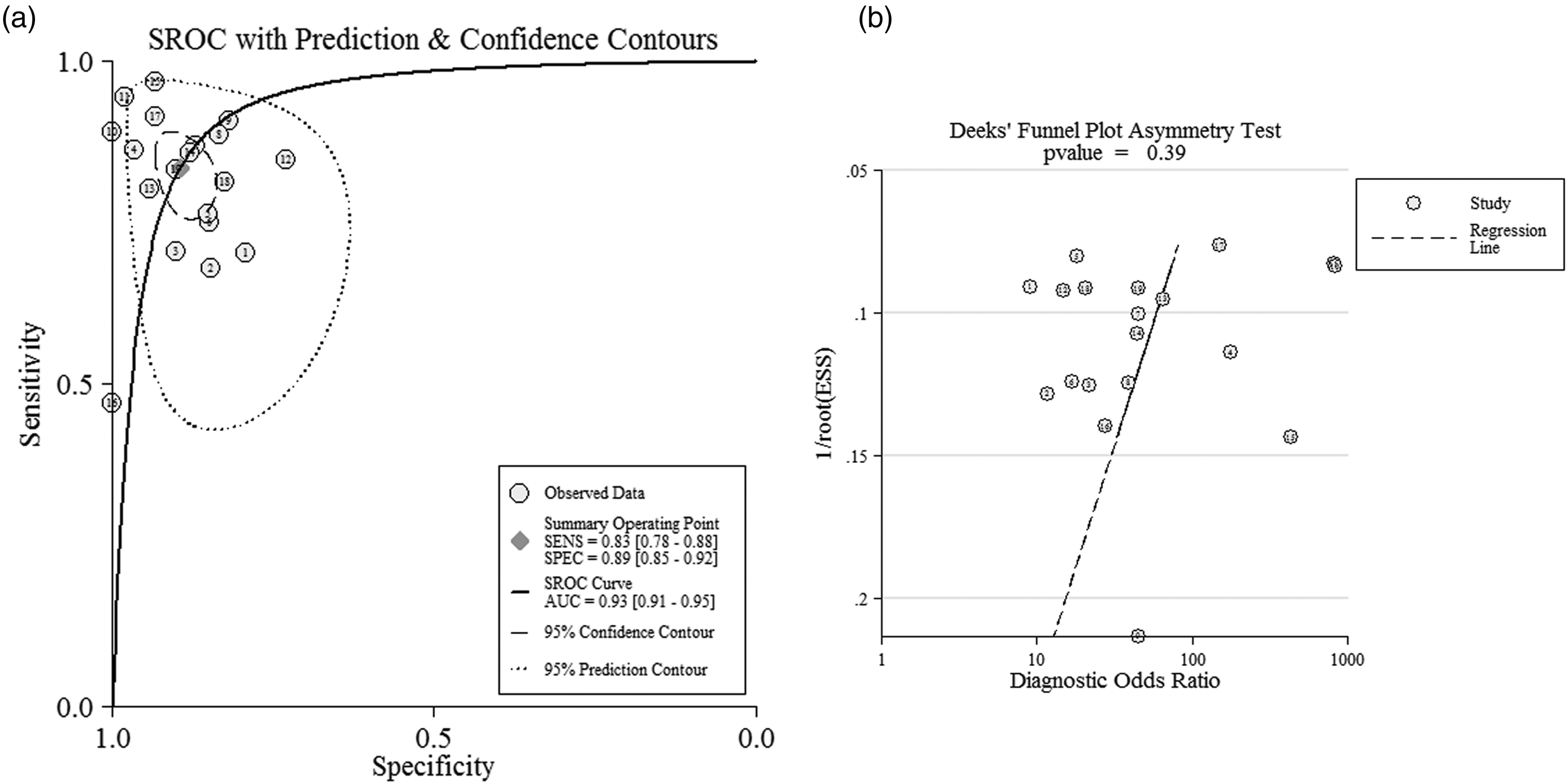
(a) SROC curve of mir-486 as diagnostic tool for lung cancer. (b) Deek’s plot for publication bias of these enrolled studies.
Publication bias and sensitivity analyses
Publication bias was detected by Deek's funnel plot, which had symmetry with P = 0.39 (Figure 2(b)), indicating no significant publication bias existed among these records. In addition, the results of sensitivity analysis also failed to find the potential heterogeneity sources (Figure S3).
Meta-regression and subgroup analysis
Results of multivariate-meta-regression suggested that type of cancer (P = 0.04) might be the heterogeneity source for SPE while no heterogeneity sources were found for SEN. Results of the subgroup analyses are summarized in Table 2. Subgroup analysis by specimen indicated that studies with both plasma and serum exhibited high diagnostic accuracy with SEN 0.8 (0.8–0.9), SPE 0.9 (0.8–0.9), and AUC 0.9 (0.8–0.9) for plasma specimen while the SEN 0.7 (0.7–0.7), SPE 0.9 (0.9–0.9), and AUC 0.9 (0.9–0.9) for serum, respectively. For ethnicity, miR-486 exhibited a higher diagnostic efficacy in the Asian population with SEN 0.9 (0.8–0.9), SPE 0.9 (0.8–0.9), and AUC 0.9 (0.9–1.0) compared to the Caucasian population with SEN 0.8 (0.7–0.9), SPE 0.9 (0.8–0.9), and AUC 0.9 (0.9–0.9), and the Caucasian/African with SEN 0.8 (0.7–0.9), SPE 0.9 (0.8–0.9), and AUC 0.9 (0.9–0.9), respectively. In the subgroup of miRNA profiling, miR-486 combined with other miRNAs obtained a better diagnostic value with SEN 0.8 (0.8–0.9), SPE 0.9 (0.9–0.9), and AUC 0.9 (0.9–1.0) than miR-486 alone with SEN 0.7 (0.7–0.8), SPE 0.8 (0.6–0.9), and AUC 0.8 (0.7–0.8). For the control type, the miR-486 could better distinguish patients with lung cancer from healthy controls with SEN 0.8 (0.7–0.9), SPE 0.9 (0.9–1.0), and AUC 1.0 (0.9–1.0) than from individuals with benign pulmonary diseases with SEN 0.8 (0.8–0.9), SPE 0.8 (0.8–0.9), and AUC 0.9 (0.9–0.9). In addition, miR-486 obtained a higher diagnostic accuracy for lung adenocarcinoma with SEN 0.9 (0.8–0.9), SPE 1.0 (0.9–1.0), and AUC 1.0 (1.0–1.0) than the lung squamous cell carcinoma with SEN 0.7 (0.6–0.8) and SPE 0.8 (0.7–0.9). For treatment, miR-486 exhibited a higher diagnostic efficacy for patients without any treatment with SEN 0.8 (0.8–0.9), SPE 0.9 (0.8–0.9), and AUC 0.9 (0.9–1.0) compared to patients with surgery with SEN 0.8 (0.7–0.9), SPE 0.9 (0.8–0.9), and AUC 0.9 (0.8–1.0). Compared with single reference miRNA, studies with multiple reference miRNAs might obtain a higher diagnostic value with SEN 0.9 (0.8–0.9), SPE 0.9 (0.9–1.0), and AUC 1.0 (0.9–1.0). In addition, there were no significant differences among different cut-off values with AUC of 0.9. No significant difference in diagnostic accuracy of miR-486 was found in other subgroups. Since only two studies was conducted for late-stage and non-circulating samples and Hessian had become asymmetric for the subgroup of early stage, so the pooled analysis for these subgroups was not conducted.
Subgroup analysis for the selected diagnostic studies.
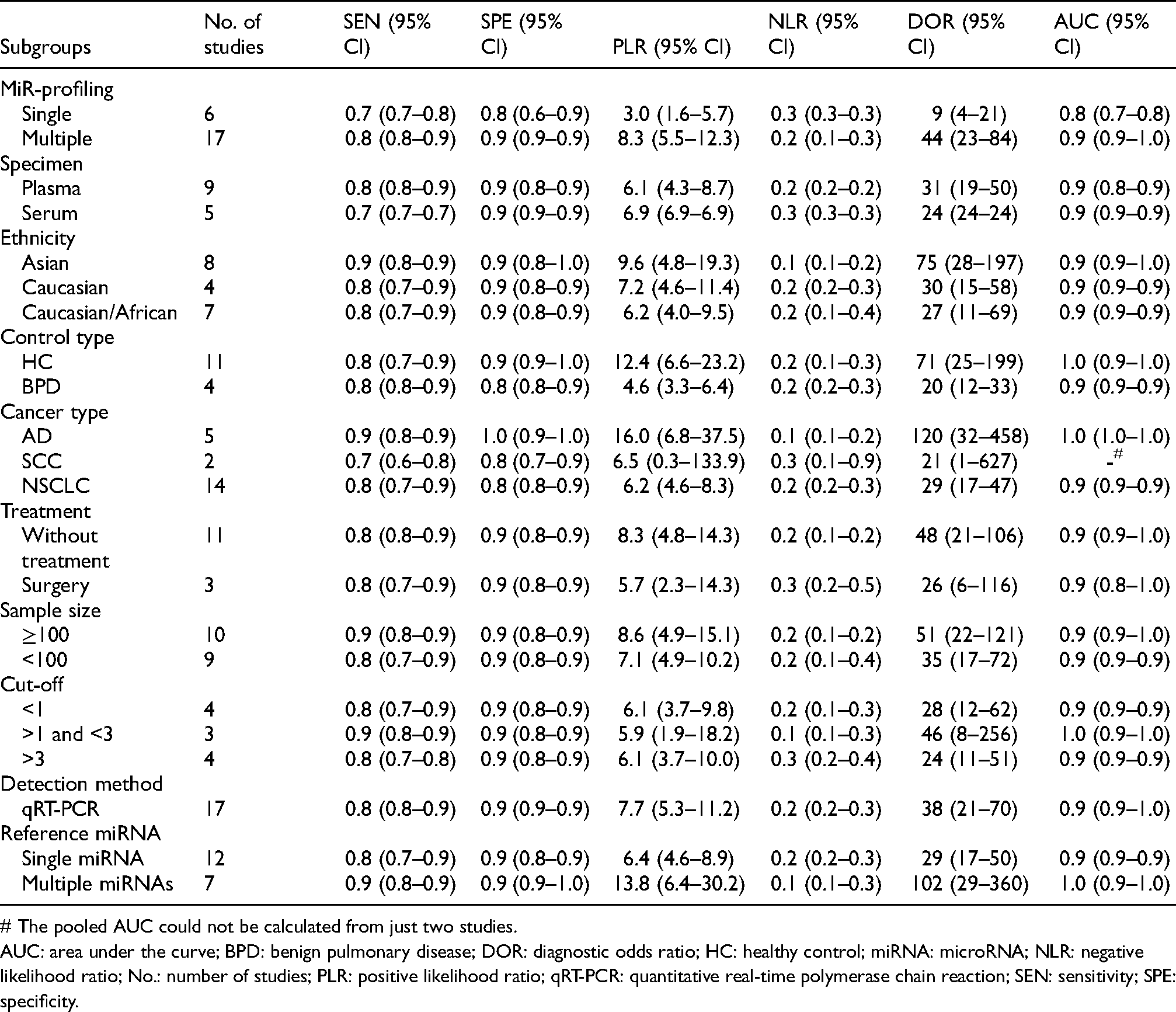
# The pooled AUC could not be calculated from just two studies.
AUC: area under the curve; BPD: benign pulmonary disease; DOR: diagnostic odds ratio; HC: healthy control; miRNA: microRNA; NLR: negative likelihood ratio; No.: number of studies; PLR: positive likelihood ratio; qRT-PCR: quantitative real-time polymerase chain reaction; SEN: sensitivity; SPE: specificity.
Prognosis meta-analysis
Study characteristics and methodological quality assessment
Finally, 691 participants with lung cancer whose average age was 61 years old from eight records on eight studies were enrolled with the main characteristics as depicted in Table 3. Most of the studies measured the miR-486 expression level by qRT-PCR from serum in China. OS, PFS, and RFS were the main outcome of these studies. The pathological type of the enrolled lung cancer was NSCLC. Evaluation for the methodological quality of the research is presented in Table 3.
The main features of eight included studies in prognostic meta-analysis.
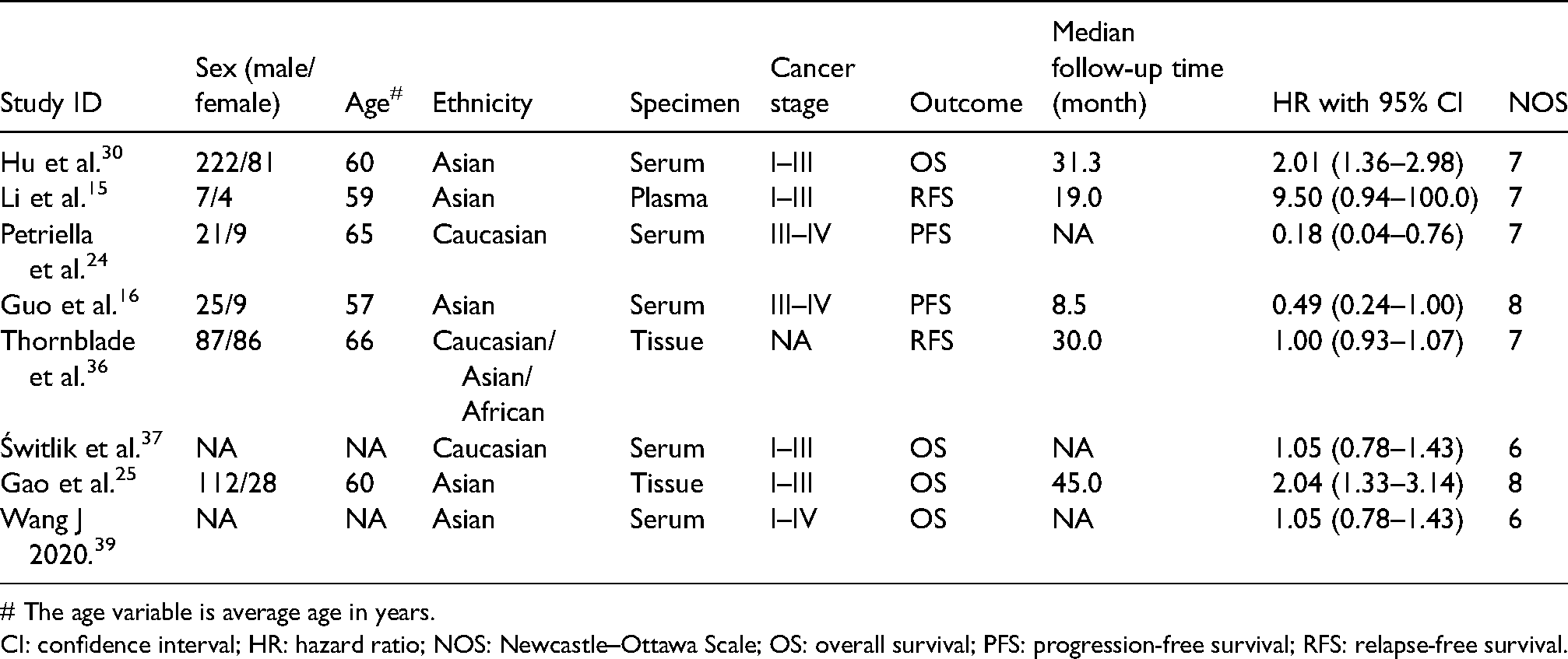
# The age variable is average age in years.
CI: confidence interval; HR: hazard ratio; NOS: Newcastle–Ottawa Scale; OS: overall survival; PFS: progression-free survival; RFS: relapse-free survival.
MiR-486 expression and survival outcomes
Since significant heterogeneity existed among the enrolled studies (I2 = 79.8, P < 0.001), we performed a random-effects model for data synthesis. The pooled HR was 1.15 (0.85–1.54) for high versus low miR-486 expression with P > 0.05, as presented in Figure S4, indicating that different miR-486 expression was not associated with the prognosis of patients with lung cancer. For patients with chemotherapy, miR-486 might be a protective factor for the prognosis with HR and 95% CI of 0.41 (0.21–0.77). However, results of subgroup analysis indicated for different ethnicities (Asian or Caucasian), cut-off less or more than 1, samples from serum or tissue, the OS or RFS, there were no statistical differences between low and high miR-486 expression with P > 0.05. However, for the overall stage subgroup, results indicated the higher the miR-486 expression, the poorer the prognosis with the HR and its 95% CI of 1.50 (1.02–2.19), P = 0.038 (Figure S5A). In contrast, for the subgroup of PFS, the result was a HR and its 95% CI of 0.41 (0.21–0.77) as shown in Figure S5B, indicating that the low miR-486 expression might be a potential risk factor for NSCLC prognosis.
Publication bias and sensitivity analyses
The Begg's funnel plot and Egger's test were conducted for the exploration of publication bias through the random-effects model, which is shown in Figure S6 with a P-value of 0.54, suggesting no significant publication bias existed. In addition, sensitivity analyses also did not find the sources of heterogeneity (Figure S7).
Discussion
Identification of accessible and suitable tools is critical and urgent for early detection, proper treatment, and precise prognosis prediction for lung cancer. MiR-486 could participate in multiple tumorigenesis and tumor progression processes. The ectopic expression of miR-486 has been reported in different cancers and would be involved in the processes of cell proliferation, apoptosis, invasion, and migration. Moreover, numerous publications have suggested that abnormal miR-486 would regulate target molecules such as PTEN, ARHGAP5, and mTOR signaling to participate in the different processes.6,18,41 Furthermore, miR-486 might inhibit epithelial-mesenchymal transition and improve NSCLC chemotherapy sensitivity. 12 In addition, miR-486 might be involved in lung tumorigenesis through the mir-660-p53-mir-486 regulatory pathway.42 In addition, the potential clinical efficacy of miR-486 in early diagnosis and precise prognosis prediction in lung cancer was reported. However, since these results were always inconsistent due to the different research conditions, a pooled analysis is urgent to ascertain a final conclusion for the clinical value of miR-486 in lung cancer.
This current study tended to explore the efficacy of miR-486 on early diagnosis and prognosis prediction for human lung cancer. Based on various research and individuals, we concluded that miR-486 might be suitable as a convenient and non-invasive biomarker for the clinical diagnosis of lung cancer with high diagnostic efficacy. This high diagnostic efficacy might be obtained by miR-486 combined with other miRNAs to distinguish lung cancer patients from healthy controls, especially for lung adenocarcinoma in the Asian population. For studies of miR-486 on prognosis in overall NSCLC, the overall HR and 95% CI were 1.15 (0.85–1.54) for high versus low miR-486 expression, indicating that high miR-486 expression was not associated with the high risk of poor prognosis. However, for the subgroup of PFS and patients with chemotherapy, the HR and 95% CI were 0.41 (0.21–0.77), indicating that the low miR-486 expression might be a potential risk factor for the PFS of NSCLC patients with chemotherapy.
To the best of our knowledge, the present study was the first meta-analysis to specifically summarize the value of miR-486 on early detection and prognosis prediction in lung cancer, though one meta-analysis has been published that focused on the clinical application of miR-486 for all kinds of cancers in 2018,43 which contained just a few lung-cancer-related articles, especially for lung cancer prognosis. Although the present meta-analysis was performed based strictly on the PRISMA guidelines, there were still several critical limitations. First, the high heterogeneity that existed among the enrolled studies should not be ignored. Although we conducted the subgroup and meta-regression analysis in order to find and reduce the heterogeneity, the results achieved little. Second, some articles not in English or Chinese or not published online were omitted. All these considerations may have caused bias during data synthesis, so long-term and larger studies would be needed to provide more robust and definitive evidence in the future.
Conclusion
This research indicated that serum or plasma miR-486 might serve as an effective tool for early diagnosis in lung cancer, especially in lung adenocarcinoma for the Asian population; moreover, circulating miR-486 may be an accessible biomarker for the prognosis prediction of PFS for NSCLC patients with chemotherapy.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155221115750 - Supplemental material for Diagnostic and prognostic value of microRNA-486 in patients with lung cancer: A systematic review and meta-analysis
Supplemental material, sj-docx-1-jbm-10.1177_03936155221115750 for Diagnostic and prognostic value of microRNA-486 in patients with lung cancer: A systematic review and meta-analysis by Xiaoyu Shen, Linlin Li, Linlin Zhang, Wenjing Liu, Yang Wu and Rui Ma in The International Journal of Biological Markers
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
