Abstract
BACKGROUND:
Few biomarkers are available for the prediction of prognosis and recurrence in lymph node (LN)-negative gastric cancer (GC) currently. miR-126 functions as a tumor suppressor in GC, however, its clinical significance in LN-negative GC remains unknown.
AIM:
To investigate the associations of tissue miR-126 level with the clinicopathological characteristics and clinical outcome of LN-negative GC patients.
METHODS:
Quantitative real-time polymerase chain reaction was performed to examine the tissue miR-126 level in 315 LN-negative GC patients who underwent curative gastrectomy with D2 lymphadenectomy. The associations of tissue miR-126 level with clinicopathological characteristics and clinical outcome were evaluated.
RESULTS:
Compared with matched adjacent non-tumor tissues, miR-126 expression was significantly down-regulated in tumor tissues. A reduced tissue miR-126 level statistically correlated with aggressive clinicopathological characteristics, including larger tumor size, deeper local invasion, and poorer prognosis. Notably, multivariate analysis identified advanced T stage and low miR-126 level as independent predictors of the unfavorable prognosis and recurrence of LN-negative GC.
CONCLUSIONS:
These results indicate for the first time that advanced T stage and low miR-126 level are predictors of unfavorable prognosis and recurrence in LN-negative GC patients. These parameters should be taken into account to stratify patients for adjuvant therapy and close follow-up.
Introduction
The incidence of gastric cancer (GC) has dramatically decreased over the past few decades in many regions of the world; however, it still remains one of the most common cancers with almost one million new cases diagnosed per year worldwide, especially in Japan, South Korea and China [1, 2]. Despite the tremendous advances in the understanding of the mechanism of pathogenesis, early detection and comprehensive cancer treatment approaches of GC, the overall 5-year survival rate for GC patients still remains unsatisfactory [3].
Currently, the prediction of prognosis of GC patients is mainly determined by the traditional Tumor, Node Status, Metastasis (TNM) classification system. And the lymph node (LN) metastasis is widely recognized as one of the most important determinants of prognosis in GC patients following curative gastrectomy [4, 5]. In theory, GC patients with localized tumor without LN and distant metastasis may be cured by R
microRNAs (miRNAs) are a novel class of small non-coding RNAs that regulate gene expression posttranscriptionally, either by translational repression or by mRNA degradation [10]. It is estimated that miRNAs may regulate about 30% genes in mammals, and govern important cellular processes, including cell differentiation, proliferation, and apoptosis [11]. Meanwhile, growing evidences demonstrate that miRNAs exert pivotal roles in the pathogenesis of various human cancers, including GC, either as oncogenes or tumor suppressors [12, 13].
Our earlier study demonstrated for the first time that miR-126 was down-regulated in GC, and ectopic expression of miR-126 could inhibit the proliferation and metastatic potential of GC cells with Crk as its direct target gene [14]. Of note, we also found miR-126 down-regulation and Crk protein up-regulation may be synergistically associated with tumor progression in GC and may predict unfavorable prognosis of GC [15]. However, the prognostic value of miR-126 in LN-negative GC patients is yet to be elucidated. The aim of this study is to identify the role of clinicopathological factors in predicting the clinical outcome of LN-negative GC patients undergoing curative resection, with particular focus on the effect of miR-126 on LN-negative GC patients’ prognosis and recurrence.
Materials and methods
Patients and tissue samples
A total of 315 LN-negative GC patients who underwent curative gastrectomy plus D2 lymphadenectomy with more than 15 LNs dissected from 2006 to 2011 at the Department of General Surgery, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine (Shanghai, China) were enrolled in this study. Inclusion criteria for this study were listed as follows: (1) Primary gastric adenocarcinoma confirmed pathologically. (2) No evidence of LN, peritoneal and distant metastasis. (3) No other organs malignancy. (4) Pathologically negative resection margins. (5) No history of neoadjuvant chemotherapy or radiotherapy in order to eliminate potential treatment-induced alterations in gene expression profiles. (6) Patient who wasn’t lost during follow-up. Primary tumor tissues and matched non-tumor tissues were collected immediately after surgery, incubated in RNAlater™ Stabilization Solution, and stored at
All the operations were performed by well-trained surgeons. LNs were meticulously dissected from the en bloc specimens by the surgeons. Dissected LNs were fixed in 10% formalin, embedded in paraffin, stained with hematorylin-eosin, then assessed independently by two gastroenterology pathologists blinded to the clinical data according to the Japanese General Rules for Gastric Cancer Study in Surgery and Pathology [16].
Follow-up of all patients was carried out for 5 years (or until death) according to our standard protocol (every 3 months in the first two years, every 6 months in the third year, and every 12 months afterwards or until death). No subject was lost to follow-up. The routine check-up during follow-up included physical examination, laboratory tests, chest X-ray, computed tomography (CT) and endoscopy. Disease-specific survival was used for evaluating the associations of miR-126 levels and clinicopathological factors with the prognosis in the LN-negative GC patients since it allowed controlling for unrelated causes of death.
Ethical approval was granted by the Ethics Committee of Ruijin Hospital, Shanghai Jiao Tong University School of Medicine (Shanghai, China). Signed informed consent was obtained from all patients for the acquisition and use of specimens and clinical data.
RNA isolation and quantitative real-time polymerase chain reaction (qRT-PCR)
RNA isolation and qRT-PCR were performed as described previously [15]. Briefly, total RNA was isolated from tissue samples using mirVana™ miRNA Isolation Kit (Ambion) according to the manufacturer’s instructions. The quality and quantity of the RNA samples were assessed by standard electrophoresis and spectrophotometric methods. The expression level of mature miR-126 was measured by qRT-PCR according to the Taqman MicroRNA Assays protocol (Applied Biosystems) and normalized using U6 small nuclear RNA (RNU6B; Applied Biosystems) by the 2
Clinical data collection
The medical records of the 315 patients were retrospectively analyzed to determine the associations of tissue miR-126 level with the clinicopathological characteristics (including age at operation, sex, tumor size, tumor site, T stage, and histologic type) and clinical outcome of LN-negative GC patients. The exact T stage was determined according to the TNM staging system of the 7
Statistical analysis
The chi-square test was used to evaluate the association between categorical variables. The cumulative probability of disease-specific survival was estimated by means of the Kaplan-Meier method. Univariate analyses of prognostic factors associated with disease-specific survival were performed using log-rank tests. Factors with significant effects on disease-specific survival in the univariate analyses were then subjected to multivariate analysis using a Cox proportional hazards model, using the forward stepwise procedure for variable selection, to identify independent prognostic factors. Univariate analyses of risk factors potentially associated with recurrence were performed using chi-square test. Factors found to be significant in univariate analysis were included in subsequent multivariate logistic regression analysis to identify independent risk factors associated with recurrence.
Results
Clinicopathological characteristics
Clinicopathological characteristics of 315 gastric cancer patients in this study were summarized in Table 1. Of the 315 patients, 218 (69.2%) were male and 97 (30.8%) were female. One hundred and thirty-four (42.5%) were 60 years of age or younger at the time of surgery and 181 (57.5%) were older than 60 years. With respects to tumor site, the tumor was found in the lower third of the stomach in 156 (49.5%) patients, in the middle third of the stomach in 110 (34.9%) patients, in the higher third of the stomach in 49 (15.6%) patients. For T staging, 65 (20.6%) patients were T1 stage, 85 (27.0%) patients were T2 stage, 97 (30.8%) patients were T3 stage, and 68 (21.6%) patients were T4 stage. In terms of histologic type, 116 (36.8%) patients presented with differentiated tumor while 199 (63.2%) patients presented with undifferentiated tumor.
miR-126 expression was down-regulated in tumor tissues of LN-negative GC
The results of real-time qRT-PCR analysis showed that the range and median of expression level of miR-126 in tumor tissues were 95.2 and 115.3, respectively. The range and median of expression level of miR-126 in matched adjacent non-tumor tissues were 162.7 and 193.5, respectively. The expression level of miR-126 was significantly lower in tumor tissues than that in matched adjacent non-tumor tissues (mean
Clinicopathological associations of miR-126 expression in LN-negative GC
Clinicopathological associations of miR-126 expression in LN-negative GC
*
Down-regulation of miR-126 expression in GC tissues compared with matched adjacent non-tumor tissues. qRT-PCR for miR-126 expression was carried out using 315 surgical specimens of GC tissues (black bar) and matched adjacent non-tumor tissues (grey bar). The mean and standard deviation of miR-126 expression levels are shown.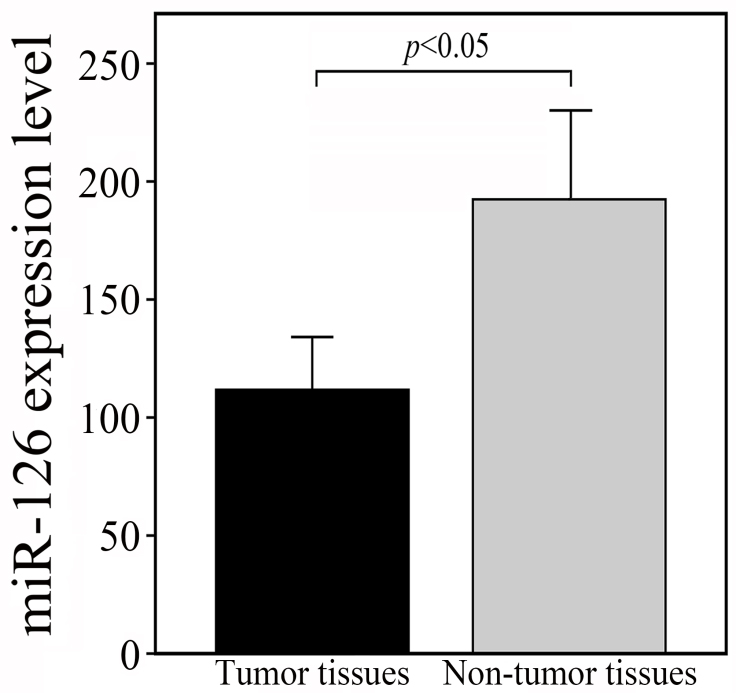
As shown in Table 1, we found that the miR-126 low-expression group favored larger tumor size, and deeper local invasion (both
Univariate survival analysis
The 5-year disease-specific survival rate of the entire cohort was 78.7%. The clinicopathological factors tested in the univariate analysis were shown in Table 2. Factors having significant influence on the survival included tumor size, T stage and tissue miR-126 level (all
Univariate analyses of prognostic factors in LN-negative GC
Univariate analyses of prognostic factors in LN-negative GC
*
Multivariate analyses of prognostic factors in LN-negative GC
*
All the three factors considered significant in univariate analysis (tumor size, T stage, and tissue miR-126 level) were included in the Cox proportional hazards model to determine the independent prognostic factors. As a result, there were two independent, statistically significant prognostic factors: T stage and tissue miR-126 level. The hazard ratio and their 95% confidence interval were listed in Table 3.
Down-regulation of miR-126 was associated with poor prognosis of LN-negative GC patients
As shown in Fig. 2, there was a significant difference in the survival between the miR-126 high-expression group and low-expression group. The 5-year disease-specific survival rate in the patients with low miR-126 level was 46.2%, whereas that in the patients with high miR-126 level was 89.5% (
Clinicopathological features associated with recurrence in LN-negative GC
Clinicopathological features associated with recurrence in LN-negative GC
*
Kaplan-Meier curves of disease-specific survival, stratified by miR-126 expression level.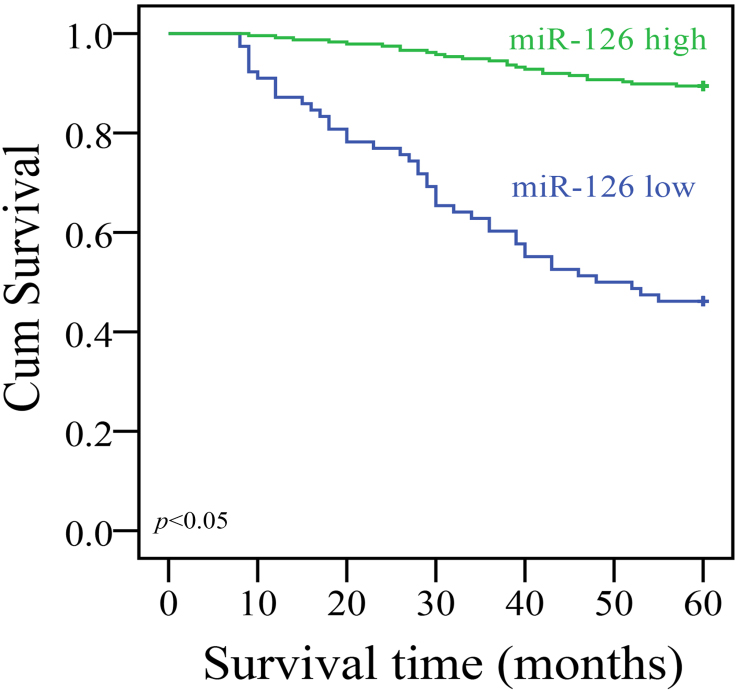
Table 5 showed the relationship between clinicopathological characteristics and recurrence. Recurrence was significantly correlated with tumor size, T stage, and tissue miR-126 level (all
Independent predictors of recurrence in LN-negative GC
Independent predictors of recurrence in LN-negative GC
*
Despite the recent advances in diagnosis, surgical techniques, perioperative management, and concept of chemotherapy, GC remains one of the most common malignant tumors and the third leading cause of cancer death worldwide [17]. The LN metastasis is generally regarded as one of the most powerful prognostic factors for GC, and GC patients without LN metastasis usually had a better survival than those with LN metastasis. It has been reported that the 5-year disease-specific survival rate of LN-negative GC varied from 73 to 86.1% [18, 19]. The 5-year disease-specific survival in our group of patients was 78.7%, which was similar to previous studies.
However, recurrence and metastasis are still noted in a subset of LN-negative GC patients after curative resection, these patients may benefit from more aggressive adjuvant therapy, which has been shown to improve survival of GC patients when compared with gastrectomy alone [20, 21]. Therefore, the identification of novel biomarkers associated with poor prognosis and recurrence in LN-negative GC patients will help to select patients after curative resection for adjuvant therapy and surveillance, and ultimately improve the prognosis. Nevertheless, biomarkers useful for progression monitoring and prognosis prediction of LN-negative GC patients are still lacking today.
The discovery that miRNAs expression is frequently dysregulated in a variety of cancers, and miRNAs have multiple target genes that are involved in cell growth and signaling pathways may broaden our understanding of the underlying mechanisms of carcinogenesis, and shed new light on prognosis prediction. As far as GC is concerned, various studies have shown that miRNAs play critical roles in the development of GC, and are promising indicators of prognosis. For example, upregulation of miR-125b [22], or downregulation of miR-338-3p is associated with poor prognosis of GC [23]. However, to date, few studies have specifically addressed the significance of miRNAs in the prognosis and recurrence of LN-negative GC.
Located on chromosome 9 within the seventh intron of the epidermal growth factor-like domain 7 (EGFL7), miR-126 is one of the most intensively studied cancer-related miRNAs in cancer patients [24]. First identified as a miRNA regulating human megakaryocytopoiesis [25], miR-126 was found to be commonly down-regulated in various cancers, and was thought to be a tumor suppressor through its ability to modulate angiogenesis, tumor growth, and metastasis. For example, miR-126 was found to be highly expressed in low-grade glioma tissues but lowly expressed in high-grade ones, and miR-126 inhibited the migration and invasion of glioma cells by suppressing GATA4 protein expression [26]. Compared to normal B cells, miR-126 was downregulated in chronic lymphocytic leukemia (CLL) cells, and overexpression of miR-126 in leukemia cell lines significantly downregulated p85
miR-126 also play vital roles in the progression of GC. For example, down-regulation of miR-126 was found to inversely correlate with an increased microvessel density (MVD) and vascular endothelial growth factor A (VEGF-A) expression in GC tissues. miR-126 could suppress tumor growth and tumor angiogenesis of GC through VEGF-A signaling [31]. Our previous studies demonstrated for the first time that miR-126 may function as a tumor suppressor in human GC through down-regulation of Crk [14]. In addition, combined miR-126-low/Crk protein-high expression in tumor tissues was found to be an independent unfavorable prognostic factor of GC [15]. But there have been no reports describing the prognostic relevance of miR-126 in LN-negative GC. Hence, in current study, we studied the clinical implication of miR-126 expression in LN-negative GC using a relatively large number of samples.
In most cases, it is the pathologist who retrieves and examines LNs. However, according to our own experience, the retrieved LNs number per specimen varies between surgeons and pathologists, surgeons retrieved more LNs than pathologists did. It has been found that increased number of retrieved LNs can reduce stage deviation and guide decision making for chemotherapy. Jiang and colleagues reported that compared with retrieval performed by pathologists, postoperative LNs retrieval performed by surgeons was associated with significant increase in the total number of LNs in stage I patients, the numbers of positive and total LNs in stage II and III patients. As a result, survival rate of stage II and III patients were significantly higher in the LNs retrieval performed by surgeons group than that in the LNs retrieval performed by pathologist group due to more accurate staging, which helped to facilitate a decision regarding the subsequent chemotherapy treatment [32]. In order to improve quality control in retrieving LNs postoperatively, and determine N stage more accurately, LNs were routinely retrieved by surgeons and then submitted to the pathologists for further examination in our department. In addition, all the patients enrolled in current study successfully underwent radical resection plus D2 lymphadenectomy with pathologically negative resection margins to ensure complete resection. Furthermore, none of patients had undergone preoperative adjuvant therapy before surgery. As a result, statistical bias was decreased and the results of current study were more objective.
Among various clinicopathological factors that have been reported to influence the survival of LN-negative GC, depth of tumor invasion is consistently reported as an adverse prognostic factor [33]. Depth of invasion, especially the presence of serosa involvement was found to be the independent negative predictor of survival [9]. Study by Lee et al. [34] demonstrated that patients without serosal invasion (T1-2) and with serosal invasion (T3-4) had a large difference in survival rates; the depth of invasion was an independent predictor of poor outcome in LN-negative GC patients. As the depth of tumor invasion increases, the tumor would be more likely to invade the blood vessels, thus increasing the likelihood of metastasis. Once tumor further perforates serosa, peritoneal seeding may occur. In addition, tumor associated-stroma secretes several factors such as stromal cell-derived factor 1 and cytokines to promote tumor invasion and stimulate cancer cell metastasis [35]. All of these factors mentioned above increased the possibility of metastasis and recurrence, leading to poorer prognosis. Our data also demonstrated that the depth of invasion was an independent prognostic factor of LN-negative GC. Of note, the findings of our study clearly showed that the down-regulation of miR-126 was significantly associated with advanced T stage. Moreover, the survival analysis demonstrated that the disease-specific survival rate of LN-negative GC patients with low miR-126 expression level was significantly lower than that of the patients with high miR-126 expression level. To eliminate the impact of mixed factors correlated with prognosis on statistical analysis, the Cox proportional hazards model was performed to establish the independent prognostic factors. The results of multivariate analysis confirmed that low miR-126 expression level and advanced T stage were independent unfavorable prognostic factors of LN-negative GC.
Unlike other solid tumors such as breast and lung cancer, tumor size is not currently included in the tumor-node-metastasis (TNM) staging system of GC, and the prognostic significance of tumor size has yet to be determined in LN-negative GC. In our current study, tumor size failed to demonstrate a significant association with survival in the multivariate analysis. Other investigators such as Lee et al. [9] also found that tumor size didn’t independently influence survival of LN-negative GC patients.
In our previous studies, we found that miR-126 expression was closely related to LN metastasis, and reduced miR-126 level was an independent predictor for the unfavorable prognosis of GC [15, 36, 37]. In current study, we found reduced miR-126 level was also an independent predictor for the unfavorable prognosis of LN-negative GC. The explanation for this phenomenon might be that down-regulation of miR-126 may be associated with LN micrometastasis in histologically LN-negative GC patients because of its strong association with LN metastasis. Due to advance in technology, such as immunohistochemistry (IHC) staining and reverse transcription-polymerase chain reaction (RT-PCR) assay, the detection of LN micrometastasis of GC has increased significantly [38, 39]. It was reported that LNs micrometastasis was detected by immunohistochemical examinations among 10%–43% patients with LN-negative GC diagnosed by routine histologic examination [40, 41]. LN micrometastasis was found to be associated with poor outcome in patients with histologically node-negative GC [42, 43]. Therefore, we speculate that miR-126 down-regulation may represent the presence of LN micrometastasis in the histologically LN-negative GC patients, which lead to the poor outcome. Therefore, in the future, we plan to analyze the association between miR-126 expression and LN micrometastasis in LN-negative GC patients to determine whether such patients with low miR-126 level tend to have more micrometastatic LNs than those with high miR-126 level.
Despite the favourable prognosis of LN-negative GC, some patients still developed recurrence. The recurrence rates were reported to vary from 9.4% to 29.4% in LN-negative GC patients [44]. The recurrence rate in our study group is 27.0%, which is consistent with other publications in this field. In addition, we found advanced T stage and low miR-126 level were the independent risk factors for recurrence.
In conclusion, although the LN-negative GC patients generally have an excellent prognosis, some patients may still have recurrence and die. To the best of our knowledge, the current study demonstrated for the first time that miR-126 expression was markedly and consistently decreased in primary tumor, and low miR-126 expression could predict the poor outcome and recurrence in patients with LN-negative GC. miR-126 thus has potential clinical utility as a promising biomarker for molecularly monitoring the progression and predicting the clinical outcome of patients with LN-negative GC. This discovery may help to better identify LN-negative GC patients with poor prognostic potentials and facilitate personalized treatment of these patients.
Footnotes
Acknowledgments
We appreciate all subjects that participated in this study. This project was supported, in part, by the grant from Shanghai Municipal Education Commission (12zz102), Yi Gong Jiao Cha Foundation of Shanghai Jiao Tong University (YG2016MS65) and Shanghai Pujiang Program (17PJ1406000).
