Abstract
BACKGROUND:
miR-126 functions as a tumor suppressor in gastric cancer (GC) by negatively regulating Crk protein expression post-transcriptionally.
OBJECTIVE:
The aim of this study was to investigate the associations of miR-126 and Crk protein expression levels, alone or in combination, with the clinicopathological characteristics and prognosis of GC patients.
METHODS:
The expression levels of miR-126 and Crk protein in 338 GC patients were analyzed by quantitative real-time polymerase chain reaction and immunohistochemistry, respectively. The relationship of miR-126 and Crk protein expression with clinicopathologic characteristics and clinical outcome was evaluated.
RESULTS:
Compared with matched adjacent non-tumor tissues, miR-126 was significantly down-regulated while Crk protein was significantly up-regulated in tumor tissues. A reduced miR-126 expression and an elevated Crk protein expression, alone or in combination, statistically correlated with aggressive clinicopathological characteristics, such as larger tumor size, deeper local invasion, more lymph node metastasis, advanced TNM stage, and poorer prognosis. Multivariate analysis showed that combined miR-126-low/Crk protein-high expression was an independent unfavorable prognostic factor of GC.
CONCLUSIONS:
These results indicate for the first time that miR-126 down-regulation and Crk protein up-regulation may be synergistically associated with tumor progression in GC and may predict unfavorable prognosis of GC.
Introduction
Gastric cancer (GC) is the fifth most common cancer and the third leading cause of cancer-related death worldwide [1]. Despite remarkable advances in early detection and comprehensive treatment approaches, the prognosis of GC patients remains poor, with a 5-year overall survival of 25% or less [2].
At present, the status and prognosis of cancer patients are mainly determined by the Tumor, Node Status, Metastasis (TNM) classification system [3]. However, the above clinical parameters have been proven to be inadequate to predict the biological behavior of cancer cells, especially those with high degree of heterogeneity like GC. Thus, it has become of prime importance to identify other simple, feasible, accessible and less costly markers for the prediction of prognosis [4], and hence prompt for proper and precise treatment plans for GC patients with dismal outcome. For instance, patients with predictable good prognosis could be spared the tedious and challenges of adjuvant therapy while on the other hand, patients with predictable poor prognosis could be further assessed and then subjected to more reasonably aggressive or comprehensive treatment options or even be offered investigational programs.
microRNAs (miRNAs) are a novel class of small non-coding RNAs that deregulate gene expression at the posttranscriptional level, either by translational repression or by mRNA degradation [5]. Notably, absolute sequence complementarity between the miRNAs and their target messenger RNAs (mRNAs) is not necessary, this flexibility implies that each miRNA could bind and regulate numerous mRNAs. It is estimated that miRNAs regulate about 30% of all protein coding genes in mammals [6], and govern various important biological functions such as cell proliferation, differentiation, development and apoptosis [7]. Meanwhile, growing evidences demonstrate that miRNAs exert pivotal roles in the pathogenesis of various human cancers, including GC, either as oncogenes or tumor suppressors [8, 9].
Located within the seventh intron of epidermal growth factor-like protein 7 gene (EGFL7) on human chromosome 9 [10], miR-126 is one of the most commonly observed cancer-related miRNAs in cancer patients. First identified as a miRNA regulating human megakaryocytopoiesis [11], miR-126 was found to be commonly down-regulated in several types of cancers and played the role of tumor suppressor by inhibiting tumor cell proliferation, migration, invasion and the EMT process. For example, miR-126 expression was found to be significantly decreased in glioblastoma tissues while over-expression of miR-126 suppressed glioma cell proliferation and invasion through targeting KRAS [12]. In addition, enhanced expression of miR-126 increased the sensitivity of non-small cell lung cancer cells to anticancer agents through negative regulation of a VEGF/PI3K/Akt/MRP1 signaling pathway [13]. Combination of circulating miR-126 and AFP is a promising noninvasive diagnostic biomarker for detection of hepatitis B virus infected hepatocellular carcinoma [14]. Furthermore, low expression of miR-126 is associated with poor prognosis in colorectal cancer [15]. Thus, aberrantly expressed miR-126 is a potentially useful biomarker for cancer screening, diagnosis, treatment response and prognosis prediction.
Our earlier study demonstrated for the first time that miR-126 was down-regulated in GC, and ectopic expression of miR-126 could inhibit the proliferation and metastatic potential of GC cells, partly through down-regulating the expression of Crk protein post-transcriptionally [16]. In line with our findings, Li et al. reported subsequently that overexpression of miR-126 inhibited GC cells invasion in part by down-regulating Crk protein expression, in addition, they found that knockdown of Crk significantly suppressed GC cells invasion [17]. Hence, miR-126 may function as a tumor suppressor in GC with Crk as its target gene. However, the prognostic value of miR-126 and Crk protein in GC patients is yet to be elucidated. The aim of this study is to further investigate the associations of miR-126 and Crk protein expression levels, alone or in combination, with the clinicopathological characteristics and prognosis of GC patients.
Materials and methods
Patients and tissue samples
Three hundred and thirty-eight patients with primary GC who underwent radical gastrectomy with D2 lymphadenectomy at the Department of General Surgery, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine (Shanghai, China) from 2010 to 2011 were included in this study. The study protocol was approved by the Ethic Committee of Ruijin Hospital and signed informed consent was obtained from all study participants.
None of the subjects underwent chemotherapy or radiotherapy prior to surgery and patients who did were excluded in order to eliminate potential treatment-induced alterations in gene expression profiles. Primary GC tissues and matched adjacent non-tumor tissues were collected immediately following surgical resection. The pathological stage was determined according to the TNM stage system of the seventh UICC/AJCC manual.
Follow-up of all patients was carried out for 5 years (or until death) according to our standard protocol (every 3 months in the first two years, every 6 months in the third year, and every 12 months afterwards or until death). No subject was lost to follow-up. The routine check-up during follow-up included physical examination, laboratory tests, chest X-ray, computed tomography (CT) and endoscopy. Disease-specific survival was used for evaluating the associations of miR-126 and Crk protein expression levels, alone or in combination, with the prognosis in the GC patients since it allowed controlling for unrelated causes of death.
RNA isolation and quantitative real-time polymerase chain reaction (qRT-PCR)
RNA isolation and qRT-PCR were performed as described previously [16]. Briefly, total RNA was isolated from tissue samples using mirVana
Immunohistochemistry
Immunohistochemistry was performed as described previously [16]. Briefly, the sample tissues were fixed in 10% neutralized formalin and embedded in paraffin blocks. Sections (4
Statistical analysis
The chi-square test was used to evaluate the association between categorical variables. The survival rates were calculated using the Kaplan-Meier method, and comparison was made by the log-rank test. The Cox proportional hazards model (using the forward stepwise procedure for variable selection) was used to determine the independent prognostic factors in a multivariate setting. A two-tailed value of
Down-regulation of miR-126 expression and up-regulation of Crk protein expression in GC tissues compared with matched adjacent non-tumor tissues. (A) qRT-PCR for miR-126 expression was carried out using 338 surgical specimens of GC tissues (black bar) and matched adjacent non-tumor tissues (grey bar). The mean and standard deviation of miR-126 expression levels are shown. (B) Immunohistochemistry staining for Crk protein expression was carried out using the same 338 surgical specimens of GC tissues (black bar) and matched adjacent non-tumor tissues (grey bar). The mean and standard deviation of Crk protein expression levels are shown.
Inverse correlation between the expression of miR-126 and Crk protein in GC
miR-126 expression was down-regulated while Crk protein expression was up-regulated in GC
Figure 1 showed the decreased expression of miR-126 (mean
Inverse correlation between the expression of miR-126 and Crk protein in GC. (A–B) Representative data of miR-126 expression levels in two paired tumor/non-tumor tissues by qRT-PCR analysis. miR-126 expression levels in tumors (T) are presented as fold changes relative to the matched adjacent non-tumor tissues (N). miR-126 expression level is lower in T1 than that in N1, and is higher in T2 than that in N2. (C–F) Representative photographs of immunohistochemical analysis of Crk protein in these two paired tumor/non-tumor tissues. Crk was detected in both the cytoplasm and the nucleus of cancer cells, as well as in adjacent normal epithelial cells with different staining intensity (magnification, 200
As shown in Table 2, we found that the miR-126 low-expression group favored larger tumor size, deeper local invasion, more extensive lymph node metastasis, and advanced TNM stage (All
Clinicopathological associations of miR-126 or Crk protein expression in GC
Clinicopathological associations of miR-126 or Crk protein expression in GC
Clinicopathological associations of combined miR-126/Crk protein expression in GC
Univariate analyses of prognostic factors in GC
Kaplan-Meier curves of disease-specific survival, stratified by combined miR-126/Crk protein expression. “dual miR-126/Crk”: miR-126-low/Crk-low or miR-126-high/Crk-high.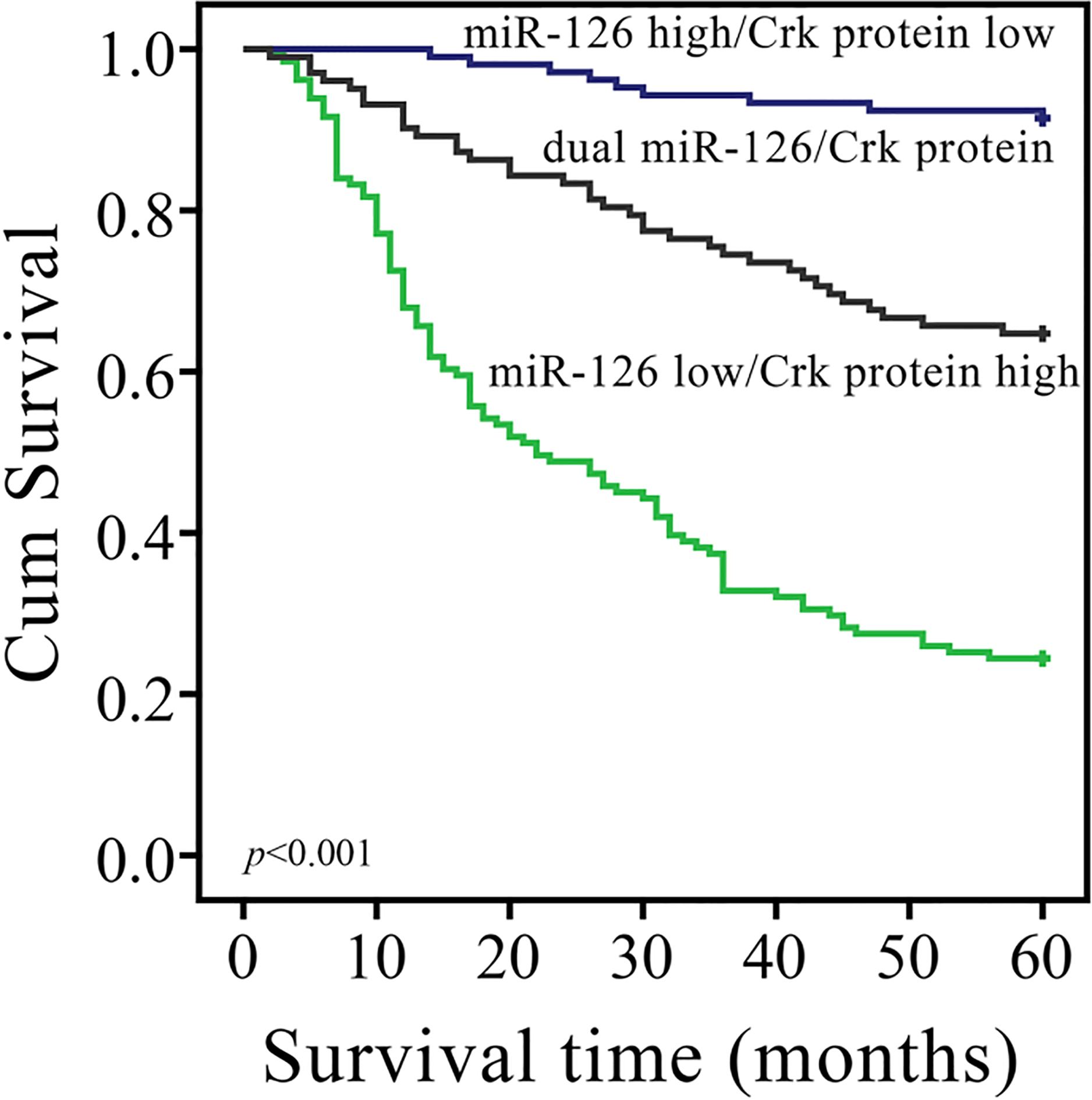
The 5-year disease-specific survival rate in the group with low miR-126 expression was significantly lower than that in the group with high miR-126 expression (34.1% vs. 82.7%,
Besides, tumor size (
As combined miR-126/Crk protein expression status was associated with disease-specific survival rate in univariate survival analysis, we further investigated whether combined miR-126/Crk protein expression status could serve as an independent prognostic marker for prognosis. Since local invasion and lymph node metastasis information was also included in the TNM stage data, the miR-126 and Crk protein expression information was included in combined miR-126/Crk protein expression data, only age, sex, tumor size, tumor site, histologic type, TNM stage, and combined miR-126/Crk protein expression status were examined by multivariate analysis for disease-specific survival rate by the Cox proportional hazards model. The results revealed that tumor size, TNM stage and combined miR-126/Crk protein expression status were independent prognostic factors (
Multivariate analyses of prognostic factors in GC
Multivariate analyses of prognostic factors in GC
Accurate prediction of prognosis for the individual GC patient is of great importance, and a precise novel prognostic biomarker would be useful in the design of more individualized and personalized treatment plan for GC as well as other types of cancers [19]. However, the prognostic biomarkers for GC are still lacking today, urgently prompting for new molecular prognostic markers.
Emerging evidences have shown that aberrantly expressed miRNAs are potentially useful biomarkers for predicting the prognosis of cancer patients including GC. For example, down-regulation of miR-451 or up-regulation of miR-630 was found to be associated with poor prognosis in GC patients [20, 21]. Particularly, miRNAs are superior to protein in clinical application due to their relatively smaller molecular weight, endogenous power of targeting broad-range regulators and the stability in clinical samples [22].
During recent years, many efforts were made to explore the association between miR-126 and cancer prognosis, and low expression of miR-126 was found to be correlated with poor prognosis in a number of cancers such as colorectal and breast cancer [15, 23]. Therefore, as a potential biomarker in predicting prognosis of cancer patients, miR-126 is attracting more and more attention. However, the prognostic value of miR-126 in GC is still largely unknown.
We have previously demonstrated for the first time that miR-126 expression was down-regulated in GC tissues compared with matched non-tumor tissues [16]. By restoring miR-126 expression in GC cells, we found that miR-126 suppressed the proliferation and metastasis potential of GC cells both in vitro and in vivo. Based on these observations, we concluded that miR-126 might function as a tumor suppressor in GC. In addition, we analyzed the relationship between miR-126 expression level and the clinicopathologic parameters in a small number of GC patients, and found that the low miR-126 expression was significantly associated with tumor size, local invasion, lymph node metastasis and TNM stage. Considering these clinicopathological parameters may affect the prognosis of gastric cancer, miR-126 might be a potential prognostic marker for patients of GC. In the present study, we further validated the relationship between tissue-based miR-126 level and clinicopathologic parameters, and analyzed its prognostic value in a larger set of GC patients.
Our results clearly showed that miR-126 expression was decreased in GC tissues compared with matched non-tumor tissues. Notably, miR-126 expression was closely related to poor prognostic features such as tumor size, local invasion, lymph node metastasis and TNM stage. These results were consistent with our previous findings that miR-126 down-regulation may be acquired in the course of tumor progression and, in particular, during the acquisition of metastatic potential.
In the present study, patients with low miR-126 expression showed a shorter disease-specific survival than those with high miR-126 expression. These findings suggested that the status of miR-126 expression might help to predict the prognosis of GC patients. Therefore, miR-126 could constitute a molecular prognostic marker additive to TNM stage for GC patients.
Because miRNAs exert functions via regulating the expression levels of their target genes, it is of great importance to identify the target genes of miRNAs. Crk gene, which is alternatively spliced to give rise to 2 well-characterized proteins (CrkI and CrkII) [24], was proved to be the direct target gene of miR-126 in GC [16, 17]. Elevated levels of CrkI and CrkII mRNA and protein are found in various human tumors such as glioblastoma and breast cancer [25, 26]. Crk influences several cellular processes including proliferation, differentiation, and migration, which are among the key dysregulated processes in malignant phenotype. Therefore, Crk is positioned in a way that alterations in its signal transduction can affect several aspects of tumorigenesis. For instance, Crk is involved in the regulation of cell growth and motility during tumor progression in ovarian cancer. Crk knockdown ovarian cancer cells exhibited disorganized actin fibers, reduced number of focal adhesion, and abolishment of lamellipodia formation with slow growth rates in culture and suppressed anchorage-dependent growth in soft agar. Tumor forming potential in nude mice was attenuated, and intraperitoneal dissemination was not observed when Crk knockdown ovarian cancer cells were injected into the peritoneal cavity [27]. Further studies showed that the Crk/DOCK180/Rac1 pathway also promoted malignant behavior of another ovarian cancer cell line SKOV3 [28]. Knockdown of CrkI/II expression by RNA interference also lead to a dramatic decrease in cell migration and invasion in multiple malignant breast and other human cancer cell lines (MDA-231, MDA-435s, H1299, KB, and HeLa), including GC cells [17, 18]. Our previous study showed that elevated Crk protein was significantly correlated with larger tumor size, more extensive lymph node metastasis, deeper local invasion and advanced TNM stage in a small group of GC patients [16]. However, the prognostic value of Crk protein in GC still remains unclear. In the present larger scale study, we proved that expression of Crk protein was up-regulated in GC tissues compared with matched non-tumor tissues, and was significantly associated with tumor diameter, local invasion, lymph node metastasis and TNM stage, suggesting that Crk might be a crucial contributor to the GC progression. More importantly, patients with higher expression level of Crk protein tended to have shorter survival than patients with lower expression level.
Based on above findings, we hypothesized that the down-regulation of miR-126 and the up-regulation of Crk protein might synergistically influence tumor progression and prognosis in GC patients. To validate this hypothesis, we further analyzed the associations between miR-126/Crk protein combined expression patterns and clinicopathological parameters and prognosis of the GC patients respectively. Our data showed that the combined miR-126 down-regulation and Crk protein up-regulation were correlated with larger tumor size, deeper local invasion, more extensive lymph node metastasis, and advanced TNM stage. Importantly, patients with miR-126 down-regulation in combination with Crk protein up-regulation had poorer prognosis than all of the other patients, and the combined miR-126/Crk protein expression was an independent prognostic factor of GC patients.
Nevertheless, at the time of our study, Yue et al. reported that miR-126 and CRK combined expression may serve as an independent predictor of overall survival in patients with advanced GC [29]. However, there are yet some differences between this study and that of Yue et al. Firstly, in Yue’s study, they analyzed the relationship between the combined expression status of miR-126/CRK mRNA and overall survival of GC patients. However, there is a likelihood of death from non-cancer related causes in their study. Therefore, a study of disease-specific survival would be more helpful in providing a comprehensive analysis of survival trends. So, in current study, we analyzed disease-specific survival which allowed controlling for non-cancer related causes of death. Secondly, unlike Yue et al. who analyzed Crk mRNA expression level in GC tissues and matched adjacent non-tumor tissues, in our study, we analyzed Crk protein expression level since we proved previously that ectopic expression of miR-126 down-regulated the expression of Crk protein post-transcriptionally. Similarly, Gong et al. proved that overexpression of miR-126 significantly down-regulated Crk protein expression level without chaning its mRNA expression level [30]. Therefore, we believe that the combined detection of miR-126/Crk protein might be more helpful to predict the clinical outcome of GC patients.
In conclusion, the findings of this current study provide concrete evidence for the first time that the dysregulation of miR-126/Crk protein axis may contribute to the aggressive progression and poor prognosis of human GC. Combined detection of their expression might be particularly helpful for surveillance of disease progression and treatment stratification. From the clinical viewpoint, the combined miR-126 down-regulation and Crk protein up-regulation might be considered as a risk factor for tumor progression, and thereby a strict systemic therapeutic strategy after surgery, such as chemotherapy with strict surveillance might improve prognosis.
Footnotes
Acknowledgments
We appreciate all subjects that participated in this study. This project was supported, in part, by the grant from Shanghai Municipal Education Commission (12zz102), Yi Gong Jiao Cha Foundation of Shanghai Jiao Tong University (YG2016MS65) and Shanghai Pujiang Program (17PJ1406000).
Conflict of interest
The authors declare no conflict of interest.
