Abstract
BACKGROUND:
Predicting lymph node metastasis (LNM) accurately is vital to design optimal treatment strategies preoperatively for gastric cancer (GC) patients. However, conventional tumor biomarkers and imaging techniques are not sufficient to predict LNM before surgery. miR-126 has been reported to play important roles in tumor metastasis which may represent a novel tumor biomarker.
OBJECTIVE:
To assess the utility of the combination of serum miR-126 and multi-detector computed tomography (MDCT) in predicting LNM preoperatively in GC.
METHODS:
Quantitative real-time polymerase chain reaction was performed to examine the serum miR-126 expression levels in 338 GC patients. MDCT was also performed. The cut-off value of preoperative serum miR-126 level for LNM was determined by receiver characteristic curve (ROC) analysis. Logistic regression analysis was used to determine independent predictors for LNM.
RESULTS:
The serum miR-126 levels of GC patients with LNM were significantly lower compared with those without LNM (
CONCLUSIONS:
These results indicate for the first time that the combination of serum miR-126 and MDCT is useful for the prediction of LNM in GC.
Introduction
In spite of the fact that the incidence of gastric cancer (GC) has declined dramatically over the past decades in many regions of the world, partially due to reduction of Helicobacter pylori infection in the population, GC remains one of the most common and lethal malignancies worldwide, with about 1 million new cases diagnosed annually [1]. In China, which is among the highest incidence areas of GC, GC is the second most common cancer and the second leading cause of cancer-associated death [2]. In some countries, such as Korea and Japan, where routine screening for GC is performed widely, GC can be diagnosed at early stage with a 5-year survival rate greater than 90% [3]. However, to date, most GC patients are still diagnosed at advanced stage and the prognosis of these patients remains poor, with a 5-year overall survival ranging between 15 and 35%, due to regional recurrence or distant metastasis [4].
It is well known that lymph node metastasis (LNM) is one of the most common metastatic patterns and one of the most significant prognostic factors of GC [5, 6]. Furthermore, stage-oriented treatment strategy is now crucial to improve the outcome of GC patients [7]. Therefore, from the view of clinical practice, preoperative determination of LNM status is vital to provide information on tumor staging and thus design more reasonable treatment strategy for this devastating illness.
Nowadays, imaging techniques especially multi-detector computerized tomography (MDCT) is widely used to evaluate LNM status preoperatively; however, many studies show that the diagnostic accuracy of MDCT is limited due to the low sensitivity and specificity [8]. Thus, MDCT alone can’t predict LNM status accurately, and addition of other non-invasive, more sensitive, specific, and less costly biomarkers better reflecting the biological behavior of GC cells are likely to improve the accuracy of LNM status prediction in GC patients.
One of the emerging fields in cancer research is microRNAs (miRNAs). As one of the gene expression regulators, miRNAs are found to deregulate gene expression at the posttranscriptional level, either by translational repression or by mRNA degradation [9], and are involved in diverse biological processes [10]. Recently, accumulating evidences suggest the critical roles of miRNAs in tumor initiation and progression, with GC no exception [11, 12, 13]. Our earlier study demonstrated for the first time that miR-126 was down-regulated in GC, and ectopic expression of miR-126 could inhibit the proliferation and metastatic potential of GC cells both in vitro and in vivo, partly through down-regulating the expression of Crk protein post-transcriptionally, thus miR-126 may function as a tumor suppressor in GC [14]. What is more, combined miR-126-low/Crk protein-high expression in tumor tissues was found to be an independent unfavorable prognostic factor of GC, which implied that miR-126 was a novel biomarker for prognosis prediction of GC patients [15]. However, whether serum miR-126 could predict the LNM status in GC patients preoperatively remains unknown.
The current study thus aimed to determine:(1) Whether serum miR-126 expression level might be a molecular biomarker for predicting the LNM status preoperatively in GC patients. (2) Whether the combination of serum miR-126 and MDCT scan may improve the accuracy of preoperative diagnosis of LNM in GC patients.
Materials and methods
Patients and clinical samples
In total, 338 patients with primary GC who underwent radical gastrectomy with D2 lymphadenectomy at the Department of General Surgery, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine (Shanghai, China) from 2010 to 2011 were recruited for this study. None of these GC patients underwent chemotherapy or radiotherapy before sample collection in order to eliminate potential treatment-induced alterations in gene expression profiles. To obtain serum samples, 10 mL of peripheral blood was drawn into separate gel tubes before surgery and then subjected within 30 min to centrifugation at 1,500 g for 10 min at 4
The study protocol was approved by the Ethic Committee of Ruijin Hospital (IRB number: 2017-171) and signed informed consent was obtained from all study participants. All participants signed an informed consent form for the use of their samples before recruitment. All specimens were handled and made anonymous according to ethical and legal standards.
RNA extraction and quantitative real-time polymerase chain reaction (qRT-PCR)
To extract total RNA in serum, 600
MDCT examination
Before MDCT scan, all patients fasted for 10–12 h. Each patient drank 800–1000 mL of warm water to establish a background for dynamic contrast-enhanced MDCT scans. Contrast media, Iohexol (300 mg I/mL, GE Healthcare Pharmaceuticals Inc., China), was administered intravenously using a power injector. The images were obtained in plain scan, arterial, venous and delayed phases with start delays of 20–25 s, 60–65 s, and 150–180 s, respectively. Criteria for diagnosing LNM are as follows: the long axis of the lymph node is greater than 1 cm, or the ratio of the short axis to the long axis is greater than 0.7, light to moderate or circumferential enhancement or clumping growth, bead-like disposition and compression of a blood vessel. A written informed consent for preoperative staging with MDCT was obtained from all patients. The MDCT findings were compared with histopathological results.
qRT-PCR for miR-126 expression was carried out using 338 GC patients’ sera. Serum miR-126 levels in GC patients with LNM (black bar) were lower compared with those in patients without LNM (grey bar). The mean and standard deviation of miR-126 expression levels are shown.
Receiver operating characteristic (ROC) curve of serum miR-126 for the prediction of LNM.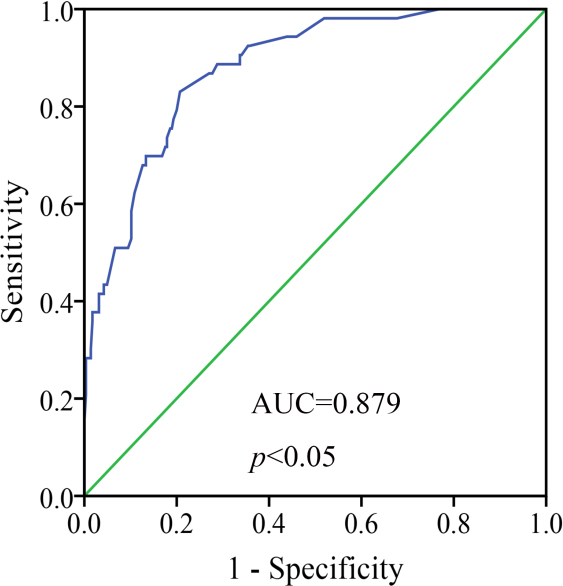
The Student’s
Results
Serum miR-126 level was reduced in GC patients with LNM
We first analyzed the serum miR-126 level according to LNM status. We found that the serum miR-126 levels in GC patients with LNM were lower compared with those in patients without LNM (mean
The predictive value of serum miR-126 level for LNM in GC
To further evaluate the ability of serum miR-126 level for predicting LNM, ROC curve analysis was generated. The area under the ROC curve of serum miR-126 was 0.879
Univariate analysis of potential risk characteristics for LNM in 338 GC patients
Univariate analysis of potential risk characteristics for LNM in 338 GC patients
LNM: Lymph node metastasis; GC: Gastric cancer. *
Multivariate analysis of potential risk factors for LNM in 338 GC patients
LNM: Lymph node metastasis; GC: Gastric cancer; CI: Confidence interval. *
The effectiveness of serum miR-126 and MDCT in predicting LNM in GC
MDCT: Multi-detector computed tomography; LNM: Lymph node metastasis; GC: Gastric cancer; Sen: Sensitivity; Spe: Specificity; Acc: Accuracy; PPV: Positive predictive value; NPV: Negative predictive value. Serum miR-126 (
Representative MDCT images predicting LNM in GC (A, C, E: Arterial phase; B, D, F: Venous phase). Patient 1 (A, B): MDCT images predicted metastatic LN (yellow arrow) which was proven to be metastatic by histopathological diagnosis. Serum miR-126 of this patient was lower than cut-off value, indicating LNM. Patient 2 (C, D): MDCT images predicted metastatic LN (yellow arrow) which was proven to be non-metastatic by histopathological diagnosis. Serum miR-126 of this patient was higher than cut-off value, indicating non-LNM. Patient 3 (E, F): MDCT images didn’t predict metastatic LN which was proven to be metastatic by histopathological diagnosis. Serum miR-126 of this patient was lower than cut-off value, indicating LNM.
The association between various clinicopathological characteristics and LNM was then analyzed using the
Multivariate analysis of potential independent clinicopathological risk factors for LNM in GC
As shown in Table 2, among factors significantly associated with the presence of LNM by univariate analysis, subsequent multivariate analysis using the logistic regression model demonstrated that deeper local invasion were significant and independent risk factor for LNM. Notably, decrease in serum miR-126 level was also significantly associated with higher probability for LNM.
Preoperative prediction of LNM by MDCT scan
All 338 GC patients underwent contrast-enhanced MDCT preoperatively after overnight fasting (Fig. 3). On comparison with the histopathological results, the accuracy, sensitivity, specificity, positive predictive value and negative predictive value for detection of LNM by MDCT scan were 69.3%, 69%, 69.2%, 84.1% and 48.6%, respectively (Table 3). At last, the combination of serum miR-126 and MDCT scan had a predictive accuracy of 86.7% for LNM.
Discussion
As one of the leading causes of cancer-related death around the world, diagnosis and treatment of GC always attract attention. Metastasis to the regional LN is widely recognized as one of the most important indicators of tumor progression as well as determinants of prognosis in GC patients following curative gastrectomy [16]. Even in early GC (EGC) whose five-year survival rate is greater than 90%, the overall incidence of LNM still exceeds 10%, and EGC patients without LNM usually had a better survival than those with LNM [17]. Therefore, LN-negative EGC patients may be curatively treated with minimally invasive endoscopic mucosal resection or endoscopic submucosal dissection in order to avoid the potentially morbid surgical procedure and preserve stomach function as well as maintain high postoperative quality of life, whereas LN-positive patients should undergo gastrectomy with LN dissection [18]. Meanwhile, neoadjuvant chemotherapy, whose beneficial effects have been proved in some randomized-controlled trials, has been recommended by increasing number of oncologists as a new treatment standard for certain subgroup of advanced GC (AGC) patients, especially for those with LNM in order to decrease TNM stage preoperatively and increase the curative resection rate [19, 20]. There is no doubt that the presence of LNM is a crucial preoperative consideration in administering this type of treatment. Thus, to ensure optimal management of GC patients, it’s critical to develop an accurate preoperative LNM assessment method.
Nevertheless, accurate assessment of LNM in GC patients before surgery seems to be very challenging. Currently, the MDCT scanning is widely performed for the preoperative evaluation once the diagnosis of GC is made. It can provide valuable information for evaluating the depth of invasion, hepatic metastasis, ascites, and peritoneal metastasis [21, 22]. However, the effectiveness of MDCT in predicating LNM in GC is limited [23, 24], possibly due to the following factors: first, the classification of LNM status is usually based on LN size, and sensitivity of MDCT for detecting LNM is low if involved LNs are smaller than 0.5 cm. Second, some enlarged LNs are in fact inflammatory hyperplasia which may cause false-positive results. Third, the clinical experience of radiologists will influence the diagnostic accuracy of MDCT imaging results. Our present study showed that MDCT scan was not ideal for the preoperative diagnosis of LNM; the sensitivity, specificity, accuracy, positive predictive value and negative predictive value were only 69.3%, 69%, 69.2%, 84.1% and 48.6%, respectively. Fairweather et al. found that CT had poor performance in identifying individual N stage of GC patients with the accuracy of only 56% [25]. Xiong et al. showed that the correct diagnosis rate of preoperative CT for LNM was 72.7% in GC patients [26]. Therefore, a combined staging approach is needed for accurate prediction of LNM before surgery for GC patients.
On the other hand, traditional tumor biomarkers for GC, such as CEA, CA19-9, and CA72-4, are inferior in sensitivity and specificity in predicting LNM with conflicting results in different studies [27, 28]. Therefore, great efforts have been made by researches to find novel molecular biomarkers including oncogenes, tumor suppressor genes, growth factors, adhesion molecules, cytokines, chemokines, proteolytic molecules and angiogenic factors to detect nodal involvement in GC patients. However, their clinical applications are limited because of high test cost, low sensitivity/specificity, or complicated technical procedure. To date, no biomarkers mentioned above have been routinely used in clinical practice to accurately predict nodal involvement in GC patients.
The discovery of miRNAs may broaden our understanding of the underlying mechanisms of metastasis, and shed new light on LNM prediction. miRNAs are 20–22 nucleotide long, single-stranded non-coding RNAs, which are involved in many important biological processes such as cellular proliferation, apoptosis and differentiation by regulating gene expression post-transcriptionally. Accumulating evidence based on published studies indicates that miRNAs also play critical roles in tumorigenesis, progression and metastasis [29]. Since first measured in peripheral blood by Mitchell et al. in 2008 [30], circulating tumor-derived miRNAs have attracted more and more attentions for its potential value as promising novel biomarkers for cancer diagnosis, LNM assessment and prognosis prediction. For example, Cheng et al. found that serum miR-106a expression levels in cholangiocarcinoma patients with LNM were significantly decreased compared with those without LNM, multivariate logistic regression analysis including serum miR-106a, CA19-9 level, tumour differentiation, neural invasion, p53, and MUC1 expression further indicated that serum miR-106a was the only independent predictor of LNM [31]. Of note, circulating miRNAs are highly stable in harsh conditions and well protected from RNases, which make the detection of circulating miRNAs more convenient and less invasive compared with detection of traditional biomarkers in tissues [30].
Studies have proven that alterations in expression of miRNAs also contribute to the pathogenesis of GC, and specific circulating miRNAs can therefore be utilized as new noninvasive diagnostic and prognostic biomarkers for GC [32]. However, previous studies mainly focused on the diagnostic and prognostic values of circulating miRNAs in GC; whether circulating miRNAs can be utilized as independent predictors of LNM and increase the predictive value of CT in diagnosing LNM remain largely unknown.
Located within the seventh intron of epidermal growth factor-like protein 7 gene (EGFL7) on human chromosome 9 [33], miR-126 is one of the most intensively studied cancer-related miRNAs. First identified as a miRNA regulating human megakaryocytopoiesis [34], miR-126 was found to be down-regulated in a variety of cancers and function as a tumor suppressor. For example, miR-126 expression was found to be significantly decreased in esophageal cancer tissues and cell lines, and restoration of miR-126 in esophageal cancer cells inhibited cell proliferation in vitro, as well as tumor growth in vivo through targeting VEGF-A [35]. Notably, circulating miR-126 may be a novel metastasis-related biomarker. For example, serum miR-126 was found to be significantly down-regulated in Stage IV non-small-cell lung cancer patients compared to Stage I/II patients and healthy controls [36]. In colorectal cancer, serum miR-126 was found to be significantly down-regulated in synchronous liver-metastatic and other organ-metastatic patients compared to localized colorectal cancer patients [37].
A series of studies indicated that down-regulation of miR-126 played pivotal roles in the progression of GC [38]. Our previous studies demonstrated for the first time that miR-126 may function as a tumor suppressor in human GC with Crk as its direct target gene [14]. Combined miR-126-low/Crk protein-high expression in tumor tissues was found to be an independent unfavorable prognostic factor of GC [15]. Moreover, GC patients with lower serum miR-126 level had worse prognosis compared with those with higher level. Low serum miR-126 level was confirmed to be an independent poor prognostic predictor in our recent study [39]. Based on these findings, we speculated that the reason why lower serum miR-126 level was strongly associated with a significantly worse survival in GC might be attributed to more LNM occurred, thus, serum miR-126 might be useful to predict LNM in GC patients.
The main findings of current study were: (1) Serum miR-126 levels were significantly lower in GC patients with LNM than those in patients without LNM. (2) Serum miR-126 level varied according to the status of LNM (the later the N stage was, the lower the serum miR-126 level was). (3) Multivariate analysis demonstrated that lower serum miR-126 level conferred a higher risk for LNM in GC patients. These findings were important because it demonstrated that serum miR-126 could be used to predict LNM preoperatively in addition to the established imaging techniques in GC patients.
We further investigated whether serum miR-126 level could improve the predictive accuracy of MDCT for LNM preoperatively. The predictive accuracy of LNM by serum miR-126 level and MDCT was 81.9% and 69.2%, respectively. The current study demonstrated clearly that the combination of serum miR-126 level and MDCT could increase the predictive accuracy of LNM to 86.7%. Therefore, the addition of serum miR-126 level may improve the predictive accuracy of MDCT.
In conclusion, this study provided the convincing evidence for the first time that the serum miR-126 expression level was markedly decreased in GC patients with LNM, and the combination of serum miR-126 level and MDCT is useful for the preoperative diagnosis of LNM in GC patients. This discovery may help to better identify GC patients with high risk of LNM and facilitate personalized treatment of GC patients.
Footnotes
Acknowledgments
We appreciate all subjects that participated in this study. This project was supported, in part, by the grant from Shanghai Municipal Education Commission (12zz102), Yi Gong Jiao Cha Foundation of Shanghai Jiao Tong University (YG2016MS65) and Shanghai Pujiang Program (17PJ1406000).
Conflict of interest
The authors declare no conflict of interest.
