Abstract
BACKGROUND:
This study investigated the use of serum amino acids and organic acids profiles as the novel metabolites for screening breast cancer (BC) patients.
METHODS:
A total of 116 subjects as training set were divided into the following three groups: BC patients (
RESULTS:
The serum amino acids and organic acids profiles significantly differed between the BC patients, BE patients and the controls. Our results demonstrate that combinations of three candidate metabolites from taurine, glutamic acid and ethylmalonic acid were found to mirror tumour burden, with AUC values ranging from 0.751 to 0.834 when comparing BC patients to the controls. The areas under the curve from the taurine, glutamic acid and ethylmalonic acid in validated study were 0.901, 0.924 and 0.749, respectively.
CONCLUSIONS:
This study shows that amino acids and organic acids profiles will be a potential screening tool for BC patients. The dysregulated metabolism of amino acids and organic acids in breast cancer might be useful for the diagnosis, therapy, prognosis and understanding the pathogenesis of breast cancer.
Introduction
Breast cancer (BC) is one of the most fearful diseases due to its increasing worldwide prevalence, not only in European-Americans, but also in Asian countries [1]. In Europe, 458,337 women were diagnosed with breast cancer in 2012 and 131,259 women died of breast cancer [2]. Worldwide, close to 1.4 million women are diagnosed with breast cancer each year and approximately one-third die from this disease. In China, the crude incidence was estimated to be 42.55 per 100,000 women [3]. Early diagnosis of breast cancer has been clearly shown to reduce mortality and improve survival [4, 5]. Several tumor markers, such as CEA and CA15-3, are known to increase in breast cancer patients [6]. However, the elevated level of the tumor markers can also be due to other causes such as megaloblastic anemia and smoking [7, 8]. With the development of many high-throughput measurement technologies, there have been many studies regarding the diagnosis of BC using high-throughput methods of analysis, such as genomics, proteomics and metabolomics. It has been reported that breast cancer may be associated with specific metabolic signatures that include significant changes in the amino acids profile, as well as large perturbation of the phospholipid metabolism [9]. Here, we focus on metabolomics, namely metabolic profiling.
Metabolomics is the most recently developed of the omics sciences and refers to the study, identification and quantification of metabolites. Metabolites are small molecules (
Thus, the aim of the present study was to investigate whether amino acids profiles and organic acids profiles, established from a simple blood draw from healthy women, benign (BE) patients and BC patients. To this end, a targeted metabolomic analysis, a highly reliable and reproducible technology, was performed in this study. Alterations in the concentrations of certain metabolites detected in the serum of these samples can be candidates as future biomarkers of BC. Likewise, alterations in these metabolites could provide new insights into the pathological heterogeneity of breast tumours.
Clinical characteristics of 116 subjects in the discovery set (mean
SD)
Clinical characteristics of 116 subjects in the discovery set (mean
Study samples
The study was approved by the Ethics Committee of Harbin Medical University and was conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from each participant. Prior to the study, a 3-day food record was obtained from all participants to exclude any significant differences between the three groups due to their dietary intake (Tables 1 and S1). The enrolled patients were not on any medications before sample collection. All blood samples were collected in the morning before breakfast and fasting blood samples were allowed to clot for 30 min to 1 h at room temperature. And then, blood samples were centrifuged at 3,000 g for 10 min at room temperature, stored at
Subjects of the discovery set
A total of 116 subjects (aged 34–56 years) were recruited from The Affiliated Tumor Hospital of Harbin Medical University, Heilongjiang Province in the North of China. Subjects were divided into the following three groups: BC patients (
Subjects of the validation set
We tested findings for validation in the independent samples (90 subjects). These subjects were also recruited from The Affiliated Tumor Hospital of Harbin Medical University and were as a subset of the original collection for the validation. The distribution of 90 subjects was as follows: BC patients (
Chemicals and reagents
Amino acids and biogenic amines standards were purchased from Sigma (St Louis, MO, USA,
Preparation of standard solutions and assessment of the method
Solutions preparation
Stock solutions of the 47 standards were prepared at 1000
Preparation of quality control sample
Quality control (QC) samples were prepared by mixing equal volume of different individual serum (10 healthy control, 10 BE patients and 10 BC patients). One QC sample was injected at the start of analytical batch and then one QC sample at every fifth injection of samples was analyzed throughout the whole analytical workflow. The replicate of QCs (
Biochemical measurements
The serum biochemistry assays were performed on full-automated clinical chemistry analyzer (AUTOLAB PM 4000; AMS Corporation, Rome, Italy) using commercial biochemical kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China), calibrated with control reagents before sample analysis. The analytes measured included glucose (GLU), total cholesterol (TC), triglyceride (TG), High-density lipoprotein cholesterol (HDL-C), and low-density lipoprotein cholesterol (LDL-C). Cancer antigen 15-3 (CA15-3) concentrations were analyzed using EIA kit (ROCHE Diagnostics, Basel, Switzerland) and expressed as Units/L. Carcinoembryonic antigen (CEA) levels were assayed by ELISA kit (ROCHE Diagnostics, Basel, Switzerland ) and expressed as
Serum amino acids and biogenic amines profiles analysis
Serum preparation for amino acids and biogenic amines profiles
Serum amino acids and biogenic amines profiles were prepared as previously describe [20]. Briefly, each 50
UPLC/MS-MS analysis
UPLC/MS-MS analysis was performed by using a Waters ACQUITY UPLC system (Waters Corporation, Milford, MA, USA) coupled to a Waters Xevo TQD Mass Spectrometer (Waters Corporation, Manchester, UK). The UHPLC/MS-MS method was performed as our previously described [20]. In briefly, a part (2
MS analyses were made use of electrospray ionization (ESI) and Selected Reaction Monitoring (SRM) scans in the positive ion mode. In each transition, cone voltage and collision energies were optimized, the ion spray voltage was 3.2 kV, and the source temperature was 150
Serum organic acid profiles analysis
Sample preparation for organic acids
Serum organic acids profiles were prepared as previously described [23]. Briefly, Aliquots (100
GC-MS/MS analysis
GC-MS/MS analysis used using a TRACE gas chromatograph with a Polaris Q mass spectrometer (Thermo Finnigan, Austin, TX, USA). Helium was used as the carrier gas. A split injector (the split ratio being 1:10) at 250
Statistical analysis
Statistical analysis
Statistical analysis was carried out using SPSS 13.0 software (SPSS, Chicago, IL, USA). All data were presented as means
Multivariate analysis
The amino acids and organic acids profiles data were analyzed by multivariate statistical analysis using SIMCA-P 13.0 software (Umetrics, Umeå, Sweden). All data were standardized to a mean of 0 and a variance of 1, according to the following formula: (X – mean(X))/std(X) for multivariate statistical analysis. Unsupervised principal component analysis (PCA) was used first in all samples to see the general separation. PCA is a bilinear decomposition method used for overview ‘clusters’ within multivariate data. The score plot of PC1 versus PC2 was used to examine separation or clusters for the three groups.
Quantitative analysis of amino acids and biogenic amines profiles in three groups
Quantitative analysis of amino acids and biogenic amines profiles in three groups
Then, partial least-squares-discriminant analyses (PLS-DA), which is a supervised classification method, is based on the PLS approach [25]. The standard PLS algorithm can be used for the dependent y vector and class labels can be used. In the two-class case the values of the dependent variable are usually assigned a 1 for one class and a 0 or
Method validation
We used QC samples to check the UPLC-MS/MS and GC-MS/MS system for reproducibility and reliability. PCA performed on QC samples and the other groups revealed that the QC samples were clustered in PCA scores plot (Supplemental Fig. S1). Fifty four metabolites from QC samples were also used to assess for method reproducibility and reliability. The relative standard deviations (RSD%) of the metabolites were showed to be 2.44–9.13% (Supplemental Table S1). Intra-day and inter-day accuracies and precisions of this method were determined by analyzing three replicates. The precisions ranged from 1.11% to 14.91%, whereas accuracies ranged from 80.14% to 118.70% (Supplementary Table S1). The above results showed that this method had good repeatability, reliability and stability for metabolite analysis.
Score plot from PCA analysis of the three groups (R2X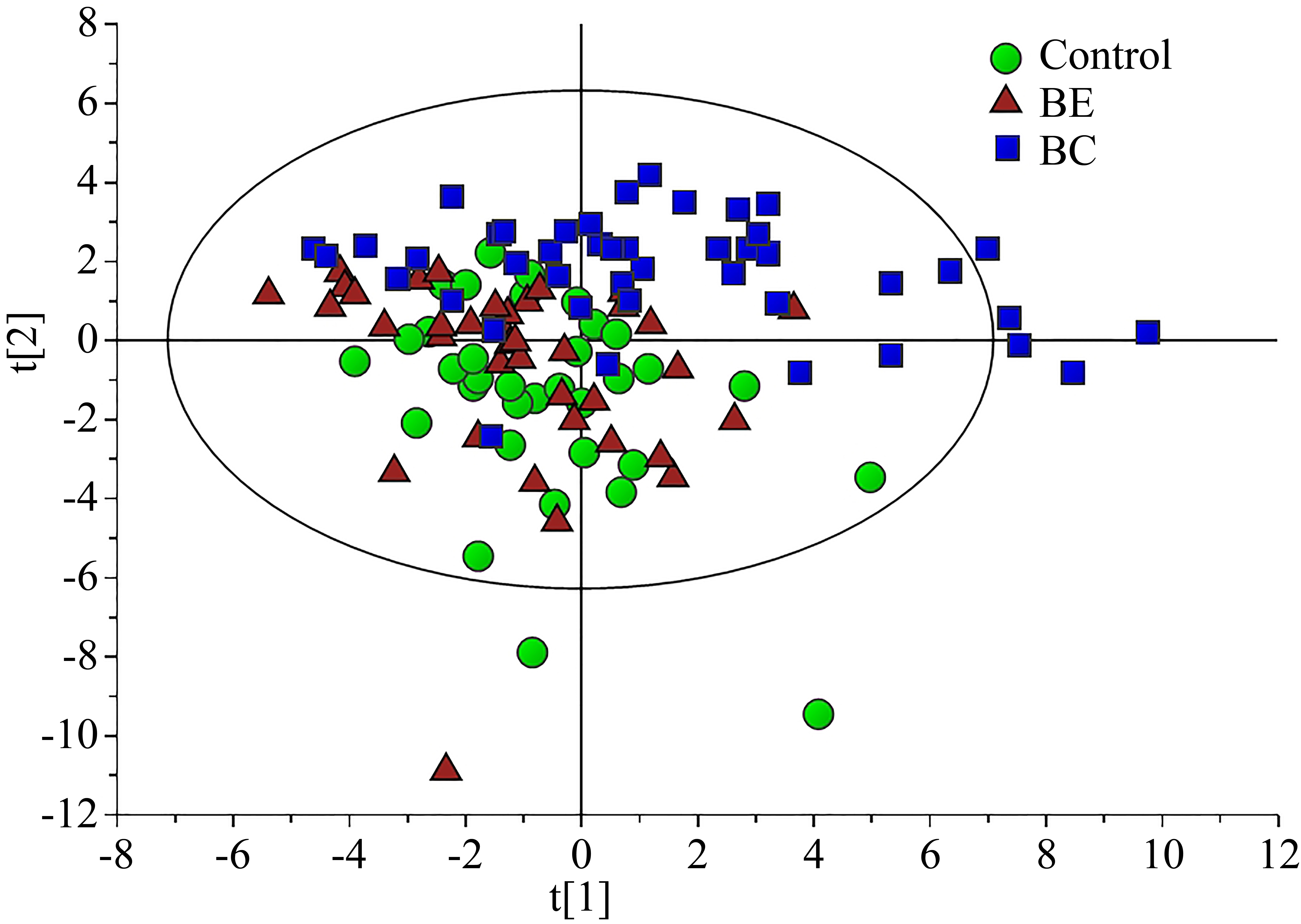
There were no significant differences between the three groups with respect to age, smoking and alcohol consumption (Table 1). Some clinical characteristics including body mass index (BMI), TG, TC, fasting blood glucose (FBG), systolic blood pressure (SBP) and diastolic blood pressure (DBP) did not differ significantly between the three groups (ANOVA,
Changes in the amino acids and biogenic amines profiles
We performed serum amino acids and biogenic amines profiles using UPLC-MS/MS in three groups. Twenty-eight metabolites were detected in all subjects (Table 2). The levels of 20 metabolites changed significantly in the BC group when compared to the control group (Fig. 1,
Changes in the organic acids profiles
The quantitative results of organic acids profiles analysis in the three groups are presented in Table 2. Comparison of 28 organic acids concentrations measured in serum from the controls, BE patients and BC patients revealed substantial differences. Ten of the organic acids (adipic acid, caproic acid, ethylmalonic acid, glycolic acid, glutaric acids, 2-hydroxybutyric acid, 2-hydroxyisocaproic acid, pyruvic acid, suberic acid, succinic acids) in the BC patients significantly decreased when compared with the healthy control. Six organic acid (adipic acid, caproic acid, glycolic acid, glutaric acids, 2-hydroxybutyric acid, succinic acids) in the BC group were lower than those of BE group. There were significant differences in three organic acids (ethylmalonic acid, 2-hydroxyisocaproic acid, succinic acids) between the healthy control and BE subjects (
PLS-DA scores plot between two sub-groups. Circle (green), healthy control; Triangle (red), BE; Box (blue), BC. (A) PLS-DA scores plot for healthy control and BE groups (R2X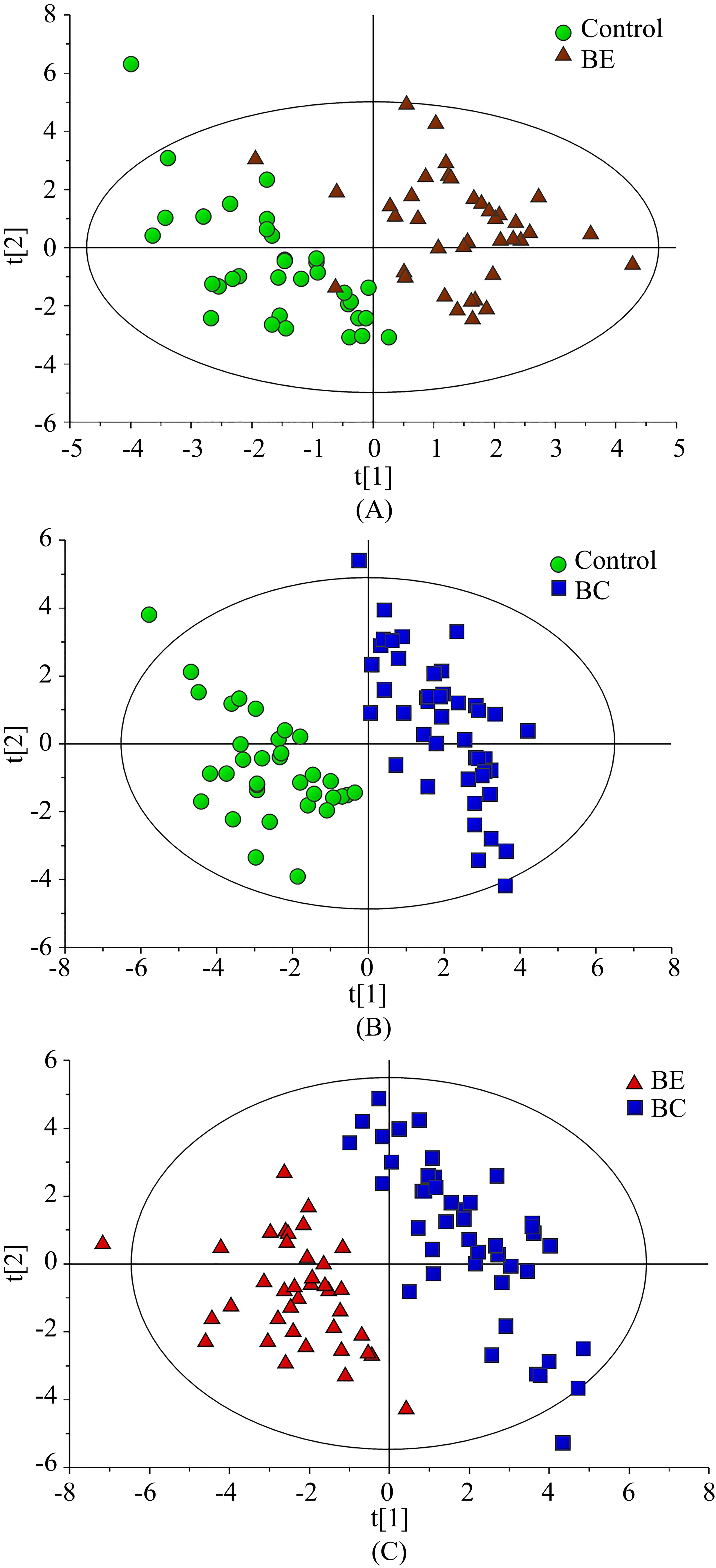
Loadings plots of free fatty acids from PLS-DA models. (A) Healthy control and BE; (B) Healthy control and BC; (C) BE and BC Corresponding loadings plot; possible biomarkers were marked with a red.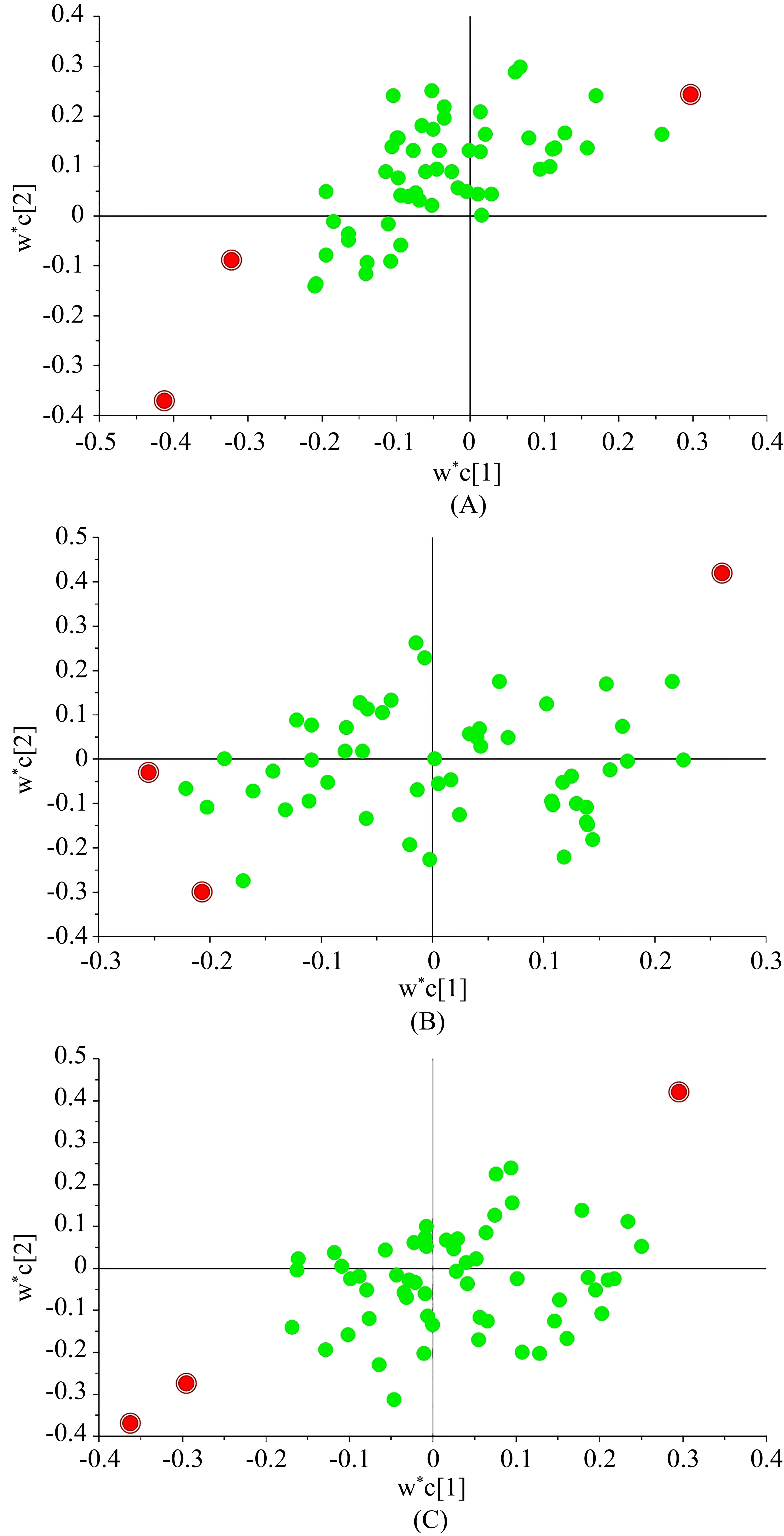
Amino acids and biogenic amines, and organic acids profiles were analyzed by PCA. The score plot of PCA represented the distribution of all samples (Fig. 1A). The first two components of the model (PC1 and PC2) explained 35.2% of the variance (R2
After crude screening by PCA, a PLS-DA model was constructed for classification of the three groups. In order to screen the important metabolites, three sub-comparisons using PLS-DA were performed to compare the healthy controls and BE patients, the healthy controls and BC patients, and BE and BC patients. The results of PLS-DA showed that the samples in BC patients versus the healthy controls and BE individuals (Fig. 2B and C) were separated clearly; there was also acceptable separation in the comparison of healthy controls and BE individuals (Fig. 2A). There were high Q2 values of in different models (Q2
Quantitative analysis of organic acids profiles in three groups
Quantitative analysis of organic acids profiles in three groups
Important metabolites based on the statistical analysis in three groups
In this work, a PLS-DA model and univariate statistical analysis was used for classification of healthy control, BE and BC patients and the identification of potential biomarkers. Firstly, the possible biomarker candidates were filtered using VIP values (VIP
Pathways modulated by breast cancer condition
Pathways modulated by breast cancer condition
Quantitative analysis of taurine, glutamic acid and ethylmalonic acid in the validation study. 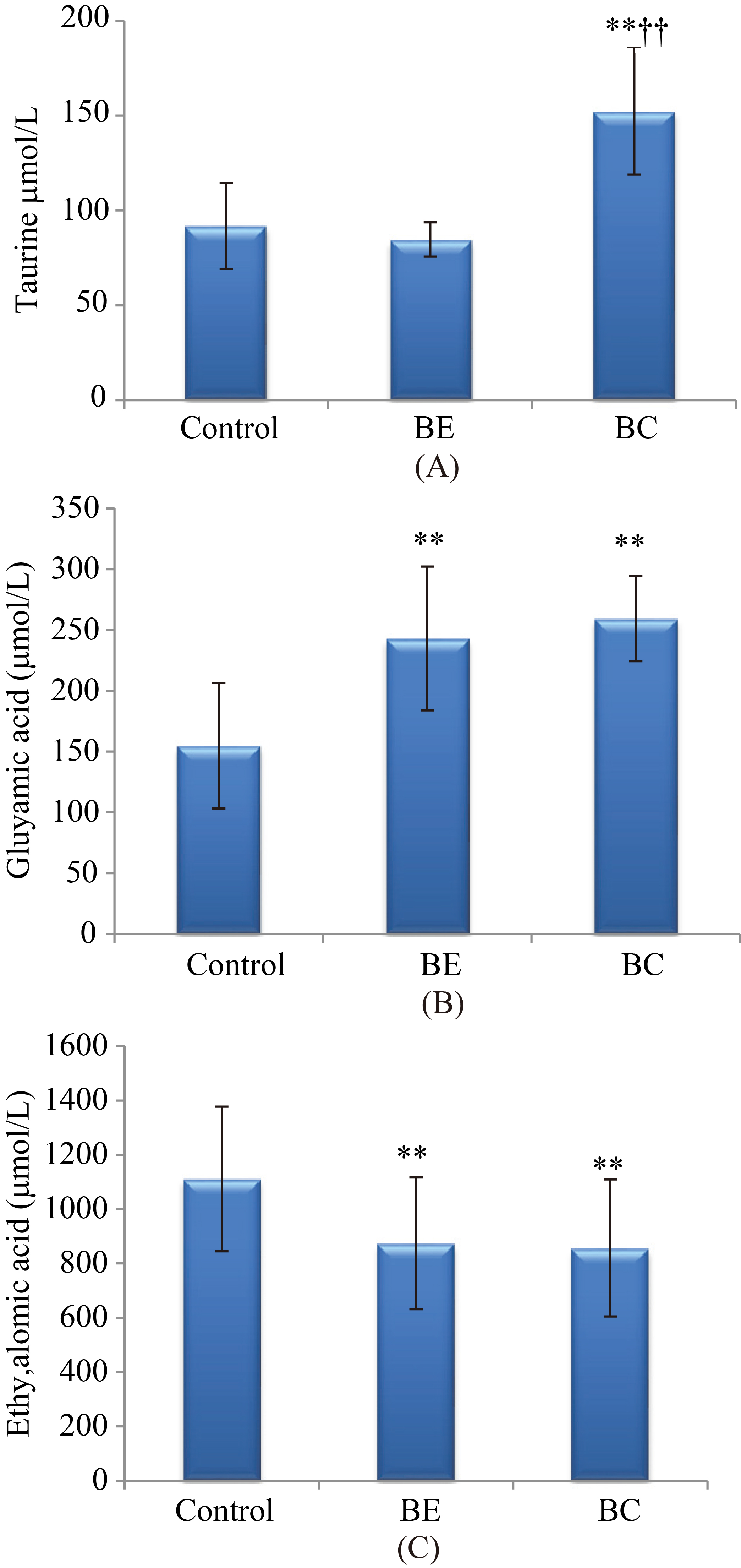
ROC analysis for discrimination of BC and the healthy control by important metabolites in the validation set.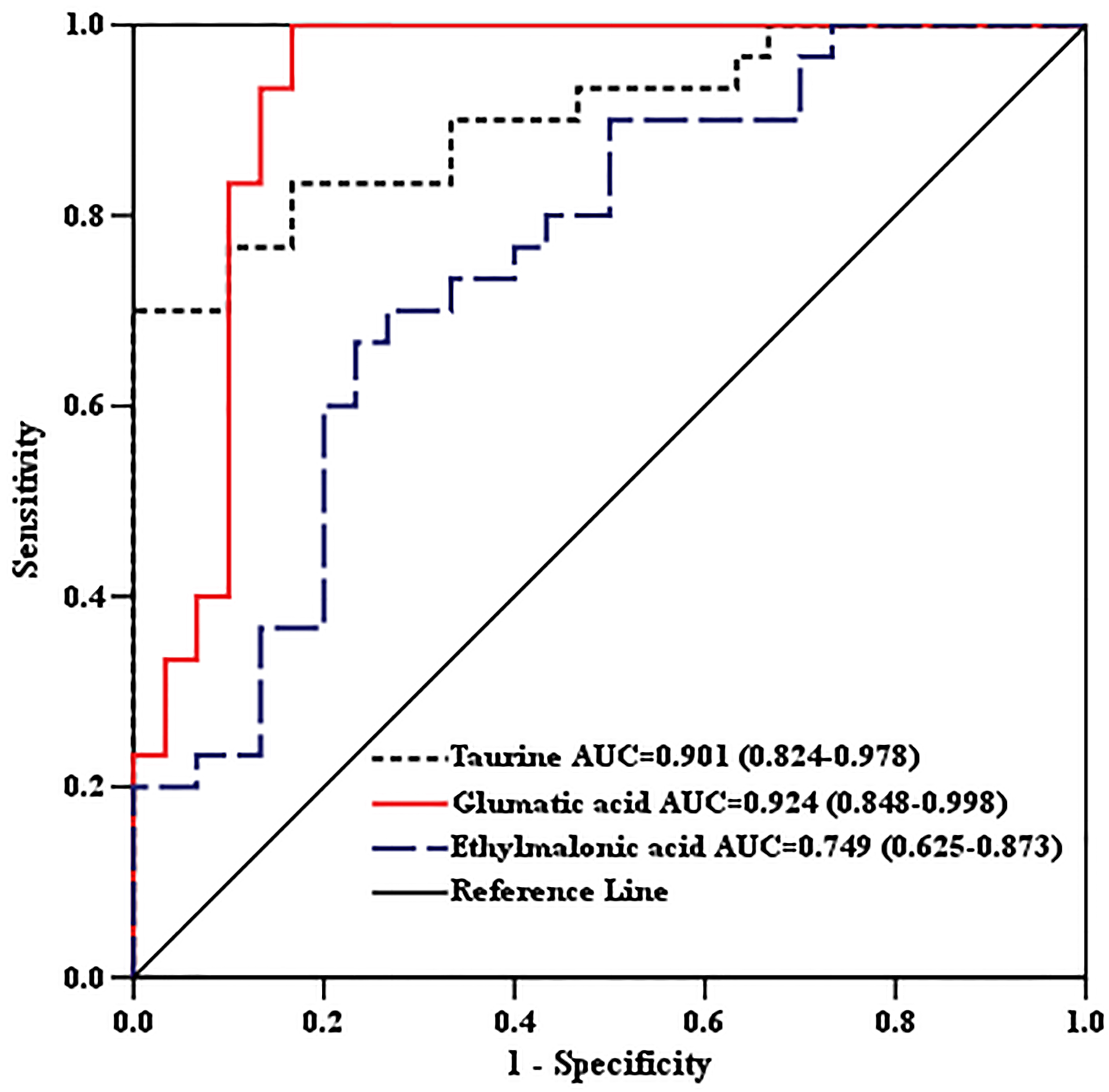
The topology analysis of pathway for the important metabolites.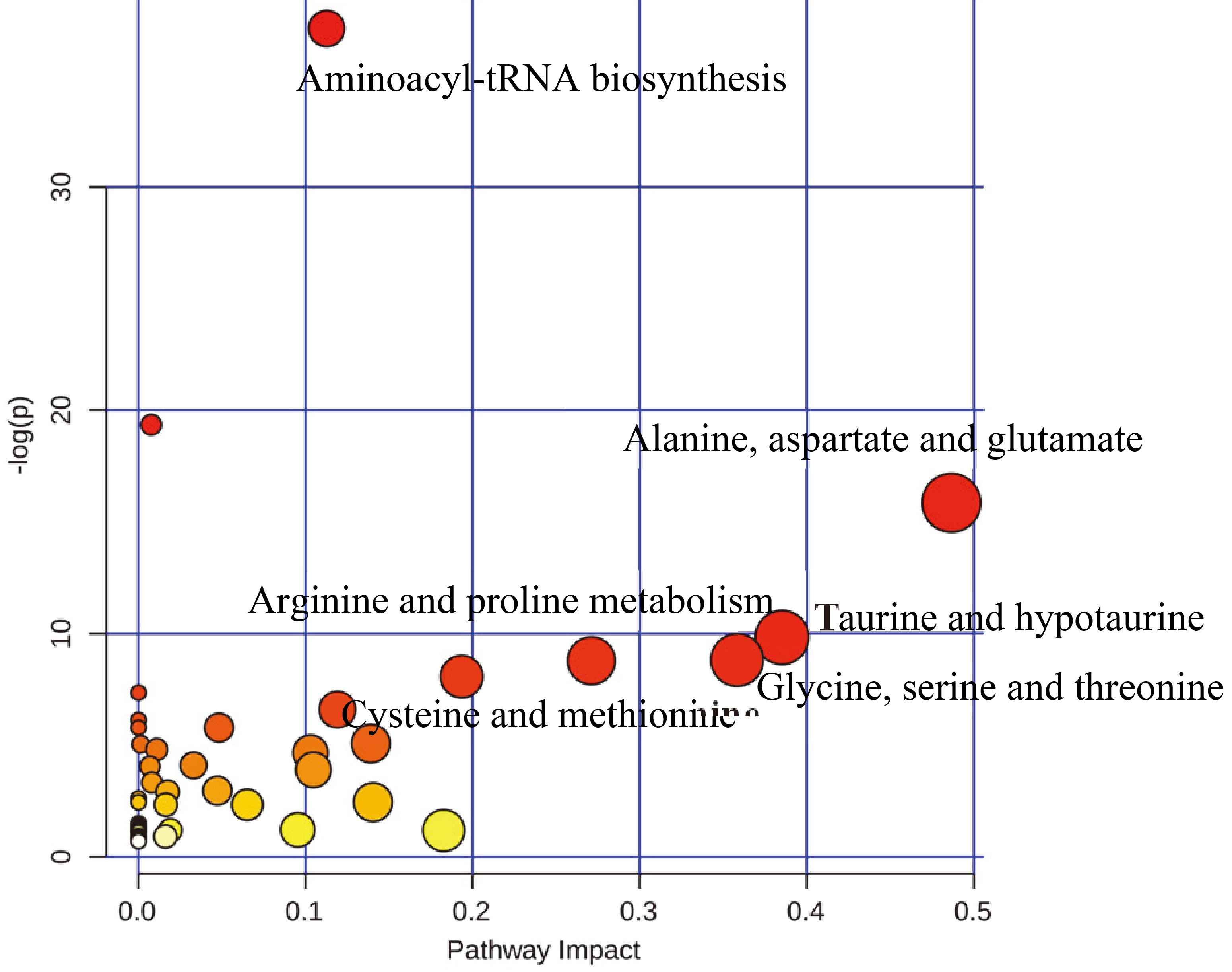
We measured the important metabolites which were able to distinguish BC patient from the other subjects in the discovery set (Table 4) from the independent healthy controls, BE patients and BC patients to validate the above results. Compared with healthy controls and BE patients, the increased taurine, glutamic acid and decreased ethylmalonic acid in BC patients was observed in Fig. 4. The results of ROC analysis (Fig. 5) showed that the AUC values for taurine, glutamic acid and ethylmalonic acid were 0.901, 0.924 and 0.749 for BC patients and the controls. The sensitivity and specificity for prediction were higher than 70% in the ROC curve (data not shown). The above results suggested that BC patients with higher level of taurine glutamic acid and the lower ethylmalonic acid were likely to be diagnosed with BC regardless of the controls.
Pathway analysis
In this work, 29 differential metabolites were imported into the MetaboAnalyst 3.0. As a result, the web-based software generated 6 metabolic pathways with a FDR,
Discussion
In this study, we demonstrated that the targeted metabolomics technology could be used to discriminate BC patients, BE patients and the controls based on serum data analyzed by UPLC-MS/MS and GC-MS/ MS. Recently, untargeted metabolomics technology has been used to analyze significant metabolic variations in BC patients. Lécuyer et al. [27] found that elevated plasma levels of lipoproteins and lipids in BC patients were observed based on the NMR metabolomics. Furthermore, Takayama et al. [28] found that polyamines have been significantly associated with rapid tumor growth and appears to be useful for the diagnosis of breast cancer patients. The above studies showed that there was the alternation of lipids and polyamines metabolism. Our results showed that some amino acids and organic acids were differently expressed in the three groups, and glutamic acid, taurine and ethylmalonic acid were identified as the potential metabolites of BC patients. Moreover, the determination of the glutamic acid, ethylmalonic acid and dimethylglycine is helpful for distinguishing the healthy control from BE patients.
One remarkable observation in this work is that the serum amino acids profiles were significantly changed in BC patients. Amino acids play essential physiological roles both as basic metabolites and metabolic regulators. Serum amino acids abundantly circulate as a medium linking all organ systems and amino acids profiles have been known to be influenced by metabolic variations in specific organ systems induced by specific diseases [29, 30]. Dowling et al. reported that the amino acids profiles characterized higher glutamate, isoleucine and proline changed in breast cancer [31]. Consistent with this study, we also found that there were the changed amino acids profiles and increased glutamate and glutamine in BC patients. Simultaneously, the result of the pathway analysis showed that the metabolic change of the alanine, aspartate and glutamate metabolism were observed in BC patients (Fig. 6). Glutamate is a product of the glutamine under the action of the phosphate-dependent glutaminase (GLS), an enzyme found within the inner mitochondrial membrane. It has been shown that there is the overexpression of GLS in both solid tumours and cell line models of cancer [32, 33]. Other groups have reported on elevated glutamate levels and associated signaling pathways in malignancies [34], and the increased rates of glutamine metabolism through the accelerated hydrolysis of glutamine to glutamate, as catalyzed by mitochondrial GLS activity in breast cancer [31]. Our findings support the metabolic changes in cancer metabolism resulted in a shift to increased rates of glutamine metabolism.
We also found taurine was also importantly different metabolite distinguishing BC patients from non-breast cancer groups (The controls and BE patients). Meanwhile, taurine and hypotaurine metabolism is importantly metabolism in breast cancer. The taurine and hypotaurine metabolism has been shown to be relevant to women cancers, such as ovarian and breast cancer [35, 36]. Here, we also have discovered that taurine and hypotaurine metabolism is also dysregulated in the blood samples of breast cancer. Many lines of evidence suggest taurine and hypotaurine metabolism is a critical pathway in tumor development [35, 36]. Taurine, which is a sulfuric amino acid, can be synthesized by the human body from cysteine. Taurine has many diverse biological functions that serve as stabilizers of cell membranes and anti-oxidants. Much evidences support the role of reactive oxygen species (ROS) and oxidative stress in development of breast cancer [37]. ROS, by attacking DNA, can induce DNA damage which in turn can either inhibit or induce replication errors and genomic instability. All these effects are known to be associated with carcinogenesis [38]. In this study, the elevated taurine, as an anti-oxidants, has high an AUC of 0.941 which diagnosed the BC patients in the validation set. The elevated content in taurine is also essential for the antioxidant glutathione [39] and taurine was increased in plasma from cancer subjects could be involved in protecting cancer cells from excessive damage by oxidative stress [40]. These studied further confirmed our results. In short, the taurine and hypotaurine metabolism significantly changed in BC patients, which is also confirmed by the other studies.
Previous metabolomics studies have shown that certain metabolites can differentiate breast cancer patients from normal control [41, 42]. For example, glutamate was found enriched and the glutamate-to-glutamine ratio was significantly correlated with estrogen receptor status in breast cancer patients [41]. Our result confirmed that there were higher glutamate and glutamate-to-glutamine ratio in breast cancer than that of the healthy control (Table 2). Moreover, histidine, glucose, and lipids were strongly correlated with breast cancer relapse with a predictive accuracy of 75% based on the serum profiles [42]. However, although TCA cycle key enzyme genes may serve as potential biomarkers to predict the outcomes of lung cancer [43], less attention is paid to the serum organic acids profiles in breast cancer. Our data suggested that the alterations of organic acids profiles could reflect underlying metabolic changes taking place during BC development. The present study has highlighted ethylmalonic acid as the organic acid with the greatest potential to act as biomarkers of BC. Animal experiments suggested that ethylmalonic acid is related to an inhibition of creatine kinase activity and induction of lipid peroxidation in hippocampus [44]. Notably, the serum ethylmalonic acid in cancer is not reported in the literature. So the reason of decreased ethylmalonic acid in BC patients will be investigated in the future study. Together, the significant increase in the serum levels of these biomarkers observed in BC patients could possibly indicate that breast cancer is accompanied by, or precipitated by, an underlying metabolic disorder affecting organic acids metabolism.
There are limitations of this study. Firstly, although our results have been validated in the independent samples, the number of studied subjects was relatively small. Thus, care must be exercised in the application of our findings to larger populations of BC patients. Secondly, the relevance of the change of organic acids profiles in the pathoetiology of BC could not be explained. Furthermore, multiple controls, such as other types of women cancer including cervical cancer, ovarian cancer, and so on, would be needed to evaluate whether the selected amino acids and organic acids biomarkers is specific to BC. Thirdly, further studies are required to explore the metabolic mechanisms of organic acids profiles in breast cancer and to validate these results in the human larger population.
In conclusion, we performed a targeted metabolomic investigation for an understanding of breast cancer metabolism. Metabolism of BC patients, as expected, differs a lot from that of BE patients and the controls. Metabolic reprogramming in BC patients, mainly characterized by changed taurine and hypotaurine metabolism, disturbed metabolism of organic acids, abnormal amino acid metabolism, is found to be related with BC development and progression. Taurine, glutamic acid and ethylmalonic acid as different metabolites are identified that could facilitate early diagnosis and clinical staging for EOC. In all, targeted metabolomics studies from breast cancer patients could be useful to describe diagnostic and/or prognosis biomarkers, as well as for monitoring treatment.
Footnotes
Acknowledgments
Natural Science Foundation of Heilongjiang Province for the returned students (No. LC2016030), The Youth Science Foundation of Liande Wu in Harbin Medical University (No. WLD-QN1704), Program for the innovation talent in Harbin Science and Technology Bureau (No. 2017RAQXJ169).
Conflict of interest
The authors declare they have no actual or potential competing conflict of interest.
