Abstract
Background
Invasive ductal carcinoma (IDC) is the most common type of breast cancer so its early detection can lead to a significant decrease in mortality rate. However, prognostic factors for IDC are not adequate and we need novel markers for the treatment of different individuals. Although positron emission tomography and magnetic resonance imaging techniques are available, they are based on morphological features that do not provide any clue for molecular events accompanying cancer progression. In recent years, “omics” approaches have been extensively developed to propose novel molecular signatures of cancers as putative biomarkers, especially in biofluids. Therefore, a mass spectrometry-based metabolomics investigation was performed to find some putative metabolite markers of IDC and potential metabolites with prognostic value related to the estrogen receptor, progesterone receptor, lymphovascular invasion, and human epidermal growth factor receptor 2.
Methods
An untargeted metabolomics study of IDC patients was performed by liquid chromatography coupled with tandem mass spectrometry (LC-MS/MS). The multivariate principal component analysis by XCMS online built a model that could separate the study groups and define the significantly altered m/z parameters. The most important biological pathways were also identified by pathway enrichment analysis.
Results
The results showed that the significantly altered metabolites in IDC serum samples mostly belonged to amino acids and lipids. The most important involved pathways included arginine and proline metabolism, glycerophospholipid metabolism, and phenylalanine, tyrosine, and tryptophan biosynthesis.
Conclusions
Significantly altered metabolites in IDC serum samples compared to healthy controls could lead to the development of metabolite-based potential biomarkers after confirmation with other methods and in large cohorts.
Introduction
Breast cancer is the most prevalent cancer and the second leading cause of mortality from cancer in women worldwide. 1 It is estimated that about 1.5 million new cases are diagnosed each year. 2 The poor survival of breast cancer patients mainly relates to cancer recurrence and metastasis, which might originate from cancer stem cells that are mostly resistant to radio- and chemo-therapies. 1 The metastasis rate of breast cancer is high and the statistics show that approximately 25% of patients may develop metastasis after tumor diagnosis, and approximately 90% of deaths related to breast cancer occur as a sequence of metastasis. The 5-year survival of breast cancer patients is more than 80% where it is only 25% for cases with distant metastasis. 3 The intra-tumor heterogeneity is the most challenging feature of breast tumors, which has a close relationship to cancer prognosis, recurrence and response or resistance to treatments. The conventional predictive factors of breast cancer include the presence of estrogen or progesterone receptors (ER and PR, respectively) which show the response to endocrine therapy and human epidermal growth factor receptor 2 (HER2) measurement, which predicts response to trastuzumab (Herceptin)/endocrine therapy and which might be used as neoadjuvant/adjuvant for advanced disease. 4 ER positive (ER+) tumors are well differentiated with less aggressiveness and better outcome after surgery. 5 PR positive (PR+) tumors are more aggressive than ER + tumors. HER2 + tumors are the most aggressive among hormone receptor positive breast cancers. However, their response to anti-HER2 therapy is satisfactory. 6 The tumors that do not express hormone receptors are called triple-negative breast cancer (TNBC). TNBC patients do not have estrogen or progesterone receptors with too little or none of the HER2 protein. TNBC is often classified as a subtype of basal-like breast cancer based on their gene expression profile. It is very invasive, and almost 50% of patients will have distant metastasis. Approximately 80% of basal-like tumors are HER2-negative (HER2−)/ER-negative (ER−). Various subtypes are also characterized for TNBC based on ESR1, PGR, and ERBB2 expression alterations in terms of DNA mutations or RNA expression such as basal-like, immunomodulatory, and mesenchymal. 7 Because the cancer cells do not have ER, PR, or HER2 protein, hormone therapy and drugs that target HER2 are not helpful, so combination therapy is the main treatment option. In addition, even though TNBC responds well to this therapy initially, it tends to recur more frequently than other breast cancers. 8 The prognosis of various breast cancer types is shown in Figure 1. Lymphovascular invasion (LVI) is another prognostic factor for breast cancer and refers to the invasion of tumor emboli to blood vessels and lymphatic spaces. 9 Several studies have demonstrated a distinct relation between the presence of LVI in breast cancer and increased risk of axillary lymph node and distant metastasis. 10 Invasive ductal breast carcinoma (IDC), accounts for approximately 80% of invasive breast cancers. It originates from the cells in the milk ducts and then spreads into the surrounding breast fibrous or fatty tissue, lymph nodes, or other areas of the body. 11 One of the currently used techniques for breast cancer detection is mammography, which has between 70% and 90% sensitivity and approximately 75% specificity for ductal carcinoma. 12 However, for dense breast tissues, the sensitivity is reduced to about 50%. Moreover, it is observer-dependent and is based on the radiologist's experience. 13 In recent years, several investigations have been carried out looking for novel molecular signatures as putative biomarkers for breast cancer. For instance, miRNAs, circulating tumor cells, or circulating tumor DNA have been proposed. 4 During the past decade, “omics” approaches (mainly genomics, proteomics, and metabolomics) have extensively been developed in the field of biomarker discovery. Among them, metabolomics— the holistic comprehensive analysis of low molecular weight (<1500 Da) metabolite content of a given biological matrix (such as biofluids, tissues, or cultured cells) in various pathophysiological conditions—has gained much attention as metabolites are closely related to phenotype and cellular functions. 14 In cancer research, these approaches not only improve our understanding of the underlying mechanisms of the disease, but also help to find putative screening or early detection markers, prognostic and theranostic markers, and potential new drug targets for more effective therapeutic interventions.15,16 Commonly used metabolomics analytical platforms include nuclear magnetic resonance (NMR) and mass spectrometry (MS) coupled with separation techniques such as gas or liquid chromatography (GC-MS and LC-MS, respectively) or capillary electrophoresis (CE-MS). Each technique benefits from various advantages depending on the application and conditions. 17 In recent years, several metabolomics-based studies have been reported in the field of breast cancer, which shows the importance of modern omics-based investigations. In their study, Matos Do Canto et al. 18 reported an untargeted metabolomic analysis of breast ductal fluid (from both the affected breasts and the unaffected contralateral breasts (as controls)) using an ultra-performance liquid chromatography coupled with a quadrupole time-of-light (UPLC-QTOF) mass spectrometer. In other study, More et al. 19 analyzed metabolomic profiles of tissue and serum samples of cancerous and control groups using targeted liquid chromatography multiple reaction monitoring mass spectrometry (LC-MRM/MS) and untargeted GC-MS approaches.

Breast cancer prognosis based on hormone receptors expression.
The most attractive population-wide screening method can be the diagnostic biomarkers in biological fluids especially those found in the blood. Blood can be a source of tumor-specific antigens and autoantibodies, nucleic acids (such as DNA and RNA), lipids, proteins, and metabolites. Previously, various studies indicated the value of each of these classes of biomolecules for the early detection of breast cancer. 20 In this study, we have performed a metabolomic profiling of 22 serum samples from invasive ductal breast cancer (14 at stage I and II and 8 at stage III according to the tumor node metastasis (TNM) staging system) compared to 10 healthy controls using targeted LC-MS/MS approach. Furthermore, serum metabolomic profiles of HER2 + /−, ER + /−, PR + /−, and LVI + /− were also acquired to identify potential diagnosis and prognosis metabolite markers. Here, we have shown that existence of distinct metabolomic profile marked for IDC, differentiated them from healthy subjects. In addition, we detected some metabolites with potential prognostic value related to ER, PR, LVI, and HER2.
Methods
Populations
A total of 22 females with invasive ductal breast cancer (14 at stage I and II and 8 at stage III according to the TNM staging system) were recruited for the study. The patients were in the age range of 30–65 years and were diagnosed with ductal breast carcinoma (IDC) based on both clinical and histopathological data. The patients had neither a history of tumor surgical operation nor chemo- or radio-therapy before sample collection. The smokers were excluded from the study. A control group composed of 10 healthy females with the same age range were also recruited in the study. This study was approved by the clinical ethics committee of Shahid Beheshti University of Medical Sciences with code (IR.SBMU.RETECH.REC.1397.821) and all the patients provided written informed consent to participate in the study. A detailed number of the study groups is listed in Supplementary Table 1. We also performed power analysis for sample size calculation. 21 The result is shown in Supplementary Figure 1. The power is calculated based on the specified difference and sample size. The green line shows the 90% line, which means there is 90% chance of detecting a difference between the two groups. The curve plots the power of the test versus the difference between the mean and the target to find the appropriate sample size for the test The purple line also shows the 80% chance of detecting a difference, which can be adequate according to some references. In this curve, the sample size is 12 for the 80% line and 16 for the 90% line. As the difference approaches 0, the power decreases and approaches α error, which is 0.05 for this analysis.
Sample collection and storage
Fasting whole blood samples were collected from patients and healthy controls in vacutainer tubes. The tubes were remained 1 h in the refrigerator and then centrifuged at 3000 g for 10 min at 4°C. The supernatant serum was stored in a freezer at −80°C until use.
Sample preparation for LC-MS/MS analysis
For LC-MS/MS analysis, serum samples were thawed on ice; 150 μL of serum was then mixed with 300 μL of cold methanol followed by vigorous shaking for 30 s. The homogenate was then kept in −20°C freezer for 10 min followed by 1 min vortexing and 10 min centrifugation at 15,000 g and 4°C. The supernatant was used for LC-MS/MS injection. We also had three pooled quality control (QC) samples, which were prepared by mixing 10 μL of each sample. One injection of QC samples was done between every 10 runs to ensure data quality.
LC-MS/MS analysis of samples
Liquid chromatography coupled with an electrospray triple-quadrupole mass spectrometer (LC-ESI-QQQ-MS/MS) was used for the analysis (Waters Quattro micro-API micromass 2695, Waters Corporation, USA). The experiment was conducted using a C18 reverse-phase Acquity BGH (100 × 2.1mm × 1.7μm Waters) with acetonitrile (ACN) and H2O as mobile phase as follows: min 1–15 ACN 5%, H2O 95%, min 15–20 ACN 95%, H2O 5%. All analyses were performed in the negative mode (ESI−). Other characteristics of the MS were as follows: capillary voltage: 3.2 KV, cone voltage: 30 V, extraction cone voltage: 3 V, drying gas: N2 at 200 L/h and 350°C, mass scan range: 50–1000, ambient autosampler, and data collection at centroid mode. An MS2 daughter scan was also performed for the significantly altered peaks from MS1 parent ions. All the chemical compounds were of analytical grade and were obtained from Sigma-Aldrich (Burlington MA, USA).
Data processing and statistical analysis
Raw spectra were collected from MassLynx v4.1 software as mzxml format and transferred to XCMS online for further processing of data and statistical analyses. By using raw chromatographic data, XCMS online first corrects the retention time deviations and aligns the chromatograms, then identifies the significantly altered ion intensities through pairwise or multi-group statistical analyses. The principal component analysis (PCA) models were built after univariate scaling and mean centering of raw data. The multivariate PCA was performed in XCMS online, which built a model that could separate the study groups and define the significantly altered m/z parameters. The PCA results showed the R2Y and Q2Y values of 0.6 and 0.4 for discrimination between the IDC and control groups. According to PCA results, those m/z ions with a P-value < 0.05 and fold change more than 1.5 were considered significant. The significantly altered metabolites were identified by searching the Human Metabolome Database (HMDB) for accurate mass to charge ratios of parent and daughter ions fragmentation patterns. The significant involved biological pathways were identified by Metaboanalyst 5.0 online platform. Supplementary Figure 2, shows the receiver operating characteristic (ROC) curves indicating the PCA model’s ability to distinguish between the groups. As can be seen in the pictures, the area under the curve was 1 for the HER2 and PR groups. This value was 0.6, 0.71, and 0.69 for the IDC-control, ER, and LVI groups, respectively.
Results
The multivariate PCA was performed to find the significantly altered metabolites between the study groups. It was shown that the QC samples had a separate cluster according to Figure 2, which manifests the good repeatability of the mass analysis. As shown in Figure 3, the first principal components, PC1 and PC2, explained 34% and 19% of the variance, respectively, (with cumulative variance of 58% for the first three components) to discriminate between IDC and healthy subjects. The PC1 and PC2 variance were 36% and 13% (with the cumulative variance of 60% for the first three components) to discriminate between ER + and ER−. According to Figure 4, PR + was separated from PR− with PC1 and PC2 of 35% and 14%, respectively, with a cumulative amount of 60% for the first three components). These amounts were 35% and 15% for the first two components (with 59% cumulative variance for the first three components) to distinguish between HER2 + and HER2−. LVI + and LVI− were also separated with PC1 and PC2 variances of 33% and 15% with 60% of the cumulative variance for the first three components. The significantly altered metabolites between IDC and healthy controls are shown in Table 1. The R2 and Q2 scores plots for PCA models and ROC curves are shown in Supplementary Figures 2 and 3. The detailed specifications of the significantly altered metabolites for all the comparisons including their average concentration ratios and P-values are also provided in Supplementary Tables 2–6. The pathway analysis results from the comparison between the control group and the IDC group showed that the most important involved pathways included arginine and proline metabolism, glycerophospholipid metabolism, and phenylalanine, tyrosine, and tryptophan biosynthesis (Supplementary Figure 4). We also performed partial least squares discriminant analysis (PLS-DA) analysis for the IDC-control groups; the result is shown in Supplementary Figure 5. The results show that the R2Y and Q2Y values are 95% and 77%, respectively. The RMSE value is 0.105 where the t1 + t2 value was 31%.
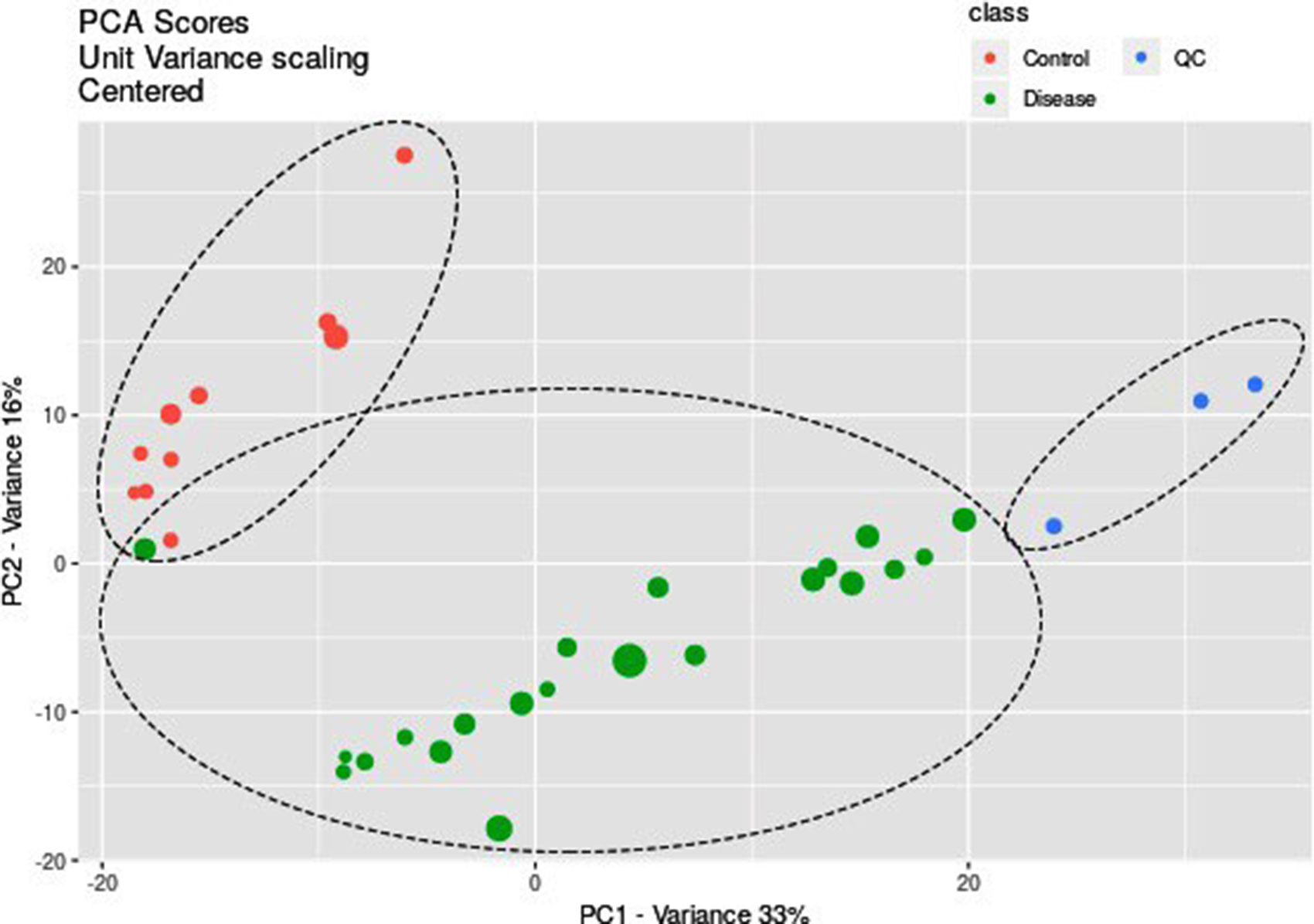
PCA scores plot shows separate clusters for disease, control and quality control (QC) samples which manifests the good repeatability of data analysis.
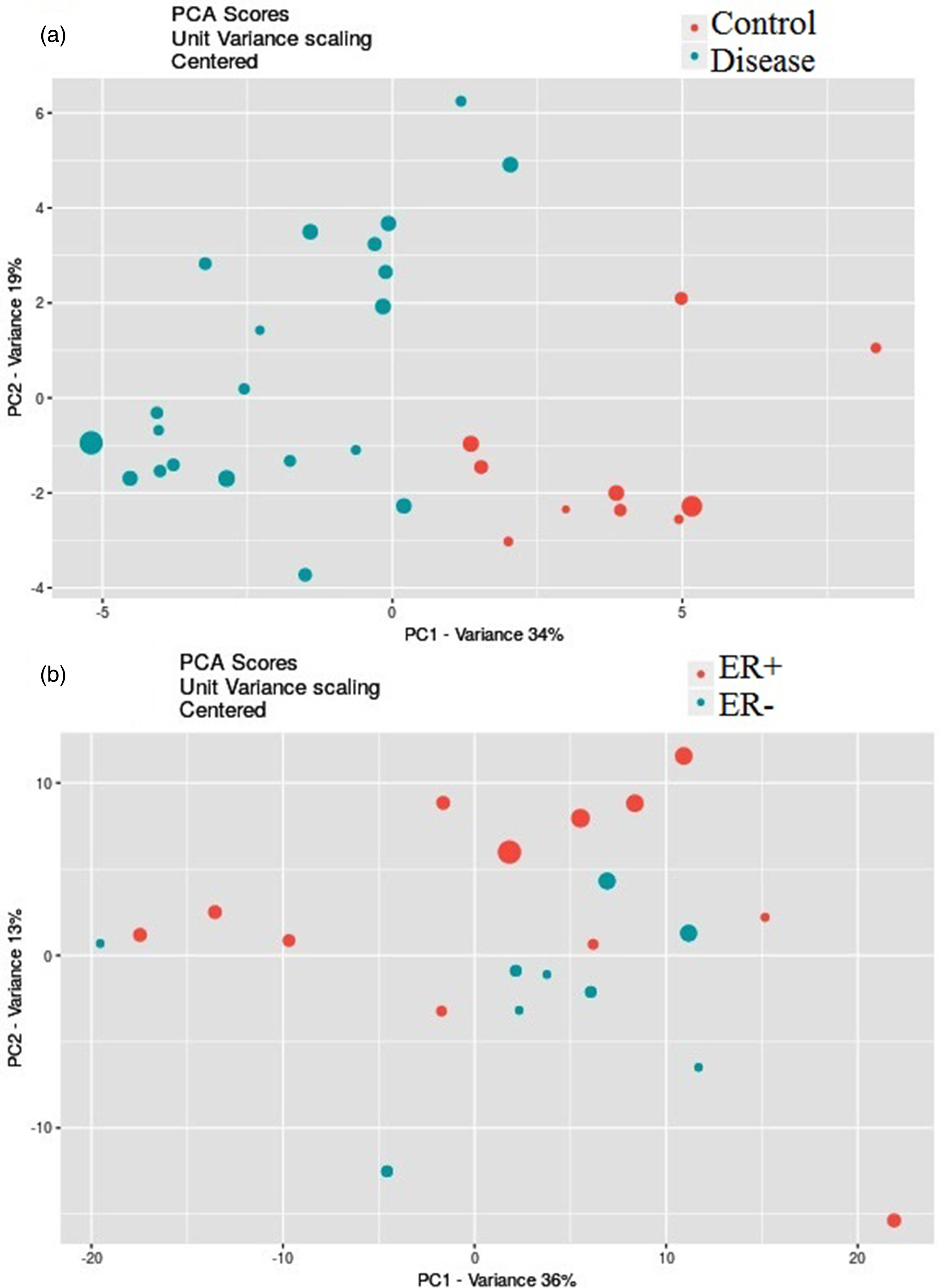
PCA scores plots produced by online XCMS for the separation in datasets: (a) IDC vs. healthy controls; (b) ER + vs. ER−.
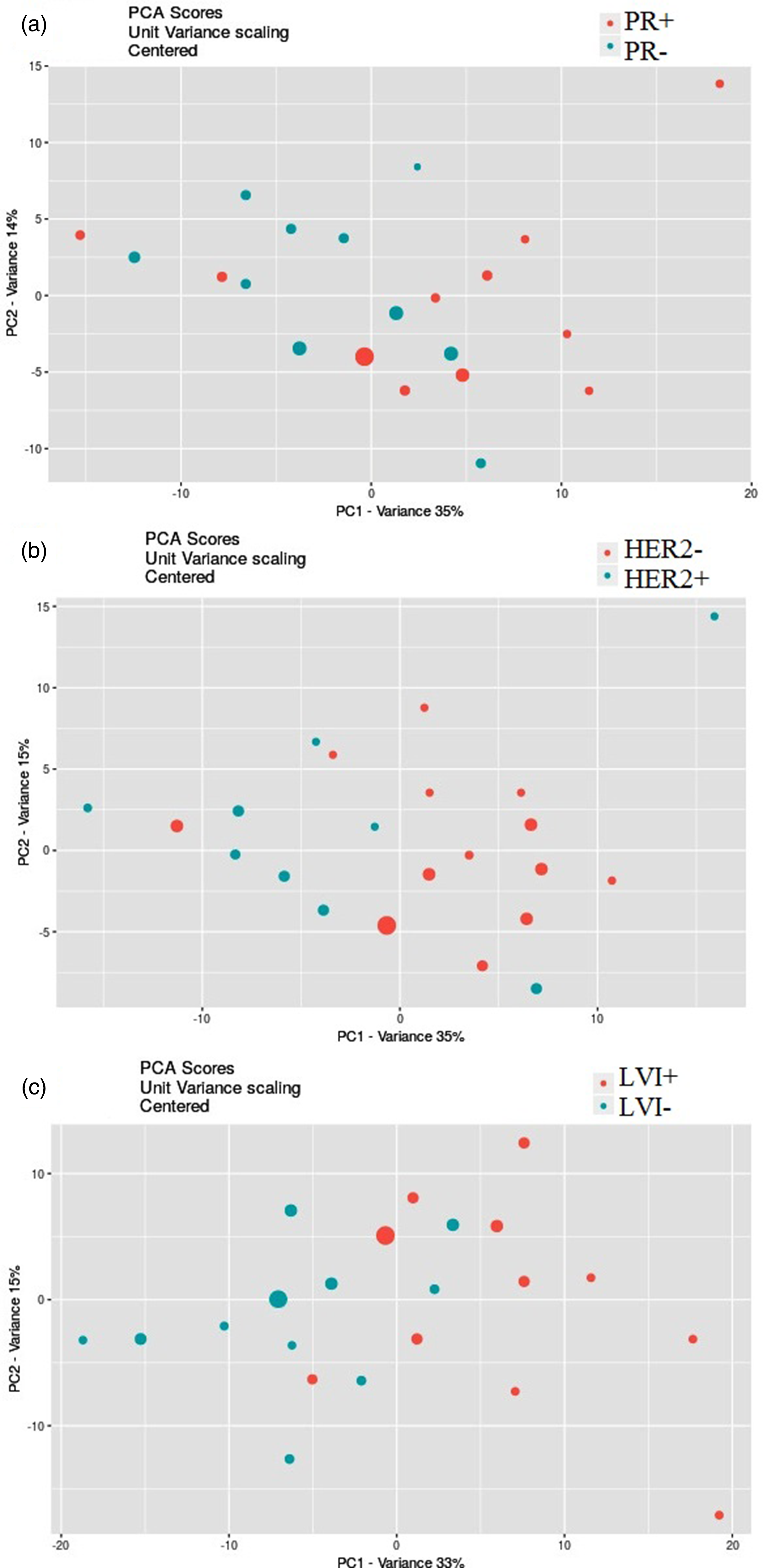
PCA scores plots produced by online XCMS for the separation in datasets: (a) PR + vs. PR−; (b) HER2 + vs. HER2−, (c) LVI + vs. LVI−.
The significantly altered metabolites between IDC and control groups.
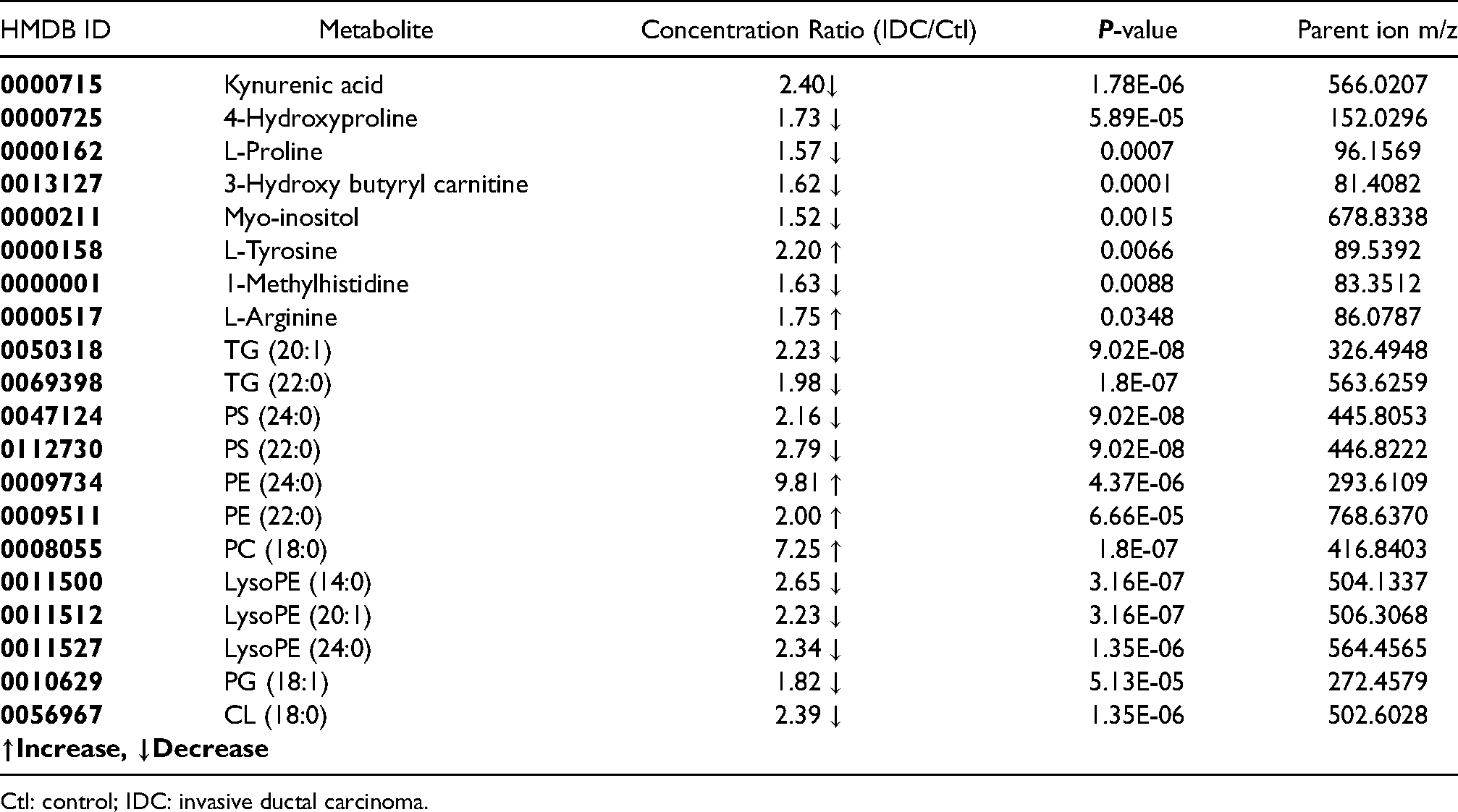
Ctl: control; IDC: invasive ductal carcinoma.
Discussion
This study was conceived with a rationale of identification of significant alteration of metabolites, which could be established as a potential biosignature of IDC compared to healthy controls. Experimentally, we analyzed and identified metabolites in the serum samples of patients with IDC that showed statistically significant differential expression compared to the healthy control group. We used the LC-MS/MS technique to explore potential serum biomarkers for early detection of IDC. Significant differential metabolites were also explored between the ER+/ER−, PR+/PR−, HER2+/HER2− and LVI+/LVI− groups as prognostic biomarkers. Comparing IDC and control groups, we found 20 significant altered metabolites between serum samples from IDC versus the control group. These metabolites were mapped to several important pathways. The most important pathways observed to be altered were arginine and proline metabolism, glycerophospholipid metabolism, and phenylalanine, tyrosine, and tryptophan biosynthesis pathways (Supplementary Figure 1). The proline metabolism plays a main role in metabolic pathway reprogramming in cancer and in related areas such as aging. The proline metabolic axis is an important position in the integration of several regulatory mechanisms and could be a novel target for adjunctive cancer therapy. 22 In addition, lipids are known as important components of cell structure and play critical roles in signal transduction and metabolism. With the introduction and development of lipidomics techniques in recent years, several investigations have shown that there is an increasing connection between lipids and different aspects of cancer. 23
Kynurenic acid (KYNA) and 4-hydroxyproline were among the most significantly altered metabolites in the IDC group compared with healthy controls. KYNA is an endogenous tryptophan metabolite. Recent studies reported the potential role of KYNA in carcinogenesis and cancer therapy. In our analysis, KYNA was reduced by 2.4-fold in the cancer samples compared to the control. Several studies indicated alterations of the KYNA level in various types of cancers and physiological conditions. Inhibition of the mitogen activated protein kinase (MAPK) pathway proteins has been raised as one of the possible anti-tumor functions of KYNA. 24 It is shown that KYNA induces cell-cycle arrest through up-regulation of the p21 Waf1/Cip expression. 25 Therefore, the presence of KYNA in the microenvironment of tumors may be helpful in tumor growth restriction. The 4-hydroxyproline and Lprolinewere other altered metabolites, which decreased by 1.73- and 1.57-fold in cancer samples compared tohealthy controls, respectively. Proline can contribute to the TCA cycle through the urea cycle and participates in reactive oxygen species (ROS) formation through oxidation by proline dehydrogenase. 22 Moreover, L-proline plays vital roles in various cellular pathways including cellular bioenergetics, cell signaling pathways, stress protection, apoptosis, and cancer cell metabolism. L-proline is also the most abundant amino acid participating in the composition of the extracellular matrix (ECM). After incorporating L-proline into collagen structure, it is hydroxylated into 4-hydroxyproline by prolyl hydroxylase enzyme. 26 Collagen is the main component of the tumor microenvironment and contributes to cancer fibrosis. Several preclinical and clinical studies have suggested collagen to be a potential prognostic biomarker associated with cancer development, cancer differentiation, cancer invasion, cancer cell metastasis and proliferation, and clinical stage of cancer patients. 27 On the other hand, elevated breast density related to collagen I, correlated to a 4- to 6-fold increased risk of breast cancer development. Collagen regulates cancer cell behavior and its progression through binding to integrin. 28 In this regard, COLXIII in breast cancer induces β1 integrin to elevate cancer invasion and drug resistance. 29 Consistent with our results, other metabolomics-based studies reported L-proline as a prognostic marker correlated with a 5-year survival rate in breast cancer patients. 30
Our data showed that other amino acids, including L-arginine and L-tyrosine, were also altered between the cancer and the control groups. Lécuyer et al. 31 reported that women characterized by higher fasting plasma levels of arginine had a higher risk of developing breast cancer. An increased arginine level was also observed in the study of Morad et al. 32 in plasma samples of breast cancer patients. They also observed alterations of proline and carnitine similar to our results. L-arginine is a semi-essential amino acid that plays a main role in cell division, immune system, and wound healing. In addition, it has a main role in breast cancer cell growth and is reported to be depleted in breast cancer patients. 33 This suggests that decreased arginine level is a special feature of the cancer development. In agreement with the result of the current study, Pietzke et al. 34 reported a significantly higher level of arginine in breast cancer as compared with all the other studied groups. Several studies in this regard detected altered arginine (up and down regulation) related to breast cancer in different bio-fluids such as serum, saliva, and plasma. 35 L-tyrosine was one of the disturbed metabolites in cancer samples compared with that in controls in this study. Numerous metabolomics studies have also reported changes in tyrosine levels between cancer patients and controls in a variety of cancers. 36 In the case of breast cancer, based on clinical metabolomics studies, tyrosine along with alanine were the highest changed metabolites, indicating that tyrosine might be an important metabolite in the diagnosis of breast cancer. Although the alteration trend of tyrosine among several investigations was contradictory, previous studies have established that tyrosine deficiency could lead to breast cancer cell growth arrest. 37 In a study performed by Suman et al., 38 tyrosine was detected as a significantly altered metabolite in early breast cancer subjects. At the same time, tyrosine was found to be higher in the breast cancer group compared with healthy controls, 39 which is in agreement with the results obtained in this study. We found 1-methylhistidine as one of the decreased identified metabolites in IDC compared with healthy controls. Histidine is a semi-essential amino acid required for growth and tissue repair in humans; it is also needed for blood cell manufacture and protects tissues against injury caused by radiation and heavy metals. In another metabolomics study based on the 1H-NMR method, serum samples from breast cancer patients were characterized by decreased concentration of histidine relative to healthy controls which is similar to the result of the present study.
Previous studies demonstrated that fatty acids and lipid levels were increased in breast cancer patients compared to healthy controls. 40 We observed an increased level of 3-hydroxybutyrylcarnitine. It is believed that there are more than 1000 types of acylcarnitine in the human body. The general function of acylcarnitine is to transport acyl-groups (organic acids and fatty acids) from the cytoplasm into the mitochondria so that they contribute to energy production. Yuan et al. 41 performed a plasma-based metabolomics study for early detection of primary breast cancer, which reported increased concentrations of the acetylcarnitine C2, the medium-chain acylcarnitines C10, and the long-chain acylcarnitines C14:1, C14:2, C16, C16:1, and C18:1 in primary breast cancer patient's plasma compared to those in healthy controls. These results indicate that cancer cells have surplus levels of acetyl-CoA that are converted to C2 by the carnitine O-acetyltransferase. 42 We observed a decreased amount of myo-inositol in IDC compared with control subjects. Myo-inositol is a carbocyclic sugar that belongs to the inositol family. 43 It is the precursor of inositol triphosphate that acts as an intracellular mediator and regulates a number of hormones such as thyroid-stimulating hormone, follicle-stimulating hormone, and insulin. 44 Previously, the lipidomic profile of different cell lines of breast cancer was compared with normal cells obtained from non-cancerous tissues by LC-MS/MS and GC-MS methods, which showed that alterations detected in breast tumor samples were mostly caused by the difference in lipid profiles of tumor cells. Martineau et al. 45 discovered the absolute content of various metabolites such as myo-inositol which revealed its usefulness as a potential biomarker. In addition, in line with our result, NMR-based metabolomics study of human breast cancer samples by Gribbestad et al. 46 reported lower levels of inositol for tumors compared to non-involved breast tissue. Recently, More et al. 19 also reported inositol as a tissue metabolite differentiating IDC from control subjects, by using a targeted liquid chromatography multiple reaction monitoring mass spectrometry (LC-MRM/MS) technique. Several lipids such as TG, PS, PE, PC, and PG also changed between the studied groups. According to previous studies, fatty acids support tumorigenesis and cancer progression. 47 This widely amplifies that fatty acids are important for cancer cells because they maintain membrane biosynthesis during hyper-proliferation and provide a critical energy source during metabolic stress. In the recent study of Diaz-Beltran et al. 48 alterations of LysoPC and LysoPE were reported in various breast cancer subtypes, which confirms our results. Recent studies have also presented that several lipid metabolic pathways are changed in cancer cells, and reprogramming in fatty acid metabolism plays a vital role in providing energy during cancer development. 49 In this study, we also found altered metabolites from the comparison of metabolite profiles between ER+/ER−, PR+/PR−, HER2+/HER2− and LVI+/LVI−, which may act as a potential biomarker panel for diagnosis, prognosis, and treatment processes in IDC disease. However, more investigations are needed to validate and use these metabolites as biomarkers. This study demonstrated the successful application of MS-based metabolomics for the studying of the metabolic changes in serum of patients with invasive ductal breast cancer. Our results indicated significant dysregulation of metabolic pathways in breast cancer patients. It was observed that the metabolite profile of patients with breast cancer was related to the reprogramming of amino acids and lipid metabolism, mainly arginine and proline metabolism. We conclude that metabolomics investigations from breast cancer patients might be useful to identify diagnostic and prognosis biomarkers, and for monitoring treatment. However, there are some limitations in our study: (a) the small sample size; although the multivariate statistical analysis compensate this shortcoming, larger sample sizes will be required to validate the results and to determine the actual metabolic differences between the different grades and stages of ductal breast carcinoma patients; (b) the lack of tumor tissue specimens for analysis with blood samples; however, the metabolomic study of body fluids—especially blood and urine that are collected through less invasive methods—has priority over the tissues. As blood serum contains the accumulation of metabolites from all body compartments, it seems difficult to find a specific blood metabolic pattern for a disease; however, metabolomics analytical methods including multivariate statistical analysis make it possible to effectively discriminate various study groups, as many studies are performed in this field, 50 and several potential biomarkers have been proposed through genomics, proteomics, and metabolomics studies.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155221123343 - Supplemental material for Metabolomic study of serum in patients with invasive ductal breast carcinoma with LC-MS/MS approach
Supplemental material, sj-docx-1-jbm-10.1177_03936155221123343 for Metabolomic study of serum in patients with invasive ductal breast carcinoma with LC-MS/MS approach by Nasrin Amiri-Dashatan, Reyhaneh Farrokhi Yekta, Mehdi Koushki, Afsaneh Arefi Oskouie, Hadi Esfahani, Salman Taheri and Elham Kazemian in The International Journal of Biological Markers
Supplemental Material
sj-docx-2-jbm-10.1177_03936155221123343 - Supplemental material for Metabolomic study of serum in patients with invasive ductal breast carcinoma with LC-MS/MS approach
Supplemental material, sj-docx-2-jbm-10.1177_03936155221123343 for Metabolomic study of serum in patients with invasive ductal breast carcinoma with LC-MS/MS approach by Nasrin Amiri-Dashatan, Reyhaneh Farrokhi Yekta, Mehdi Koushki, Afsaneh Arefi Oskouie, Hadi Esfahani, Salman Taheri and Elham Kazemian in The International Journal of Biological Markers
Supplemental Material
sj-docx-3-jbm-10.1177_03936155221123343 - Supplemental material for Metabolomic study of serum in patients with invasive ductal breast carcinoma with LC-MS/MS approach
Supplemental material, sj-docx-3-jbm-10.1177_03936155221123343 for Metabolomic study of serum in patients with invasive ductal breast carcinoma with LC-MS/MS approach by Nasrin Amiri-Dashatan, Reyhaneh Farrokhi Yekta, Mehdi Koushki, Afsaneh Arefi Oskouie, Hadi Esfahani, Salman Taheri and Elham Kazemian in The International Journal of Biological Markers
Supplemental Material
sj-docx-4-jbm-10.1177_03936155221123343 - Supplemental material for Metabolomic study of serum in patients with invasive ductal breast carcinoma with LC-MS/MS approach
Supplemental material, sj-docx-4-jbm-10.1177_03936155221123343 for Metabolomic study of serum in patients with invasive ductal breast carcinoma with LC-MS/MS approach by Nasrin Amiri-Dashatan, Reyhaneh Farrokhi Yekta, Mehdi Koushki, Afsaneh Arefi Oskouie, Hadi Esfahani, Salman Taheri and Elham Kazemian in The International Journal of Biological Markers
Supplemental Material
sj-docx-5-jbm-10.1177_03936155221123343 - Supplemental material for Metabolomic study of serum in patients with invasive ductal breast carcinoma with LC-MS/MS approach
Supplemental material, sj-docx-5-jbm-10.1177_03936155221123343 for Metabolomic study of serum in patients with invasive ductal breast carcinoma with LC-MS/MS approach by Nasrin Amiri-Dashatan, Reyhaneh Farrokhi Yekta, Mehdi Koushki, Afsaneh Arefi Oskouie, Hadi Esfahani, Salman Taheri and Elham Kazemian in The International Journal of Biological Markers
Supplemental Material
sj-docx-6-jbm-10.1177_03936155221123343 - Supplemental material for Metabolomic study of serum in patients with invasive ductal breast carcinoma with LC-MS/MS approach
Supplemental material, sj-docx-6-jbm-10.1177_03936155221123343 for Metabolomic study of serum in patients with invasive ductal breast carcinoma with LC-MS/MS approach by Nasrin Amiri-Dashatan, Reyhaneh Farrokhi Yekta, Mehdi Koushki, Afsaneh Arefi Oskouie, Hadi Esfahani, Salman Taheri and Elham Kazemian in The International Journal of Biological Markers
Supplemental Material
sj-png-7-jbm-10.1177_03936155221123343 - Supplemental material for Metabolomic study of serum in patients with invasive ductal breast carcinoma with LC-MS/MS approach
Supplemental material, sj-png-7-jbm-10.1177_03936155221123343 for Metabolomic study of serum in patients with invasive ductal breast carcinoma with LC-MS/MS approach by Nasrin Amiri-Dashatan, Reyhaneh Farrokhi Yekta, Mehdi Koushki, Afsaneh Arefi Oskouie, Hadi Esfahani, Salman Taheri and Elham Kazemian in The International Journal of Biological Markers
Supplemental Material
sj-jpg-8-jbm-10.1177_03936155221123343 - Supplemental material for Metabolomic study of serum in patients with invasive ductal breast carcinoma with LC-MS/MS approach
Supplemental material, sj-jpg-8-jbm-10.1177_03936155221123343 for Metabolomic study of serum in patients with invasive ductal breast carcinoma with LC-MS/MS approach by Nasrin Amiri-Dashatan, Reyhaneh Farrokhi Yekta, Mehdi Koushki, Afsaneh Arefi Oskouie, Hadi Esfahani, Salman Taheri and Elham Kazemian in The International Journal of Biological Markers
Supplemental Material
sj-jpg-9-jbm-10.1177_03936155221123343 - Supplemental material for Metabolomic study of serum in patients with invasive ductal breast carcinoma with LC-MS/MS approach
Supplemental material, sj-jpg-9-jbm-10.1177_03936155221123343 for Metabolomic study of serum in patients with invasive ductal breast carcinoma with LC-MS/MS approach by Nasrin Amiri-Dashatan, Reyhaneh Farrokhi Yekta, Mehdi Koushki, Afsaneh Arefi Oskouie, Hadi Esfahani, Salman Taheri and Elham Kazemian in The International Journal of Biological Markers
Supplemental Material
sj-jpg-10-jbm-10.1177_03936155221123343 - Supplemental material for Metabolomic study of serum in patients with invasive ductal breast carcinoma with LC-MS/MS approach
Supplemental material, sj-jpg-10-jbm-10.1177_03936155221123343 for Metabolomic study of serum in patients with invasive ductal breast carcinoma with LC-MS/MS approach by Nasrin Amiri-Dashatan, Reyhaneh Farrokhi Yekta, Mehdi Koushki, Afsaneh Arefi Oskouie, Hadi Esfahani, Salman Taheri and Elham Kazemian in The International Journal of Biological Markers
Supplemental Material
sj-png-11-jbm-10.1177_03936155221123343 - Supplemental material for Metabolomic study of serum in patients with invasive ductal breast carcinoma with LC-MS/MS approach
Supplemental material, sj-png-11-jbm-10.1177_03936155221123343 for Metabolomic study of serum in patients with invasive ductal breast carcinoma with LC-MS/MS approach by Nasrin Amiri-Dashatan, Reyhaneh Farrokhi Yekta, Mehdi Koushki, Afsaneh Arefi Oskouie, Hadi Esfahani, Salman Taheri and Elham Kazemian in The International Journal of Biological Markers
Footnotes
Acknowledgements
The authors would like to thank Basic Sciences Department, Shahid Beheshti University of Medical Sciences, for their support.
Author contributions
NA-D, MK, and RY contributed to the conducted research, statistical analysis and wrote paper. HE, ST, and EK conducted research. AO designed the research and the data analysis, and had primary responsibility for the final content. All authors read, provided feedback, and approved the final manuscript.
Availability of data and materials
The data used to support the findings of this study are included within the article. Additional data or information can be requested by contacting the corresponding author.
Declaration of conflicting interests
The authors have indicated that they have no conflicts of interest regarding the content of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shahid Beheshti University of Medical Sciences, (grant number IR.SBMU.RETECH.REC.1397.821)
Patient consent statement
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
