Abstract
BACKGROUND:
The treatment strategies of astrocytoma have not changed considerably due to the restricted appreciation of its biology.
OBJECTIVES:
This study aimed to evaluate the expression of the stem cell-related proteins (CD133 and Sox2) and their prognostic value in astrocytic glioma.
METHODS:
The immunohistochemical expression of CD133 and Sox2 in 40 patients with an astrocytic glioma of different grades was studied. The recorded data on the overall survival (OS), progression-free survival (PFS) and the response to the therapeutic protocol were collected and lastly analyzed.
RESULTS:
CD133 expression was observed in 87.5% of the cases, while positive Sox2 expression was found in all the studied cases. There was a significant association of CD133 expression with the histological grade and the tumor size (
CONCLUSIONS
: Astrocytoma with CD133 and Sox2 overexpression had an unfavorable prognosis and poor clinical response to the current therapeutic protocol.
Introduction
Astrocytoma is the most prevalent intracranial tumor that accounts for about 75% of neuroepithelial tumors. They are classified according to the WHO scale into low-grade astrocytoma (GI-II), anaplastic astrocytoma (GIII), and glioblastoma multiforme (GIV) [1]. In Egypt, astrocytic glioma is the most common glial tumor (79.4% of all gliomas) [2].
Regardless of the recent developments, the majority of astrocytic glioma in remains resistant to therapy. Recent studies suggested that the tumor biology and the resistance to treatment are attributed to the presence of cancer stem cells (CSCs). CSCs are defined as a subpopulation of cancer cells that have the capacity for indefinite self-renewal and the generation of different types of progeny through differentiation [3].
The importance of CSCs in estimating the prognosis of patients with gliomas has been evaluated using several markers closely related to their presence [4], and the search for novel prognostic or predictive molecular parameters are still the primary target for the improvement of its clinical management [5].
CD133 is the marker of choice for identifying cancer stem cells in gliomas [6]. It plays a fundamental role in cancer progression by the maintenance of stem cell properties of cancer cells, such as their ability to self-renew. It has been supported that the expression of CD133 is controlled by epigenetic factors [7].
Sox2 is a transcription factor belonging to the sex-determining region Y-box family, which is expressed in a wide array of tissues and play important roles in the regulation of organ development, cell type specificity, and in the pluripotency maintenance of CSCs of self-renewal and differentiation [8, 9].
Sox2 overexpression has been reported in a growing list of tumors, including esophageal carcinoma, pancreatic carcinoma, lung cancer, breast cancer, ovarian carcinoma, hepatocellular carcinoma and head and neck cancers [1]. Particularly, the Sox2 expression is important for the maintenance and development of the CNS tumors and positively associated with the tumor grade of brain tumors [10, 11].
Therefore, the idea of the present study was to evaluate the expression level of these CSC-related proteins in different astrocytic glioma grades and to assess their predictive role regarding the survival and response to therapy.
Materials and methods
Patients and tumor specimens
We searched the database of the Pathology department and identified 40 Formalin-fixed, paraffin-embedded (FFPE) tissue specimens of primary intracranial astrocytoma. Surgical resection was done in the Neurosurgery department of the same institution, during the period from January 2013 to December 2015. The clinical data of the studied cases were collected from the patients’ medical files, including the age, sex, preoperative Karnofsky performance status (KPS) score, treatment strategies, follow-up data, postoperative survival data, and records of the response to therapy. We exclude patients that had a recurrent tumor, incomplete clinical information or uncertain follow-up data. The hematoxylin and eosin stained slides were reviewed and the cases were graded in accordance with the 2016 World Health Organization classification of tumors of the central nervous system [12]. The study was approved by the ethics committee of Zagazig University Hospitals.
According to the treatment protocol, patients with grade II astrocytoma get a three-dimensional conformal radiotherapy of 45–54 Gy in 1.8–2 Gy fractions. Grade-III astrocytoma received a three-dimensional conformal radiotherapy of 60 Gy in two Gy fractions, while GIV astrocytoma (GBM) received a chemo-radiotherapy regimen including temozolomide 75 mg/m
Follow-up of the patients was done by magnetic resonance image (MRI) every 3–4 months to detect the patients’ response. A complete response (CR) was defined as a complete disappearance of the tumor; partial response (PR) was
Immunohistochemistry
Immunohistochemical staining was carried out on a Dako autostainer universal staining system (Dako Denmark A/S, Glostrup, Denmark) by the avidin-biotin method. One paraffin-embedded block of astrocytoma tissue was selected from each case and cut into 4-
Assessment of the immunohistochemistry
Immunohistochemical staining for CD133 and Sox2 was scored for the tumor cells. The number of positively stained cells in ten representative microscopic fields was counted and the percentage of positive cells was calculated. The results were interpreted by two independent pathologists who were blind to the clinicopathological and follow up data.
A) Diffuse astrocytoma (GII) (H&E, 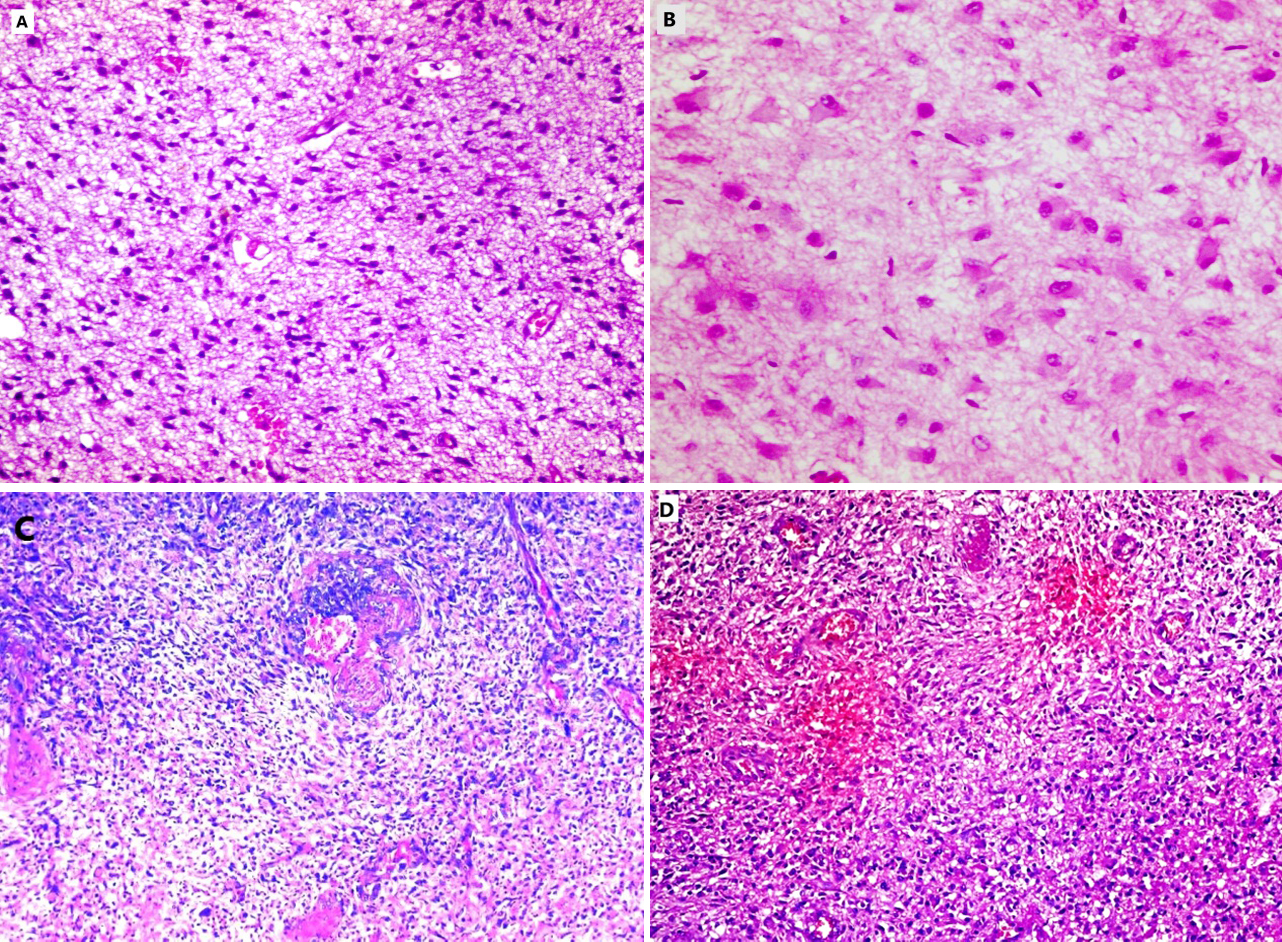
The cytoplasmic/or membranous staining of CD133 was considered as a positive expression. CD133 staining was semi-quantitatively graded for the percentage of tumor cells: negative
Sox2 scoring
We classified the percentage of Sox2 expression in tumor cells as low (
Statistics
Continuous variables were expressed as the mean
Clinicopathological features of 40 astrocytic glioma patients
Clinicopathological features of 40 astrocytic glioma patients
Patients’ characteristics
The present study included forty patients; 27 males and 13 females. The mean age of the patients at the initial diagnosis was 46.47
After the treatment, according to the protocol an overall response (OAR) of 40% of the studied cases was noted, while 60% exhibited no clinical response (NR). The median follow-up duration was 21.5 months (range 8–48 months), during which 17 patients (42.5%) died and 10 cases out of 24 cases (41.7%) that revealed no response had a progression. The clinicopathological features and the treatment regimen of our cases were presented in Table 1.
Continuous variables were expressed as mean
The association of CD133 and Sox2 expression with the clinicopathological features
The cytoplasmic/or membranous expression of CD133 was detected in 87.5% of the studied cases, where 60% revealed diffuse immunostaining. There was a significant up-regulation of CD133 expression with the higher astrocytoma grade (
The nuclear expression of Sox2 was observed in all the studied cases, where 42.5% revealed high expression. There was a significant up-regulation of Sox2 expression with the higher astrocytoma grade (
There was a significant association between SOX2 and CD133 expression (
Relations between clinicopathological features and CD133 expression
Relations between clinicopathological features and CD133 expression
Categorical variables were expressed as number (percentage); continuous variables were expressed as mean
CD133 cytoplasmic immunoexpression in astrocytic glioma cases; A) Focal CD133 immunoexpression in a case of diffuse astrocytoma (IHC, 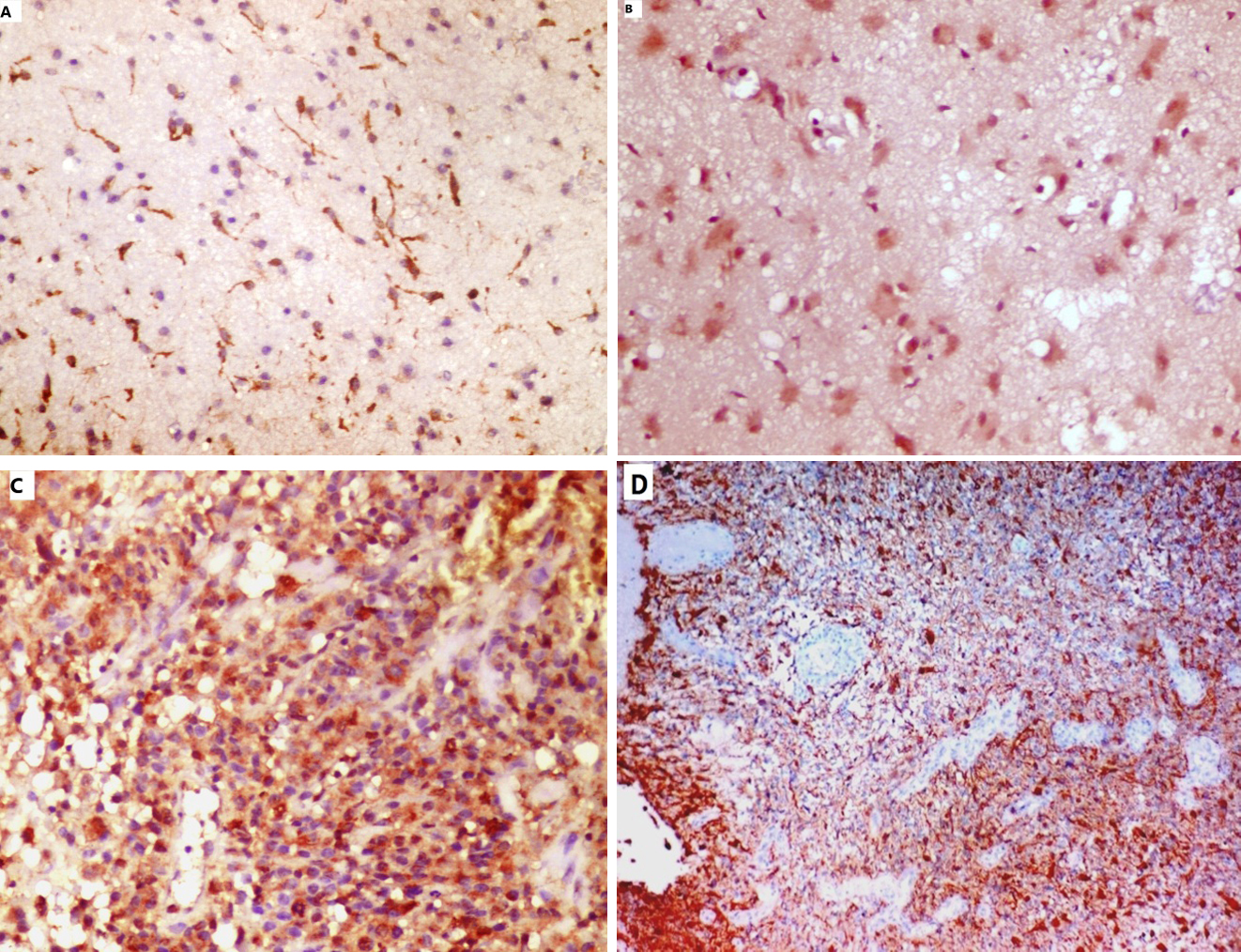
Relations between clinicopathological features and Sox2 expression
Categorical variables were expressed as number (percentage); continuous variables were expressed as mean
Sox2 nuclear immunoexpression in astrocytic glioma cases; A) Low Sox2 immunoexpression in a case of diffuse astrocytoma (IHC, 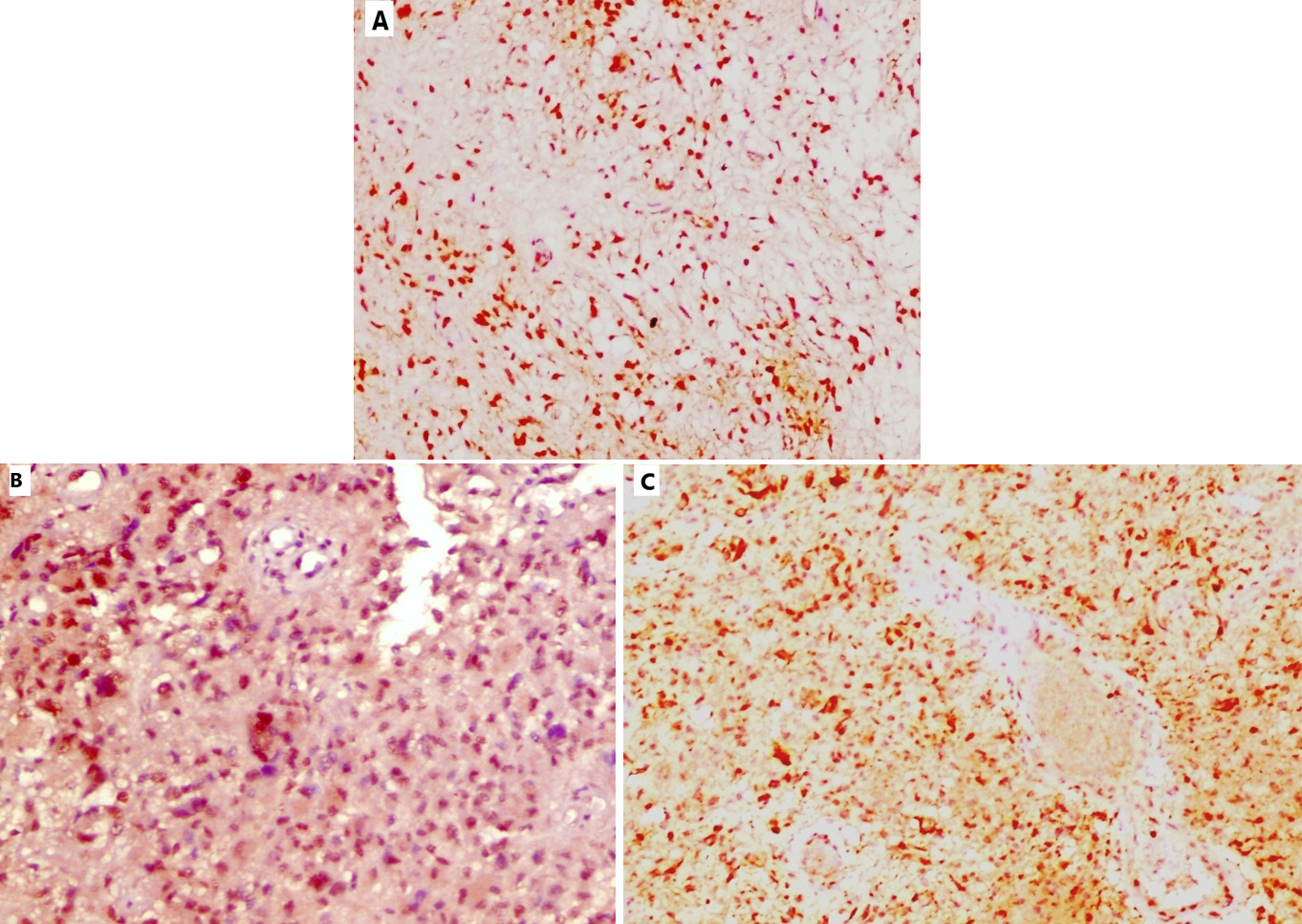
Relations between CD133 expression and treatment outcome
Categorical variables were expressed as number (percentage); NR: Not reach yet; § Chi-square test for trend; † Log-rank test;
Relations between Sox2 expression and treatment outcome
Categorical variables were expressed as number (percentage); NR: Not reach yet; § Chi-square test for trend; † Log-rank test;
Kaplan-Meier plot for Progression Free Survival (PFS); A) Stratified according to CD133 IHC staining (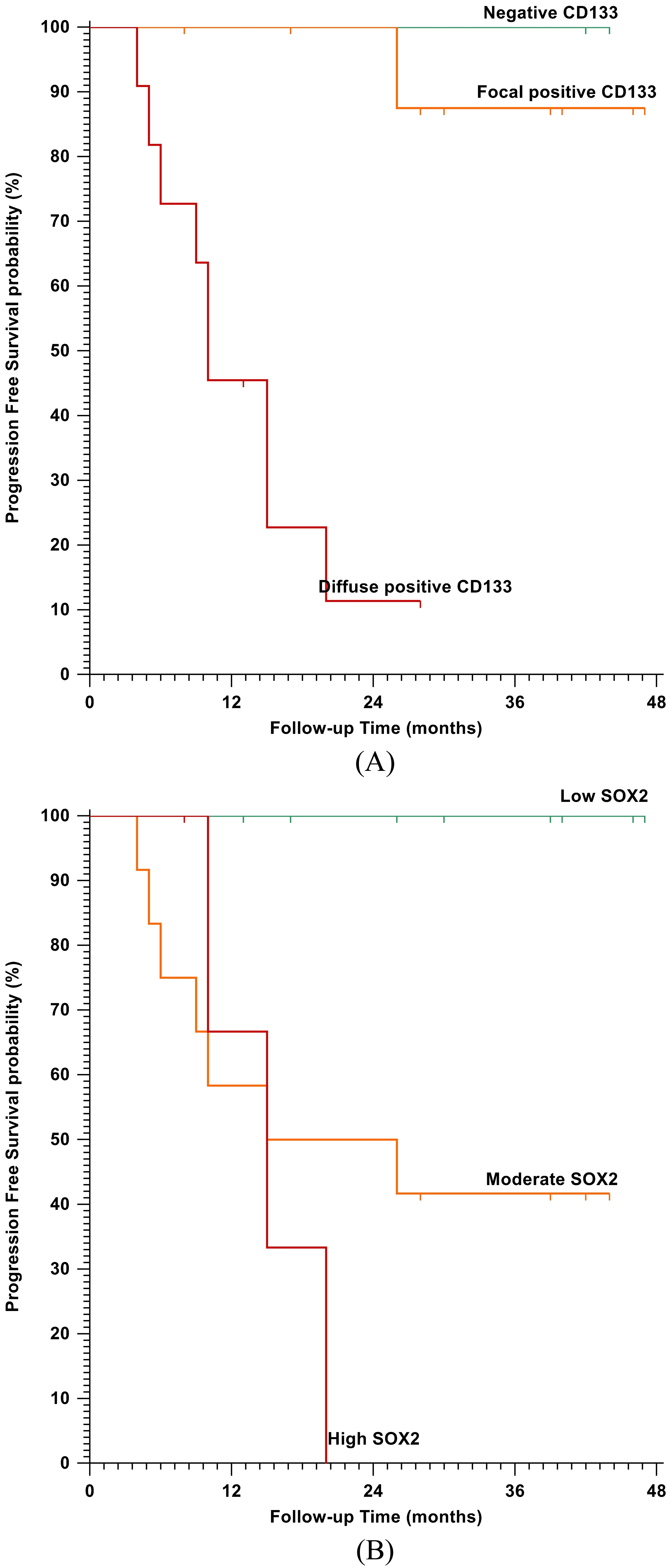
Kaplan-Meier plot for Overall Survival (OS): A) Stratified according to CD133 IHC staining (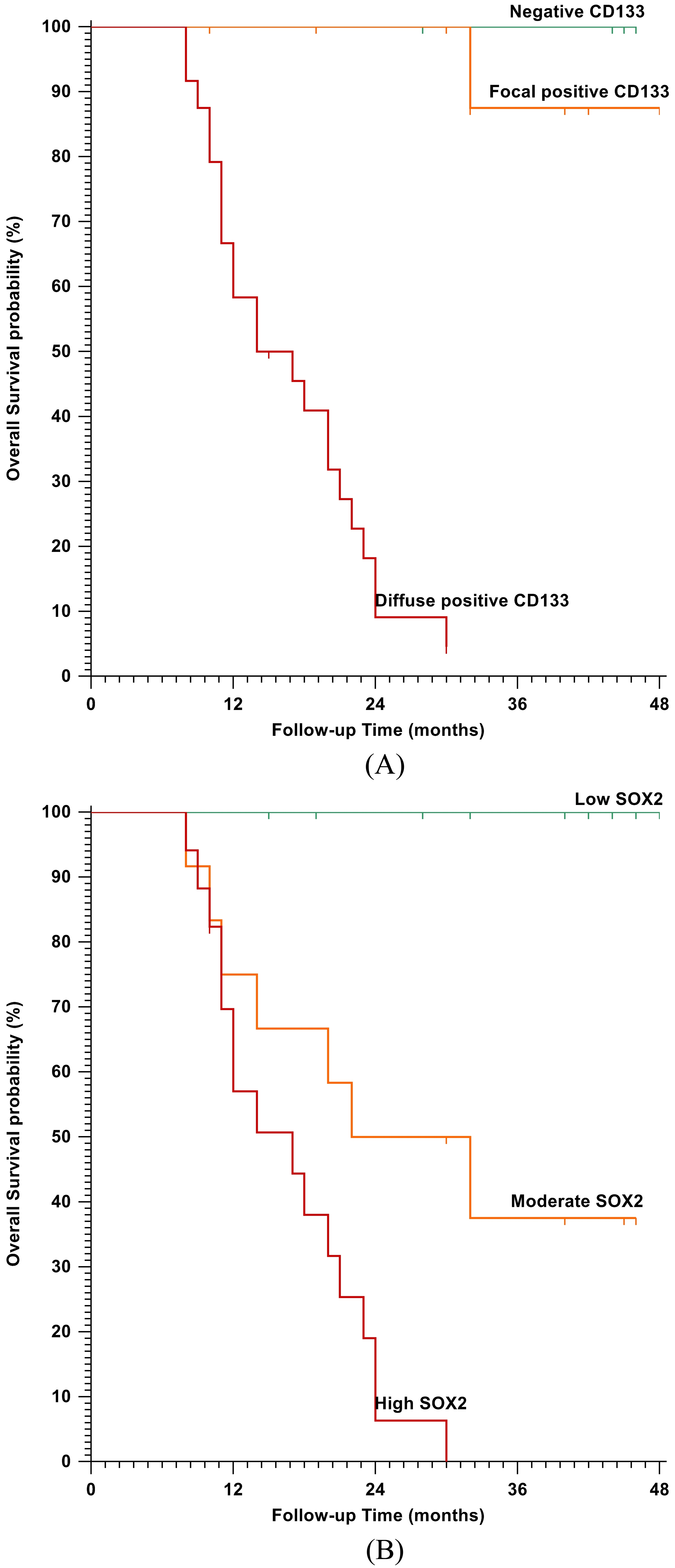
Regarding the response to the therapy, only 3 cases (7.5%) achieved a complete clinical response (CR), 13 cases (32.5%) had a partial response (PR) while the remaining cases exhibited no response (NR). CD133 up-regulation showed a significant association with the poor response to therapy (
CD133 overexpression was significantly associated with shorter OS (
Discussion
Glioma is the most common type of primary brain tumor. Among them, anaplastic astrocytoma and glioblastoma are highly malignant with unfavorable prognosis. Only 10% of high-grade tumors survive for more than five years despite the current advances in the research and therapy [15, 16]. Therefore, improved understanding of the tumor biology, the molecular basis of invasion and spread, and resistance to standard therapeutic modalities may aid to improve patients’ outcome and the prediction of the future recurrence [17].
The identification of stem cells in brain tumors provides a powerful tool to investigate the tumorigenic process in the central nervous system and to develop targeted therapies to these cells. However, the methods for the isolation/enrichment of CSCs remain still imperfect and require improvements, as a consequence of the lack of universal markers [18].
In the last years, the treatment approaches for astrocytic glioma have not changed markedly because of the limited understanding of the biology of the tumor. The poor prognosis of the patients is attributed to the presence of drug-resistant cells which are insensitive to the current strategies [19]. Cancer stem cells (CSCs) comprise a subpopulation of tumor cells with a self-renewal capacity and ability for tumor initiation. CSCs are also responsible for the tumor recurrence and resistance to the current therapeutic modalities. Therefore, the identification of these cells seems to be fundamental for understanding the process of carcinogenesis, and for the development of targeted therapies [20].
In our study, CD133 expression was noted in 87.5% of the studied cases. Previously [21, 22], CD133 expression was reported in 77.6 and 96.2%, respectively of their astrocytoma cases. These different results may be attributed to the different specificity and sensitivity of the commercially available antibodies used in these studies. It was reported that different CD133 antibody clones identify different epitopes [11]. In addition, we observed a significant up-regulation of CD133 expression with astrocytoma grade and the tumor size as reported before [23]. Therefore, we supposed that CD133 overexpression is a marker of aggressive astrocytic glioma and a crucial factor for tumor progression.
In the current work, a significant association was present between CD133 overexpression and poor OS and PFS, where the patient survival declines significantly in those with diffuse CD133 expression. These results go with a previous investigation [23], which reported that the patients whose tumors expressed a high-level of CD133 had a mean survival time of 25.7 months compared to 45.7 months for patients whose tumors expressed a low-level of CD133. Similarly, another study reported a significant association with poor overall survival (OS) (
Angelastro and Lamé supported the hypothesis that CD133 plays an antiapoptotic role in protecting CSCs from chemotherapeutic effect [29]. Moreover, CD133-positive glioma stem-like cells have the ability to survive high-dose irradiation leading to recurrent glioma [30]. Therefore, targeting CSCs could eradicate CNS tumors and protect against recurrent tumors. SirT1, which plays an important role in governing the radiosensitivity of glioma cells, is a downstream target of CD133 [31].
Our work reported a poor response to the therapy protocol in the cases that revealed diffuse CD133 expression, and this result matched with the previous reports [32, 33], which found that CD133
Another marker evaluated in the present study was Sox2, an established stem cell regulator highly expressed in multiple tissue stem cells, including various types of neural stem cells and progenitor cells [36]. In addition, Sox2 has been demonstrated to be a glioma-specific marker [4].
Detectable Sox2 expression was found in all cases of the studied astrocytoma with different intensity grades, suggesting a role for increased Sox2 expression in glioma development, which was in agreement with Annovazzi et al. study [37]. Schmitz et al. found that Sox2 is overexpressed in all the investigated malignant glioma ranging from 6 to 66% stained tumor cells [38].
Our results revealed a positive relation between Sox2 expression and malignancy grade in astrocytoma which was consistent with other investigators [14, 39]. Similarly, Annovazzi et al. reported that the percentage of intensely Sox2 stained nuclei in glioblastoma were higher than diffuse astrocytoma [37]. These findings suggest the substantial contribution of Sox2 expression in the tumor progression.
Knockdown experiments have indicated that Sox2 is required to sustain the aggressive growth and infiltrative behavior of GBMs [40, 41]. In particular, Gangemi et al. reported that Sox2-silenced glioblastoma tumor-initiating cells have been shown to inhibit proliferation and tumorigenicity in immune-deficient mice, indicating that Sox2 may be a target for glioblastoma therapy [40]. Furthermore, Alonso et al. showed that the knockdown of the Sox2 gene in the GBM cell line reduces cellular proliferation and colony formation [42]. Several strategies are starting to use Sox2 directly or indirectly to target GSCs [43]. Therefore, these data suggest that Sox2 is a key gene that maintains the stemness of glioma stem cells [44].
In contrast, Phi et al. concluded that the expression of Sox2 did not correlate with malignancy grade in astrocytic tumors [45]. This discrepancy may due to heterogeneous nature the astrocytic tumors. Brain tumors are heterogeneous with respect to genetic and histological properties of cells within the tumor tissue [46]. Sox2 is known to be involved in the development and progression of multiple types of tumor [47, 48, 49].
High levels of Sox2 have been associated with tumor aggressiveness and worse prognosis [50]. Wang et al. [39], verified that patients with positive Sox2 expression exhibited a significantly reduced survival rate, compared with those with negative Sox2 expression (
Regarding the response to therapy, our finding was similar to the preceding studies [43], where the cases with elevated Sox2 expression were more resistant to the therapy. However, Lemke et al. reported that the Sox2 positive fraction of glioma-initiating cell cultures was sensitive to irradiation [53]. The resistance to chemotherapy might be due to the prominent role of Sox2 in the regulation of GSCs [43]. Furthermore, Sox2 overexpression in cancer cells promoted evasion of apoptosis in tumor cells through antiapoptotic signaling [54]. Therefore, targeting its activity may offer an attractive therapeutic approach to treat glioblastoma.
The prior studies in ovarian cancer cell lines and breast cancer cell lines [55, 56], have shown that exogenous elevation of Sox2 can promote resistance to chemotherapeutics currently used. In addition, silencing Sox2 efficiently suppressed the expression of drug-resistance and anti-apoptotic genes and increased the sensitivity of the cells to radiation in oral squamous cell carcinoma [57].
Immunotherapy represents a promising treatment alternative to improve the clinical outcome of patients suffering from glioblastoma [43]. Schmitz et al. recognized Sox2 as a glioma-associated antigen abundantly and specifically overexpressed in glioma cells. In addition, they identified an immunogenic HLA-A*0201-restricted T-cell epitope derived from Sox2 that effectively activated tumor-directed cytotoxic T lymphocytes. Therefore, they suggested Sox2 as a promising novel strategy based on immunotherapy [38].
Interestingly, Song et al. found that Sox2 regulates tumor-initiating and drug-resistant properties in CD133-positive glioblastoma stem cells, suggesting that crosstalk between stem cell markers is required for CSC properties [58]. Although the high Sox2 level is associated with poor prognosis in many cancers including prostate [54], breast [56] and ovarian cancers [59], low Sox2 level in gastric cancer [60], and squamous cell lung cancer [61] was linked to poor prognosis. The reasons for the divergent results for Sox2 levels in different cancers remain to be investigated. Consequently, it is evident that there is a clear need for further investigations into the clinical implications of Sox2 expression, particularly how Sox2 levels influence tumor progression and patient survival.
In conclusion, we recognized that patients with CD133 and Sox2 overexpression had an unfavorable prognosis and poor clinical response to the current protocol of therapy suggesting the ineffectiveness of the current therapeutic modalities in these patients. Further investigations are needed to assess whether reversing the expression of these stem cell markers could be a novel therapeutic approach to prolong survival for this high-risk group of patients.
Footnotes
Conflict of interest
All of the authors declare that they have no conflict of interest regarding this paper.
