Abstract
Early diagnosing of skin cancer and investigation of metastatic potential of cancer cells are very important to treat it appropriately. Infrared spectroscopy of biological tissues is an emerging technique which gives the spectral differences between healthy and diseased cells. In this work, we have demonstrated that attenuated total reflectance Fourier transform (ATR-FTIR) spectroscopy can be used in diagnostic of skin cancer and in differentiating metastatic potential of cancer cells. Using IR spectroscopy, we can identify various types of cancer such as basal cell carcinoma, malignant melanoma, nevus and metastatic potential by alternations in hydration level and molecular changes. We examined biopsy of different types of cancer cells to diagnose skin cancer at early stages by using FTIR spectroscopy. To differentiate metastases we examined two human melanoma cells of same patient but at different metastatic potential and two murine melanoma cells with common genetic background but different metastatic potential.
Our findings revealed that melanoma changes the permeability of cell membrane and higher metastatic potential is related to the hydration level of cell membrane. Thus, ATR-FTIR spectroscopy is a potential technique to help in early diagnosing of skin cancer and to differentiate different metastatic potentials.
Introduction
Skin protects our body from unhealthy environmental effects. Ultraviolet radiations from sun and other ionizing radiations cause sun burns, melanoma, skin carcinoma and premature skin aging. Cases of cutaneous melanoma are increasing largely with every passing year and its worst condition metastatic cancer is a major cause of deaths. Cutaneous melanoma (MM), Basal cell carcinoma (BCC) and squamous cell skin (SCC) are the three major types of skin cancer [1, 2, 3, 4, 5, 6]. Cutaneous melanoma is directly related to the sun exposure and studies show that sun shield sites show aggressive behavior as compared to sun exposed areas. A low serological level of vitamin D is observed in sun shield sites as compared to non shield sites and this low level of vitamin D is related to the low percentage of BRAF mutation in melanoma patients [7]. BCC and MM skin cancers are mostly localized and probability of metastatic stage is rare. However, SCC cancer can spread all around developing a metastatic stage which becomes a cause of death. While treating a cancer patient, it is very important that metastatic potential of the tumor should be correctly identified. Usually morphological and lymph node states methods are used for the classification of tumor. But these methods are not reliable in all types of cancer such as breast and pancreatic tumors. Other methods for metastatic classifications are time taking and cost effective such as genetic test for tumor [8, 9, 10].
Hence, the objective of this research was to propose an easy and new diagnostic technique for cancer and different metastatic potentials by FTIR spectroscopy. This technique is also used to identify other kinds of melanoma and non melanoma skin cancer. Mid infra range spectroscopy is a very useful technique for biological molecules and their relation with the surroundings. This technique has potential to investigate differences in biological cells but water contents in biological samples limit the efficiency of mid-IR spectroscopy. This problem can be overcome by equipping FTIR spectrometer with attenuated Total Reflectance (ATR) element. Using this method, we can obtain spectra of live biological cells in solution [11, 12, 13, 14, 15]. The ATR infrared spectroscopy enables total internal reflection phenomenon and large number of internal reflections produce evanescent wave. If we place a sample in contact with ATR than that evanescent wave loses its energy at similar frequencies to that of sample’s absorbance. This technique enables the direct measurement of absorption spectra of biological samples placed in contact with ATR element [16]. Previous research showed that the higher movement of metastatic cells and the fluidity of the cell’s membrane; both can be linked with each other [17, 18, 19, 20, 21, 22].
Methodology
For the study of different types of skin cancer we took different samples from 6 patients with BCC on the head (6 specimens), 4 patients with MM on the back (8 specimens) and 2 patients with nevus (NEV) on neck (3 specimens). For different stages of cancer, B16-F1 and B16-F10 cells (grown in growth medium composed of DMEM (D5796, Sigma-Aldrich, St. Louis, MO, USA)) were produced from the murine melanoma B16 by injecting into C57 mice. B16-F1 cells produced as lung metastases of B16 cells. Another cell B16-F10 with different metastases was generated by re-injecting into mice for several rounds of selection. This selection method gave two cell lines with a genetic background in common but at different metastatic potential. B16-F10 cells are at higher metastatic potential as compared to B16-F1 cells. Similarly, two human melanoma cells are taken from primary tumor but different metastases of same patient. These cells were spherical in medium solution having medium ratio 0.99
FT-IR spectra of skin tissues: normal healthy tissue (N), basal cell carcinoma (BCC), melanoma (MM) and nevus (NEV), in the region 4000–400 cm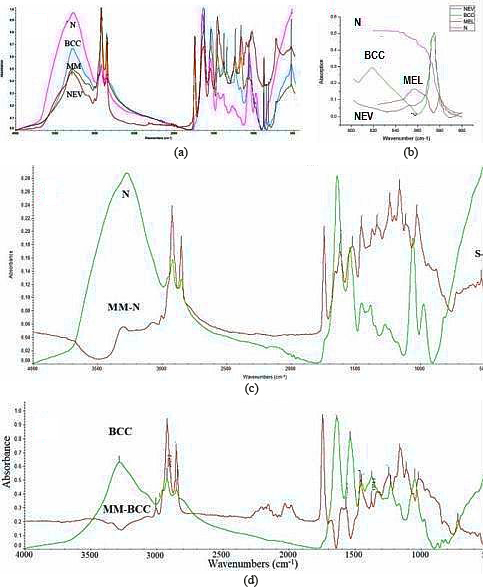
Early diagnosing of cancer
FTIR spectra of normal (N), NEV, BCC and MM biopsies are given in Fig. 1a. This spectrum gives intensities measurements and shifts in frequencies of normal and cancerous tissues in spectral range 4000–400 cm
From our results it is clear that diseases effects the structure of skin at molecular level. To understand the behavior and spectral difference in diseases we subtracted the spectra of normal tissue (N) from melanoma (MM) (Fig. 1c). In the subtracted the spectra we observed
The graph represents absorption versus wave number in the range of 1000–1700 cm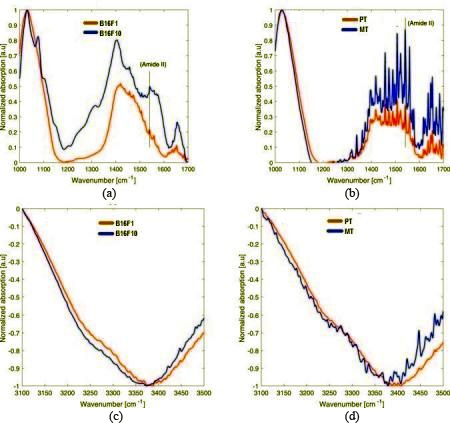
To analyze the ability of ATR-FTIR between the different metastatic potential of similar tumor we used a pair of human and mouse melanoma cells. The B16-F1 and B16-F10 cells from the same parental murine but at low and high metastatic potential respectively. Similarly, we investigated two biopsies of same patient such as PT (Primary tumor) and MT (metastatic Tumor), whereas PT is primary tumor and MT are at lower and higher metastatic potential respectively. For FTIR spectra, we used live cell solution and Germanium ATR element. The solution was homogenous, as the cells were hung in the medium. For background measurements, a small drop of solution was placed on Germanium ATR and measurements were made. That homogenous stage spectrum was subtracted from each measured spectrum of different cells. Metastatic cells are more mobile in the tissue and fluidity level in plasma membrane is higher in comparison with cells at less metastatic potential [17, 18, 19, 20, 21, 36]. Increase in fluidity level in plasma membrane cause an increase in hydration level [37, 38, 39]. So, metastatic of cancerous cell might be due to hydration levels and can be observed by absorption intensities of protein in different regions such as amide A, amide B, amide I, II, III and amide IV [40]. It is clear from previous study that the intensity of absorption peak in protein increases with the increase in hydration level [14]. It was assumed that protein of membrane show similar effects by an increase in hydration. Normalization of intensities of different peaks dominant in various experiments is a good way to compare the protein related absorption intensities. We normalized amide II peaks intensity of 1540 cm
A boxplot description of the distribution of values of ratio between the Gaussian centered around 3400 cm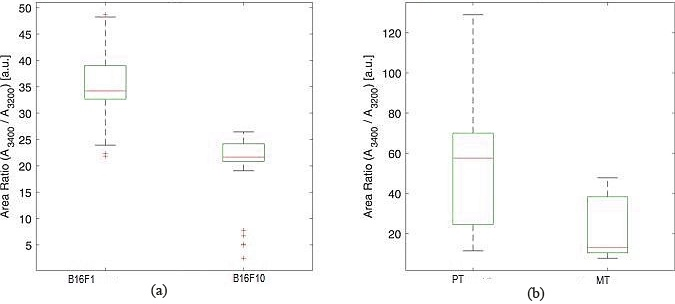
In this study, we describe a promising ATR-FTIR method to measure the spectra of biological cells. We have analyzed the hydration level of the plasma membrane by using two human melanoma cells and two variant of murine B16 to investigate their metastatic potential. We have analyzed two main evidences that show relation of metastatic potential with level of hydration of plasma membrane. In first observation, the higher level of absorption intensity in amide II region for higher metastatic potential show the higher hydration level of plasma membrane. Second evidence is that higher metastatic state had more HDW molecules in cell’s membrane. Both of our tests gave regular results that may allow us to identify metastatic potential of various cells by examining spectral differences and relate to previous literature. Ghimire et al. [45] differentiated metastases of prostate cancer in lymphoma spectra and melanoma serum of mouse model by using FTIR spectroscopy. The major differences found between the two serum samples were absorption values of protein, carbohydrates and nucleic acid. Wu et al. [46] used FTIR spectroscopy to classify the metastatic and non metastatic lymph nodes. Using FTIR spectroscopy along with Support vector machine they found accuracy, sensitivity and specificity 88.9%, 75.0% and 95.3% respectively. Hands et al. [47] used 1
In this research we found remarkable differences between ATR-FTIR spectra of cells at different metastatic potential which may lead to develop a clinical diagnostic tool by analyzing the metastatic biomarker such as hydration level and high density water in cell membrane. Clinical FTIR spectroscopy may provide an accurate and improved diagnostic method for cancer by replacing the difficult and expensive existing techniques.
The ATR-FTIR method is a potential tool to diagnose various types of cutaneous melanoma by spectral differences. This method can also be used to identify metastatic potential of cancer cells. The comparison of less metastatic and more metastatic cells showed that hydration level of plasma membrane is a major difference between both states of cancer. ATR-FTIR method is a useful technique to identify different type of cancer cells and their metastatic potentials.
