Abstract
BACKGROUND:
MicroRNA-34a (miR-34a), as a tumor-suppressive miRNA, has been found to induce cell apoptosis in acute myeloid leukemia (AML). However, the diagnostic and prognostic significance of miR-34a in AML remains largely unknown.
OBJECTIVE:
We aimed to explore its associations with clinical characteristics and prognosis of AML patients.
METHODS:
This study detected serum miR-34a level in 117 diagnosed AML patients and 60 control subjects by using qRT-PCR, and results were compared to clinical features and patient outcome. Since cytogenetically-normal AML (CN-AML) has a good uniformity of cytogenetics and provides a perfect platform for detection of AML biomarkers, we further analyzed miR-34a expression in 56 CN-AML subjects.
RESULTS:
We found that miR-34a was significantly downregulated in AML and CN-AML patients. MiR-34a underexpression was commonly observed in AML patients with intermediate/poor risk cytogenetic, and M5 subtype. ROC analysis demonstrated that serum miR-34a could well identify AML/CN-AML patients from healthy individuals. More importantly, miR-34a expression was found negatively correlated with aggressive clinical variable, and served as an independent prognostic indicator. In addition, AML/CN-AML patients with low miR-34a expression displayed shorter overall and recurrence free survival.
CONCLUSIONS:
Altogether, miR-34a might have an application as a diagnostic and prognostic indicator for AML patients.
Introduction
Acute myeloid leukemia (AML), a genetically and clinically heterogeneous disease, is one of the most frequent fatal malignancy worldwide [1, 2]. Cytogenetically-normal AML (CN-AML), which accounts for nearly half of AML patients, has a good uniformity of cytogenetics and provides a perfect platform for detection of AML biomarkers [3]. The survival rate in adult AML patients is age-dependent, and 90% patients older than 60 failed to reach long-term survival [4, 5]. Early diagnosis of AML can increase survival rate of patients with this disease. Thus, identification of biomarker highly specific to AML is desperately needed.
MicroRNAs (miRNAs) are small, non-coding RNAs that negatively regulate target genes by binding to their 3’-untranslated regions [6]. Different miRNAs can function as oncogenes or tumor suppressors, and their expression is associated with various biological processes, including cell growth, development, proliferation, differentiation and apoptosis [7, 8]. Previous reports have revealed that some miRNAs, such as miR-155, miR-210, miR-96, miR-328, contributed significantly to the initiation and progression of AML [9, 10, 11, 12].
MiR-34a belongs to the miR-34 family and located at chromosome 1p-36.23 [13]. Some previous studies has explored the role of miR-34a in AML. For instance, Liu et al. showed miR-34a was markedly reduced in leukemia cell lines. Moreover, enforced miR-34a expression significantly stimulated apoptosis and suppressed autophagy in AML cells by negatively regulating HMGB1 [14]. Similarly, Wang et al. provided in vitro evidence to confirm the anti-oncogenic role of miR-34a in AML immuno-based therapy, and PD-L1 expression was inversely correlated with miR-34a expression [15]. However, the clinical significance of miR-34a expression in AML has not yet been reported and remained poorly known. Here, we examined the expression of miR-34a in blood samples from all participants to evaluate its significance for AML.
Correlation between miR-34a expression and clinical parameters of 117 AML patients
Correlation between miR-34a expression and clinical parameters of 117 AML patients
PLT, platelet; WBC, white blood cells; BM, bone marrow; FAB, French-American-British classification.
Patients and samples
This study included 117 newly diagnosed AML cases, with classification of favorable-risk cytogenetic (21 subjects), intermediate-risk cytogenetic (59 subjects) and poor-risk cytogenetic (37 subjects). Of the 117 AML cases, 56 patients were classified as CN-AML. Additionally, 60 healthy volunteers without any type of malignancy or other benign disease were enrolled as controls. All study protocols were approved by the Ethics Committee Board of Zhangzhou Affiliated Hospital of Fujian Medical University and informed consent was obtained from each participant. Detailed clinical characteristics of the patients were presented in Table 1.
Up to 5 ml whole blood was withdrawn from the controls and AML subjects before receiving any therapy. All serum specimens were centrifuged at 3000 rpm for 5 min, then centrifuged at 12000 g for 5 min at 4
RNA extraction and quantitative reverse-transcription PCR (qRT-PCR)
RNA was isolated from the serum using the miRNeasy extraction kit (Qiagen, CA, USA). RNA sample concentration was quantified by NanoDrop ND-1000 (Thermo Fisher scientific, Waltham, MA, USA). Total RNA was reversely transcribed using the Prime-Script RT reagent kit (TaKaRa, Dalian, China). Quantitative RT-PCR was performed in an Applied Biosystems 7500 Detection System (Applied Biosystems, CA, USA). The relative expression of miR-34a was calculated by the comparative 2
Statistical analyses
The statistical analyses in this study were processed using GraphPad Prism 6.0 (GraphPad Software, San Diego, CA, USA) and Statistical Program for Social Sciences 18.0 software (SPSS, Inc., Chicago, IL, USA). Comparisons of the expression level of plasma miR-34a among groups were performed using Kruskal-Wallis test. X tile software (
(A) MiR-34a levels were significantly lower in AML and CN-AML patients than controls. (B) For all AML cases, miR-34a levels in favorable risk cytogenetic group were greatly elevated compared to intermediate or poor risk cytogenetic group. (C) MiR-34a levels expression significantly lower in AML patients with M5 subtype.
(A) ROC analysis using miR-34a for differentiating AML patients from controls. (B) ROC analysis using miR-34a for differentiating CN-AML patients from controls.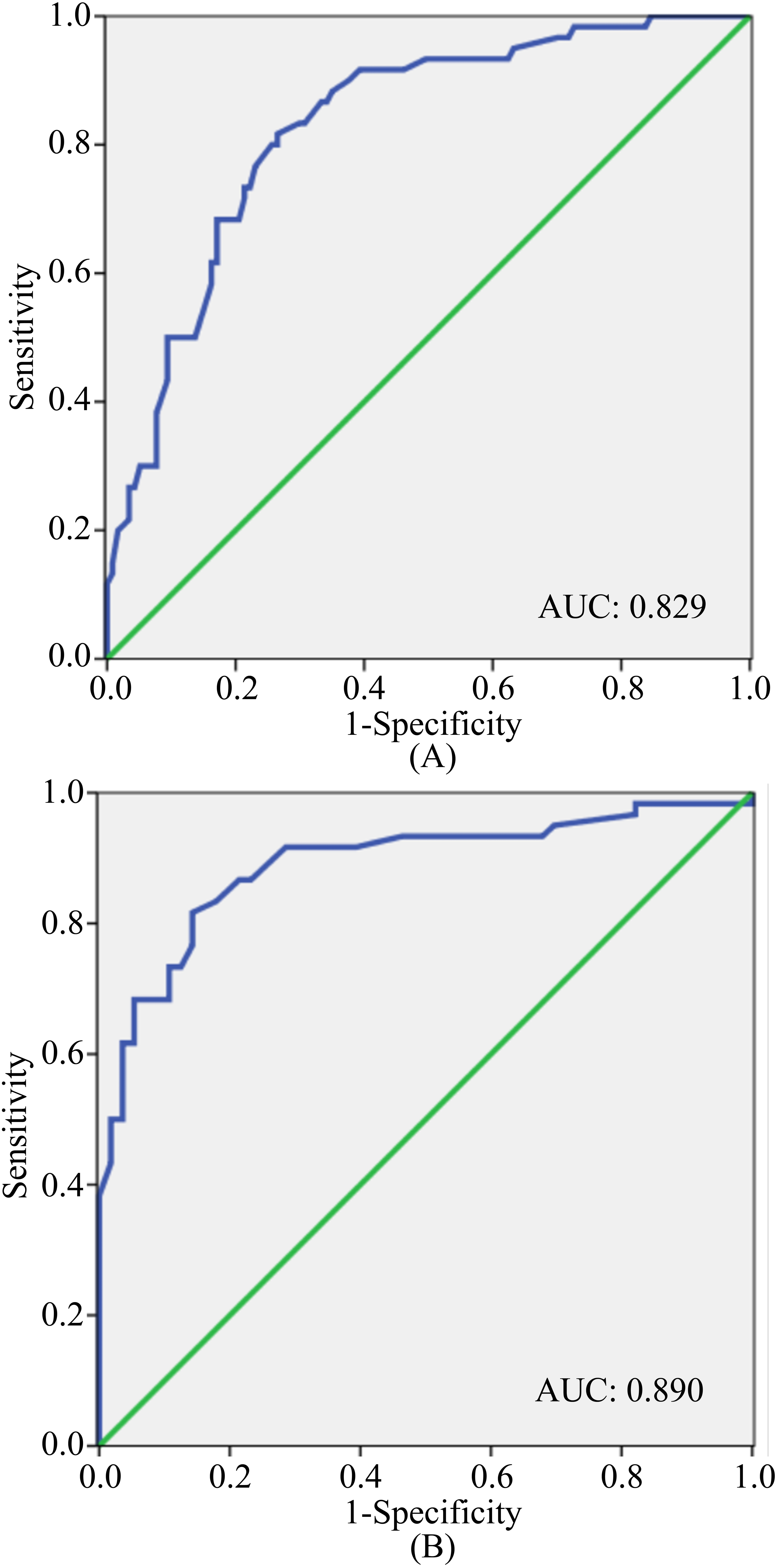
(A) AML subjects in high miR-34a expression group had longer OS. (B) AML subjects in high miR-34a expression group had longer RFS.
(A) CN-AML subjects in high miR-34a expression group had longer OS. (B) CN-AML subjects in high miR-34a expression group had longer RFS.
MiR-34a levels were greatly decreased in AML and CN-AML patients
MiR-34a expression was detected in serum samples by qRT-PCR. The data presented in Fig. 1A found miR-34a expression levels were highly downregulated in AML/CN-AML patients compared with those in healthy individuals (both
ROC curve analysis revealed serum miR-34a could distinguish AML patients from controls with AUC value of 0.829, and the specificity and sensitivity were 81.7% and 73.5%, respectively (Fig. 2A). Also, serum miR-34a could serve as a reliable biomarker for differentiating CN-AML subjects from controls with 81.7% specificity, 85.7% sensitivity, and an AUC value of 0.890 (Fig. 2B).
Association between miR-34a expression and clinical variables of AML
To evaluate the correlation between miR-34a expression and clinical variables, all AML patients were divided into high expression group (
Prognostic Value of miR-34a expression in AML and CN-AML
For the survival analysis, the AML patients in high miR-34a expression group had a significantly longer five-year OS (
Multivariable analysis of the indicators for OS and RFS in 117 AML patients
Multivariable analysis of the indicators for OS and RFS in 117 AML patients
In this study, we demonstrated that down-regulation of miR-34a was a common event in AML/CN-AML. In addition, miR-34a expression was markedly decreased in AML patients with intermediate/poor risk cytogenetic, and M5 subtype. Then, ROC analysis showed that serum miR-34a could well screen AML/ CN-AML patients from normal controls. Low miR-34a expression was strongly linked to aggressive clinical features, as well as inferior clinical outcome (OS/RFS). MiR-34a also confirmed to be an independent predictor of AML. Collectively, these results indicated that miR-34a might function as a tumor suppressor in AML. In agreement with previous data obtained in cell models, miR-34a was a direct target of p53, which is a known tumor suppressor. In addition, TP53 alterations were associated with repressed miR-34a expression in AML, indicating that loss of p53-miR-34a signaling pathway might be important of leukemogenesis [16]. MiR-34a could suppress the expression of B-Myb and E2F1, leading to cell cycle rest in malignant cells [17]. Interestingly, C/EBP
Besides AML, miR-34a acting as a tumor suppressor has also been extensively studied in various cancer types. For instance, Tang et al. discovered that increased miR-34a expression restrained the migration and invasion of pancreatic cancer cell lines in vitro and tumor growth in vivo by regulation of Snail1 and Notch1 [19]. MiR-34a was decreased in the cervical cancer tissue and its downregulation was closely associated with poor overall survival. Moreover, both Bcl2 and c-Met were identified as its regulatory target [20]. In a study carried out by Zhao et al., reduced miR-34a expression greatly promoted the migration and invasive ability of cancer cells by upregulating CD44 in osteosarcoma [21]. Likewise, another study revealed restoration of miR-34a expression inhibited the tumorigenic properties of osteosarcoma cells in vitro and the metastasis in vivo through targeting c-Met [22]. Wu and colleagues verified that miR-34a overexpression markedly suppressed multiple myeloma stem cell proliferation, colony formation and stimulated cell apoptosis in vitro, as well as inhibited carcinogenesis and lytic bone lesions in mice through negatively regulating TGIF2 expression [23]. In hepatocellular carcinoma (HCC), miR-34a levels were dramatically decreased in HCC tissues compared with normal tissues, and miR-34a downregulation was closely associated with shorter overall survival. Forced miR-34a expression inhibited the oncogenic activities and induced apoptosis of cancer cells [24, 25]. In breast cancer, loss of miR-34a was found both in cancer tissues and cell lines. In addition, elevated miR-34a significantly increased cell proliferation, invasion and migration via degrading Wnt1 [26], Bcl-2 and SIRT1 [27]. Hui et al. indicated that miR-34a was reduced in gastric cancer cell lines and tissues, and low miR-34a levels were positively correlated with worse prognosis of patients with gastric cancer [28]. Another study showed high miR-34a expression could decrease cell proliferation of gastric cancer, increase cell sensitivity to cisplatin, and vice versa. MET was the target gene of miR-34a [29]. In ovarian cancer, cancerous tissues possess greatly decreased levels of miR-34a in comparison with adjacent normal tissues. MiR-34a suppression stimulated cancer proliferation and motility through regulating AXL [30], and Snail [31]. Duan and colleagues found miR-34a levels were greatly downregulated in prostate cancer tissues, and its overexpression resulted in cell growth arrest and increased cell cycle arrest by targeting SIRT1 [32]. Shi et al. reported that miR-34a expression was reduced in esophageal cancer tissues, and its upregulation attenuated cancer cell proliferation and invasive capability in vitro [33]. In head and neck squamous cell carcinoma, Kumar et al. noted that miR-34a expression was significantly reduced both in tumors and cell lines, and in vitro and in vivo data showed ectopic expression of miR-34a strongly repressed the tumorigenicity by silencing survivin expression via E2F3a [34]. In colon cancer, miR-34a levels were significantly lower in cancer tissues and cell lines than normal tissues and cells, and its underexpression markedly promoted cell proliferation and migration via targeting Fra-1 [35], and PDGFRA [36]. In non-small cell lung cancer, Shi and colleagues reported miR-34a inhibition induced clonal and clonogenic properties of cancer cells and promoted tumor development in vivo [37]. In addition, enforced miR-34a expression was strongly correlated with advanced glioma grade, and high miR-34a expression was independent prognostic factor affecting the progression-free survival and overall survival of glioma patients [38].
Whereas, these results were not in line with recent findings of some reports. In papillary thyroid carcinoma (PTC), Ma et al. confirmed that miR-34a expression was increased in cancerous tissues. MiR-34a upregulation significantly enhanced cellular viability, colony formation, and suppressed cell apoptosis by directly targeting GAS1, while silencing of miR-34a showed opposite effects [39]. Similarly, miR-34a expression was dramatically elevated in early stage colorectal cancer (CRC) patients compared to healthy individuals. Moreover, miR-34a was highly expressed in adenoma patients compared to normal/polyp subjects. Additionally, ROC analysis demonstrated that miR-34a could discriminate CRC patients from normal/benign disease groups [40]. Therefore, miR-34a seems to play a key role in human malignancies with a cancer-type dependent manner.
Conclusion
Taken together, miR-34a expression is significantly decreased in AML patients. Low miR-34a expression is strongly associated with unfavorable clinical variables and poor prognosis. Thus, miR-34a might be used as a potential indicator for the prognosis of AML.
Footnotes
Conflict of interest
None.
