Abstract
BACKGROUND:
Sensitive and specific biomarkers of Pancreatic Ductal Adenocarcinoma (PDAC) are desperately needed to allow early diagnosis and improve patient’s survival. Ezrin autoantibodies were recently described as present in 93% of PDAC patients and 40% of healthy subjects who later developed PDAC. However, another prospective study failed to replicate these findings. Both studies were based on the use of a solid phase ELISA immunoassay.
OBJECTIVE:
We aimed at re-evaluating the usefulness of Ezrin autoantibodies as PDAC biomarkers using the Luciferase Immuno Precipitation System (LIPS), an alternative immunoassay format that found successful application for the measurement of autoantibodies against pancreatic autoantigens.
METHODS:
We produced a Nanoluciferase™ tagged Ezrin (NLuc-Ezrin). NLuc-Ezrin was then used as antigen in LIPS to test for Ezrin autoantibodies patients affected by PDAC (
RESULTS:
Overall, binding in liquid phase to Ezrin by serum antibodies was rare and low titer. Furthermore, we did not find statistically significant differences in the prevalence of Ezrin autoantibodies between patients affected by either PDAC or OPD compared to control.
CONCLUSIONS:
Our results do not confirm the usefulness of Ezrin autoAbs as biomarker of PDAC.
Keywords
Introduction
Pancreatic ductal adenocarcinoma (PDAC) constitutes roughly 90% of exocrine pancreas tumors [1, 2]. PDAC is mostly discovered when the patient presents with acute clinical manifestations, i.e. when the underlying disease process has reached a relatively advanced stage [3, 4]. As a consequence, while less frequent than several other solid tumors, PDAC is a leading cause of death due to cancer [5].
To improve patient’s survival, novel biomarkers associated with the earlier stages of PDAC are actively sought. Among the candidates under investigation, immunological signatures, like circulating autoantibodies (autoAbs), might provide such an early signal of pancreatic cancer. This concept is derived from the experience of other clinical settings, like that of paraneoplastic neurological syndromes (PNS). In PNS, autoimmune manifestations at the level of the central nervous system are associated with specific autoAbs, that can be indicative of the potential presence of either ovarian or small cell lung carcinoma [6, 7, 8].
In PDAC, the presence of autoAbs to tumor associated antigens (TAA) has been reported by several papers [9, 10, 11, 12, 13, 14, 15]. Recently, the presence of autoAbs to Ezrin, a structural component of the plasma membrane and cell cytoskeleton overexpressed in several kinds of cancers [16, 17, 18, 19, 20], was reported in the serum of both genetically engineered mice that spontaneously develop PDAC and in 93% of patients with advanced stages of PDAC [10]. In addition, Ezrin autoAbs were found also in 40% of those who went on to develop PDAC from a small cohort of subjects enrolled in the European Prospective Investigation into Cancer and Nutrition (EPIC) study [21], suggesting that Ezrin autoAbs might precede PDAC discovery by several years.
However, another group failed to be replicate these latter findings in a much larger cohort from the Southern Community Cohort Study (SCCS) study [11]. Both the original report and the follow-up study used a solid phase ELISA immunoassay to detect Ezrin autoAbs. In light of the sometimes problematic performance of ELISA for the measurement of autoAbs in other clinical settings [22, 23, 24, 25], we decided to validate this candidate autoAb PDAC biomarker by developing a novel test for Ezrin autoAbs. Our Ezrin autoAb test is based on the Luciferase Immuno Precipitation System (LIPS) [26], a liquid phase immunoassay format, that proved sensitive and specific for the measurement of diverse antibody responses, in particular those against pancreatic type 1 diabetes autoantigens [27, 28, 29, 30, 31].
Materials and methods
Patients
Serum samples were collected at the San Raffaele Scientific Institute from 40 patients with pancreatic ductal adenocarcinoma (PDAC), 50 patients with other pancreatic diseases (OPD), and 60 healthy control subjects (HC), whose demographics are shown in Table 1. The majority of the PDAC cohort consisted of stage IIA (
Demographics of subjects tested in the study
Demographics of subjects tested in the study
Clinical features of PDAC patients
The complete coding sequence of Ezrin (GenBank NP001104547) was amplified by PCR from human pancreatic islet cDNA using specific primers containing the EcoRI and Not I restriction sites, and cloned in frame downstream of a Nano-luciferase (NLuc) reporter (Promega, Madison WI, USA) into a modified pCMVTnT plasmid expression vector (Promega). All plasmid clones were verified by automated sequencing. The recombinant chimeric NLuc-Ezrin was expressed in Expi293F eukaryotic cells using the Expi293™ Expression System (Life Technologies, California, USA), according to the manufacturer’s instructions; 48 hours after transfection, we quantified antigen production in transfected cell lysates by measurement of the luciferase activity. Briefly, cells were lysed using Passive Lysis Buffer (Promega) and serial dilutions in PBS PH 7.4
Luciferase immune precipitation system (LIPS) assay for detection of Ezrin autoantibodies
We used NLuc-Ezrin as antigen in LIPS to test for the presence of anti-Ezrin autoantibodies. Briefly, for each LIPS test, 4
Statistical analysis
Differences in median autoantibody titers between cohorts were assessed by the Mann-Whitney test. The ability of the immunoassays to discriminate cases from controls was assessed using the Fisher exact test and the area under the curve of the receiver operator characteristics analysis (ROC-AUC). All analyses and graphs were made using the GraphPad Prism v5 software (GraphPad Software, Inc., La Jolla, CA, USA). For all tests the p-value
Immunoprecipitation of NLuc-Ezrin with monoclonal antibody. Panel A: bar plot of LIPS raw data after immunoprecipitation of recombinant luciferase tagged NLuc-Ezrin using the Ezrin specific EP886Y monoclonal antibody and a healthy human serum as negative control (Neg). Panel B: standard curve used to convert LIPS results in AU. Serial dilutions of the EP886Y specific monoclonal are shown on the graph together with the AU attributed to each point and the R square value of the regression line.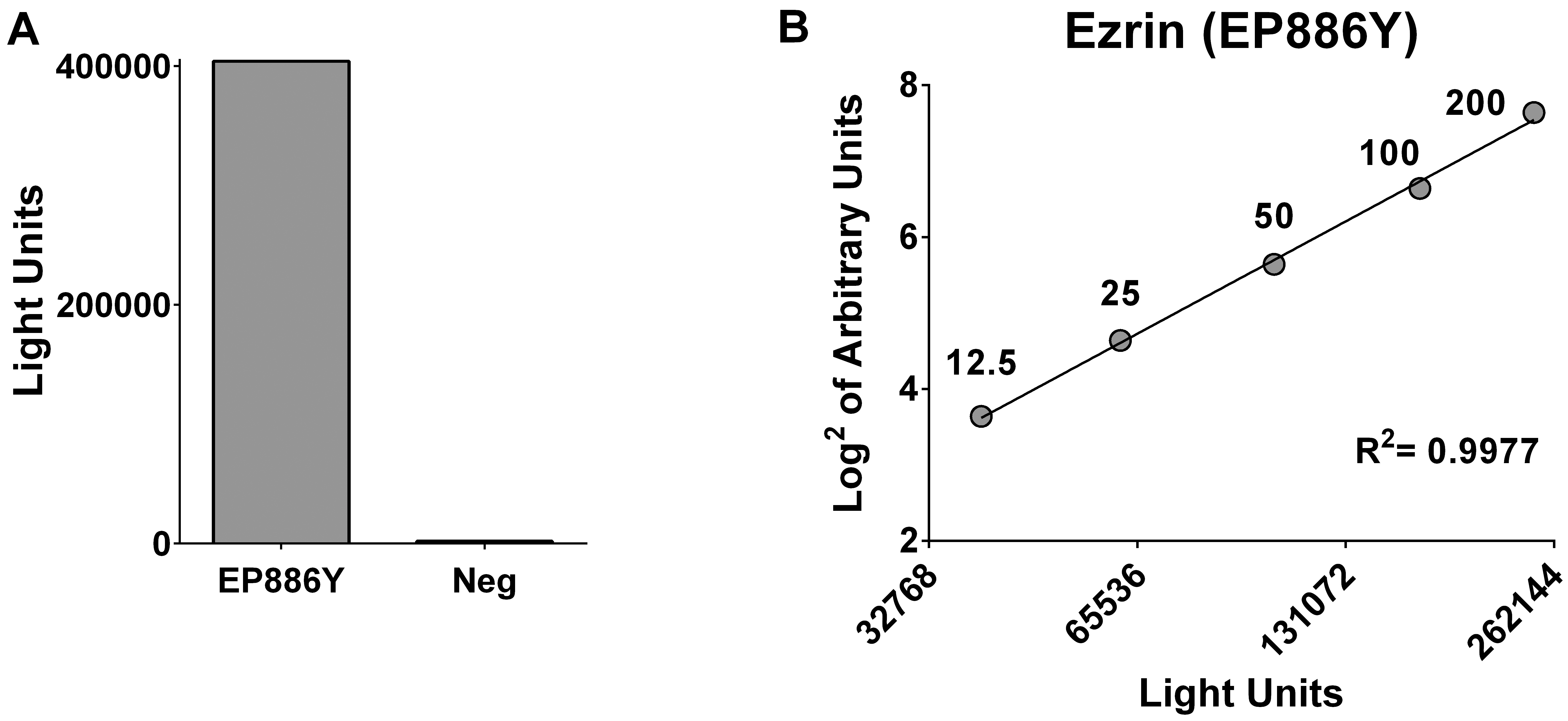
Immunoprecipitation of luciferase tagged Ezrin
We performed a preliminary evaluation of recombinant NLuc-Ezrin in LIPS using the rabbit monoclonal EP886Y, raised against a synthetic peptide corresponding to residues near the carboxyl-terminus of human Ezrin. NLuc-Ezrin was successfully immunoprecipitated by the EP886Y monoclonal antibody (Fig. 1A). Serial dilutions of EP886Y in normal human serum were subsequently used to establish a reference standard curve allowing the conversion of LIPS results into arbitrary units (Fig. 1B).
Anti-Ezrin autoantibody LIPS results. Panel A: scatter plot of Ezrin autoantibody LIPS results in patients with PDAC, OPD, and HC. The number of subjects in each group is indicated in parentheses. The dotted line indicates the mean plus 3 standard deviation of values recorded in the 60 HC. Panel B: ROC curve analyses of LIPS results for PDAC (left graph) or OPD (right graph) patients analyzed using the HC as control group. The dashed line indicates the line of identity.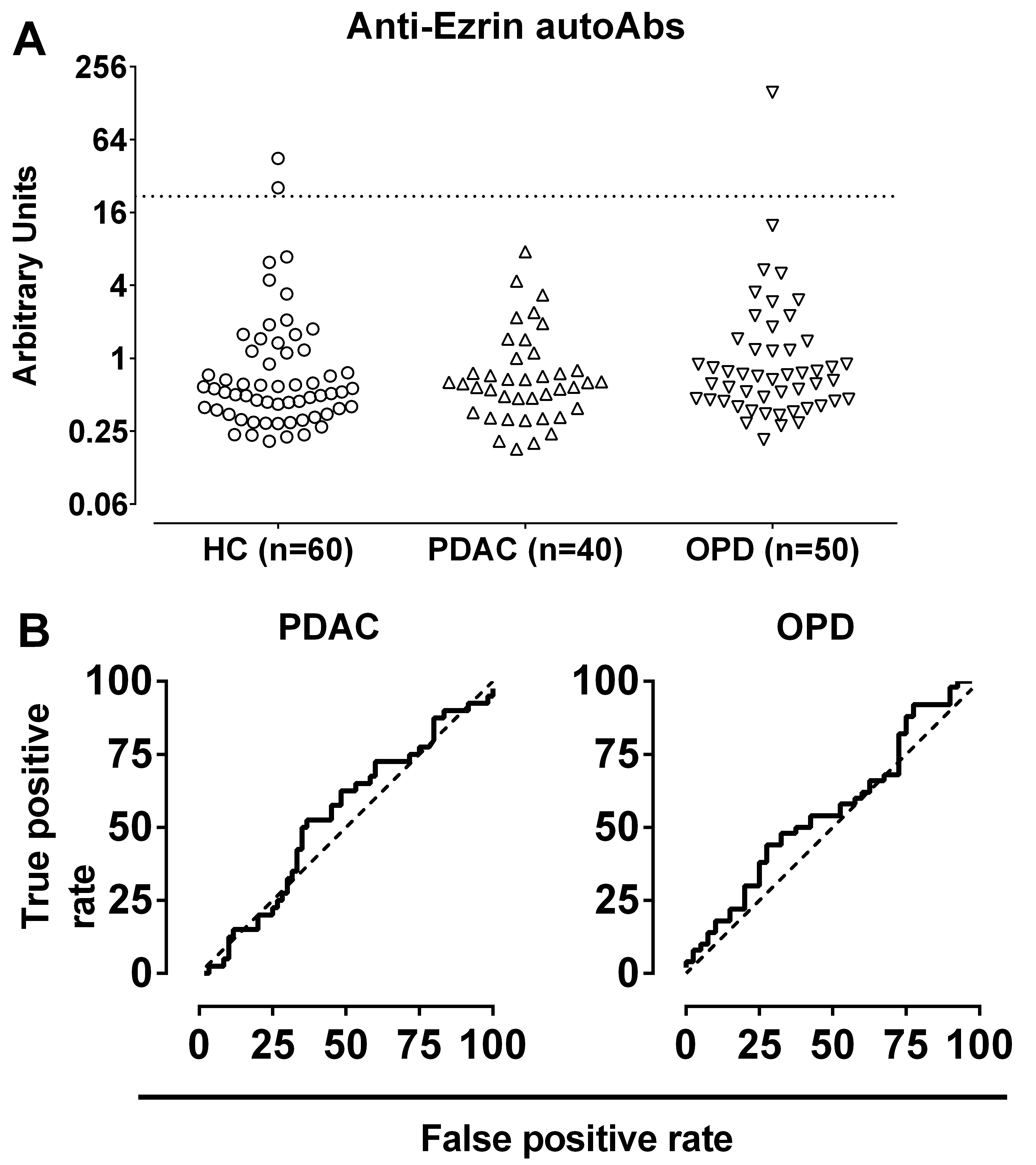
We investigated the presence of anti-Ezrin autoAbs in a selection of patients with PDAC (
Discussion
Our study investigated the antibody response against Ezrin, a candidate autoantibody PDAC biomarker with a reportedly large prevalence in PDAC patients [10]. Currently, the monitoring of antibodies against tumor associated antigens (TAAs) has found only a limited application in the cancer diagnostic of paraneoplastic neurological syndromes, in which specific autoAbs can precede by several years the appearance of lung and ovarian cancers [35, 36, 37, 38]. While the presence of autoAbs to candidate TAAs has been reported also in other cancers [12, 39, 40, 41, 42, 43, 44], including PDAC [9, 10, 11, 12, 13, 14, 15], none of these proposed biomarkers has been thoroughly validated and introduced in standard diagnostic routines or screening programs for PDAC.
Among the reported candidate targets of antibodies in PDAC, Ezrin looked a particularly promising, since Ezrin autoAbs were found in a very large fraction of patients with PDAC and appeared to be predictive of future PDAC development in a prospective cohort of subjects from the EPIC study. However, the striking 93.2% sensitivity for PDAC of the Ezrin autoantibody test was achieved using a threshold for positivity based on a ROC curve analysis that maximized accuracy, and at the cost of the test specificity that was only of 75% [10].
Of notice, it was previously demonstrated, using endoscopic ultrasonography as a test model [45], that any efficient screening strategy for PDAC should achieve a specificity of at least 90%. However, in that study, the inclusion in the screening strategy of subjects at higher baseline risk of PDAC [46] slightly relaxed the specificity requirements of the test. Any screening strategy for PDAC to be implemented in the general population, where the prevalence of the disease is quite low (0.02%), would instead demand a far more stringent specificity, and closer to the 100% mark.
Using a more stringent threshold of 95% specificity, a reassessment of the original ROC curve analysis results for Ezrin autoAbs suggests that the sensitivity for PDAC of the Ezrin autoantibody ELISA might be around 50% in advanced stage patients and 43% in subjects from the EPIC study that developed PDAC.
While still very promising, this revised sensitivity for PDAC of the Ezrin autoantibody test would nevertheless fail to resolve the discrepancy with a follow-up study [11] that did not observe differences in Ezrin autoantibody levels in a cohort of age, sex, and race matched PDAC cases (
We suspected that technical differences may play a role in the discrepancy between published Ezrin autoantibody measurement, and re-evaluated Ezrin autoantibodies in a relatively large cohort of PDAC patients and controls using a novel LIPS assays. In LIPS, antigen-antibody binding happens in liquid phase unlike on a solid phase as in ELISA. In fact, the adsorption of antigens to the polystyrene surface of ELISA plates is known to lead to partial antigen denaturation [47, 48, 49, 50] and to the exposure hydrophobic residues. In turn, considering the large pool of polyreactive serum antibodies normally found in every individual [51, 52], it is not surprising that several ELISA assays showed a problematic specificity [24, 53, 54, 55, 56, 57, 58]. In addition, common ELISA assay practices contribute additional confounding factor that might impact on test specificity, like for example the common use as blocking reagent of BSA, a known target of relatively common antibodies in normal sera [55, 59, 60].
On the contrary, liquid phase immunoassays were repeatedly proved empirically to be more sensitive and specific than solid phase assays in several international workshops aimed at the standardization of type 1 diabetes autoantibodies [22, 23, 24, 25].
Using our novel liquid phase LIPS test, we analyzed the presence of Ezrin autoAbs in a cohort of patients that showed a PDAC stage distribution that turns towards earlier stages, in comparison with PDAC stages of patients from Capello’s study. Our cohort of PDAC patients represents the ideally target population that we want to detect with early biomarkers.
Our LIPS test showed that the Ezrin antibody response was overall remarkably low in titer and generally indistinguishable from the background levels of the assay. Moreover, the few instances in which antibody binding to the chimeric nanoluciferase- antigen were slightly elevated in some sera were not able to discriminate PDAC or OPD patients from healthy controls. Furthermore, this inability to discriminate cases from control was independent of the selected threshold for positivity, as demonstrated by the ROC curve analysis of the results which again proved that antibodies to Ezrin measured by a liquid phase assay were not disease specific.
Therefore, despite the use of an alternative technology, we could not confirm Ezrin autoAbs as a useful biomarker for discrimination of PDAC from any of the control groups analyzed.
In addition to further insight on Ezrin autoantibodies and pancreatic cancer, our work should raise the awareness that the search for robust circulating antibody biomarkers of PDAC and other cancers would benefit by the adoption of liquid phase immunoassays either in the identification or validation of antibody responses to candidate tumor antigens.
Footnotes
Acknowledgments
D. Liberati is enrolled as PhD student in the San Raffaele Vita-Salute University and conducted this study as partial fulfillment of the requirements for a PhD degree in Molecular Medicine. This study was supported by the Associazione Italiana per la Ricerca sul Cancro (AIRC, bando 5
