Abstract
PURPOSE:
Plasma and serum cell-free DNA (cfDNA) are useful sources of tumor DNA, but comparative investigations of the tumor mutational status between them are rare.
METHODS:
we performed droplet digital PCR assay for representative hotspot mutations in metastatic breast cancer (MBC) (ESR1 and PIK3CA) in serum and plasma cfDNA concurrently extracted from the blood of 33 estrogen receptor-positive MBC patients.
RESULTS:
ESR1 mutations in plasma cfDNA were found in 7 of the 33 patients; ESR1 mutations in serum cfDNA were detected in only one out of 7 patients with ESR1 mutations in plasma cfDNA. PIK3CA exon 9 and exon 20 mutations in plasma cfDNA were found in 3 and 7 out of the 33 patients, respectively; PIK3CA exon 9 mutations in serum cfDNA were detected in 2 out of 3 patients with PIK3CA exon 9 mutations in plasma cfDNA; PIK3CA exon 20 mutations in serum cfDNA were detected in 2 out of 7 patients with PIK3CA exon 20 mutations in plasma cfDNA.
CONCLUSIONS:
Here we show the higher frequency of ESR1 and PIK3CA mutations in the plasma than in the serum in 33 MBC patients; therefore, serum samples should not be considered the preferred source of cfDNA.
Keywords
Introduction
Analysis of cell-free DNA (cfDNA) circulating in serum and plasma has been shown as a potential alternative procedure for studying tumor tissue DNA in metastatic breast cancer (MBC). The detection of cfDNA is not easy but the introduction of new techniques, such as digital PCR (dPCR), has overcome many challenges. In addition, qualitative and quantitative changes of tumor mutational status in circulating cfDNA have been shown in various therapeutic conditions [1]. Therefore, analysis of the tumor mutational status in cfDNA from MBC patients can be used not only as a monitoring tool of disease progression, but also as a predictor of tumor response to therapy [2].
In alteration frequency in metastatic versus primary tumors in the BOLERO-2 cohort, Hortobagyi et al. demonstrated that PIK3CA mutations had the highest frequency in metastatic and primary breast tumors, and that ESR1 mutations had higher frequency in metastatic breast tumors than in primary breast tumors [3]. Digital PCR (dPCR) assays on plasma cfDNA of two phase III cohorts recently demonstrated that the difference of clinical features between the hotspot mutations in MBC, ESR1 and PIK3CA mutations. The BOLERO-2 cohort recently revealed that progression-free survival (PFS) benefit of everolimus was maintained irrespective of PIK3CA mutations, but it decreased according to the presence of ESR1 mutations [4, 5]. On the other hand, the PALOMA-3 cohort revealed that PIK3CA and ESR1 status did not affect PFS benefit of palbociclib [6, 7]. Plasma is currently recommended as a source of cfDNA, however, serum samples still remain the preferred candidate molecular sources especially in retrospective studies using biobanked samples [8].
To verify whether serum and plasma could show similar tumor mutational results, we assayed the representative hotspot mutations in MBC, ESR1 and PIK3CA [3], in serum and plasma cfDNA concurrently extracted from the same blood of 33 estrogen receptor (ER)-positive MBC patients.
Materials and methods
A total of 33 MBC patients, treated at Kumamoto University Hospital between 2007 and 2016, were enrolled in this study. Cases were selected if archival paired plasma and serum were available. Informed consent was obtained from all patients before biopsy or surgery. The Ethics Committee of Kumamoto University Graduate School of Medicine (Kumamoto, Japan) approved the study protocol. The treatment of MBC patients was performed in accordance with the National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology [9]. Basically, MBC patients were assessed monthly for clinical response at the Kumamoto University Hospital.
Blood collected in EDTA K2 tubes and serum separating tubes was processed as soon as possible and was centrifuged at 1,467
The nonparametric Mann-Whitney U test and the chi-square test or Fisher’s exact test were adopted for statistical analysis of the associations of the presence of cfDNA ESR1 and PIK3CA mutations mutations between plasma and paired serum. Differences were considered significant when a
Patients’ characteristics
Patients’ characteristics
Abbreviations: HR, hormone receptor; ER
A total of 33 MBC patients were enrolled in this study. All tumor plasma and serum samples contained sufficient DNA for this study. The demographics and baseline characteristics of MBC patients are shown in Table 1. The median age of the patients at blood biopsy was 58 years (range, 31–84). A total of 29 (87.9%) out of 33 patients had been treated with hormonal therapy, and a total of 26 (78.8%) out of 33 patients had been treated with aromatase inhibitors before the blood draw. The median duration of follow-up was 146 months (range, 15–284 months). Figure 1A shows the concentrations of cfDNA of paired plasma and serum. There were no statistically significant differences between the concentrations of cfDNA in the plasma (median: 117 pg/
A. Concentrations of cfDNA (pg/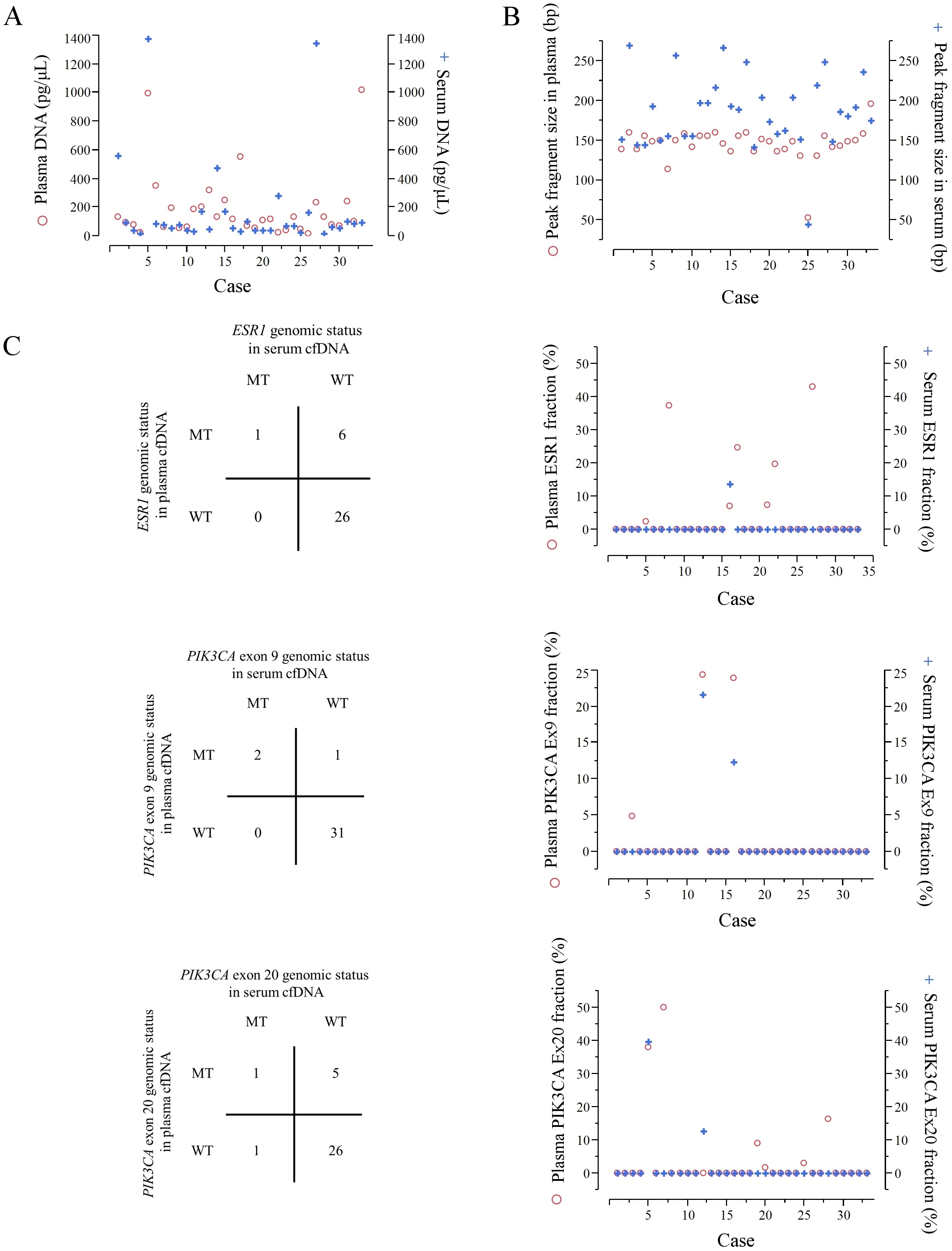
We investigated the presence of ESR1 and PIK3CA mutations in cfDNA extracted from plasma and serum samples of 33 MBC patients since the different frequency of ESR1 and PIK3CA mutations between plasma and serum have not been fully investigated. Although the concentrations of cfDNA between the plasma and serum were similar, we found differences in the presence of ESR1 and PIK3CA mutations. ESR1 mutations in serum cfDNA were detected only in one out of 7 patients with ESR1 mutations in plasma cfDNA; PIK3CA exon 9 mutations in serum cfDNA were detected in 2 out of 3 patients with PIK3CA exon 9 mutations in plasma cfDNA; and PIK3CA exon 20 mutations in serum cfDNA were detected in 2 out of 7 patients with PIK3CA exon 20 mutations in plasma cfDNA.
This difference may be explained by that serum samples are easily affected by DNase activity and contamination from lysis of white blood cells (WBCs). Barra et al. reported that serum cfDNA was more affected by degradation than EDTA-treated plasma cfDNA, since DNase activity was inhibited by EDTA; a 14.9-fold higher DNase activity was detected in the serum compared with EDTA-treated plasma [14]. Several groups reported that serum cfDNA is contaminated by genomic DNA from white blood cells (WBCs). Lee et al. showed that the extra DNA in serum is generated during the process of clotting due to WBC lysis [15]. Gautschi et al. indicated that serum cfDNA concentration strongly correlated with WBC counts [16]. In addition, some groups suggested that longer DNA fragments originate from WBCs in serum samples than in plasma samples [17, 18]. Indeed, in our study the peak of the fragment size of serum cfDNA was consistently larger than that of paired plasma cfDNA. Thus, serum cfDNA can lose potentially important diagnostic information, such as tumor-derived genetic alterations. However, the present study has some limitations; it is a retrospective, single-institute study with a relatively small patient cohort.
However, it is unlikely that the following two results could be explained only by such reasons. First, according to the Fig. 1C showing the actual MAF of cfDNA, almost half of such cases represented very large MAF (
Conclusions
Our results showing a higher frequency of ESR1 and PIK3CA mutations in the plasma than in the serum in 33 MBC patients suggest that the use of serum as a source of cfDNA may be discouraged.
Footnotes
Acknowledgments
This work was supported in part by a grant-in-aid (project numbers 17K16551) for scientific research from the Ministry of Education, Science and Culture of Japan.
Conflict of interest
All the authors declare that they have no actual, potential, or perceived conflict of interest with regard to the manuscript submitted for review.
