Abstract
BACKGROUND:
Accumulating studies have reported the abnormal expression of microRNA-136 (miR-136) in numerous types of human cancer, and its involvement in cancer initiation and progression. However, there are no investigations of miR-136 in osteosarcoma (OS).
OBJECTIVE:
To explore the expression pattern, clinical significance and potential roles of miR-136 in OS.
METHODS:
miR-136 expression in clinical OS tissues and human OS cell lines were detected by qPCR, and its associations with clinicopathological characteristics of OS patients were statistically analyzed. Then, the effects of miR-136 on OS cell proliferation, migration and invasion were assessed in vitro. Its underling mechanisms were also investigated.
RESULTS:
miR-136 expression in OS tissues and cells were dramatically decreased compared with corresponding non-cancerous tissues and cells, respectively. Low miR-136 expression was significantly associated with aggressive clinical features, including the advanced clinical stage, the presence of lung and distant metastasis (all
CONCLUSION:
Our findings provide the first evidence that the aberrant expression of miR-136 may be implicated into carcinogenesis and cancer progression of OS. Functionally, miR-136 may inhibit the proliferation, migration and invasion of OS cells via negatively regulating its target gene MTDH. Thus, miR-136-MTDH axis may be a potential therapeutic targets for the treatment of OS.
Keywords
Introduction
Osteosarcoma (OS) represents the third most common primary malignant bone tumor in childhood and adolescents [1]. It often occurs in the proximal tibia and humerus, as well as the metaphyseal regions of distal femur [2]. Typical symptoms and signs of this malignancy contain localized swelling, joint movement limitations, pain and trabecular bone destruction [3]. OS is characterized by the high degree of malignancy, as well as the increasing incidence of recurrence and metastasis [4]. Therapeutic strategies for OS therapy include surgical resection combined with radiotherapy and chemotherapy [5]. Despite many efforts have been made, the prognosis of OS is still unsatisfied. The five-year survival rate of OS patients is as low as 60–70% globally. What is more, the clinical outcome of patients with metastatic disease is usually far worse (less than 20%) [6]. Therefore, it is urgent to explore the underlying mechanisms of OS and identify novel biomarkers for improving the prognosis of OS patients.
Associations of miR-136 expression with various clinicopathological characteristics of OS patients (qPCR analysis was performed to detect the expression levels of miR-136 in 100 OS tissues. The associations between miR-136 expression and various clinicopathological characteristics were assessed by the chi-square or Fisher’s exact tests)
Associations of miR-136 expression with various clinicopathological characteristics of OS patients (qPCR analysis was performed to detect the expression levels of miR-136 in 100 OS tissues. The associations between miR-136 expression and various clinicopathological characteristics were assessed by the chi-square or Fisher’s exact tests)
Note: ‘NS’ refers to the difference without statistical significance.
MicroRNAs (miRNAs), a group of single stranded, non-coding and small RNAs with 18
To explore the expression pattern, clinical significance and potential roles of miR-136 in OS, in the current study, expression levels of miR-136 in clinical OS tissues and human OS cell lines were detected by reverse transcription quantitative polymerase chain reaction (qPCR). Associations of miR-136 expression with clinicopathological characteristics of OS patients were statistically analyzed. Then, the effects of miR-136 on OS cell proliferation, migration and invasion were assessed in vitro. A putative target of miR-136, metadherin (MTDH), was obtained from miRTarBase, and was verified by luciferase activity assay. After that, the effect of MTDH loss on miR-136 downregulated in OS cells was further observed.
Patients and tissue samples
Our study was performed in compliance to the National Research Council’s guide and approved by the Research Ethics Committee of Beijing Luhe Hospital Affiliated to Capital Medical University and Hangzhou Cancer Hospital. The written informed consent was obtained from all the patients with Osteosarcoma prior to collection of samples.
A total 100 histologically diagnosed OS and adjacent noncancerous bone tissues (located over 3 cm away from the tumor) were obtained from 100 patients with primary OS between April 2010 to March 2015 in the Department of Orthopedics, Beijing Luhe Hospital Affiliated to Capital Medical University and Department of Orthopedic Oncology, Hangzhou Cancer Hospital. Each pair of osteosarcoma and adjacent tissue was collected from the same patient after pathological verification. No patient received immunotherapy, radiotherapy or chemotherapy before surgery. Tissue specimens were immediately frozen in liquid nitrogen until further use. The clinicopathological features of these patients were summarized in Table 1.
Cell culture and transfection
Two OS cell lines MG-63 and U2OS, as well as human osteoblasts hFOB1.19 were obtained from the American Type Culture Collection (Hill, NJ, USA), and were maintained in Dulbecco’s Modified Eagle’s Medium (DMEM, Life Technologies, South Logan, UT, USA) supplemented with 10% fetal bovine serum (FBS, Life Technologies, Carlsbad, CA, USA), 100 U/ml penicillin, and 100
When the MG-63 and U2OS cells were grown in the logarithmic growth phase, the cells were cultured in a 24-well plate with 2
RNA extraction and qPCR
Total RNA (including miRNA) was extracted from clinical tissues or cells by using TRIZOL reagent (Invitrogen Life Technologies, Carlsbad, CA, USA) and purified with miRNeasy mini kit (Qiagen, Hilden, Germany). The RNA concentration was quantified on a NanoDrop 2000 (Thermo Scientific, USA) by O.D. 260/280 nm absorbance. Reverse transcription assay and real time PCR assay were performed using PrimeScript RT reagent Kit (TaKaRa, Dalian, China) and SYBR Premix Ex TaqII (Tli RNaseH Plus) kit (TaKaRa, Dalian, China) respectively. The miR-136 expression was normalized with U6 small RNA as internal control. The primer sequences used in this study were as following: miR-136, forward 5’-ACA CTC CAG CTG GGA CTC CAT TTG TTT TG-3’, reverse 5’-CTC AAC TGG TGT CGT GGA GTC GGC AAT TCA GTT GAG TCC ATC AT-3’; U6, forward 5’-CTC GCT TCG GCA GCA CA-3’, reverse 5’-AAC GCT TCA CGA ATT TGC GT-3’. The relative expression was analyzed using 2
Cell proliferation assay
The CCK-8 assay was performed to evaluate the cell proliferation of MG63 and U2OS cells transfected with miR-136/NC mimics or miR-136/NC inhibitor. At 48 h post-transfection, the cells were collected and seeded into 96-well plates at a density of 3,000 cells/well. Then, the cells were cultured at 37
Decreased expression of miR-136 in human osteosarcoma (OS) tissues and cells. (A) miR-136 expression was significantly decreased in OS tissues compared with matched normal tissues (OS vs. non-cancerous 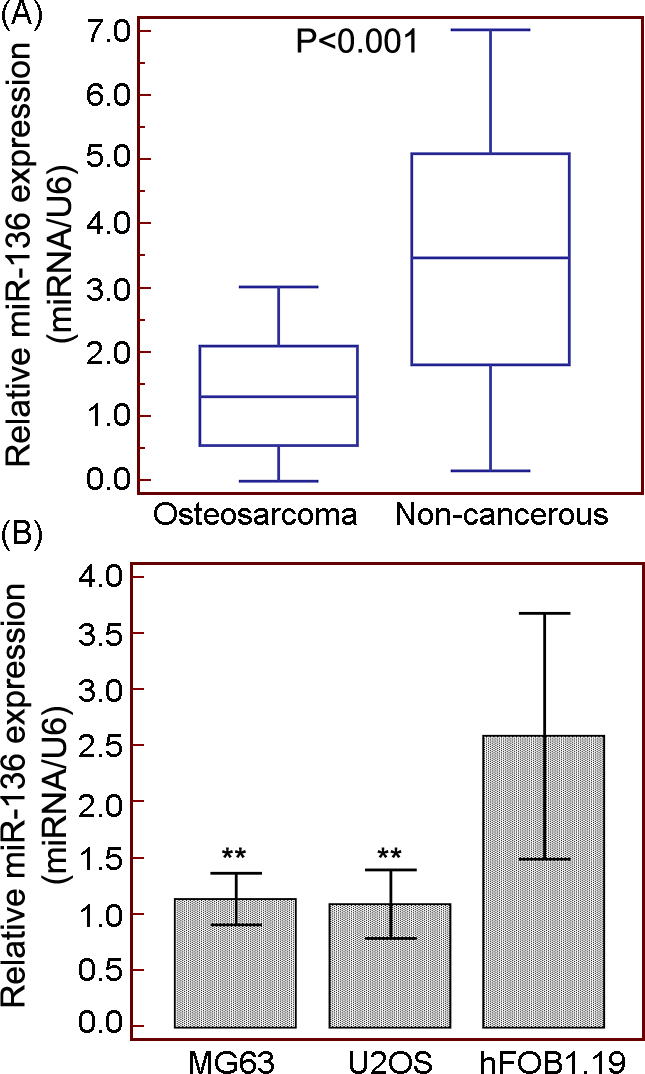
miR-136 expression suppresses the cell proliferation, migration and invasion of OS cells in vitro. (A and B) Relative expression levels of miR-136 in blank, miR-136-mimic and NC-mimic-transfected MG63 and U2OS cells, respectively. (C and D) The cell viabilities of blank, miR-136-mimic and NC-mimic-transfected MG63 and U2OS cells, respectively. (E and F) The migrated and invasive capacities of blank, miR-136-mimic and NC-mimic-transfected MG63 and U2OS cells, respectively. **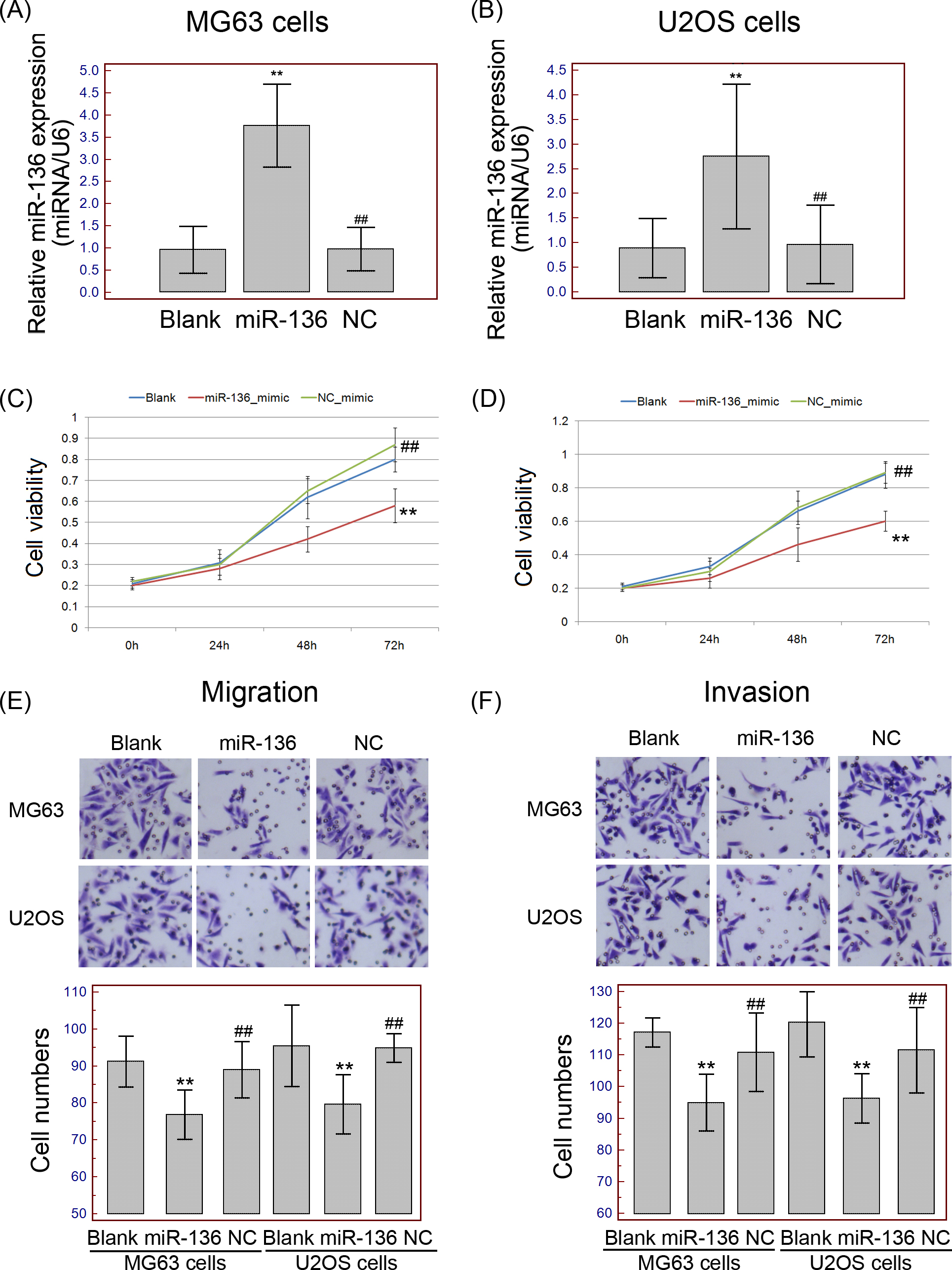
The transwell migration assay was performed to assess the migration abilities of MG63 and U2OS cells transfected with miR-136/NC mimics or miR-136/NC inhibitor. Following the transfection, the cells were harvested and resuspended in Eagle’s minimum essential medium. Cell suspension (100
Matrigel coated cell culture chambers (8
The migrated or invasive MG63 and U2OS cells adherent to the lower surface of the filter were fixed with methanol, stained with 0.5% crystal violet for 20 min, photographed, and counted. The number of migrated or invasive MG63 and U2OS cells was counted at 200
Western blot analysis
MTDH protein expression levels in MG63 and U2OS cells transfected with miR-136/NC-mimics, or siRNA-MTDH/siRNA-NC were detected by western blot analysis. Briefly, total protein in OS cells of different groups was extracted using 500
Dual luciferase reporter gene assay
According to the data of miRTarBase (
MTDH is a direct target of miR-136 in OS cells. (A and B) Relative expression of MTDH protein in both MG63 and U2OS cells in different groups. **
Loss of MTDH abrogates the tumor suppressive roles of miR-136. (A and B) Relative expression of MTDH protein in both MG63 and U2OS cells in different groups. **
Our data analysis were performed using SPSS version 11.0 software for Windows (SPSS Inc, IL, USA). Data were showed as mean
Results
Decreased expression of miR-136 in human OS tissues and cells
To assess the potential clinical relevance of miR-136 to carcinogenesis of OS, we firstly detected the expression levels of miR-136 in OS tissues and cell lines using qPCR. As shown in Fig. 1A, miR-136 expression was significantly decreased in OS tissues compared with matched non-cancerous bone tissues (OS vs. non-cancerous
Decreased expression of miR-136 associates with aggressive progression of OS patients
To evaluate the associations of miR-136 expression with various clinicopathological characteristics of OS patients, all 100 patients were divided into low miR-136 expression (
miR-136 expression suppresses the cell proliferation, migration and invasion of OS cells in vitro
To evaluate the potential significance of miR-136 in malignant phenotypes of human OS cells, we transfected miR-136 mimic and non-targeting control mimic (NC mimic) into MG63 and U2OS cells, respectively. Following the transfection, the expression levels of miR-136 in miR-136-mimic-transfected MG63 and U2OS cells were both significantly increased compared with that in NC mimic group (both
MTDH is a direct target of miR-136 in OS cells
According to miRTarBase (
Loss of MTDH abrogates the tumor suppressive roles of miR-136
To validate whether the tumor suppressive roles of miR-136 in OS cells were associated with its regulation on MTDH, we transfected miR-136-inhibitor alone or combined with si-MTDH into two OS cell lines. As a result, the expression levels of MTDH protein was significantly reduced by the co-transfection of miR-136-inhibitor and si-MTDH in comparison with those in OS cells transfected with miR-136-inhibitor alone (both
Discussion
miRNAs with multifaceted activities regulate the corresponding target gene expression and exhibit direct or indirect oncogenic or tumor suppressive properties in different types of cancers. In the current study, we observed the decreased expression of miR-136 in both clinical OS tissues and human OS cell lines for the first time. Importantly, low miR-136 expression was significantly associated with aggressive clinical features, including the advanced clinical stage, the presence of lung and distant metastasis. Moreover, we found that the enforced expression of miR-136 obviously inhibited the proliferation, migration and invasion of OS cells in vitro. Mechanistically, MTDH was predicted and verified as a target gene of miR-136, and further functional experiments indicated that the loss of MTDH abrogated the tumor suppressive roles of miR-136 in OS cells.
Recent studies has indicated that miR-136 may play tumor suppressive roles in breast cancer, ovarian cancer and colon cancer, or a oncogenic role in non-small cell lung cancer [14, 15, 16, 17, 18]. Here, our data suggested that miR-136 may function as an anti-oncogenic regulator in human OS. Since miRNAs exert their roles though directly regulating target genes, in this study, our results obtained from dual luciferase reporter gene assay suggest that the fragment at the 3’-UTR of the MTDH mRNA may be the complementary site for the miRNA-136 seed region. Thus, MTDH may be one of the direct targets of miR-136. MTDH, also known as Astrocyte elevated gene-1 (AEG-1) and Lysine-rich CEACAM1 co-isolated (LYRIC), has been indicated as a crucial oncogene with multifunctional roles in cancer development and progression via involved into various oncogenic signaling pathways, such as the ERK/mitogen-activated protein kinase and Wnt/
In conclusion, our findings provide the first evidence that the aberrant expression of miR-136 may be implicated into carcinogenesis and cancer progression of OS. Functionally, miR-136 may inhibit the proliferation, migration and invasion of OS cells via negatively regulating its target gene MTDH. Thus, miR-136-MTDH axis may be a potential therapeutic targets for the treatment of OS. Further studies are needed to validate these findings in vivo and clinical experiments.
Footnotes
Conflict of interest
None.
