Abstract
OBJECTIVE:
Osteosarcoma is the most common primary malignant skeleton tumor that derives from mesenchymal cells. Emerging evidences have identified the vital role of long non-coding RNAs (lncRNAs) in the development of osteosarcoma. In this study, we aimed to investigate the role of lncRNA gastric carcinoma highly expressed transcript 1 (GHET1) in osteosarcoma progression.
METHODS:
The expression levels of relevant genes in clinical samples and cell lines were determined by quantitative real-time PCR. Cell proliferation was determined by CCK8 and cell colony formation assays. Transwell assay was used to detect the invasion and migration of osteosarcoma cells. Cell apoptosis and cell cycle were detected by flow cytometry. Protein levels were detected by western blot. In vivo tumor growth was investigated in the xenograft nude mice model. To determine whether growth inhibition and apoptosis are responsible for antitumor activity of silencing GHET1, immunohistochemistry for proliferation and TUNEL assay was performed in xenograft tissues. In vivo lung metastasis was performed to detect the effect of GHET1 on cell metastasis ability.
RESULTS:
Our results revealed that GHET1 was up-regulated in osteosarcoma tissues compared to normal tissues. GHET1 was also increased in osteosarcoma cell lines compared to normal osteoplastic cell line. The up-regulation of GHET1 was significantly associated with TNM stage, distant metastasis and lymph node metastasis in patients with osteosarcoma. In vitro studies showed that silencing GHET1 in MG-63 and U2OS cells inhibited cell proliferation, cell invasion and migration and epithelial-to-mesenchymal transition (EMT), promoted cell apoptotic rate, and also caused an increase in cell population at G
CONCLUSION:
GHET1 was up-regulated in osteosarcoma tissues and cell lines, inhibited cell apoptosis, promoted cell proliferation, invasion and migration by affecting EMT in vitro, and was correlated with the tumor growth and metastasis in vivo. GHET1 may be a potential therapeutic target of osteosarcoma treatment.
Introduction
Osteosarcoma is a common primary bone tumor, accounting for the most frequent malignant bone tumor in children and adolescents [1]. Approximately 80% of osteosarcoma patients have metastatic disease at the time of diagnosis and metastasis is a consistent problem in tumor prognosis and treatment [2, 3]. With the development of combinatorial chemotherapy, the prognosis of osteosarcoma patients has been improved in the nearest statistics with nearly 60–70% of 5-year survival rate [4, 5]. While the molecular mechanisms of osteosarcoma have gained considerable attention, the mechanisms underlying its initiation and progression remain unclear. Further exploration of osteosarcoma will help in the development of effective strategies in the diagnosis, treatment and prognosis of osteosarcoma.
Long non-coding RNAs (LncRNAs) are a class of transcripts that are
Epithelial-mesenchymal transition (EMT) is able to promote cancer cell invasion and metastasis. EMT is a process for epithelial cells transforming into mesenchymal cells under specific pathological conditions [17, 18]. During this process, tumor cells lose epithelial characteristics, E-cadherin, occluding, and other cellular tight junctions and transform into mesenchymal cells accompanying with increasing mesenchymal cell biomarkers, ZEB2, Snail, N-cadherin and vimentin [6, 19]. Growing evidences indicate that lncRNAs were involved in osteosarcoma invasion and metastasis by regulating EMT [20]. Although several lncRNAs have been demonstrated involved in osteosarcoma tumor progression by regulating EMT [21], the specific role of novel lncRNA GHET1 in osteosarcoma are not studied preciously.
In this study, we assessed the expression levels of relevant genes in clinical samples and osteosarcoma cell lines. We also analyzed the relationship between GHET1 expression and clinicopathological features of osteosarcoma patients. In addition, we investigated the effect of silencing GHET1 on the proliferation, invasion, migration, apoptosis, cell cycle and EMT of osteosarcoma cells in vitro. Moreover, the effects of GHET1 on in vivo tumor growth, lung metastasis, cell proliferation and cell apoptosis were also explored.
Correlation between GHET1 expression and clinical features of osteosarcoma patients
Correlation between GHET1 expression and clinical features of osteosarcoma patients
Clinical patient data and tissue sample
This study was approved by Ethics Committee and Institutional Review Board of The Affiliated Tumor Hospital of Zhengzhou University. Tissue specimen collections were made with full informed consent of the patients, all the research protocols had been examined by Ethics Committee and Institutional Review Board of The Affiliated Tumor Hospital of Zhengzhou University and complied with the ethical statement.
A total of 60 osteosarcoma patients were recruited in our study who undergo surgical operation at The Affiliated Tumor Hospital of Zhengzhou University. The osteosarcoma diagnosis was histopathologically confirmed. None of them received radiotherapy or chemotherapy before surgery. Written informed consents had been obtained from all the enrolled patients. The clinical information for all the samples is detailed in Table 1. All the osteosarcoma tissues and paired adjacent non-tumor tissues were obtained from patients after surgical excision and were confirmed by two experienced pathologists. Then, the tissues were collected and frozen in liquid nitrogen until use.
Cell culture and transfection
Human osteosarcoma cell lines U2OS and MG-63 were obtained from the American Type Culture Collection (Manassas, VA, USA). Human U2OS cells were cultured in McCoy’s5A medium (Thermo Fisher Scientific, Waltham, MA, USA) with 10% heated-inactivated fetal bovine serum (FBS; Thermo Fisher Scientific). NHOst, KHOS and MG-63 cells were grown in DMEM (Thermo Fisher Scientific) supplemented with 10% FBS. The cells were cultured at 37
The small interference RNAs for GHET1 (si-GHET1 #1 and si-GHET1#2) and the scrambled siRNA (si-NC) were purchased from Ribobio (Guangzhou, China). The pcDNA3.1 and the GHET1 overexpressing plasmid (named pcDNA3.1-GHET1) were obtained from the GenePharma (Shanghai, China). Different concentrations of small interfering RNAs (siRNAs) or plasmids were transfected into cells using Lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions.
Quantitative real-time PCR (qRT-PCR)
Total RNA from cell and tumor tissues was isolated using TRIzol reagent (Invitrogen, Carlsbad, USA) according to the manufacturer’s instructions. The quality and quantity of total RNA sample were tested using a microplate spectrophotometer (Multiskan MK3, Thermo Fisher Scientific, Waltham, MA, USA). Individual RNA samples were reversely transcribed into cDNA by the PrimeScipt RT Master Mix kit (Takara, Dalian, China). The relative levels of relevant genes were determined by quantitative real-time PCR using the SYBR Premix Ex TaqTM (Takara) and the specific primers in the IQ5 Multicolor Real-Time PCR Detection system (Bio-Rad Laboratories, Hercules, China). Primers for GHET1 were: forward 5’-CCC CACAAATGAAGACACT-3’ and reverse 5’-TTCCC AACACCCTATAAGAT-3’; ZEB2: forward 5’-TGAG GATGACGGTATTGC-3’ and reverse 5’-ATCTCGTT GTTGTGCCAG-3’; Snail: forward 5’-TGTTGCAGT GAGGGCAAGAA-3’ and reverse 5’-GACCCTGGTT GCTTCAAGGA-3’; N-cadherin: forward 5’-CGAGC CGCCTGCGCTGCCAC-3’ and reverse 5’-CGCTGC TCTCCGCTCCCCGC-3’; vimentin: forward 5’-TAC AGGAAGCTGCTGGAAGG-3’ and reverse 5’-ACCA GAGGGAGTGAATCCAG-3’; E-cadherin: forward 5’-TACGCCTGGGACTCCACCTA-3’ and reverse 5’-C CAGAAACGGAGGCCTGAT-3’; GAPDH: forward 5’-CGGAGTCAACGGATTTGGTCGTAT-3’ and reverse 5’-AGCCTTCTCCATGGTGGTGAAGAC-3’. Changes in the expression were calculated using the 2
CCK-8 assay
After transfection with si-NC, si-GHET1#1 or si-GHET1#2, equal number of MG-63 and U2OS cells were seeded in 96-well plates respectively, and at 0, 24, 48, 72 h post-transfection, cell proliferation was measured with a Cell Counting Kit-8 kit (CCK8, R&S Bio-Technology, Shanghai, China) according to the manufacturer’s instructions. The OD values were measured at a wavelength of 450 nm with a microplate spectrophotometer (Multiskan MK3).
Cell colony formation assay
Cell proliferation ability was also detected by colony formation assay. Human osteosarcoma cells were seeded into 6-well plates at a density of 1000 cells/ml, 2 ml/well. The cells were transfected with si-NC, si-GHET1#1 or si-GHET1#2. The medium was replaced with fresh medium every 3 days. After incubation for 10 days, the cells were then fixed with 70% ethanol and stained with 0.1% crystal violet for 30 min at 37
Cell apoptosis and cell cycle assay
Flow cytometry was performed for cell apoptosis assay. Cells were seeded into 6-well plates at density of 1
Transwell assays for migration and invasion
The migration and invasion potential of osteosarcoma cells was evaluated by transwell assay. At 48 h after transfection, cells were seeded in the upper chamber of an insert (8
Western blot analysis
Total protein was extracted from cells using total protein extraction kit according to the manufacture’s instruction (KeyGen, Rockville, MD, USA). The protein concentrations of the lysates were measured by the Protein BCA Assay Kit (Bio-Rad, Hercules, CA, USA). For the western blotting assay, 20
In vivo tumor xenograft model
All animals were cared for in strict compliance with the Guide for Care and Use of Laboratory Animals (Ministry of Science and Technology of China, 2006). All experiments were approved by the Institutional Animal Care and Use Committee of The Affiliated Tumor Hospital of Zhengzhou University.
Four-week-old male Balb/c nude mice were purchased from Shanghai Jiesijie Experimental Animal Co. Ltd. (Shanghai, China). Animals in this study were housed under a 12 h light/12 h dark cycle at controlled temperature (22–26
At 48 h after transfection (si-NC, si-GHET1#1), the transfected MG-63 cells were subcutaneously injected into 4-week-old male Balb/c nude mice. The mice were then randomly divided into two groups, si-NC group and si-GHET1#1 group,
LncRNA GHET1 was over-expressed in osteosarcoma tissue and associated with poor prognosis. (A) Expression of GHET1 in 60 cases of osteosarcoma tissue and adjacent non-tumor tissue detected by qRT-PCR. (B) Overall survival of osteosarcoma specimens with high and low expression of GHET1. The correlation between GHET1 expression and prognosis of patients with osteosarcoma was analyzed by Kaplan-Meier method analysis (log-rank test). (C) Expression of GHET1 in three osteosarcoma cell lines (KHOS, MG-63, U2OS) were detected by qRT-PCR compared to one normal human osteoplastic cell line (NHOst). 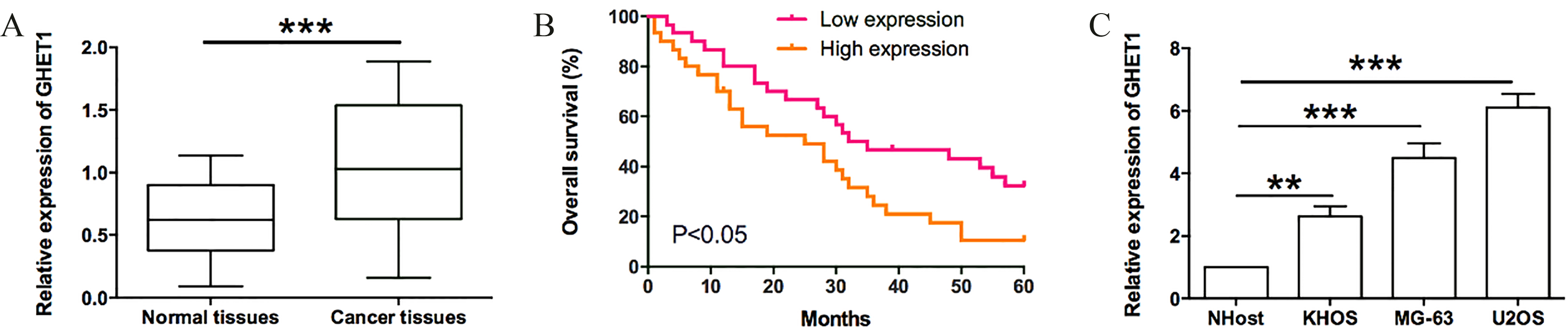
MG-63 cells were pre-transfected with si-NC or si-GHET1#1 at 4
Immunohistochemistry
Three
TUNEL assay
An in-situ Cell Death Detection Kit (ROCHE, USA) for TUNEL was applied in this study. The slides were dewaxed in xylene, hydrated using graded ethanol. De-paraffinized sections were washed with PBS and incubated with proteinase K for 30 min at 37
Statistical analysis
The statistical analysis was performed by using GraphPad Prism Software Version 5.0. All data in the study were represented as mean
Silencing GHET1 suppressed the proliferation of osteosarcoma cells in vitro. (A, B) MG-63 and U2OS cells were transfected with GHET1 specific siRNA for 48 h and the transfection efficiency was measured by qRT-PCR. (C, D) CCK-8 assay showed the proliferation ability of MG-63 and U2OS cells transfected with si-NC, si-GHET#1 or si-GHET#2 for 24 h, 48 h and 72 h. (E, F) Colony formation assay was performed to measure the effect of GHET1 knockdown on cell growth. MG-63 (E) and U2OS (F) cells were transfected with si-NC, si-GHET#1 or si-GHET#2 for 10 days. 
Silencing GHET1 induced cell cycle arrest and promoted the apoptosis of osteosarcoma cells. (A, B) Effect of GHET1 on apoptosis of osteosarcoma cells. MG-63 (A) and U20S (B) cells were transfected with si-NC, si-GHET#1 or si-GHET#2 for 48 h, cell apoptosis was examined by flow cytometry assay. (C, D) Influence of GHET1 on cell cycle progression in osteosarcoma cells. MG-63 (C) and U20S (D) cells were transfected with si-NC, si-GHET#1 or si-GHET#2 for 48 h, cell cycle was detected by flow cytometry method. 
Silencing GHET1 inhibited invasion and migration of osteosarcoma cells. (A) Effect of GHET1 on cell invasion in osteosarcoma cells. MG-63 (A) and U20S (B) cells were transfected with si-NC, si-GHET#1 or si-GHET#2 for 48 h. The transwell invasion assay was used to measure the capacity of cellular invasion. (B) Effect of GHET1 on cell migration in osteosarcoma cells. MG-63 (C) and U20S (D) cells were transfected with si-NC, si-GHET#1 or si-GHET#2 for 48 h, cell migration was detected by transwell migration assay. 
LncRNA GHET1 was over-expressed in osteosarcoma tissue and associated with poor prognosis
To determine the biological function of GHET1, the expression of GHET1 in 60 pair osteosarcoma tissues and corresponding adjacent normal tissues was measured by qRT-PCR. According to the results, the expression of GHET1 was significant up-regulated in cancerous tissues compared with normal counterparts (
Silencing GHET1 suppressed the proliferation of osteosarcoma cells in vitro
To investigate the effect of GHET1 on the cell proliferation in osteosarcoma, CCK8 and cell colony formation assays were performed. GHET1 siRNA was transfected into MG-63 and U2OS cells. To ensure the efficiency of interference an avoid off-target effects, we used two validated effective interference target sequence of GHET1. QRT-PCR results revealed that GHET1 level was apparently down-regulated in MG-63 (Fig. 2A) and U2OS (Fig. 2B) cells after transfected with si-GHET1# and si-GHET1#2 (
Silencing GHET1 induced cell cycle arrest and promoted the apoptosis of osteosarcoma cells
For further investigation, we sequentially verified the effect of GHET1 on apoptosis and cell cycle of osteosarcoma cells using flow cytometry. Results showed that down-regulation of GHET1 in MG-63 and U2OS cells obviously increased the percentage of apoptotic cells when compared to control group (
Silencing GHET1 regulated EMT-related gene expression. (A, B) Relative mRNA expression levels of ZEB2, Snail, N-cadherin, E-cadherin and vimentin in MG-63 (A) and U2OS (B) cells transfected with si-NC, si-GHET#1 or si-GHET#2 for 48 h. The mRNA levels were measured by qRT-PCR analysis. (C, D) Western blotting analysis of protein levels of ZEB2, Snail, N-cadherin, E-cadherin and vimentin in MG-63 (C) and U2OS (D) cells transfected with si-NC, si-GHET#1 or si-GHET#2 for 48 h. 
Silencing GHET1 suppressed the tumor growth, lung metastasis, cell proliferation and caused cell apoptosis in vivo. (A) The volume of MG-63-xenograft tumors in nude mice. MG-63 cells transfected with si-NC or si-GHET#1 were subcutaneously injected into nude mice. When the tumors were established, the tumor volumes were examined every 5 d for 35 d. Average tumor volumes were represented starting from the first injection and continuing until sacrifice 35 d later. (B) The weight of MG-63-xenograft tumors. After 35 d, the mice were euthanized, the tumors were removed and weighed. (C) The average numbers of metastasis nodules observed in the lungs of mice injected with MG-63 cells intravenously. Animals were sacrificed at 8 weeks after cells inoculation, pulmonary metastasis was assessed by counting the number of nodules from a macroscopic view. The images of lung and the haemotoxylin and eosin staining for the metastatic nodules were shown in left panel.(D) The relative expression of GHET1 in the tumors. The relative levels of GHET1 expression in vivo were determined by qRT-PCR. (E) The relative expression of ZEB2, Snail, N-cadherin, E-cadherin and Vimentin in the tumors. The relative expression levels of these genes were determined by qRT-PCR. (F) Effect of GHET1 on the marker of proliferation in tumors of MG-63-exografted mice. The proliferation cells in the tumor specimens were detected by Ki-67 Immunohistochemistry. (G) Influence of GHET1 on the apoptosis in tumors of MG-63-exografted mice. A TUNEL assay was performed to determine the level of apoptosis. 
We next undertook transwell analyses to determine the role of GHET1 in osteosarcoma cells migration and invasion. The transwell invasion assay showed that the number of invaded cells in osteosarcoma cells transfected with si-GHET1#1 or si-GHET1#2 was significantly decreased when compared to the cells transfected with si-NC (
Overexpression of GHET1 promoted proliferation, invasion and migration of osteosarcoma cells
As shown in Supplemental Fig. S1A and B, the expression of GHET1 was significantly up-regulated in MG-63 and U2OS cells after transfected with pcDNA3.1-GHET1. Further CCK8 assay, colony formation assay, cell invasion and migration assay showed that overexpression of GHET1 significantly promoted osteosarcoma cell proliferation, invasion and migration (Supplemental Fig. S1C–J). These results indicated that overexpression of GHET1 could promote the proliferation, invasion and migration of osteosarcoma cells.
Silencing GHET1 regulated EMT-related gene expression
The effects of GHET1 on the expression of EMT-related markers including ZEB2, Snail, N-cadherin, vimentin and E-cadherin were determined by qRT-PCR and western blotting assay. The qRT-PCR results revealed that osteosarcoma cells transfected with GHET1 siRNA had lower mRNA expression levels of ZEB2, Snail, N-cadherin, vimentin, and higher mRNA expression level of E-cadherin (
Silencing GHET1 suppressed the tumor growth, lung metastasis, cell proliferation and caused cell apoptosis in vivo
To investigate the effect of GHET1 on osteosarcoma tumorigenesis in vivo, xenograft assay was performed. MG-63 cells transfected with si-NC or si-GHET1#1 were subcutaneously injected into the nude mice. After injection, the dynamic growth of implanted tumors was monitored. As shown in Fig. 6A, after injection for 25 d, the mean volume of the tumors in si-GHET1#1 group was significantly smaller compared with the si-NC group (
To evaluate the metastatic potential of GHET1 on MG-63 cells, we cultured the cells transfected with si-NC or si-GHET1#1 and compared their rates of metastasis to the lungs. We injected the cells into the tail vein of mice and the mice were killed 8 weeks later. The number of tumor nodules formed by MG-63 cells transfected with si-NC was significantly more than that formed by MG-63 cells transfected with si-GHET1#1 (
QRT-PCR was performed to detect the expression levels of GHET1 and EMT-related gene expression in tumors. As a result, GHET1 level was significantly down-regulated in the si-GHET1#1-transfected group compared with the si-NC group (
To determine whether growth inhibition is responsible for observed antitumor activity of silencing GHET1, immunohistochemistry for proliferation was performed in xenograft tissues. Immunohistochemistry results showed that silencing GHET1 effectively attenuated the expression of proliferation marker Ki-67 (
Discussion
In the present study, we first reported that lncRNA GHET1 could be a potential target in osteosarcoma progression. We demonstrated that GHET1 was up-regulated in osteosarcoma tissues and cell lines. The up-regulation of GHET1 was significantly related to TNM stage, distant metastasis and lymph node metastasis. Silencing GHET1 was able to suppress the proliferation, invasion, migration of human osteosarcoma U2OS and MG-63 cells, and promote cell apoptosis in vitro, as well as inhibit the in vivo tumor growth and metastasis.
Osteosarcoma remains one of the most prevalent malignancies and has poor overall survival rates [22]. Despite with the development of diagnosis and treatment of osteosarcoma, the prognosis is still poor because of the tumor recurrence, metastasis and drug resistance [10]. Therefore, further investigation of the mediator in tumor progress and development of new therapeutic strategies are needed. Here we focused on lncRNA GHET1.
Long non-coding RNAs (LncRNAs) are reported to regulate gene transcription by binding promoter regions and changing histone markers and chromatin state [19]. In addition, lncRNAs interact with the proteins that may be important for cancer biology. Relative studies have found that lncRNAs could influence the development of tumor by affecting the epigenetic, promoting tumor invasion and metastasis including osteosarcoma [8, 23]. LncRNA CRNDE promotes osteosarcoma cell proliferation, invasion and migration by regulating Notch1 signaling and epithelial-mesenchymal transition [24]. LncRNA GAS5 suppresses cell growth and epithelial-mesenchymal transition in osteosarcoma by regulating the miR-221/ARHI pathway [11]. LncRNA MALAT1 promotes the proliferation and metastasis of osteosarcoma cells by activating the PI3K/Akt pathway [25]. LncRNA ZFAS1 sponges miR-486 to promote osteosarcoma cells progression and metastasis in vitro and vivo [26]. Gastric carcinoma high expressed transcript 1 (GHET1) is a novel lncRNA located in an intergenic region in gastric carcinoma [27], which has been reported to be overexpressed in several human cancer types including colorectal cancer [14], esophageal cancer [15], non-small cell lung cancer [16], gastric cancer [13], head and neck cancer [28] and pancreatic cancer [29], while its expression and function in osteosarcoma has not been reported previously. In our present study, GHET1 was overexpressed in osteosarcoma tissues in comparison with adjacent normal tissues. Kaplan-Meier survival analysis indicated that high GHET1 expression in osteosarcoma tissues closely related to poor overall survival of osteosarcoma patients. In addition, we demonstrated that high expression of GHET1 in osteosarcoma patients was significantly associated with TNM stage, distant metastasis and lymph node metastasis.
Previous studies indicated that GHET1 could promote breast cancer cell [12], HCC cell [30], NSCLC cell proliferation, invasion and migration. GHET1 was found to promote gastric carcinoma cell proliferation by increasing c-myc mRNA ability [31], and to promote multidrug resistance in gastric cancer cells [32]. In hepatocellular carcinoma, GHET1 silencing could inhibit the proliferation, migration, invasion and EMT of SMMC-7721 and HepG2 cells in vitro [12]. In NSCLC, down-regulation of GHET1 in A549 and SK-MES-1 cells suppressed cell proliferation, invasion and EMT process [16]. In pancreatic cancer, inhibition of GHET1 expression inhibited the proliferation capacity of tumor cells, arrested cell cycle in G
EMT is important for tumor invasion and the metastasis process [21]. During EMT, primary site epithelial cells undergo morphological and genetic alterations that take on a mesenchymal character, resulting in secondary tumor formation at another site [33]. The EMT-related metastasis has been demonstrated in various studies for osteosarcoma. Thus, controlling of EMT is considered a promising approach to inhibition of metastasis [20]. EMT can be observed by detecting the epithelial markers, such as E-cadherin, and mesenchymal markers, such as vimentin and N-cadherin [34]. It has been reported that ZEB2 binds to the E-BOX sequence in the E-cadherin promoter, leading to down-regulation of E-cadherin expression, thereby inducing EMT [35, 36]. Snail has traditionally been implicated in promoting EMT in various system of tumor progression, which is able to stimulate angiogenesis, cell proliferation and invasion in human tumor malignancies including OS [18, 37]. In our present study, we found that silencing GHET1 reduced MG-63 and U2OS cells invasion and migration capability through regulation of EMT-related marker protein expressions. QRT-PCR and western blot were used to detect the expression of EMT-related markers in experimental cells, and our results confirmed that silencing GHET1 suppressed EMT in osteosarcoma. Osteosarcoma cells transfected with GHET1 siRNA had lower mRNA and protein expression levels of ZEB2, Snail, N-cadherin, vimentin, and higher expression level of E-cadherin. In the present study, it is a limitation that we only examined the effects of GHET1 silencing on the expression of EMT-related genes, and future study may perform gene profile experiments such as the RNA-seq to further elucidate the mechanisms of GHET1-meidated osteosarcoma progression.
Distant metastasis is commonly observed in patients with osteosarcoma after surgery. It is estimated that metastasis has been found in 85% of patient with osteosarcoma [38]. The most common site of osteosarcoma metastasis is the lung. Metastatic osteosarcoma is difficult to control, and respiratory symptoms appear only in the setting of extensive involvement [39]. Osteosarcoma also metastasizes to other bone and soft tissue locations. Death from osteosarcoma is usually a result of pulmonary metastasis and respiratory failure because of widespread progression [28, 39]. In our experiments, we conducted an in vivo lung metastasis model by injecting MG-63 cells into the tail vein of nude mice, and counted the tumor nodules of the lungs. The number of tumor nodules formed by MG-63 cells transfected with si-NC was significantly more than that formed by MG-63 cells transfected with si-GHET1, which mean inhibition of GHET1 could suppress the metastasis of osteosarcoma.
Conclusions
GHET1 was up-regulated in osteosarcoma tissues and cell lines, inhibited cell apoptosis, promoted cell proliferation, invasion and migration by affecting EMT in vitro, and was correlated with the tumor growth and metastasis in vivo. GHET1 may be a potential therapeutic target of osteosarcoma treatment.
Footnotes
Conflict of interest
None.
Supplementary data
Overexpression of GHET1 promoted the proliferation, invasion and migration of osteosarcoma cells. (A, B) MG-63 and U2OS cells were transfected with pcDNA3.1 or pcDNA3.1-GHET1, at 48 h after transfection, the expression of GHET1 was measured by qRT-PCR. (C, D) CCK-8 assay showed the proliferation ability of MG-63 and U2OS cells transfected with pcDNA3.1 or pcDNA3.1-GHET1 for 48 h. (E, F) Colony formation assay was performed to measure the effect of GHET1 overexpression on cell growth. (G, H) Effect of GHET1 overexpression on cell invasion of osteosarcoma cells as measured by transwell invasion assay. (I, J) Effects of GHET1 overexpression on cell migration of osteosarcoma cells as measured by transwell invasion assay. 
