Abstract
BACKGROUND:
Epidemiological data have revealed that colorectal cancer (CRC) risk is increased in patients with Metabolic syndrome.
OBJECTIVE:
To explore the expressions of IGF-1, ERK, GLUT4, IRS-1 in MS patients with CRC and their associations with the clinical characteristics of CRC.
METHODS:
We investigated the expressions of IGF-1, ERK, GLUT4 and IRS-1 in greater omental adipose tissues of 168 MS patients with/without CRC, 85 CRC patients without MS and 98 healthy controls by RT-PCR, and analyzed the relationships between their expressions and clinical characteristics of CRC.
RESULTS:
The expression levels of IGF-1 and ERK in MS patients with/without CRC were higher while the expression levels of GLUT4 were lower compared with CRC patients without MS and healthy controls (
CONCLUSIONS:
Expressions of IGF-1, ERK and GLUT4 in greater omental adipose tissues might be useful biomarkers and predictive targets in the diagnosis of CRC.
Introduction
Metabolic syndrome (MS) is a complex syndrome of metabolic disorders and typically characterized by glucose intolerance, central obesity, hypertension and dyslipidemia. Recent findings indicated that metabolic syndrome, diabetes mellitus (DM) and colorectal cancer (CRC) shared common risk factors, such as insulin resistance and inadequate physical activity, etc. [1]. Epidemiological data revealed that colorectal cancer risk was increased in patients with Metabolic syndrome [2, 3]. Till now, hyperinsulinemia/insulin resistance was accepted as the basic mechanism responsible for metabolic syndrome. The associations between metabolic syndrome and cancer also were focused on obesity and insulin resistance.
Insulin is a major anabolic hormone which can stimulate both normal and malignant cell proliferation and its effect on cell proliferation is suggested to be with insulin-like growth factor 1 (IGF-1) stimulation [4]. Yoon et al. investigated the relationships between IGF-1 and colorectal cancer in a study of 32,826 women, they found that those with higher levels of IGF-1 were at increased risks of advanced colorectal cancer [5]. Intracellular signaling pathways starting from these receptors are also overlapping transducing signals through insulin receptor substrates 1 and 2 (IRS-1 and IRS-2) to the phosphatidylinositol-3 kinase/AKT (PI3K/Akt) and Raf-miogen-activated protein kinase (MAPK)/extracellular signal-regulated kinase (ERK) kinase MEK-ERK MAPK (Ras/Raf/MEK/ ERK) cascades, universally dysfunctioning in cancer and involved in several cancer-related processes [6, 7]. Hyperinsulinemia could increase IGF-1 bioavailability by decreasing hepatic secretion of IGF-binding protein-1 and 2 [8]. IGF receptor was overexpressed in breast and colon cancers and its activation activated ras/MAPK pathway and PI3k/AKT pathway for cell proliferation. Ras-Raf-MAPK cascade was an important center link in inducing tumor cell proliferation and tumor development. Expressions and activities of ERK and MAPK protein in colorectal cancer as well as the signal transduction pathway were closely related to the occurrence, development and treatment of colorectal cancer [9]. It was also found that insulin/IGF signaling might be involved in the maintenance of stem cell phenotype in colorectal cancer [10].
It has been accepted that visceral adipose tissue is an endocrine tissue in addition to its role in fat storage, and can release a wide range of inflammatory cytokines and other secreted proteins associated with insulin resistance and central obesity, which have also been described as prognostic factors for CRC [11, 12, 13]. ERK, IGF-1, glucose transporter 4 (GLUT4) and IRS-1 all played important roles in insulin resistance and obesity. In the present study, we investigated the expressions of IGF-1, ERK, GLUT4 and IRS-1 in greater omental adipose tissues of metabolic syndrome patients with/without colorectal cancer, CRC patients without metabolic syndrome and healthy controls, and further analyzed the relationships of IGF-1,ERK, GLUT4, IRS-1 expressions and clinical characteristics of colorectal cancer, to explore the associations between the expressions of IGF-1,ERK, GLUT4, IRS-1 and colorectal cancer.
Materials and methods
Patients
Metabolic syndrome patients with/without colorectal cancer, CRC patients without metabolic syndrome and healthy controls were enrolled in the present study from March 2014 to December 2016. All patients were treated in the affiliated hospital of Qingdao University (Qingdao, China). This study was approved by the Committee for Medical Ethics in the affiliated hospital of Qingdao University and all patients provided written informed consent. The study conformed with the Code of Ethics of the World Medical Association (Declaration of Helsinki). All cancer patients underwent radical surgery with lymph node dissection. Standard adjuvant chemotherapy was administrated to patients with presence of lymph nodes involvement. All metabolic syndrome patients were divided into two subgroups depending on the presence of CRC: with CRC and without CRC. Patients only with CRC were in CRC group. Subjects who were in Emergency Department for selective operation were as healthy controls. These patients were healthy before and without metabolic syndrome, colorectal cancer, or other diseases. We got their omental fat samples during their operations.
The criteria for inclusion in the group with metabolic syndrome taking into account the recommendations of the International Diabetes Federation (2005) was the presence of abdominal type of obesity (waist circumference greater than 85 cm for men and 80 cm for women – in China) in combination with at least two of the four additional criteria: increase blood serum triglyceride level of more than 1.7 mmol/l or receiving drug therapy for hypertriglyceridaemia; reduction in high density lipoproteins less than 0.9 mmol/L for men and 1.1 mmol/L for women; high blood pressure (systolic over 130 mmHg, or diastolic more than 85 mmHg, or receiving hypertension therapy); increase in fasting blood glucose more than 100 mg/dL, or receiving drug therapy for hyperglycaemia.
The diagnosis of colorectal cancer was confirmed by preoperative biopsy or postoperative pathologic results. Pathological diagnosis was performed by pathologists who were unaware of clinical findings or patient medical histories. Patients with CRC were treated primarily by surgical resection with adjuvant chemotherapy for node-positive patients and node-negative patients with adverse pathological features according to the National Comprehensive Cancer Network guidelines. Tumor staging of CRC was performed according to the sixth edition of the American Joint Committee on Cancer staging manual. Informations regarding tumor location, TNM staging, histological differentiation of tumors, vascular invasion and treatment options were collected from pathological and colonoscopic sample analyses. Advanced neoplasia was defined as either an adenoma with a diameter of at least 10 mm, a tubulovillous or villous adenoma, an adenoma with high-grade dysplasia, or lesions with invasive features.
Inclusion criteria: (1) Patients with colorectal cancer were diagnosed according to pathological results, and did not receive radiotherapy, chemotherapy or other treatment for cancer before operation and all CRC presented as adenocarcinomas; (2) Oral drugs for hypertriglyceridaemia, hypertension and hyper-glycaemia were not used before and during operation; (3) Diet and weight-loss drugs were not received in the past 3 months; (4) All patients provided informed consent. Exclusion criteria: (1) Patients with acute inflammation, chronic liver and kidney damage, acromegaly, hyperthyroidism, polycystic ovary syndrome, Cushing syndrome and other endocrine diseases, and patients taking glucocorticoids, thyroid hormones, non steroidal anti-inflammatory drugs, diuretics and other drugs that affect blood sugar and insulin; (2) Familial adenomatous polyposis, hereditary nonpolyposis colorectal and anal cancer cases; (3) Patients had a history of familial malignancy or other synchronous malignancy (such as ovarian cancer, prostate cancer, breast cancer), were excluded.
Clinical data
Detailed clinical data were conducted within 2 weeks before operation. Data collection included history of smoking, alcohol consumption, history of DM and hypertension, weight, height, waist circumference, blood pressure, aspartate aminotransferase (AST) and alanine aminotransferase (ALT), high density lipoprotein (HDL), fasting insulin and C-peptide, low density lipoprotein (LDL), fasting plasma glucose (FPG), glycosylated hemoglobin (HbA1c), triglyceride (TG), total cholesterol, urea nitrogen, creatinine and carcinoembryonic antigen (CEA). Participants were asked to fast for at least 10 hours and to avoid smoking on the morning of the examination. FPG and PPG levels were measured by an enzymatic glucose oxidase/peroxidase colorimetric method (cat no. ECS000016; OneTouch
Tissues
Fresh omental adipose tissues were obtained from patients during surgery. For mRNA expression study tissues samples were immediately immersed in RNAlater (Ambion Inc., USA) and stored at
RNA extraction
Total RNA from omental adipose tissues was extracted using RNeasy Mini Kit (Qiagen). The concentration and quantity of total RNA were determined based on the absorbance at 260 nm using a NANO DROP spectrophotometer (ThermoScientific, USA). To assess RNA integrity, RNA integrity numbers (RIN) was measured using 2200 TapeStation Instrument and R6K ScreenTape (Agilent Technologies, Inc., Santa Clara, USA). Thermo Scientific RevertAid First Strand cDNA Synthesis Kit was used for efficient synthesis of first strand cDNA from RNA templates.
mRNA expressions of ERK, IGF-1, GLUT4 and IRS-1 by RT-qPCR. The mRNA expression levels of IGF-1 and ERK in metabolic syndrome patients with/without CRC were higher while the mRNA expression level of GLUT4 was lower than CRC patients without metabolic syndrome and healthy controls (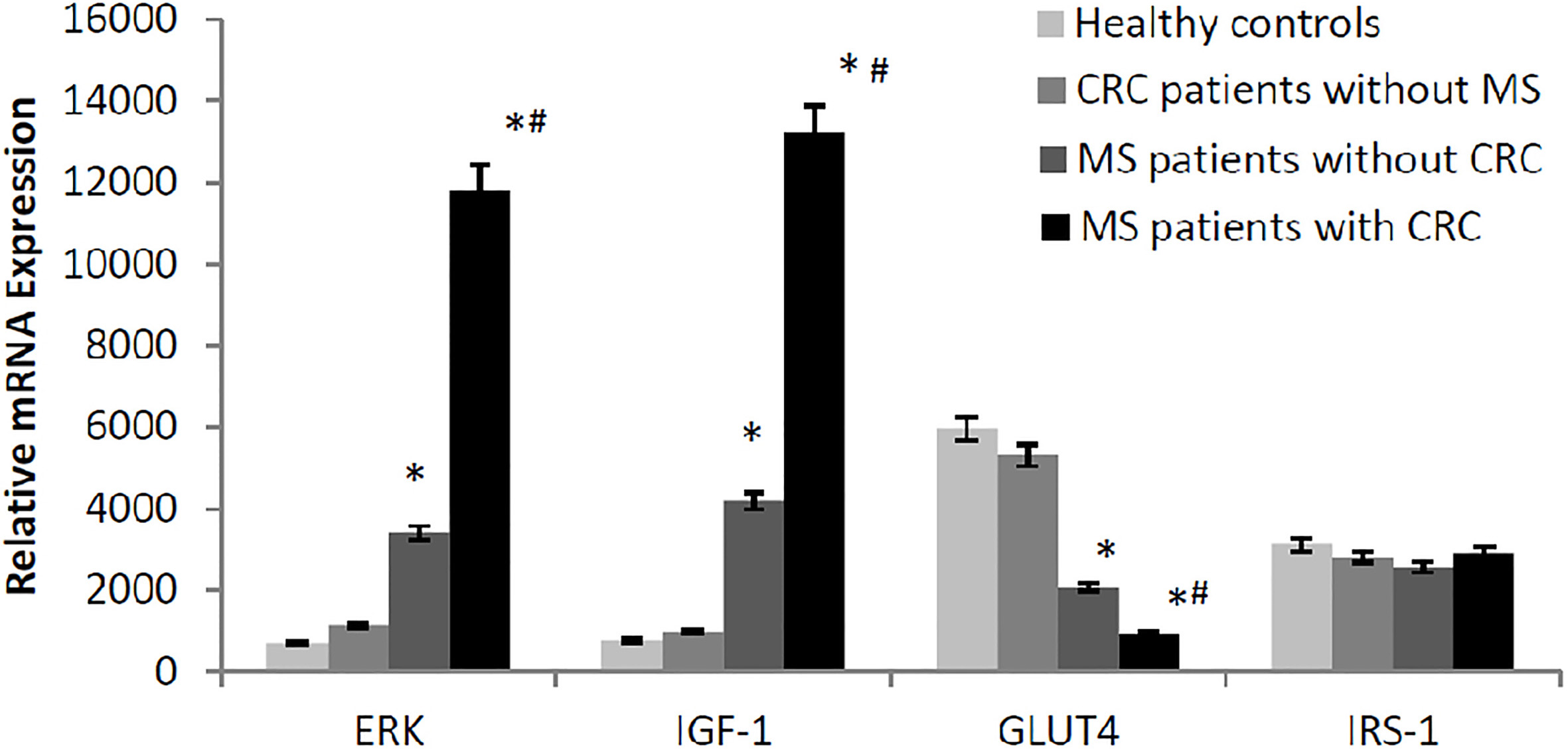
PCR was conducted in 25
A ratio of specific mRNA/Actin (Actin as a respective control) amplification was then calculated (
Statistical analysis
Statistical analysis was performed using SPSS 19.0 software. All data were expressed as mean
Results
Patients characteristics
One hundred and sixty-eight metabolic syndrome patients (including 84 patients with CRC and 84 patients without CRC), 85 CRC patients without metabolic syndrome and 98 healthy controls were initially enrolled in the study. BMI, waist circumference, hip circumference, systolic blood pressure (SBP), diastolic blood pressure (DBP), TG, TC, FPG and fasting insulin of patients with metabolic syndrome were significantly higher than CRC patients without metabolic syndrome and healthy controls (showed in Table 1). TG, FPG and fasting insulin of metabolic syndrome patients with CRC were significantly higher compared with metabolic syndrome patients without CRC (
Clinical characteristics in all subjects
Clinical characteristics in all subjects
Compared with CRC patients without MS and healthy controls,
The mRNA expression levels of IGF-1 and ERK in metabolic syndrome patients with/without CRC were higher while the mRNA expression level of GLUT4 was lower than CRC patients without metabolic syndrome and healthy controls (
Associations between expressions of ERK, IGF-1, GLUT4 and CRC
The correlation analysis between mRNA expressions of ERK, IGF-1, GLUT4 and fasting insulin (FINs) in metabolic syndrome patients with CRC
As shown in Fig. 2, in metabolic syndrome patients with CRC, the levels of FINs were positively correlated with the mRNA expression of ERK (
Associations between expressions of ERK, IGF-1, GLUT4 and clinical characteristics of metabolic syndrome patients with CRC
The associations between the expressions of ERK, IGF-1, GLUT4 and clinical parameters of metabolic syndrome patients with CRC were further analyzed. Positive associations were found between expression levels of ERK and tumor size (
The associations between relative mRNA levels of ERK, IGF-1, GLUT4 and clinical parameters of metabolic syndrome patients with CRC
The associations between relative mRNA levels of ERK, IGF-1, GLUT4 and clinical parameters of metabolic syndrome patients with CRC
The correlation analysis between mRNA expressions of IGF-1, ERK, GLUT4 and FINS. A: the correlation analysis between mRNA expressions of IGF-1 and FINS. B: the correlation analysis between mRNA expressions of ERK and FINS. C: the correlation analysis between mRNA expressions of GLUT4 and FINS. D: the correlation analysis between mRNA expressions of ERK and IGF-1. The greater 
Clinical characteristics of metabolic syndrome, such as obesity, dyslipidemia, and insulin resistance, have been linked to an increased risk of CRC in several studies [14, 15, 16]. It was deduced that insulin resistance, hyperinsulinemia, or resultant dysregulation of IGF axis participated in the possible mechanisms of metabolic syndrome on CRC [17]. In the present study, we investigated the relationships between the expressions of ERK, IGF-1, GLUT4, IRS-1 in omental adipose tissue of metabolic syndrome patients with/without CRC, CRC patients without metabolic syndrome and healthy controls to testify the above deduction, and further analyzed the associations between these factors and the clinical characteristics of CRC. We found that expression levels of IGF-1 and ERK in metabolic syndrome patients with CRC were higher while expression level of GLUT4 was lower compared to metabolic syndrome patients without CRC. This indicated that IGF-1, ERK and GLUT4 were associated with metabolic syndrome patients with CRC. Expression levels of ERK, IGF-1, GLUT4 were associated with clinical characteristics of CRC, including tumor size, distant metastasis and advanced stages (III/IV).
Insulin has been shown to affect the growth of both normal and neoplastic epithelial cells and promote mitosis [18, 19]. High insulin levels might increase the risk of colorectal cancer by stimulating cell proliferation and/or inhibiting apoptosis through increasing the bioactivity of IGF-1 [19, 20]. Another possible mechanism by which metabolic syndrome might influence colorectal neoplasm development was through obesity-related inflammation and oxidative stress [21]. Inflammation and cytokine production by visceral adipose tissues might lead to alternation of I
Hyperinsulinaemia might downregulated insulin-like growth factor binding proteins (IGFBPs) to indirectly enhance IGF-1 bioavailability [17]. This also happened via insulin binding to IGF-1 receptors (IGF1Rs). Kushlinskii et al. found IGFBP-3 could enhance p53-dependent apoptosis after DNA damage in colorectal cancer [24]. Loss of IGFBP-3 could promote the development of colorectal adenomas with retained wild-type p53 function by inhibiting p53-dependent apoptotic signals and promoting survival of abnormal cells and tumourigenesis. In our results, increase level of IGF-1 in great omental adipose tissue of metabolic syndrome patients with CRC was consistent with previous findings.
Previous studies also found that the effect of insulin and IGF-1 was partly mediated by the activation of ras pathway and this also could enhance cellular sensitivity to other growth factors [25]. Ras-Raf-MAPK cascade was an important center link in inducing tumor cell proliferation, cells differentiation, blood vessel formation and tumor development [26, 27]. ERK was an important part of intracellular signaling pathways of Ras-Raf-MAPK cascade [28, 29]. Expressions and activities of ERK and MAPK protein in colorectal cancer as well as the signal transduction pathway were closely related to the occurrence, development and treatment of colorectal cancer [30, 31]. In the present study, expressions of ERK in great omental adipose tissue of metabolic syndrome patients with CRC significantly increased, FINs and mRNA expression of ERK was significantly positively correlated in metabolic syndrome patients with CRC. These results indicated that ERK was positively associated with metabolic syndrome and CRC.
GLUT4 is considered to be an important signal factor of insulin resistance [32, 33]. It had been shown that levels of GLUT4 in 3t3-L1 adipocytes were decreased under the effect of insulin for a long time [34]. Our previous studies have shown that mRNA expression of IRS-1 and GLUT4 in greater omentum adipose tissues of metabolic syndrome patients were significantly reduced. The present study also showed that expressions of GLUT4 in metabolic syndrome patients with CRC obviously decreased, and fasting serum insulin and GLUT4 were negatively correlated. This indicated that hyperinsulinia and GLUT4 played their roles in the development of CRC, although the exact mechanism remained to be elucidated.
Insulin receptor substrates (IRS) are important mediators of insulin signal transduction pathway. The present study showed that there were no differences in the expression levels of IRS-1 between four groups. However, previous studies had shown that expressions of IRS-1 in subcutaneous and omental adipose tissues in patients with type 2 diabetes were lower than normal controls [35, 36]. The possible reason might be the limited number of samples in the present study. The relationship between IRS-1 and metabolic syndrome and CRC still needed further investigations.
Conclusions
In conclusion, we found that expression levels of IGF-1 and ERK in omental adipose tissue of metabolic syndrome patients with CRC were higher while expression level of GLUT4 was lower compared to metabolic syndrome patients without CRC. This indicated that IGF-1, ERK and GLUT4 were associated with metabolic syndrome patients with CRC. Expressions of IGF-1, ERK and GLUT4 in greater omental adipose tissues might be useful biomarkers and predictive targets in the diagnosis of CRC. A further investigations about the specific mechanisms with more in-depth analyses would be performed.
Footnotes
Acknowledgments
We appreciate all subjects that participated in this study.
Conflict of interest
The authors declare that they have no conflict of interest.
