Abstract
BACKGROUND:
The significance of serum dipeptidyl peptidase-IV (DPP-IV) in papillary thyroid carcinoma (PTC) has not been elucidated.
OBJECTIVE:
This study aimed to assess the role of serum DPP-IV in the carcinogenesis and prognosis of PTC.
METHODS:
The serum DPP-IV concentration was measured in 171 male patients with PTC, 81 male patients with a benign thyroid nodule (BTN), and 52 male healthy controls (HCs). Multivariate logistic regression and Cox regression analyses were performed to evaluate the correlations between variables. Receiver operating characteristic (ROC) curves were used to calculate the diagnosis accuracy.
RESULTS:
The ROC curve indicated a good performance of DPP-IV for discriminating PTC from BTN, with an area under the curve (AUC) of 0.881 (95% CI, 0.840–0.922). Serum DPP-IV demonstrated a modest performance in predicting nonstructurally persistent disease/recurrent disease (NSPRD) survival, with an AUC of 0.778 (95% CI, 0.635–0.922). A serum DPP-IV level
CONCLUSIONS:
Serum DPP-IV may be a predictive biomarker for PTC diagnosis and prognosis in Chinese male patients.
Keywords
Introduction
Thyroid carcinoma is the most common endocrine malignancy, and its incidence has increased rapidly in recent years [1, 2]. In particular, papillary thyroid carcinoma (PTC) accounts for 80–85% of all thyroid malignancies [1, 3]. Currently, effective and reliable biomarkers allowing the early prediction and prognostic intervention for PTC are not available, although several biomarkers, such as high-mobility group A1, long noncoding RNA cancer susceptibility candidate 2, and survivin DEx3, have been studied for thyroid carcinomas [4, 5, 6]. Therefore, it is very important to identify novel biomarkers that can accurately diagnose PTC and predict its prognosis.
Dipeptidyl peptidase IV (DPP-IV), also referred to as CD26 (EC 3.4.14.5) [7], is a glycosylated transmembrane type II glycoprotein [8, 9, 10]. It is able to cleave N-terminal dipeptides with either L-proline or L-alanine at the penultimate position [11]. A soluble form of DPP-IV exists in bodily fluids [12], and the tissue origin of circulating DPP-IV includes bone marrow cells [13], adipocyte cells [14], and human skeletal muscle cells [15]. DPP-IV is implicated in a variety of biological processes, including lipid metabolism [16], glucose regulation [17], autoimmunity and inflammation [18], carcinogenesis [9], and aggressiveness of cancer [19]. In addition, overexpression of DDP-IV has been found in thyroid cancers and other malignancies [1]. Moreover, DPP-IV is involved in various aspects of carcinogenesis, including tumor cell proliferation and invasion [20], metastasis [10, 21], and prognosis [22], and has been studied as a therapeutic target [7, 9]. The diverse biological activities of DPP-IV largely depend on the cell type and the microenvironment of the carcinoma [11]. Both in vitro and in vivo experiments have verified that DPP-IV accelerates endometrial carcinoma progression [9]. Furthermore, DPP-IV has been implicated in the pathogenesis of thyroid carcinomas as it exhibits significantly increased activity in PTC tissue than in follicular thyroid adenoma, thyroid nodular hyperplasia, and nontumorous tissue [23]. DPP-IV staining in tissue for the diagnosis of PTC has yielded a sensitivity of 97–100% [24] and a specificity of 95% [25]. Additionally, high DPP-IV expression in PTC tissue has been positively correlated with extrathyroidal extension and an advanced tumor stage, and negatively correlated with no evidence of disease during follow-up [1]. Given a certain role of tissue DPP-IV in the diagnosis and prognosis of various carcinomas, there is growing interest in the potential of serum DPP-IV for predicting carcinomas; however, various studies have yielded conflicting results. For example, circulating DPP-IV has been shown to be overexpressed in patients with pancreatic ductal adenocarcinoma [26]. In addition, serum DPP-IV, as an independent prognostic factor for colorectal cancer, has been found to be correlated with an unfavorable overall and disease-free survival [22]. In contrast, another study found that, compared with healthy controls, the serum DPP-IV level was lower in patients with esophageal squamous cell carcinoma, and a low DPP-IV level predicted a poor prognosis [27]. Furthermore, for patients with Ewing sarcoma, there is no significant difference in serum DPP-IV activity between healthy controls and patients with osteosarcoma [28]. Thus, for PTC, the diagnostic applicability of circulating DPP-IV has not been established.
In this study, we investigated the role of the serum DPP-IV level in patients with PTC versus patients with a benign thyroid nodule (BTN) and healthy individuals, and its predictive performance for the diagnosis and prognosis of PTC was also assessed.
Materials and methods
Study population
Between March 2012 and March 2013, male patients with PTC at The First Hospital of Jilin University were consecutively enrolled. In addition, consecutive subjects with a BTN from the Physical Examination Department were recruited. The diagnosis of PTC or BTN was based on radiological evidence from ultrasound, computed tomography (CT) scanning, and magnetic resonance imaging as well as histopathological results of fine-needle aspiration biopsy or thyroid tissue after operation. Volunteers without thyroid diseases or malignant diseases were enrolled as healthy controls (HCs) and donated their blood samples at the Physical Examination Department. All the patients with PTC had newly diagnosed primary PTC. The exclusion criteria included the following: 1) carcinomas other than PTC; 2) metastases from another carcinoma to the thyroid; 3) radiation therapy at any time before admission; 4) severe comorbidities, such as renal failure, heart failure, liver cirrhosis, and malignant hematologic disease; 5) insufficient data. Approval was obtained from the Ethics Committee of The First Hospital of Jilin University, and all subjects gave their informed consent to participate.
Tumor, lymph node, metastasis (TNM) stages of PTC were graded according to the 7
Patients were routinely followed up every three months after surgery during a four-year follow-up until March 2017. All patient data were originated from a thyroid carcinoma database. During follow-up, neck ultrasound and lung CT were used to monitor for PTC recurrence. Both serum thyroglobulin (Tg) and thyroglobulin antibody (TgAb) levels were measured every three months. The disease status during the follow-up was defined based on the modified criteria as follows [1]: 1) no evidence of disease, Tg
Blood samples and determination of serum DPP-IV concentration
Venous blood samples were taken from each patient with PTC at the time of diagnosis and from the controls (BTN and HCs). Blood samples were placed at room temperature for 30 min for coagulation, followed by centrifugation at 3000
As described previously [29], the assay for the determination of DPP-IV enzymatic activity was conducted in triplicate in 50 mM Hepes buffer (200 mM NaCl, 10 mM ZnCl
Clinicopathological characteristics of the male subjects,
(%)
Clinicopathological characteristics of the male subjects,
BTN, benign thyroid nodule; HC, healthy control; PTC, papillary thyroid carcinoma; CLNM, central lymph node metastasis; LLNM, lateral lymph node metastasis; TNM, tumor, lymph node, metastasis.
The correlation between the serum DPP-IV level and clinicopathological characteristics by binary univariate logistic regression analysis
DPP-IV, dipeptidyl peptidase-IV; OR, odds ratio; CI, confidence interval; PTC, papillary thyroid carcinoma; BTN, benign thyroid nodule; CLNM, central lymph node metastasis; LLNM, lateral lymph node metastasis.
Diagnostic performance of serum DPP-IV in differentiating PTC from BTN and/or HC
DPP-IV, dipeptidyl peptidase-IV; AUC, area under the curve; BTN, benign thyroid nodule; HC, healthy control; PPV, positive predictive value; NPV, negative predictive value; LR
SPSS (version 18.0) and GraphPad Prism software (version 5.01) were used for statistical analyses. Only male subjects were enrolled to avoid the influence of gender. Continuous data are expressed as the mean
Results
Patient characteristics
In this study, a total of 171 patients with PTC, 81 with a BTN, and 52 HCs were included. The clinicopathological characteristics are summarized in Table 1. Subjects with a BTN (53.30
Comparison of serum DPP-IV levels among groups
The serum DPP-IV concentration was 207.9
Correlation between the serum DPP-IV level and clinicopathological characteristics
Binary univariate logistic regression analysis showed that the serum DPP-IV level was positively correlated with malignancy, capsule invasion, CLNM, vascular invasion, and TNM stage (III
Serum dipeptidyl peptidase-IV (DPP-IV) levels in healthy controls (HCs), patients with a benign thyroid nodule (BTN), and patients with papillary thyroid carcinoma (PTC). The black horizontal lines indicate the mean values of measurements, and the error bars indicate standard deviations. Data were statistically analyzed by the ANOVA test.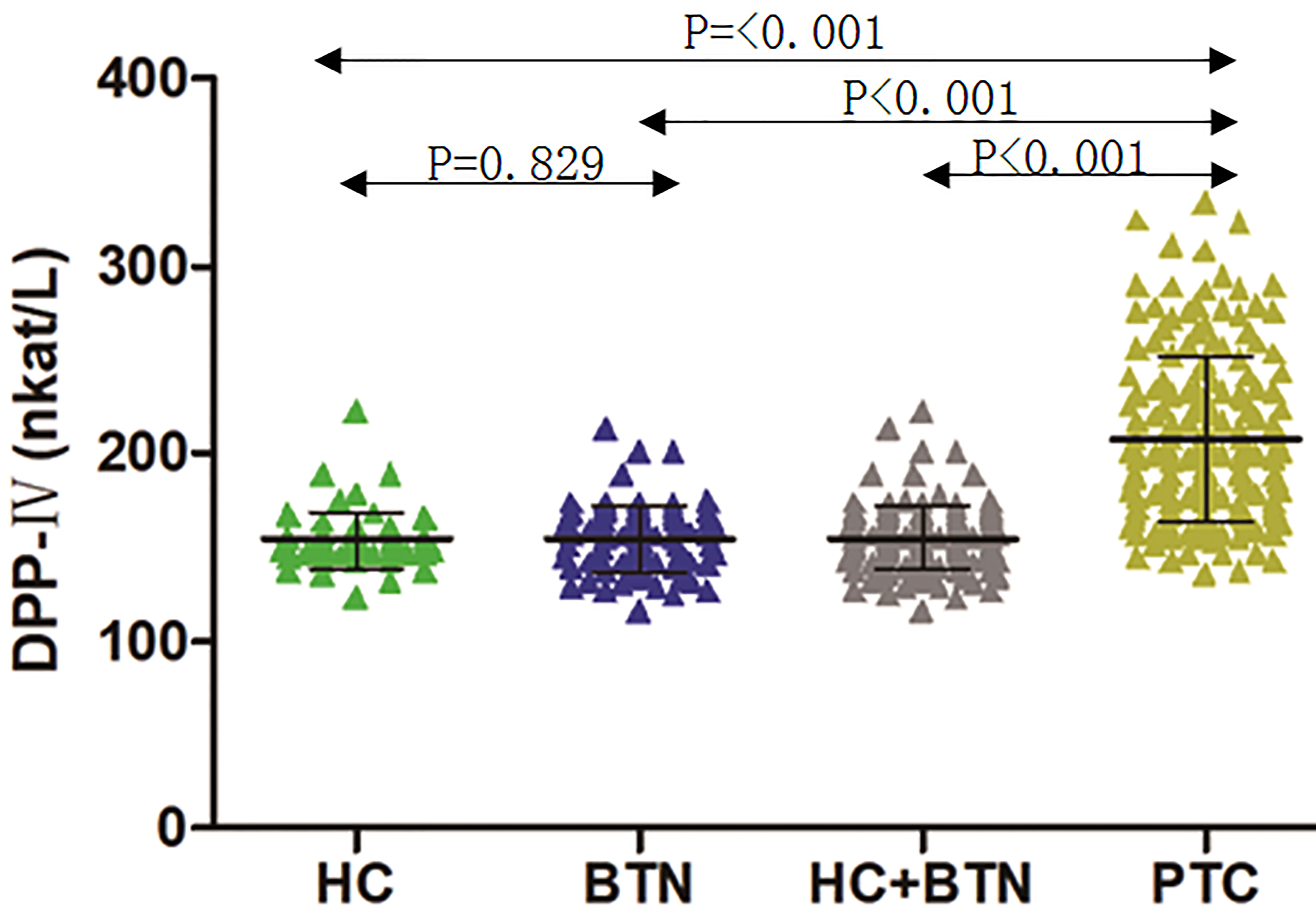
The capability of serum DPP-IV in the differential diagnosis between PTC vs. HCs and/or BTN by ROC curves. A. PTC vs. HC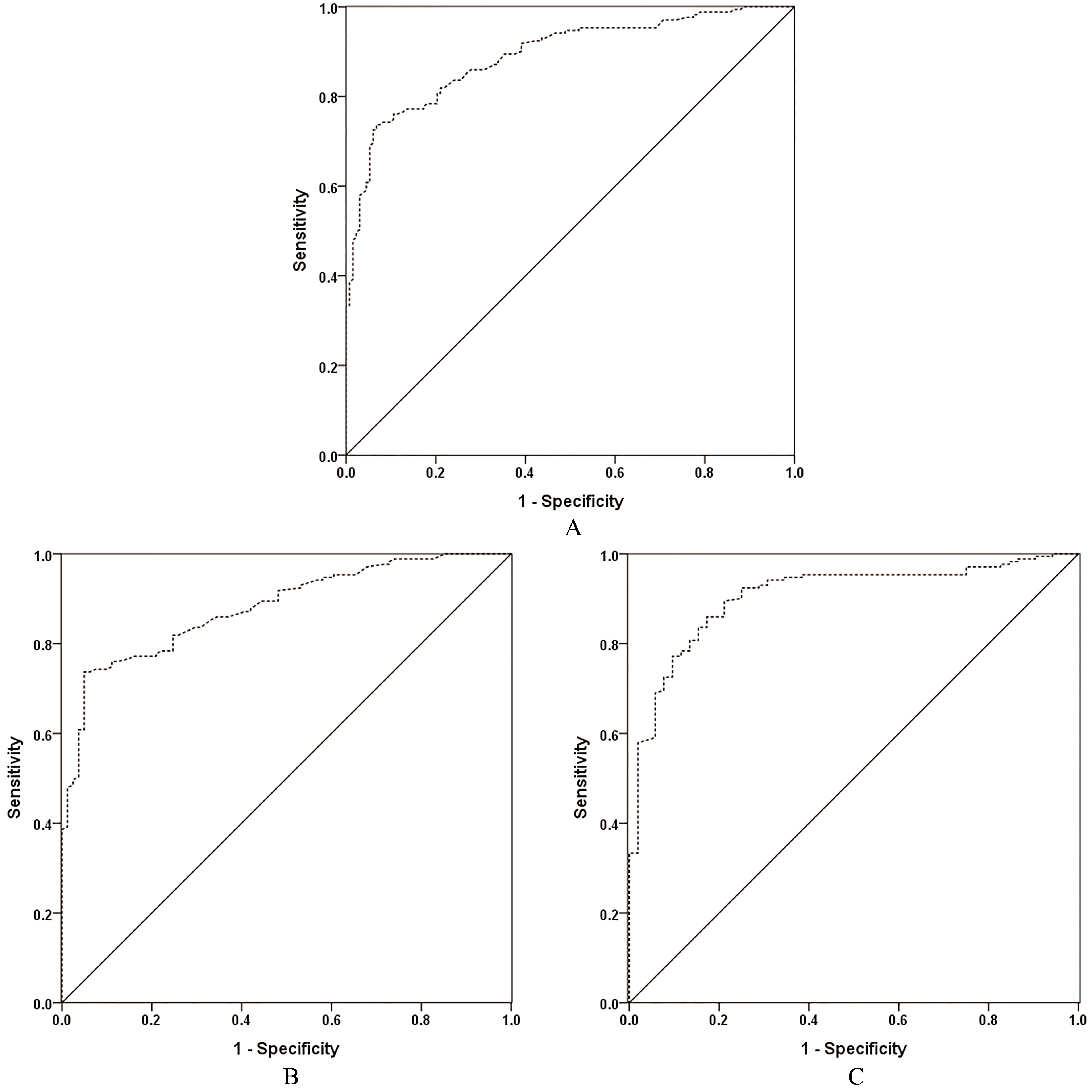
ROC curves were used to assess the validity of the serum DPP-IV level in diagnosing PTC (Table 3, Fig. 2). ROC curves revealed that serum DPP-IV yielded an AUC of 0.881 (95% CI, 0.840–0.922) at the cutoff value of 174.70 nkat/L for differentiating PTC from BTN. Moreover, at the same cutoff value, DPP-IV yielded an AUC of 0.891 (95% CI, 0.855–0.926) for the differential diagnosis of PTC from BTN
Clinicopathological features of patients with PTC by serum DPP-IV stratification
Considering the optimal cutoff value of DPP-IV as 174.70 nkat/L for differentiating PTC from BTN, patients with PTC were stratified into two subgroups: those with a low level of serum DPP-IV (
Clinicopathological features of patients with PTC by serum DPP-IV stratification at 174.70 nkat/L,
(%)
Clinicopathological features of patients with PTC by serum DPP-IV stratification at 174.70 nkat/L,
DPP-IV, dipeptidyl peptidase-IV; CLNM, central lymph node metastasis; LLNM, lateral lymph node metastasis; TNM, tumor, lymph node, metastasis.
Serum DPP-IV levels in the patients with CLNM or different TNM stages. A. Patients with CLNM vs. without CLNM; B. Patients with an early TNM stage (I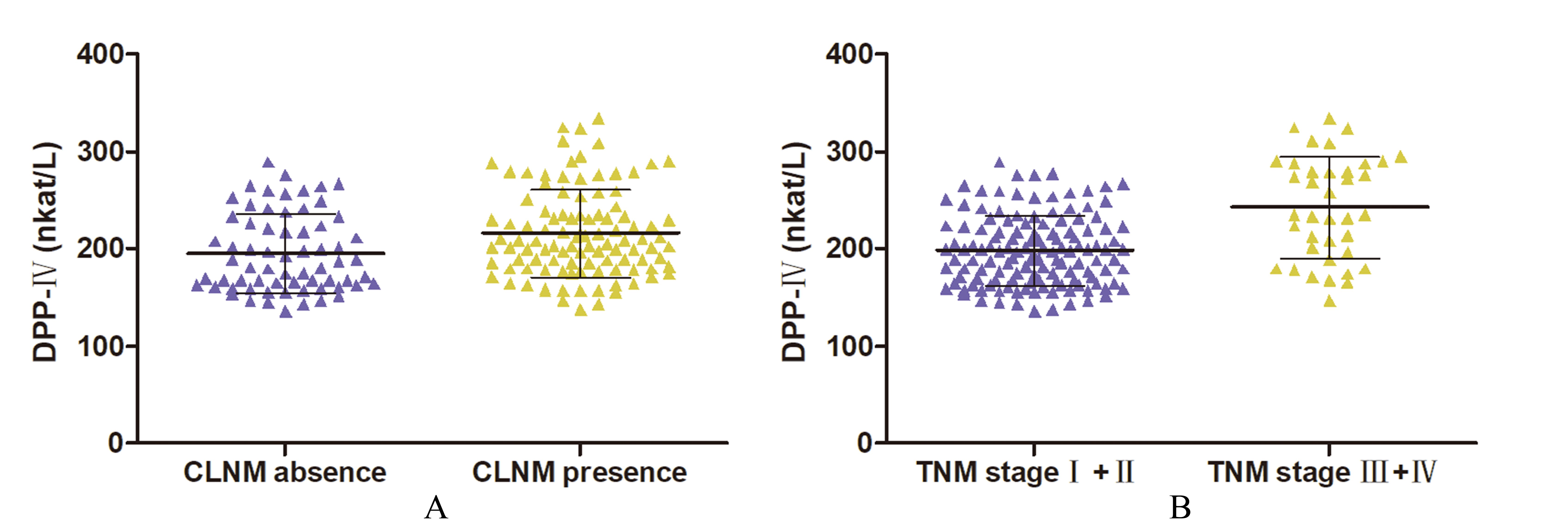
Receiver operating characteristic curve analysis for structurally persistent disease and recurrent disease in patients with papillary thyroid carcinoma predicted by the level of serum dipeptidyl peptidase-IV (AUC 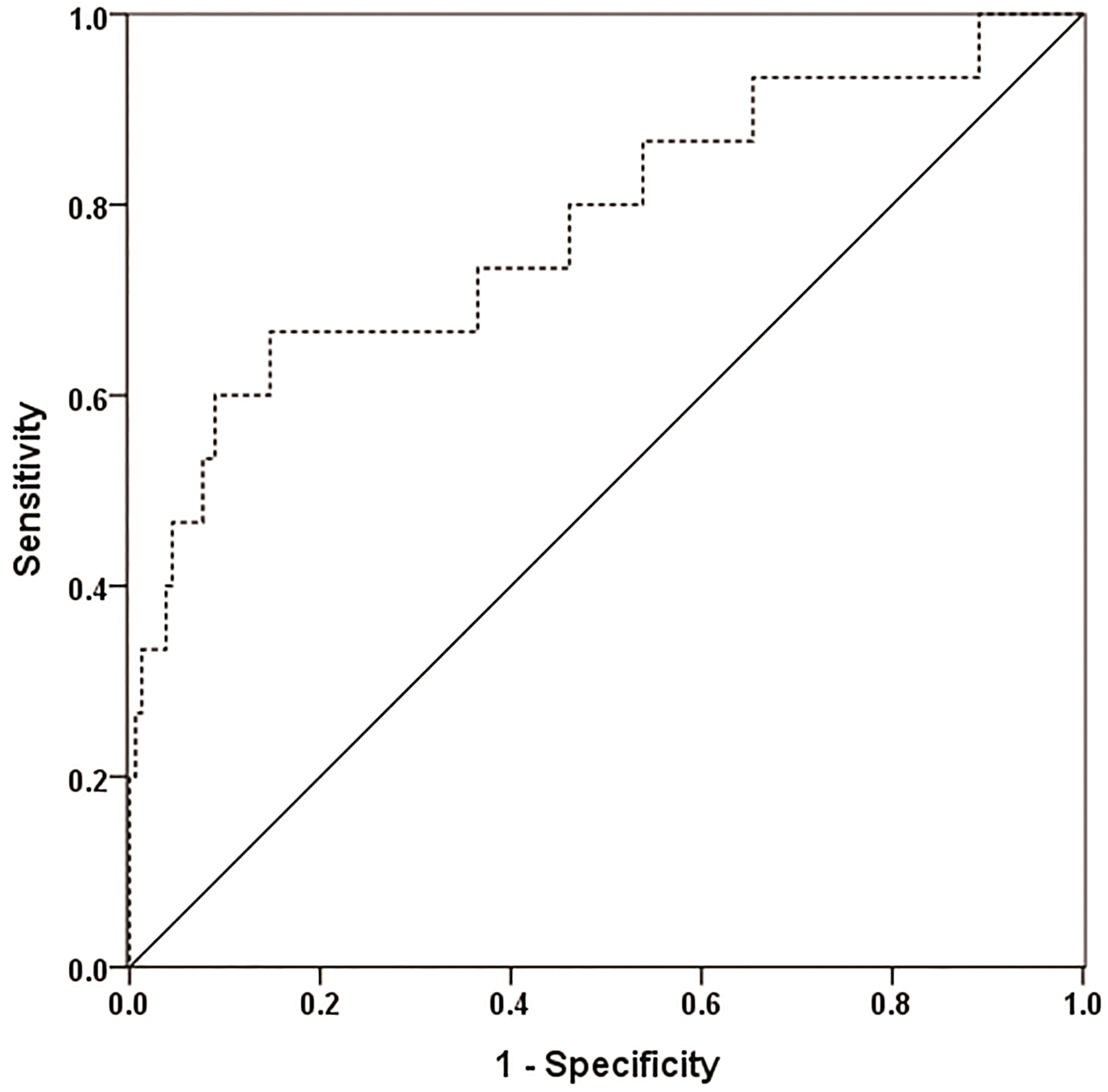
Patients with CLNM had a higher DPP-IV level than patients without (215.80
The patients were followed up for four years. By ROC curve analysis, serum DPP-IV demonstrated a moderate performance in predicting SPRD at the cutoff value of 250.0 nkat/L, with an AUC of 0.778 (95% CI, 0.635–0.922), a sensitivity of 66.7%, a specificity of 85.3%, a positive predictive value of 66.7%, a negative predictive value of 85.3%, a positive likelihood ratio of 4.537, and a negative likelihood ratio of 0.390 (Fig. 4).
Patients with PTC had a mean NSPRD survival time of 46.65
Clinicopathological features of patients with SPRD or NSPRD,
(%)
Clinicopathological features of patients with SPRD or NSPRD,
CLNM, central lymph node metastasis; LLNM, lateral lymph node metastasis; SPRD, structurally persistent disease/recurrent disease; NSPRD, nonstructurally persistent/recurrent disease.
Nonstructurally persistent disease/recurrent disease (NSPRD) survival time curve in the male patients with papillary thyroid carcinoma based on the cut-off value of dipeptidyl peptidase-IV (DPP-IV) at 250.0 nkat/L. SPRD was observed in 17 out of 171 subjects, among which 5 of 138 patients had a DPP-IV level 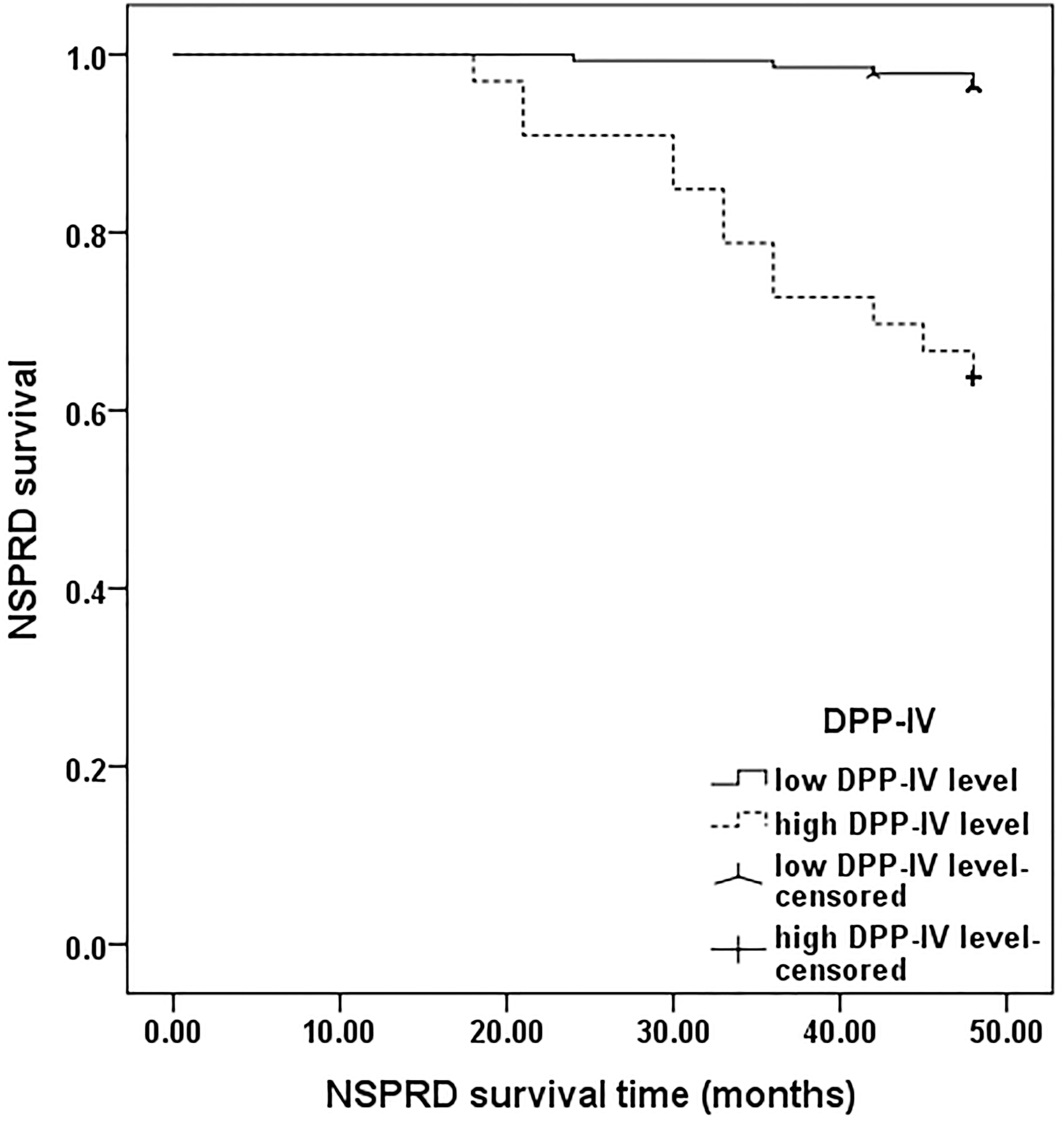
Older patients (
Risk factors for predicting SPRD in patients with PTC by Cox proportional hazards regression analysis
SPRD, structurally persistent disease/recurrent disease; PTC, papillary thyroid carcinoma; OR, Odds ratio; CI, confidence interval; BMI, body mass index; CLNM, central lymph node metastasis; LLNM, lateral lymph node metastasis.
Analyzed by the univariate Cox proportional hazard regression model, age (
Discussion
Although PTC typically has a good prognosis, there is a risk of it differentiating into aggressive and lethal thyroid carcinomas, e.g. poorly differentiated thyroid carcinoma or anaplastic thyroid carcinoma [31]. Moreover, PTC has shown a high recurrence rate (30%) and a high cancer-related mortality rate (8%) over a long-term follow-up period [32]. Therefore, early diagnosis and intervention is of pivotal significance. In this study, we found that the serum DDP-IV expression was significantly higher in patients with PTC than in patients with a BTN and HCs. Serum DPP-IV cutoff values of 174.70 nkat/L for PTC diagnosis and 250 nkat/L for SPRD were identified. Patients were more likely to develop SPRD if they had a larger tumor size (
Tissue DPP-IV can be utilized as a diagnostic biomarker for discriminating benign neoplasms from malignancies. An immunohistochemistry assay has demonstrated that DPP-IV has a sensitivity of 82.81% for the diagnosis of epithelial ovarian carcinoma, which was increased to 97.67% if detected by mRNA analysis [33]. In addition, tissue DPP-IV is associated with cancer progression and aggressiveness. In colorectal cancer, DPP-IV activity is positively associated with an advanced TNM stage, the degree of differentiation, and the development of metastasis [34]. Moreover, a higher tissue DPP-IV level predicts a worse overall survival as compared to a lower level in colorectal cancer or PTC patients [34]. Taken together, these findings verify the importance of DPP-IV expression in carcinogenesis, carcinoma aggressiveness, and prognosis of diverse cancers.
However, the use of molecular analysis of tumor tissues and cells is restricted in daily clinical work due to its invasive properties and unavailability. Herein, relative to the detection of tissue DPP-IV, determination of the serum DPP-IV level is more convenient, less invasive, and more cost-efficient. Owing to the heterogeneity of previous studies, no consensus has been achieved regarding the level of serum DPP-IV in the prediction of carcinogenesis and progression. Some studies have revealed unfavorable outcomes attributed to a high DPP-IV level, while others have supposed inauspicious consequences resulting from a low DPP-IV level. In colorectal carcinoma, serological DPP-IV activity has been inversely correlated with worse overall and disease-free survivals [22]. On the contrary, patients with metastatic prostate cancer had a reduced serum DPP-IV activity compared to those with localized disease or HCs [35]. In esophageal squamous cell carcinoma, a high serum DPP-IV level represented a survival advantage compared with a low serum DPP-IV level [27]. In this study, serum DPP-IV was positively associated with PTC occurrence, aggressiveness, and prognosis. Especially, serum DPP-IV was able to serve as a biomarker for PTC diagnosis at a cutoff value of 174.70 nkat/L and for prognostic prediction at a cutoff value of 250 nkat/L. Busek et al. found that newly diagnosed diabetic patients with pancreatic ductal adenocarcinoma have higher serum DPP-IV levels than those of patients with diabetes mellitus only [26]. This result was similar with ours, which showed that the serum DPP-IV level was higher in PTC patients than in controls. Agreement was achieved from another study showing that an elevated serum DPP-IV level was associated with poor outcomes in colorectal cancer prognosis [22]. However, different results from ours were found in a study performed by Boccardi et al. [36], which indicated that a decrease in the serum DPP-IV level could serve as an early diagnostic marker for gastric cancers. These discrepancies may be explained by the fact that the investigators studied different types of malignancies with various assessment tools among a heterogeneous population (e.g., gender) [37]. In addition, the presence of diabetes mellitus also affects the serum DPP-IV level [26]. Therefore, diverse proportions of diabetic subjects among studies may partially contribute to the discrepancies. The findings from our study helped to make a timely diagnosis of PTC and to predict the prognosis of such patients. However, a further study with a larger sample size is needed to verify our current data.
Although DPP-IV has been implicated in carcinogenesis, the molecular mechanism is still ambiguous. Several molecular mechanisms have been studied to determine the involvement of DPP-IV in carcinogenesis. Nuclear accumulation of DPP-IV is inhibited by the loss of tumor suppressor P53, which consequently incurs plasma-membrane-associated DPP-IV-dependent lipid peroxidation, resulting in ferroptosis [16]. A high DPP-IV level is associated with the BRAF mutation in PTC, and a positive BRAF mutation is found in approximately 50% of PTC patients [38]. DPP-IV promotes epidermal growth factor-induced epithelial cell transformation and mammary carcinogenesis through induction of PIN1 expression [39]. In addition, in in vitro experiments of malignant mesothelioma cells, DPP-IV has demonstrated its tumor cell invasion ability through its interaction with alpha5beta1 integrin [11]. Further investigation is needed to elucidate the exact mechanism underlying the role of DPP-IV in carcinogenesis in the future.
This study has several strengths. First, to the best of our knowledge, it is the first study to analyze the significance of serum DPP-IV in PTC diagnosis and prognosis. Second, only male subjects were enrolled to avoid the influence of gender. Third, the eligible patients were consecutively enrolled to minimize a selection bias. Our study also has some limitations, including a relatively small sample size and the lack of female participants. Additionally, due to the lack of a standard serum biomarker for PTC, we did not set a control with a known serum PTC biomarker. Finally, considering that this was a single-center study conducted in China, caution must be exercised when generalizing the results of this study in different populations.
In conclusion, serum DPP-IV may be a predictive biomarker for male patients with PTC for both diagnosis and prognosis. Confirmation of these findings will require further investigation in a prospective study with a larger sample size, including female participants. Also, the mechanisms by which DPP-IV is involved in the progression and aggressiveness of PTC need to be elucidated.
Footnotes
Acknowledgments
This study was supported by Special Projects for Health in Jilin Province (2018SCZWSZX-039; 2018 SCZWSZX-050), Projects of the Education Department of Jilin Province (20150125; 2016453; 2016482), and a Project of the Health Department of Jilin Province (20150125).
Conflict of interest
The authors have no conflicts of interest to declare in relation to this article.
