Abstract
PURPOSE:
The evaluation of molecular targets in gastric cancer has demonstrated the predictive role of HER2 amplification for trastuzumab treatment in metastatic gastric cancer. Besides HER2, other molecular targets are under evaluation in metastatic gastric tumors. However, very little is known about their role in resected tumors. We evaluated the expression of HER2, EGFR, MET, AKT1 and phospho-mTOR in resected stage II–III adenocarcinomas.
METHODS:
Ninety-two patients with resected stomach (63%) or gastro-esophageal adenocarcinomas (27%) were evaluated. Antibodies anti-HER2, EGFR, MET, AKT1 and phospho-mTOR were used for immunostaining of formalin-fixed paraffin-embedded slides. Using FISH, HER2 amplification was evaluated in cases with an intermediate (
RESULTS:
EGFR overexpression (11%) was a poor prognostic factor for overall survival (3-year OS: 47% vs 77%; Log-Rank
CONCLUSIONS:
EGFR is confirmed a poor prognostic factor in resected gastric cancers. We firstly describe a mutually exclusive overexpression of EGFR and AKT1 with potential prognostic implications, suggesting the relevance of this pathway for the growth of gastric cancers.
Introduction
The incidence of gastric cancer in Europe has been estimated in 140 new cases per 100,000 persons [1]. Although the new diagnoses of stomach cancer are declining in western countries, the relative incidence of adenocarcinomas of the gastro-esophageal junction (GEJ) is increasing [2, 3]. In Italy, stomach and GEJ cancers are the fifth more lethal neoplasms [1]. Surgery is the mainstay of treatment in localized tumors but often is not curative and most of stage II and III patients recur despite a complete resection (R0). Perioperative chemotherapy is helpful to reduce the risk of relapses and prolongs the overall survival of patients with gastric cancer [4, 5]. Tumors of the GEJ may benefit from the addition of a neoadjuvant radiation treatment [6, 7]. Metastatic tumors are treated with palliative chemotherapy and those with HER2 amplification are candidates to receive trastuzumab since it can prolong their survival (median OS 13.8 vs 11.1 months; hazard ratio (HR) 0.74, 95% CI, 0.60–0.91;
However, in locally advanced (stages II–III) gastric and GEJ cancers, limited data are available regarding the expression of tyrosine kinase receptors and the activation of the related intracellular signaling pathways. Moreover, an integrative analysis of the expression of multiple markers is missing in this setting of patients. Therefore, we evaluated the expression of HER2, EGFR, MET, AKT1 and phospho-m-TOR in a series of 92 patients who underwent to surgical resection for stages II and III gastric and GEJ adenocarcinomas. These biomarkers have been chosen with the idea of a possible targeted treatment since inhibitors are available in the clinical and research settings.
Materials and methods
Patients
This retrospective evaluation included 92 consecutive patients who underwent to surgical resection at University Hospital of Pisa. Evidence of distant metastases and macroscopic residual disease (R2) after surgical resection were exclusion criteria. All tumors were adenocarcinoma and originated from the stomach and the GEJ. The histological diagnosis was assessed according to Laurén’s classification, whereas stage and the grade of differentiation were defined according to 7th edition of the American Joint Committee on Cancer (AJCC) staging system (TNM). Patients’ Eastern Cooperative Oncology Group (ECOG) performance status (PS) was assessed. The ethical committee of the northwest Tuscany approved this protocol and patients gave their written consent to this molecular characterization that was conducted in accordance to the Declaration of Helsinki.
Immunohistochemistry
Formalin-fixed paraffin-embedded (FFPE) surgical specimens were retrieved and a pathologist (A.P.) reviewed the diagnosis and assessed the archival material for quantity and quality. Four
Fluorescent in-situ hybridization
Fluorescent in-situ hybridization (FISH) was performed to determine HER2 amplification in cases with a 2+ positive immunohistochemistry staining. The pharmDx test kit (Dako) was employed according to vendor instructions. The data were analyzed according to what previously reported by Hofmann et al. [10].
Statistical analysis
The correlation between protein expression and clinical features has been analyzed using the Fisher’s exact or the
Results
Patients
The median age at the diagnosis was 67.5 years (range 33–85; patients’ characteristics are summarized in Table 1). All tumors were adenocarcinoma, 25 (27%) occurred in the GEJ and 67 (73%) in the stomach. GEJ tumors were more common in males (34%) than in female (15%) with a trend towards significance (Chi-Square
Patients’ characteristics
Patients’ characteristics
Nc
(A) Flow chart of the performed tests. The flow chart indicates the number of patients evaluable for each protein, the cases with a positive staining and the tumors with overexpression of HER2, MET, EGFR, AKT1 and phospho-mTOR. 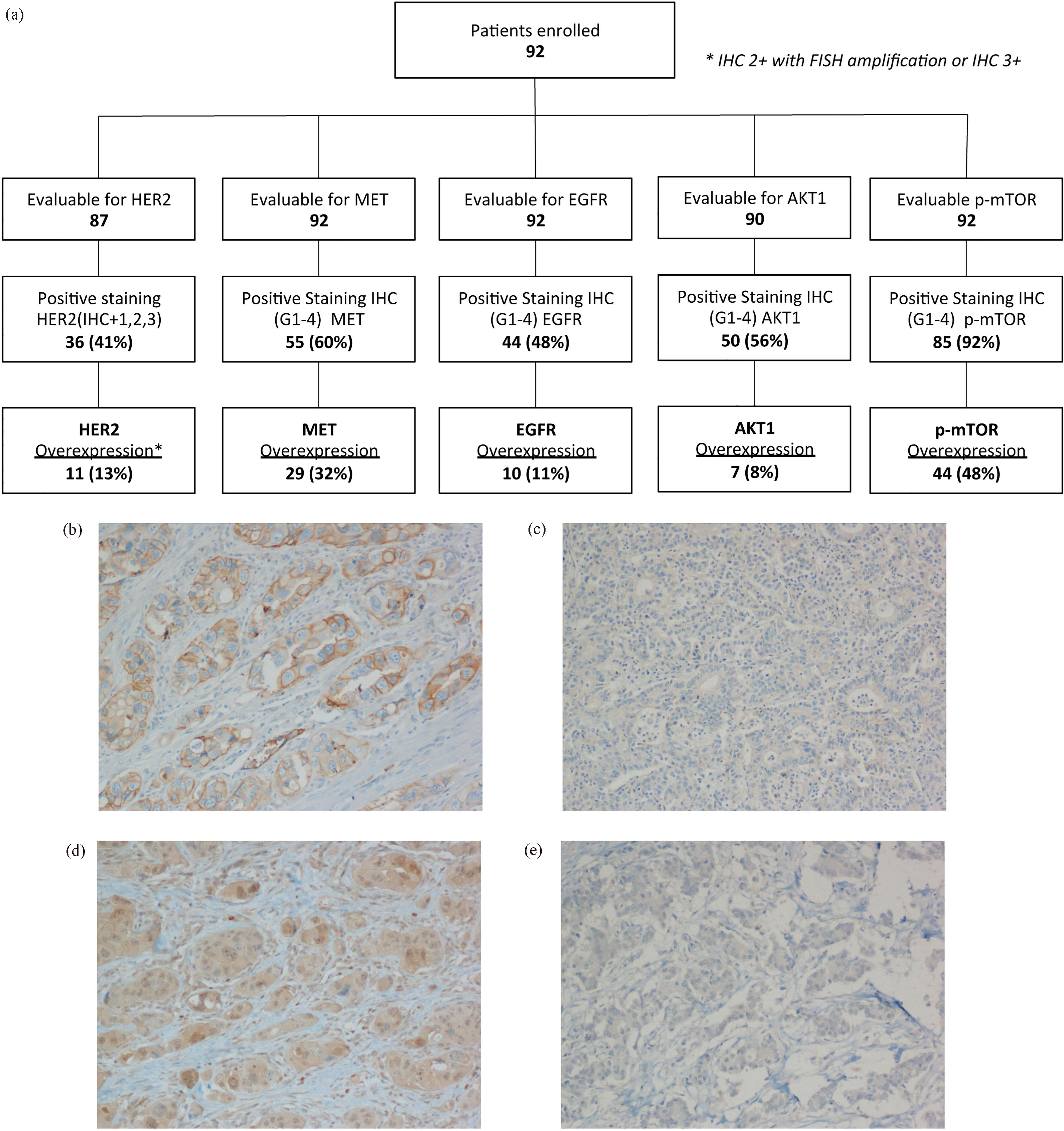
EGFR was overexpressed in 11% of the patients (Fig. 1A–C). Patients with EGFR overexpression had a worse OS than those without (3-year OS: 47% and 77%, respectively; Log-Rank
Kaplan-Meier curves for overall survival and disease free survival of tumors with (EGFR+) and without (EGFR-) EGFR overexpression (a and b, respectively); with (HER2+) and without (HER2-) HER2 amplification (c and d, respectively); with (MET+) and without (MET-) MET overexpression (e and f, respectively).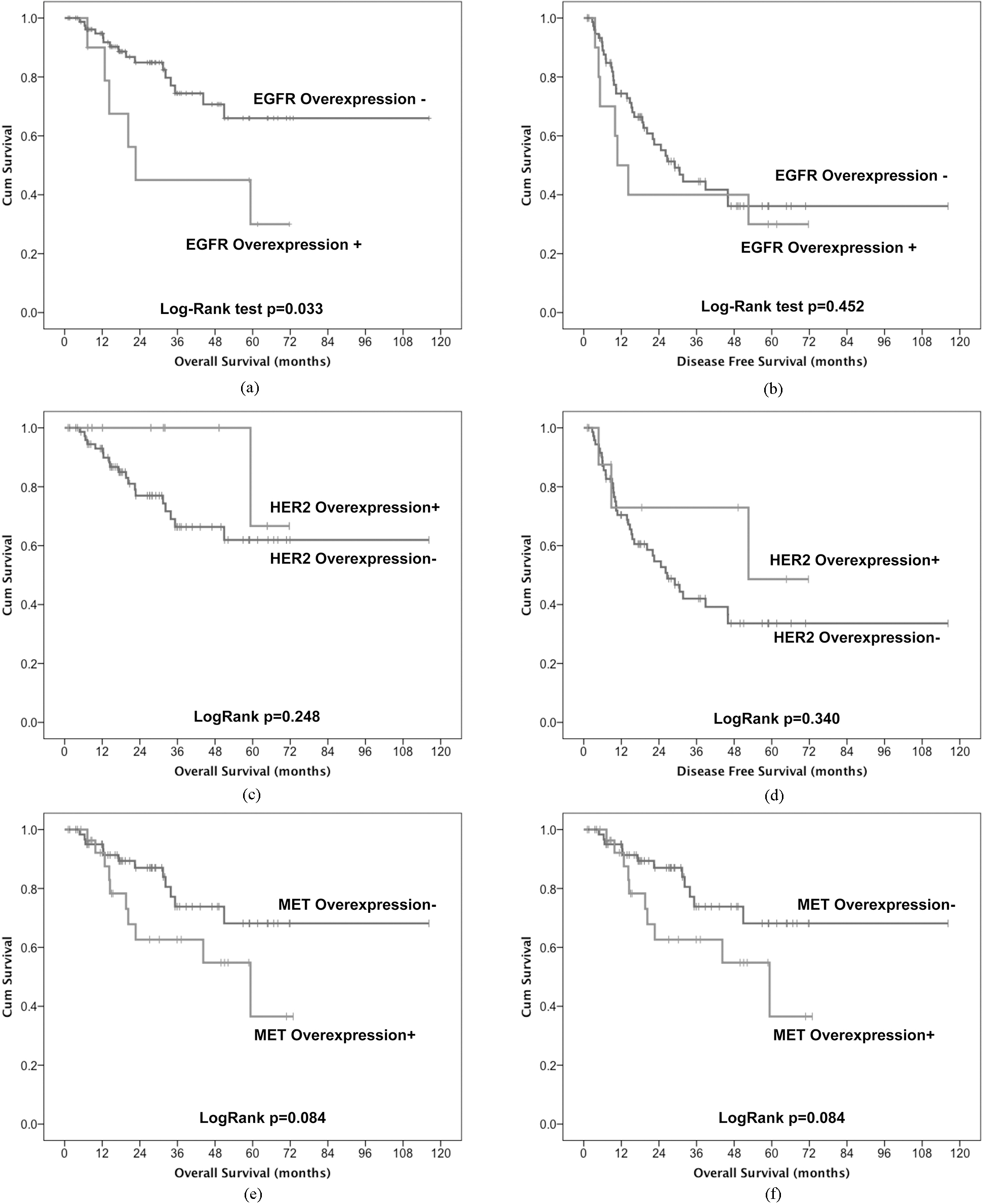
Univariate and multivariate survival analysis for OS
HER2 staining was strongly intense (
MET was overexpressed in 32% of the patients. A trend for a worse survival was appreciated in tumors with MET overexpression for OS (3-year OS MET+
AKT1 was overexpressed in only 8% of the tumors (Fig. 1D and E). There were not significant differences in OS (Fig. 3a) and in DFS (Fig. 3b) between patients expressing or not AKT1. The low number of tumors with AKT1 overexpression limited the interpretation of survival analysis.
Kaplan-Meier curves for overall survival and disease free survival of tumors with (AKT1+) and without (AKT1-) AKT1 overexpression (a and b, respectively); with (AKT1orEGFR Overexpression+) or without (AKT1orEGFR Overexpression-) EGFR or AKT1 overexpression (C and D, respectively).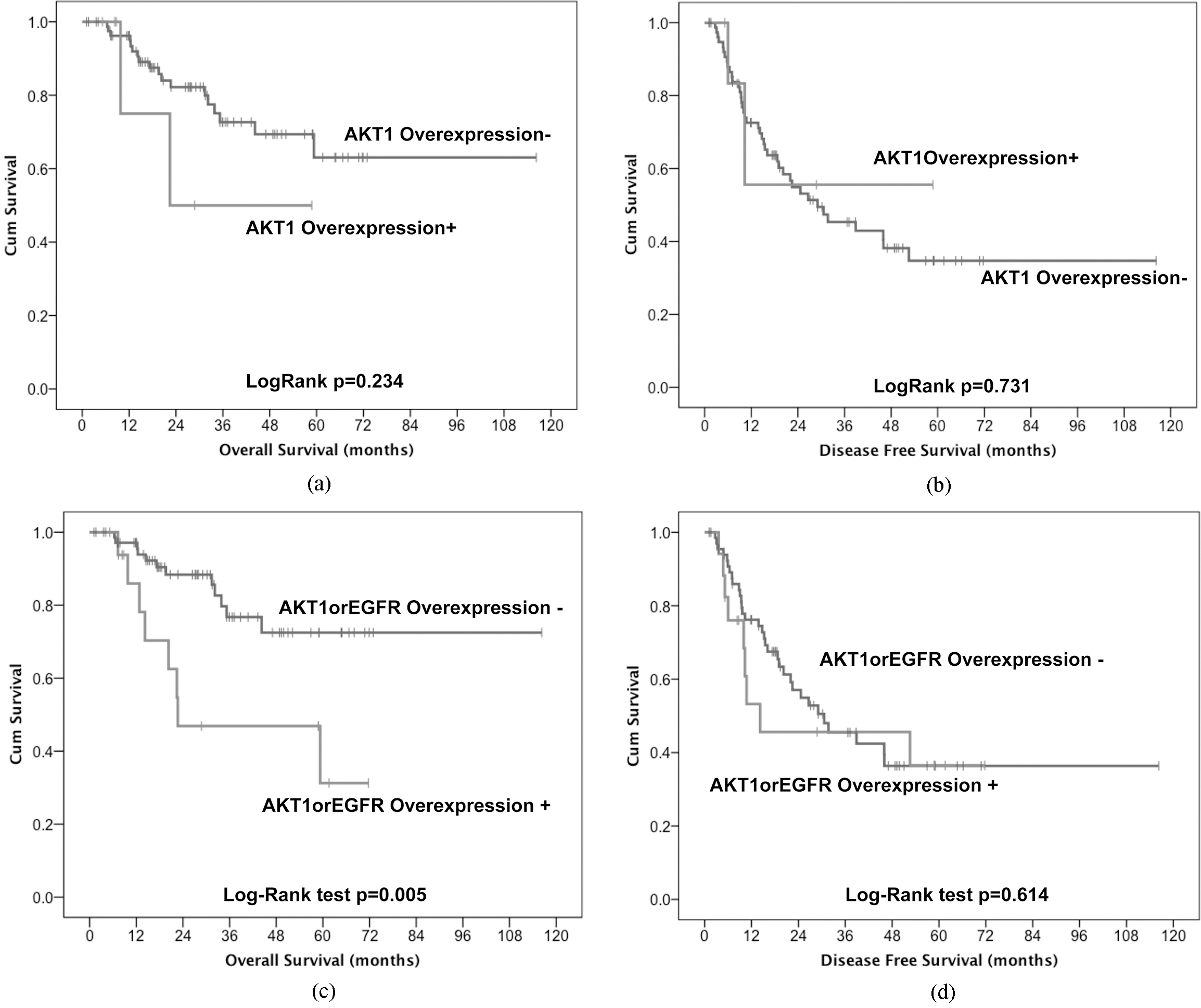
Phospho-mTOR was overexpressed in 48% of tumors. P-mTOR overexpression did not correlate with none of the patient characteristics, with survival or with the overexpression of the other biomarkers studied (p-mTOR/EGFR
The combined analysis of the 5 biomarkers demonstrated that the overexpression of EGFR and AKT1 were mutually exclusive. The overexpression of EGFR/ AKT1 was a strongly significant poor prognostic factor for OS (3-year OS 52% in overexpressed and 79% in not overexpressed, Log-Rank
Discussion
The overexpression of EGFR was a poor prognostic factor in completely resected stages II–III gastric and GEJ cancers. EGFR and AKT1 overexpression were mutually exclusive and identified a subset of patients with a poor prognosis probably indicating the importance of this axis in determining aggressiveness of these tumors. Moreover, tumors with MET overexpression had a trend for a worse OS.
Similarly to what previously described by Rudiger Siewert et al. [20], we observed a prevalence of GEJ adenocarcinomas in males possibly related to the different exposure to risk factors such as alcohol and smoking habits. Only tumor stage was a prognostic factor in our series with implication for the construction of multivariate models (Table 2). The limited number of patients contributes to explain these limitations since the reduced statistical power of the test performed. With a median follow-up of 32 months (95% CI: 25–39) the median overall survival was not reached with 65% of patients alive after 5 years. The median DFS was 29 months with a 93% rate of complete resections (R0) indicating a clinical outcome slightly above the expectations. Indeed, comparable results have been described in phase III clinical trials of perioperative treatment from Ychou et al. that describes a 5-year OS of 58–51% [5] and from Cunningham et al. of 36.3%–23% [4], for patients who received perioperative chemotherapy or surgery alone, respectively. Given the retrospective nature of the analysis patients received different schedules of perioperative or post-operative chemotherapy. The heterogeneity of the treatment reflects the variable clinical condition of the patients and the multiple adjuvant schedules of reference changed during the long time lapse of accrual.
HER2, MET, EGFR, AKT1 and mTOR can be targets for treatment with specific drugs. Therefore, the expression of them has been largely evaluated in metastatic gastric tumors. On the contrary, limited data are available in locally advanced gastric cancers. HER2 and EGFR belong to the ErbB family of membrane tyrosine kinase receptors. HER2 ectodomain does not bind to the ligand and the receptor does not homodimerized under physiological conditions but under pathological overexpression [19]. HER2 has been largely evaluated in gastric cancers being overexpressed in about 20% of metastatic tumors [21]. The amplification of HER2 locus is observed in 7–34% of gastric cancers and is responsible for most of the cases of protein overexpression [10, 22, 23]. The prognostic role of HER2 overexpression remains controversial, even if some reports describe a poorer survival of HER2 positive metastatic gastric cancers [23, 24]. In previous reports, about 20% of the resected gastric adenocarcinomas overexpress HER2, most of them presented an intestinal histotype according to Laurén classification [23, 25, 26], similarly to what observed in our series. Therefore, it is not surprising the trend for a longer OS and DFS of tumors with HER2 overexpression/amplification. In the TOGA trial, trastuzumab has prolonged the progression free survival (PFS) and the OS of patients when given in combination with chemotherapy during the first line treatment of metastatic gastric tumors with HER2 amplification [8]. This result demonstrates that HER2 amplification is a target for therapy in gastric cancer. However, the role of trastuzumab in adjuvant therapy is still under investigation.
EGFR overexpression has been described in 9–45% of gastric cancers and has been associated with a poor prognosis [10, 27, 28, 29]. Our data confirm previous observations, since EGFR was overexpressed in 11% of completely resected gastric adenocarcinomas and was prognostic for a shorter OS. Only 2.3–16% of gastric cancers present an amplification of EGFR locus; therefore, epigenetic modifications have been claimed to explain the overabundance of EGFR overexpression [10, 19, 30, 31, 32]. EGFR mutations have been described in few cases of gastric adenocarcinoma [33], thus only 4% out of 932 cases have been reported in COSMIC database. Anti-EGFR treatments have failed to demonstrate a clinical benefit in randomized trials and showed even a possible detrimental effect [34, 35].
Copy number gains of MET locus have been described using RT-PCR techniques in 10% of stage II/III gastric adenocarcinomas [36]. Copy number gains of MET define a more aggressive subset of tumors with a shorter DFS and OS [36]. There was an inverse correlation between a higher number of MET copies and a poorer survival. Using FISH, author demonstrated that the copy number gains of MET were linked to a polysomic chromosome 7 rather than a focal amplification of MET locus [36]. Only 4 tumors out of 170 have been described with a real amplification of MET [37]. There is a direct correlation between the number of copies of MET and its protein overexpression. Indeed, MET overexpression has been described in 8–24% of gastric carcinomas [38, 39]. Similarly, we observed MET overexpression in 29% of locally advanced gastric cancer. The trend for a more aggressive behavior of MET overexpressing tumors is in line with previous reports that describe a worse prognosis for these patients [36, 37, 38, 39].
The activation of MET, EGFR or HER2 can activate the axis PI3K-AKT1-mTOR in vitro and determine cell proliferation [40]. The hyperphosphorylation of mTOR has been described in 46.5% of stage II/III gastric adenocarcinoma and associated with the presence, at the diagnosis, of more metastatic lymphnodes and a poorer survival [41, 42]. We observed overexpression of phospo-mTOR in 48% of tumors. There was not a correlation between mTOR phosphorylation and patients’ outcome. In different kind of tumors, the level of mTOR phosphorylation can represent a predictive marker of response to treatment with everolimus [43]. There was not a correlation between the level of mTOR phosphorylation and the overexpression of HER2, EGFR, MET and AKT1. A possible explanation is that the phosphorylation of mTOR is not an evidence of PI3K-11-mTOR pathway activation. Indeed, there are 2 multi-protein complexes that include mTOR: mTORC1 and mTORC2. Besides mTOR, the first complex includes RAPTOR, MLST8, PRAS40 and DEPTOR; whereas, the second RICTOR, G
The activation of AKT1 induces cell proliferation through the stimulation of cell cycle progression and inhibits the intrinsic apoptotic pathway. Hyper phosphorylation of AKT1 has been described in 29–68% [44, 45] of gastric adenocarcinomas and it is associated a poor prognosis [45]. Although the role of AKT1 phosphorylation has been largely elucidated in the transduction of proliferative signals, also the amount of AKT1 protein can stimulate the proliferation of neoplastic cells. Indeed, copy number gains of AKT1 have been described in gastric cancers [46]. In our series, AKT1 was overexpressed in 8% of tumors and associated with a trend for a poor overall survival. Interestingly, we noticed that AKT1 and EGFR overexpression were mutually exclusive and identified a subgroup of patients with a significantly poor overall survival. There are limited evidences that EGFR and AKT1 overexpression are sufficient to drive tumor growth in experimental model of gastric cancer. Mutually exclusive aberrations, such as EGFR and KRAS mutations or EGFR mutation and ALK rearrangement in lung cancer, affect genes able to drive the cancer growth. Therefore, the mutually exclusive overexpression of EGFR and AKT1, may suggest an important role of EGFR-AKT1 axis in tumors of the stomach and GEJ. Because of EGFR and AKT1 are two genes involved in the same pathway, the overexpression of joust one of them could be sufficient to increase proliferation making the other redundant and therefore not enriched during the clonal evolution of the tumor. Another possibility is that tumors with EGFR and AKT1 overexpression belong to different subtypes of gastro-esophageal cancers. Indeed, genome wide analyses of somatic mutations have shown the presence of 4 different types of gastric cancer (positive for Epstein-Barr virus, with microsatellite instability, genomically stable and with chromosomal instability), each one with a peculiar array of genomic aberrations [47, 48]. The overexpression of AKT1-EGFR was a prognostic factor independent from tumor stage and the overexpression of MET. A trend for a worse DFS was observed in patients with EGFR/AKT1 overexpression (median DFS 14.2 months, 95% CI: 0–59.7), compared to those without (median DFS 30.6 months, 95% CI: 15.2–46); this difference was not significant (Log-Rank
In conclusion, our results confirm the wretched role of EGFR overexpression also in resected stage II–III gastric tumors and suggest similar conclusions for MET. Moreover, we firstly describe the mutually exclusive overexpression of EGFR and AKT1 with potential prognostic implications.
Footnotes
Acknowledgments
ARCO (Association for Research in Clinical Oncology) founded this research.
Conflict of interest
The authors declare that they have no conflict of interest.
