Abstract
BACKGROUND:
Gastric cancer (GC) remains a huge challenge to the heathy of human beings, largely due to lacking of effective therapeutic measures. Though an oncogenic role for circular RNAs (circRNAs) circ_0067997 in the progression of GC has been described recently, the molecular modulatory mechanism of it still remains to be further explored. The aim of present study is to examine the molecular network of circ_0067997 in GC.
METHODS:
qRT-PCR was carried out to determine the mRNA levels of circ_0067997, miR-615-5p and AKT1 in cisplatin (DDP)-insensitive or sensitive GC tumor tissues and cells, while the correlations among the contents of these molecules were determined by statistical analysis. The expression of circ_0067997 was manipulated by short-hairpin RNA and lentiviral-mediated approaches, while that of miR-615-5p was achieved by the application of its inhibitor or mimic. The in vivo action of circ_0067997 on tumor formation was determined by measuring tumor weight/volume/size and analyzing tumor apoptosis through TUNEL staining in mouse xenograft model and, while the in vitro effects of this circRNA and its target miR-615-5p on the cell survival and death were separately evaluated by CCK-8 assay and flow cytometry. Additionally, luciferase reporter assays were executed to determine the sequentially regulatory relationships of circ_0067997, miR-615-5p, and AKT1.
RESULTS:
Our data demonstrated that the level of circ_0067997 level was increased in DDP-insensitive GC tissues and cell line, while miR-615-5p presented the opposite results. Moreover, the relationships between circ_0067997 and miR-615-5p levels, circ_0067997 and AKT1 contents presented negative and positive correlations in clinic samples, respectively. Importantly, circ_0067997 was found to repress miR-615-5p expression, consequently leading to increased growth while reduced apoptosis of GC cells in the presence of DDP. Furthermore, the validated sequential regulation was circ_0067997 modulating miR-615-5p adjusting AKT1.
CONCLUSIONS:
This study demonstrated that circ_0067997 functioned as a sponge of miR-615-5p to target AKT1 expression, thereby enhancing the growth and restricting the apoptosis of DDP-insensitive GC cells. These new findings offered a valuable target for the detection and management of GC.
Keywords
Introduction
As one of the frequent cancers, gastric cancer (GC) occupies the second position of cancer-related mortality worldwide [1, 2]. The emergence of about 1 million new occurring GC cases has been documented annually [3]. Though the application of surgery and chemotherapy alone or combined contributed to a significant improvement or reduction of GC-related incidence or death [4], these measures also have inherent shortcomings. For instance, the former strategy can only be used in GC early stage, while the latter approach like the first-line cisplatin (DDP)-based chemotherapy often leads to drug resistance following repeated and long-time usage [5, 6, 7]. Additionally, the current diagnostic approaches often fail to diagnose GC at the early stage, thereby resulting in a late therapeutic intervention or poor prognosis [4, 6, 8]. These facts highlight the urgent requirements to clarify the pathogenic mechanisms and identify novel molecular biomarkers and effective therapeutic targets for the patients with GC.
Growing evidence indicates that circular RNAs (circRNAs) are tightly associated with DDP resistance in various cancers [5, 6, 9, 10, 11]. circRNAs are a class of non-coding RNAs (ncRNAs) [12], with the properties including its specifically forming manner and its covalently closed-loop structure [13, 14]. circRNAs have been found to achieve their functions through serving as the sponge of microRNAs (miRNAs), which in turn regulate target mRNAs of specific genes [14]. Importantly, the functions of circRNAs as important tumor promoters or suppressors in the development of various cancers have been verified by accumulating studies [15, 16, 17, 18, 19, 20]. Furthermore, the involvement of circRNAs in the growth and apoptosis of GC cells and the dysregulation of these RNA molecules in the progression of GC have also been confirmed [21, 22]. For instance, circ_0006470 was identified to enhance the growth and migration of GC cells via targeting miR-27b-3p and repressing autophagy pathway [22]. Similarly, circ_0067997 has been identified as an oncogenic molecule in the progression of GC by a recent study [23]. However, the biological action and molecular mechanism of this circRNA in the context of GC tumors with or without DDP resistance has not been addressed.
The current work intends to study the action of circ_0067997 on the growth and survival of GC cells either sensitive or insensitive to DDP, analyze its expression alteration in clinical GC tumor tissues, and address the underlying mechanism.
Materials and methods
Human samples
Each of 25 cases for gastric cancer tissues and gastric mucosal tissues that are sensitive and insensitive to the first-line chemotherapy therapeutic cisplatin (Cisplatin, DDP) were separately collected. qRT-PCR was applied to evaluate the level of circ_0067997 in these tissues Written and signed consent was provided by all patients. This project obtained the approval from the independent ethics committee of The Affiliated Changzhou No.2 People’s Hospital of Nanjing Medical University and complied with the Declaration of Helsinki. The evaluation of chemosensitivity results (for patients who choose cisplatin as the first-line treatment plan) was based on: the unified standard established by WHO for the efficacy of neoadjuvant chemotherapy, and the followup of tumor sizes determined by CT/PET-CT detection method Accordingly, the comprehensively curative effect was judged and classified into: complete remission (CR) meaning no tumors found by clinical means; partial remission (PR) representing tumor reduction
Cell culture
Cisplatin-resistant and sensitive human gastric cancer cell lines SGC-7901/DDP and SGC-7901 used in this study were provided by the cell bank of Shanghai Biology Institute (Shanghai, China) and maintained in DMEM (Trueline, Kaukauna, WI, USA) plus 10% FBS (Thermo Fisher Scientific), 1% penicillin/streptomycin antibiotics (Solarbio, Beijing, China), 2 mM l-glutamine, and in an incubator with 5% CO
qRT-PCR
TRIzol Reagent (Invitrogen, Waltham, MA, USA) prepared total RNAs from various samples were utilized to produce cDNA with a corresponding kit (Thermo Fisher Scientific, Waltham, MA, USA) following the instructions of the manufacturer. qRT-PCR was performed with the conditions: 95
Western blot (WB)
The concentration of RIPA buffer plus Protease inhibitor Cocktail (11697498001, Roche, Heidelberg, Germany) prepared whole protein lysates from distinct samples was estimated by BCA method with a corresponding kit (PICPI23223, Thermo, USA). 25
Interfering and enhancing the expression of hsa-circ_0067997
To interfere hsa-circ_0067997 expression, short hairpin RNAs (shRNA) specific to human hsa-circ_0067997 (shcirc-1: 5’-GAGAAGAAT TAGAATGTAA-3’, site: 155–173; shcirc-2, 5’-GGAAGGCACCAATTGACAA-3’, site: 347–366; and shcirc-3 5’-GAAAGATCTTAGA CCAGCA-3’, site: 177–196), plus a negative control shRNA (shNC, 5’-UUCUCCGAACGUGUC ACGUTT -3’), were produced by a commercial company and inserted into lentiviral plasmid (pLKO.1).
To enhance the expression of this circRNA, target cells were subjected to transfection of pLO5-ciR lentiviral plasmid containing hsa-circ_0067997 (NM_022763) cDNA or corresponding empty vector as a negative control (oeNC) by using Lipofectamine 2000 according to the instructions of the manufacturer, and then cultured for 48 h before analysis.
Dual luciferase reporter gene assay
TargetScan and Starbase programs were applied to analyze potential binding sites between hsa-circ _0067997 and has-miR-615-5p, has-miR-615-5p and AKT1. Wild type and mutant (Mut) binding sequences were generated based on the prediction and cloned into luciferase reporter vectors (pGL3-Basic) to obtain corresponding WT 3’ UTR and Mut 3’UTR. These reporter constructs, along with has-miR-615-5p miNC or mimics were introduced into 293T cells and their luciferase activities were determined using dual luciferase reporter gene kit (CP002, Beijing Yuanpinghao Biotechnology Co., Ltd.) 48 h post transfection.
Cell proliferation
Cell Counting Kit-8 (CCK-8) assay (Signalway Antibody, Maryland, USA) was conducted to examine cell growth. Accordingly, 0, 24, 48 and 72 h cultured cells were incubated with CCK-8 reagent (1:10) for 1 h and subjected to the analysis of optical densities (ODs) on a microplate reader (DNM-9602, Pulangxin, Beijing, China) at 450 nm wavelength. Repeat experiments were carried out thrice.
Flow cytometry
Cells collected 48 h post transfection were subjected to the Annexin V-fluorescein isothiocyanate (C1062, Beyotime, Beijing, China) staining and the measurement of apoptosis on a flow cytometry (CytoFLEX, Beckman, USA). Cell populations with Annexin V
RNA pull-down assay
Biotinylated-probe (RiboBio, Guangzhou, China) complementary to the junction area of hsa-circ_006 7997 (5’-TGGACTTTCCTGTGGACCCCA-3’) was utilized for RNA pull-down assay. Lysates of SGC-7901/DDP cells prepared with corresponding lysis buffer plus RNase inhibitor (Promega, Madison, WI, United States) were subjected to the sequential incubation with probes for 2 h and then DynabeadsTM M-280 Streptavidin (Invitrogen) for 4 h. Following this, qRT-PCR assay was executed on the RNAs from treated cells as described above.
Animal experiments
Association between circ_0067997 expression and clinicopathological characteristics in human gastric cancer patient,
50
Association between circ_0067997 expression and clinicopathological characteristics in human gastric cancer patient,
Data was analyzed by using chi-square test.
In vivo study obtained the approval from the ethics committee of The Affiliated Changzhou No.2 People’s Hospital of Nanjing Medical University. The handling and experimentation of animals completely complied with the guidelines of Institutional Animal Care and Use Committee (IACUC). Five nude mice (Vitalriver Company, Beijing, China) were included in individual group. 4- to 6-week-old mice received equal numbers of shNC- or shcirc -transfected SGC-7901/DDP cells (
TUNEL staining was conducted with ApopTag kit (Intergene) as described by the manufacturer. At least three samples were included in each group.
Statistical analysis
All statistical analyses were done with GraphPad Prism software Version 7.0 (La Jolla, CA, USA). Shown data were mean
Results
Circ_0067997 downregulation suppressed the growth and tumor inducing capabiliy of human DDP-resistant GC cell SGC-7901/DDP
Knockdown of circ_0067997 boosted the DDP effect on the proliferation of human Cisplatin-resistant gastric cancer cell SGC-7901/DDP. (A) The levels of circ_0067997 in para-cancerous and gastric cancer tumor tissues (sensitive and non-sensitive) was revealed by qRT-PCR, n=25 for each group. 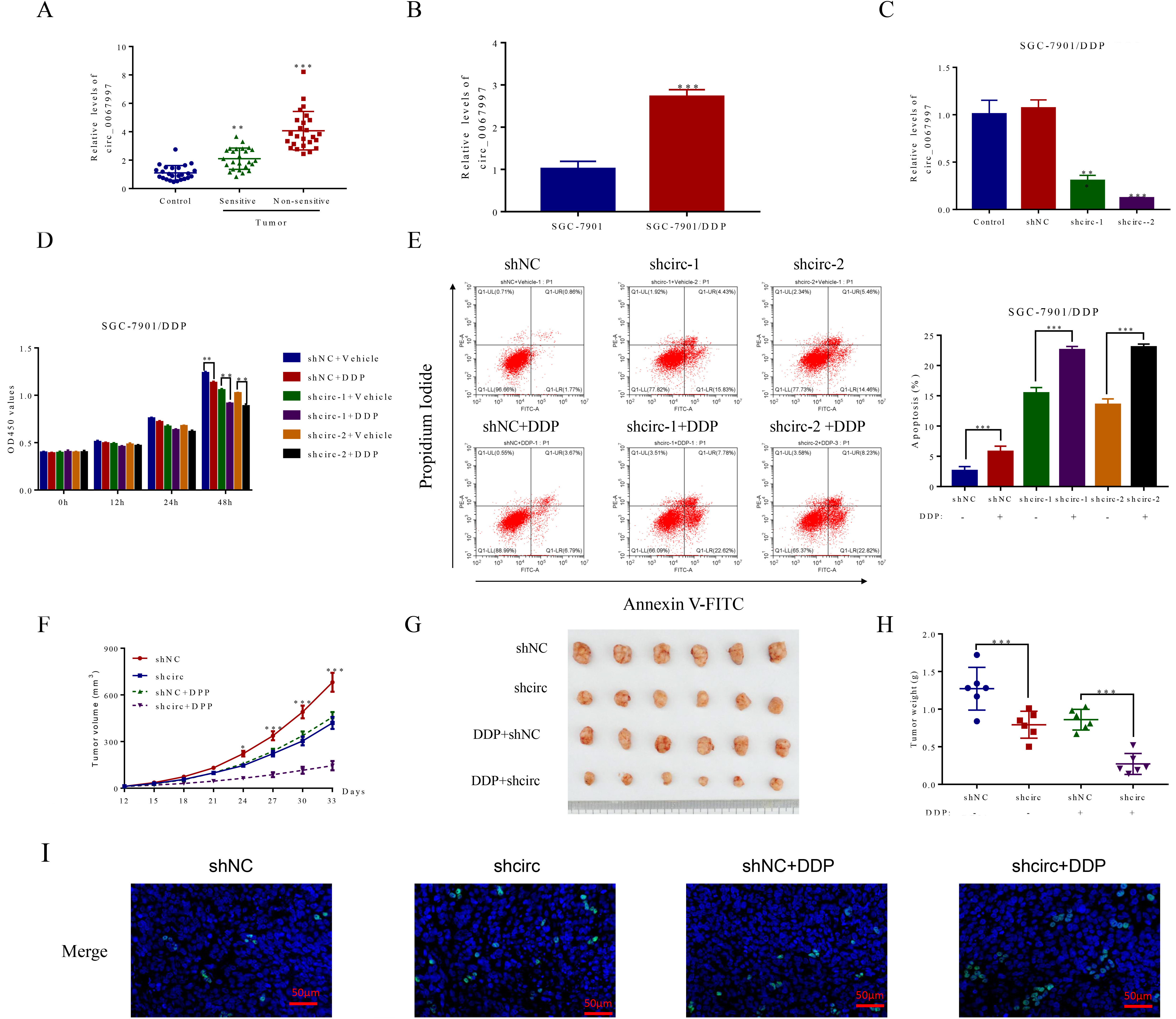
To establish the relevance of circ_0067997 to the development of GC, its expression in the para-cancerous (as the control) and GC tumor tissues (including sensitive or insensitive to DDP) was assessed by qRT-PCR. As indicated in Fig. 1A, relative to the control, the levels of circ_0067997 in the GC tumor tissues were pronouncedly increased, with higher content in DDP insensitive tissues than in DDP sensitive ones. The correlation of circ_0067997 content with clinicopathologic features was further analyzed in enrolled patients. The data shown in Table 1 demonstrated the tight link of the circ_0067997 expression level to GC severity, such as histological type, clinical stage and lympth node metastasis, rather than gender and age. Likewise, similar phenomenon was also observed in GC cells, with increased level of this circRNA in DDP insensitive SGC-7901/DDP cells as relative to that in DDP sensitive SGC-7901 cells (Fig. 1B).
To investigate the association of altered circ_0067997 with GC progression, its expression was artificially manipulated by short-hairpin RNA (shRNA)-mediated method. Indeed, pronouncedly reduced circ_0067997 expression resulting from its corresponding shRNAs (shcric-1 and shcric-2) was validated in SGC-7901/DDP cells, as relative to either untreated control or scramble shRNA (shNC) treated control cells (Fig. 1C). Next, the consequences of downregulated circ_0067997 on GC cell growth and apoptosis were separately evaluated. As indicated in Fig. 1D and E, the proliferation of circ_0067997 downregulated SGC-7901/DDP cells was remarkedly restricted, while the apoptosis of these cells was boosted upon the addition of DDP, as relative to corresponding control cells. To confirm the in vivo functional significance of altered circ_0067997 level, the capability of its shRNA treated SGC-7901/DDP cells to induce tumors was evaluated in mice. Intriguingly, the tumor inducing ability of SGC-7901/DDP cells with reduced circ_0067997 content was considerably compromised especially with DDP, as reflected by the reductions in tumor volume, size and weight, as compared to the corresponding control (Fig. 1F–H). Meanwhile, an enhanced apoptosis in tumor tissues from mice receiving circ_0067997 shRNA treated cells was seen particularly in the presence of DDP, as relative to the control (Fig. 1I). Collectively, the results reveal that circ_0067997 knockdown can boost the effect of DDP on GC cell growth & survival and tumor forming ability, suggesting a positive role of this circRNA in the progression of GC.
miR-615-5p interacted with circ_0067997. (A) The levels of circ_0067997 target microRNA miR-615-5p in para-cancerous and gastric cancer tumor tissues (sensitive and non-sensitive) were revealed by qRT-PCR, 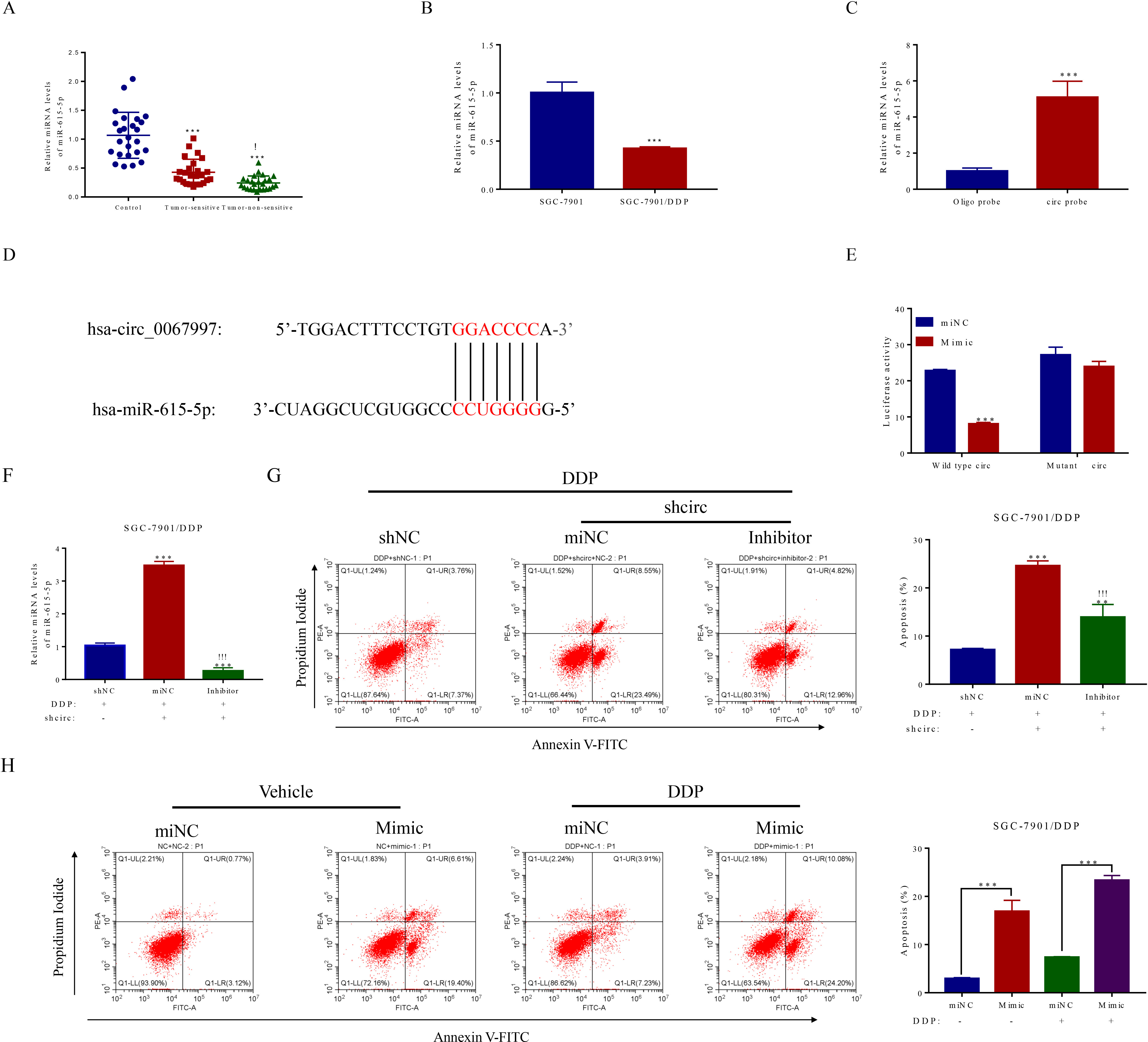
circ_0067997 overexpression at least partly abolished DDP’s action on the growth of SGC-7901 cells. (A) circ_0067997 overexpression was achieved in SGC-7901 cells by using lentiviral-mediated method. 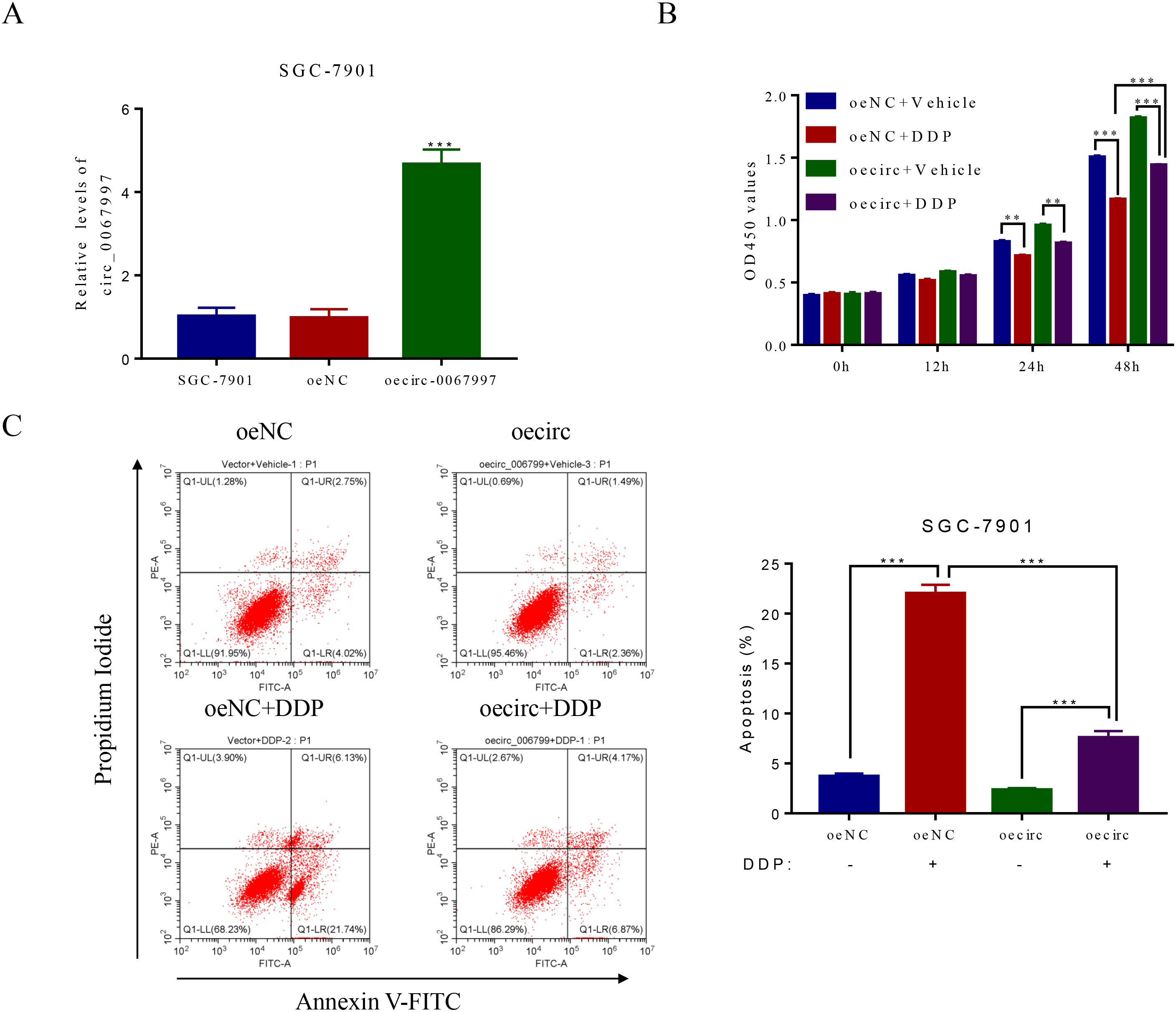
miR-615-5p regulated AKT1 expression through interacting with its 3’UTR and the presence of a negative correlation was identified among these molecules in human gastric cancer tumor tissues. (A) The mRNA levels of AKT1 in para-cancerous and gastric cancer tumor tissues (sensitive and non-sensitive) were revealed by qRT-PCR, 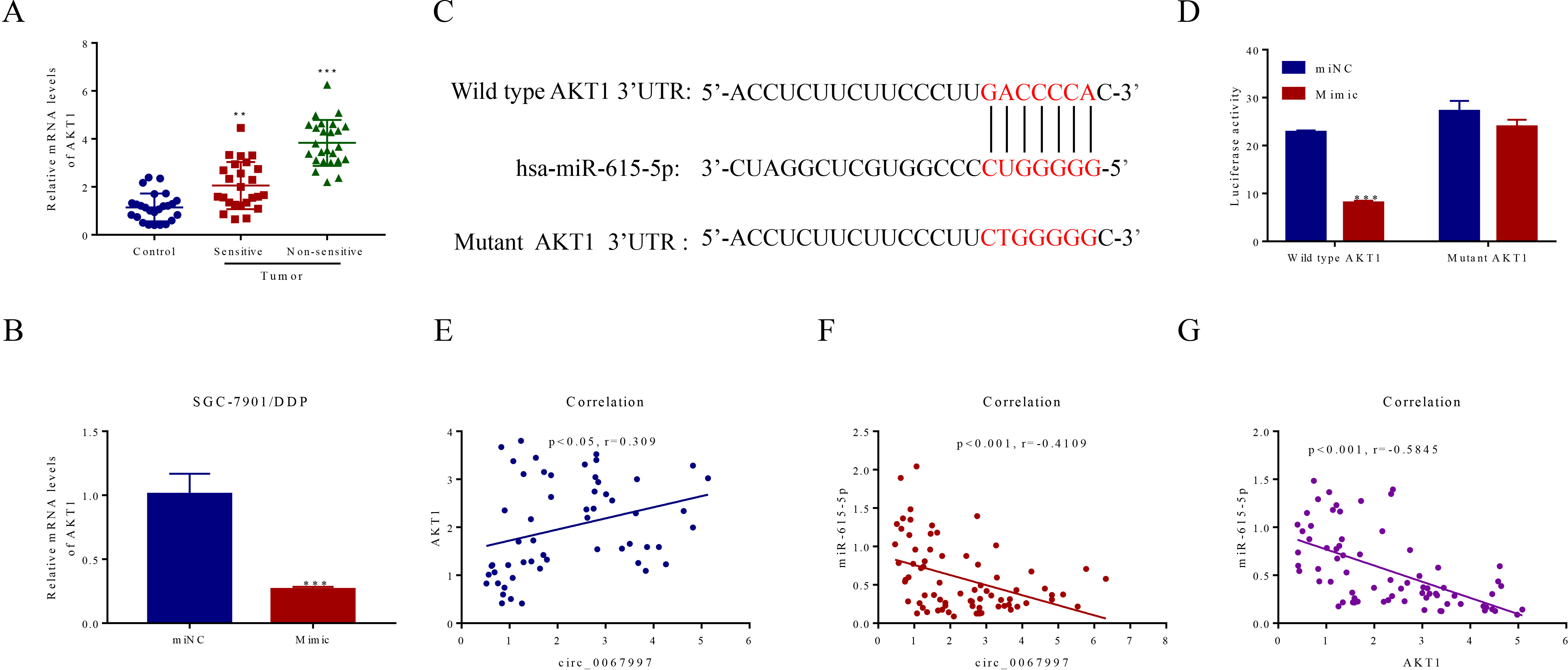
Having found and validated the in vitro and in vivo functions of circ_0067997 in terms of GC pathogenesis, we next explored the action mechanism of this circRNA under the condition of GC disorder. It is suggested and demonstrated that circRNAs mainly serve as sponge for corresponding miRNAs. Therefore, we analyzed potential miRNAs related to circ_0067997 through circRNA interactome prediction approach by using circinteractome database (
To provide further substantial evidence of a vital role for circ_0067997 in GC progression, we performed a gain-of-function experiment. Lentiviral-mediated overexpression of circ_0067997 was verified in SGC-7901/ DDP cells receiving circ_0067997 expressing vector (oecirc_0067997) rather than cells receiving an empty vector (oeNC) (Fig. 3A). More significantly, compared with the control SGC-7901cells receiving vehicle and oeNC, DDP application alone led to reduced cell growth, while circ_0067997 overexpression alone resulted in increased proliferation. Interestingly, the inhibitory effect of DDP on cell growth was compromised by circ_0067997 overexpression, as reflected in cells receiving DDP and oecirc_0067997 simultaneously, indicating that circ_0067997 overexpression at least partially revoked the action of DPP on the growth of SGC-7901cells (Fig. 3B). Similarly, the promoting effect of DDP on cell apoptosis was also restricted by circ_0067997 overexpression (Fig. 3C). Therefore, these evidences further validate the critical action of circ_0067997 for GC progression.
miR-615-5p repressed the expression of AKT1 via binding to its mRNA 3’UTR
To address how miR-615-5p regulates its predicted target AKT1, potential binding site for this miRNA on the 3’UTR of AKT1 was identified by bioinformatic analysis. Subsequently, the expression of AKT1 was examined in GC tumor tissues (both sensitive and insensitive to DDP) and adjacent tissues. As indicated in Fig. 4A, increased level of AKT1 in the GC tumor tissues was seen, as relative to the adjacent tissues, with the highest one found in the DDP-insensitive tumor tissues. More interestingly, the expression of AKT1 was strongly repressed by the presence of miR-615-5p mimic but not scramble control (Fig. 4B). To establish the direct connection between miR-615-5p and AKT1, luciferase reporter constructs containing WT or Mut AKT1 3’UTR sequences were generated for this purpose (Fig. 4C). Indeed, the luciferase activity of reporter construct containing AKT1 WT 3’UTR was evidently supressed by the application of miR-615-5p mimic to the cells, while this inhibitory effect exerted by miR-615-5p was largely vanished in the presence of mutant miR-615-5p binding site on the AKT1 3’UTR (Fig. 4D). Finally, the corrections of expression levels among circ_0067997, miR-615-5p and AKT1 were analyzed in GC tumor tissues. A positive correlation (
Discussion
GC remains an important healthy issue and poses huge socioeconomic burden to human beings, as reflected by over 1 million newly occurring cases and over 0.7 million death cases annually [24, 25, 26]. Meanwhile, the efficiencies and shortcomings of current therapeutic measures urgently appeals for understanding of the underlying mechanisms responsible for GC initiation and progression, thereby facilitating the improvement of diagnostic approaches and the exploration of effective targets. In this work, we provided robust evidence to demonstrate that increased level of circ_0067997 was linked to the progression of GC and reduced sensitivity to DDP, while altered content of this circRNA had an important impact on the growth and survival of GC cells, furthermore, this circRNA was identified to regulate miR-615-5p/AKT1 axis, thus suggesting that circ_0067997 and its regulated signal pathway play a critical role in GC pathogenesis.
Accumulating evidence has revealed that aberrant levels of circRNAs are often related to the development of numerous cancers, such as cervical cancer, pancreatic cancer, liver cancer [27, 28, 29]. Furthermore, as biomarkers, different circRNAs have been employed to appraise cancer cell properties including metastasis, growth, survival [30, 31]. For instance, the upregulated levels of circ_0072995 and circ_CEP128 was found in breast cancer and bladder cancer, respectively. The alterations of these circRNAs were associated with the behaviors like invasion, growth and apoptosis of cancer cells [32, 33]. Likewise, dysregulated circRNAs were observed in GC. Furthermore, the impact of some circRNAs on GC cell behaviors has also been validated [34, 35]. For example, GC cells exhibited higher level of circ_0067997, as also validated in GC tissues. Moreover, increased level of this circRNA was closely linked to worse heathy status in GC patients, and also facilitated the growth and invasion of GC cells [23]. As a new finding, the current work further revealed that GC tissues insensitive to DDP had higher level of this circRNA than those sensitive to this reagent. Additionally, a series of in vivo and in vitro evidence demonstrated that circ_0067997 boosted the effect of DDP on the growth and apoptosis of DDP-insensitive GC cells. Therefore, these new finding, together with previous results [23], highlight the promoting role of circ_0067997 in the pathogenesis of GC including the context of DDP sensitivity and suggest its potential as a therapeutic target of GC.
Numerous previous studies suggested and confirmed that circRNAs serve as the sponge for miRNAs, thereby achieving the modulation of various physiopathological processes [36, 37]. Furthermore, the miRNA and mRNA axes were verified to be inactivated and activated by circRNAs in distinct tumor cells, consequently, circRNAs had a forward or reverse impact on the progression of cancers [38, 39]. For instance, circ_0006006 was found to accelerate the progression of non-small cell lung cancer, as indicated by its promoting or inhibitory actions on the growth, migration and apoptosis of lung cancer cells, respectively, these functions of this circRNA were associated with its modulation on miR-924/SRSF7 axis [39]. In contrast, circ_0069117 was validated to have anti-osteosarcoma (OS) function, since it could impair the proliferation and migration of OS cells, this action of circ_0069117 was ascribed to its regulation on miR-875-3p/PF4V1 axis [38]. The current study demonstrated that circ_0067997 sponged miR-615-5p to regulate AKT1 in the setting of GC cells insensitive to DDP, as supported by both loss and gain-of-function evidence. Distinguishing from previous finding showing that circ_0067997 stimulated the progression of GC by targeting miR-515-5p and X chromosome-lined inhibitor of apoptosis (XIAP) [23], this work indicated that the boosting action of this circRNA on GC development was attributed to targeting miR-615-5p/AKT1 axis. The possible explanations for this might be due to the difference in the objects studied, including tumor tissues and cell lines, or due to versatile functions of circRNAs [40]. Regardless of these, the finding provided here indicated that circ_0067997 contributes to the progression of GC in a boosting way through regulating various pathways, suggesting this circRNA and its downstream molecules could be suitable for the development of new therapeutics.
It is well known that miRNAs modulate the expression of target mRNA in a post-transcriptional manner through binding to the complementary sites within the 3’UTR [41]. The bioinformatic analysis revealed that AKT1 is a potential target of miR-615-5p. The key role of AKT1 in the progression of different cancers has been addressed by previous studies [42, 43]. In this regard, miR-615-5p was found able to affect the expression AKT1 at the post-transcription level, as validated by luciferase reporter assay. Interestingly, miR-615-5p/AKT1 axis was found to be regulated by long non-coding RNA under the condition of lung cancer [44]. Thus, our finding, together with previous study, demonstrated the importance of that ncRNAs including circ_0067997 and miR-615-5p/AKT1 axis in the pathogenesis of GC and other cancers.
In summary, this study provided ample evidence to demonstrate that circ_0067997 boosted the growth while suppressed the apoptosis of GC cells even in the presence of DDP, suggesting that this circRNA plays a promoting action on the progression of GC. Moreover, this action of it was mediated by sponging miR-615-5p to repress AKT1 expression. Therefore, circ_0067997 and its regulated miR-615-5p/AKT1 axis could be considered as a useful molecule and therapeutic target for the detection and management of GC.
Data availability statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Funding statement
This research was supported by the Changzhou Sci&Tech (CJ20200097), Young Science & Technology Project of Changzhou Health (QN202033) and National Nature Science Foundation of China (81902391).
Authors contributions
Conception: LT.
Interpretation or analysis of data: YJ and YF.
Preparation of the manuscript: LT, YJ and YF.
Revision for important intellectual content: YG and GW.
Supervision: SC, GC and SW.
Ethics approval and consent to participate
Written and signed consent was provided by all patients. This project obtained the approval from the independent ethics committee of The Affiliated Changzhou No.2 People’s Hospital of Nanjing Medical University on 21th, Oct. 2020 and complied with the Declaration of Helsinki (Approval No. [2020]KY212-01).
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-220145.
Footnotes
Acknowledgments
We sincerely acknowledged the support given by the DaLian Medical University, DaLian 116000, China and Department of General Surgery; The Affiliated Changzhou No.2 People’s Hospital of Nanjing Medical University.
Conflict of interest
None.
