Abstract
Background:
Ramucirumab in combination with paclitaxel has demonstrated substantial antitumor activity in the treatment of advanced human epidermal growth factor receptor-2 (HER2)-negative gastric cancer (GC) or gastro-esophageal junction cancer (GEJC). However, the cost-effectiveness of this regimen in this patient population remains uncertain, particularly within the Chinese healthcare context.
Objective:
This study aimed to assess the cost-effectiveness of ramucirumab plus paclitaxel as a switch maintenance regimen compared to continuing first-line chemotherapy for patients with advanced HER2-negative GC or GEJC, from the perspective of the Chinese healthcare system.
Design:
A health economic evaluation was conducted to compare two treatment strategies.
Methods:
A partitioned survival model was developed to project the disease progression of HER2-negative GC or GEJC. Data for overall survival and progression-free survival were extracted from the ARMANI trial and were extrapolated to project long-term survival outcomes. Direct medical costs and utility values were gathered. The main outcome measures, including the cost, utility, and incremental cost-utility ratio (ICUR), were used to determine the cost-effectiveness of the ramucirumab plus paclitaxel as switch maintenance regimen. Sensitivity analyses, including one-way sensitivity analysis (OWSA) and probabilistic sensitivity analysis (PSA), were performed to evaluate the robustness of the findings.
Results:
The base-case analysis revealed that the ICUR for ramucirumab plus paclitaxel as a switch maintenance regimen was ¥1,097,535 per quality-adjusted life-year (QALY) compared to the continuation of first-line chemotherapy. OWSA showed that the ICUR was sensitive to variations in the cost of ramucirumab, patient weight, and the cost of subsequent treatments in the continuation of first-line chemotherapy group. Results from the PSA indicated that the switch maintenance regimen had a very low probability of cost-effectiveness, at just 0.4%. In contrast, the continuation of the first-line regimen demonstrated a high likelihood of being cost-effective, with a 99.6% probability.
Conclusion:
The cost-effectiveness analysis suggested that from the Chinese healthcare system perspective, the switch maintenance regimen at current price is unlikely to be an advantageous regimen in terms of cost-effectiveness for patients with advanced HER2-negative gastric or GEJC at a willingness-to-pay threshold of ¥287,247/QALY.
Plain language summary
Ramucirumab plus paclitaxel is effective for advanced HER2-negative gastric or gastro-oesophageal junction cancer, but its cost-effectiveness in China is unclear. This study compared this combination as switch maintenance therapy with continuing first-line chemotherapy from the Chinese healthcare perspective, using data from the ARMANI trial and a health-economic model to assess costs, quality-adjusted life years (QALYs), and incremental cost-utility ratio (ICUR), with sensitivity analyses to verify results. The combination therapy cost Â¥1,097,535 per QALY gained, far exceeding China’s willingness-to-pay threshold of Â¥287,247/QALY, and had a 0.8% chance of being cost-effective versus 99.2% for continuing first-line treatment. At current prices, this switch maintenance regimen is unlikely to be cost-effective for these patients in China, though lowering ramucirumab costs might improve its cost-effectiveness.
Keywords
Introduction
Gastric cancer (GC) or gastro-esophageal junction cancer (GEJC) poses a substantial disease burden and stands out as the principal contributor to cancer-associated mortality in the affected population on a global scale. According to the latest GLOBOCAN 2022 estimates, approximately 968 thousand newly diagnosed cases and 659 thousand stomach cancer-related deaths were recorded, making it the fifth leading cause of cancer-related deaths worldwide. 1 GC still remains a major health burden worldwide, especially in Eastern Asia and Eastern Europe. 1 In Asia, China was the most affected country, accounting for nearly half of all GC-related deaths worldwide. 2 In 2022, the new cases and death cases in China were 358,672 and 260,372 individuals, respectively. 3
Early-stage GC has no particular signs or symptoms, which leads to delayed diagnosis and poor 5-year survival rate. 4 In the management of advanced GC and GEJC, a variety of pharmacotherapeutic strategies have been employed, such as chemotherapy, targeted drug therapy, and immune checkpoint inhibitors. Notably, for patients with HER2-negative advanced GC or GEJC and with absent or low PD-L1 expression, fluoropyrimidine in combination with platinum-based chemotherapy represents the first-line treatment regimen recommended by clinical guidelines.5–7 Nevertheless, in contemporary clinical trials, the median progression-free survival (PFS) linked with doublet chemotherapy is merely around 6 months, and the overall survival (OS) hardly exceeds 12 months.6,8 Hence, it is crucial to exploring the initial treatment approaches promptly. Ramucirumab is a monoclonal antibody that targets the vascular endothelial growth factor receptor-2, playing a significant role in inhibiting angiogenesis, which is crucial for tumor growth and metastasis. 9 This therapeutic agent has been extensively studied across various types of cancers, demonstrating its potential in improving clinical outcomes when used alone or in combination with other treatments. The RAINBOW trial, a pivotal phase III study, demonstrated the efficacy of ramucirumab combined with paclitaxel as a second-line treatment, showing significant improvements in OS (median 9.6 vs 7.4 months, hazard ratio (HR) = 0.807, p = 0.017) and PFS (median 4.4 vs 2.9 months, HR = 0.635, p < 0.0001) compared to placebo plus paclitaxel. 10 This underscored the potential of this combination therapy in specific patient populations. A strategy of switch maintenance with alternative regimens that are not cross-resistant could delay clinical deterioration and prolong the benefit of the initial treatment strategy, which was proved to improve efficacy in advanced non-small-cell lung cancer. 11 This strategy was also tried in other studies, such as the ARMANI trial. This is a randomized, open-label, multicenter phase III trial, which assessed the efficacy of switch maintenance therapy with ramucirumab plus paclitaxel versus the continuation of oxaliplatin-based chemotherapy in patients with advanced HER2-negative GC or GEJC. 12 The trial found that switch maintenance regimen significantly improved PFS compared to the continuation of first-line chemotherapy (median 6.6 vs 3.5 months, HR = 0.61, p = 0.0002), suggesting that it could be a switch maintenance or early second-line treatment strategy for patients who are not eligible for immunotherapy or other targeted agents. However, the cost-effectiveness of ramucirumab plus paclitaxel for patients with HER2-negative advanced GC or GEJC is uncertain. To date there has not been any pharmacoeconomic evaluation of this switch maintenance treatment strategy in China. Performing economic analyses to clarify the cost-effectiveness is meaningful and helpful for physicians, oncologists, or healthcare decision-makers, particularly in situations where resources are limited. We therefore perform this cost-effectiveness research and evaluate whether ramucirumab plus paclitaxel schedule as a switch maintenance strategy was more cost-effective than continuation of first-line oxaliplatin-based chemotherapy for HER2-negative GC or GEJC from the perspective of Chinese healthcare system.
Materials and methods
The Consolidated Health Economic Evaluation Reporting Standards (CHEERS), initially released in 2013 and subsequently updated in 2022, serves as a comprehensive framework to enhance the transparency and reproducibility of health economic evaluations. This study’s economic evaluation rigorously followed the CHEERS 2022 guidelines, as detailed in Supplemental Table 1.13,14
Model structure
A decision-analytic model was constructed to assess the cost-effectiveness of ramucirumab plus paclitaxel as switch maintenance regimen compared with the continuation of first-line oxaliplatin-based chemotherapy, analyzed from the perspective of the Chinese healthcare system. A partitioned survival model (PSM) was utilized to simulate disease progression in patients. The simulated patient cohort consisted of individuals with ramucirumab plus paclitaxel as switch maintenance versus continuation of first-line oxaliplatin-based chemotherapy, in accordance with the criteria used in the ARMANI trial. 12 The PSM comprised three mutually exclusive health states: progression-free (PF), progressed disease (PD), and death. Patients entered the model in the PF state and could transition to the PD or death state based on survival probabilities derived from clinical survival data; and the PSM was employed to calculate the proportion of cohorts in each health state at any given model time, utilizing independently estimated parametric functions for PFS and OS curves. A 4-week model cycle was implemented to simplify cost estimation, and the 5-year time horizon was determined to ensure that high proportion of patients reached the absorption state, enabling the assessment of the long-term therapy outcomes. Details of the decision tree and model structure are provided in Figure 1.
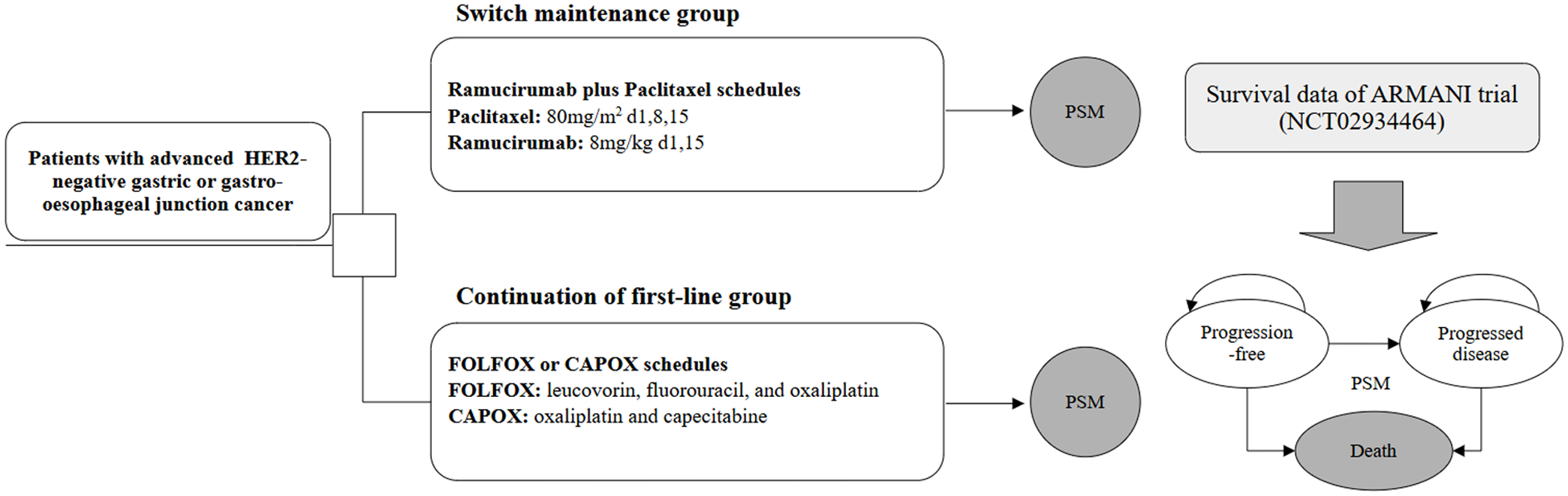
The decision tree and PSM structure overview.
Clinical data
The current analysis relied on data from the ARMANI trial, which provided detailed information on PFS, OS, and safety outcomes. Despite its robust design, the trial’s follow-up duration was limited, necessitating the extrapolation of survival data to enable long-term horizon modeling. Generally, such extrapolation requires individual patient data (IPD) from clinical trials, allowing survival curves to be fitted to appropriate parametric distributions. However, due to the limited accessibility of trial IPD, we employed a pseudo-IPD reconstruction method using an algorithm proposed by Guyot. 15 This method has gained widespread acceptance in survival analysis research for generating reliable pseudo-IPD. After generating the pseudo-IPD, we tested multiple parametric survival distributions, including Weibull, Log-logistic, Log-normal, exponential, and Gompertz distribution. 16 The optimal survival distribution was chosen and validated through the Akaike Information Criterion (AIC), Bayesian Information Criterion (BIC), and visual assessments of fit. Survival data used in this analysis were obtained from the survival data provided in the ARMANI trial.
In addition to survival outcomes, the analysis considered treatment-related adverse events (AEs). Given that grade 1 and 2 AEs were generally well-managed, our primary focus was on grade 3 or higher AEs with incidence rates exceeding 5%. Table 1 provides a detailed summary of the survival curve shape parameters and the incidence rates of AEs.
Key clinical data.
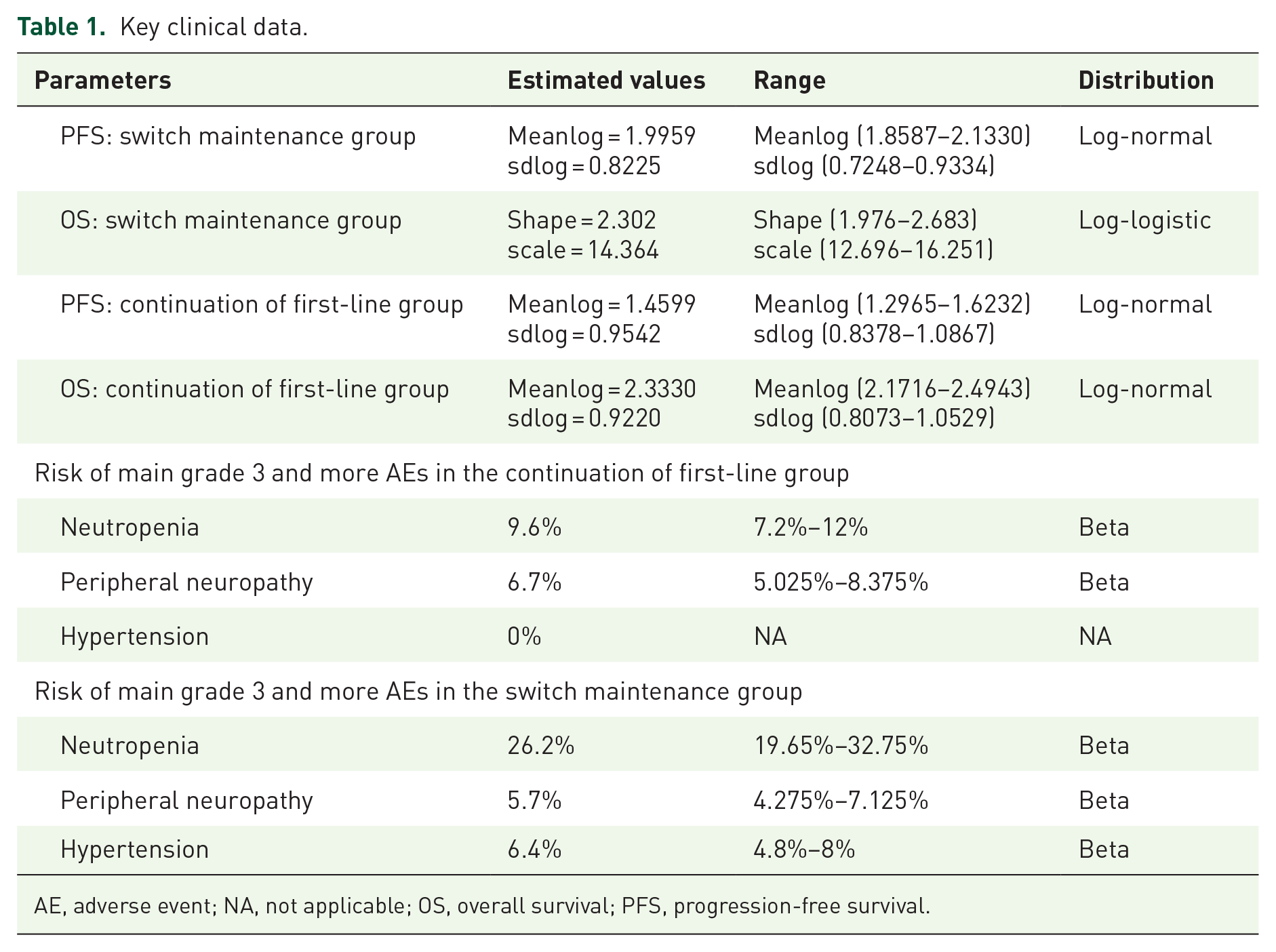
AE, adverse event; NA, not applicable; OS, overall survival; PFS, progression-free survival.
Treatment regimens and resource use
In this analysis, the simulated cohort was divided into two groups based on the treatment regimen: (1) Continuation of first-line group: CAPOX, which accounted for 13.2% of the cohort, or FOLFOX, making up 86.8% of the cohort; (2) Switch maintenance group: ramucirumab plus paclitaxel. The dosing strategies for both groups were in accordance with the doses recommended by the CSCO Clinical Guidelines. 7 The FOLFOX schedule consisted of leucovorin, fluorouracil, and oxaliplatin. Oxaliplatin was administered at a dose of 85 mg/m2 on day 1 of each 14-day cycle for a total of six cycles. Leucovorin was given at a dose of 400 mg/m2 on day 1 of each 14-day cycle for six cycles. Fluorouracil was first administered intravenously at a dose of 400 mg/m2 and then followed by 2400–3600 mg/m2 via intravenous infusion over 46 h, on day 1 of each 14-day cycle for six cycles. The CAPOX schedule included capecitabine and oxaliplatin. Therein, oxaliplatin was administrated at a dose of 130 mg/m2 on day 1 of each 21-day cycle for four cycles; and capecitabine was administered at a dose of 1000 mg/m2 orally, twice daily, from day 1 to day 14 of each 21-day cycle for four cycles. Paclitaxel was administrated intravenously at a dose of 80 mg/m2 of body surface area on days 1, 8, and 15 of each 28-day cycle. Ramucirumab was administrated intravenously at a dose of 8 mg/kg on days 1 and 15 of each 28-day cycle. In both study groups, treatment was continued until disease progression and unacceptable toxic effects. The post-discontinuation systemic therapies were implemented in accordance with the regimens reported by the ARMANI trial, which are presented in Supplemental Table 2.
Costs and utilities
This analysis was conducted from the perspective of the Chinese healthcare system, considering only direct medical costs. These included expenses related to therapeutic drugs, intravenous administration, management of grade 3 or higher AEs, follow-up, and palliative care. The follow-up costs encompassed various components such as imaging, diagnostic tests, and disease management. Drug acquisition costs were sourced from a local pricing database,17,18 while costs for intravenous administration, palliative care, follow-up visits, and subsequent treatment were extracted from published literatures or calculation.19,20 Management expenses for severe AEs graded as level 3 or higher were also derived from published studies. Expenditures associated with the management of severe AEs were gathered from published studies.
In the cost-effectiveness analysis, health utility values were essential for calculating cumulative quality-adjusted life-years (QALYs). These QALYs quantified the patients’ health-related quality of life (HRQoL) associated with different stages of tumor progression. Each health state, such as PF and PD, was assigned a specific utility value. These values were derived from a published study on advanced GC patients, which assessed HRQoL using the EQ-5D questionnaire. In addition, a scenario analysis using utility values from the RAINBOW trial was also conducted to provide further economic evidence. Both cost and utility data were discounted at a 5% annual rate to reflect the time value of money and quality of life adjustments. Comprehensive parameter details are summarized in Table 2.
Key model inputs costs, utility estimates, and other parameters.

In this table, the costs of AEs presented were paid on a per-event basis.
AEs, adverse events; PD, progressed disease; PFS, progression-free survival.
Analyses
In the base-case analysis, we evaluated the cost-effectiveness of two treatment regimens. The incremental cost-effectiveness ratio (ICER) was employed to calculate the additional cost per extra life-year (LY) obtained between the two regimens. Meanwhile, the incremental cost-utility ratio (ICUR) served to measure the incremental cost per QALY. A regimen was regarded as “cost-effective” when its ICUR was lower than the willingness-to-pay (WTP) threshold. In this analysis, the WTP threshold was determined as three times the per capita gross domestic product (GDP). In 2024, this value was equivalent to 287,247 China Yuan (CNY), and it was used as a crucial benchmark for assessing the cost-effectiveness of the treatment regimens within the Chinese context.
To validate the credibility of our research outcomes, an array of uncertainty analyses, including one-way deterministic sensitivity analyses (DSA) and probabilistic sensitivity analyses (PSA), were carried out. In the DSA, we explored the effect of each input’s uncertainty on the ICUR. The annual discount rate was set to vary between 0 and 8%. Meanwhile, other model inputs were adjusted within the reported 95% confidence intervals or reasonable ranges (specifically, ±25% of the base-case value). Regarding the PSA, Monte Carlo simulations with 1000 iterations were adopted. Key parameters were simultaneously sampled according to predefined probability distributions. Costs were assigned Gamma distributions, and the incidence rates of AEs along with utilities were sampled using Beta distributions. To comprehensively evaluate the cost-effectiveness of switch maintenance regimen at various thresholds, the cost-effectiveness acceptability curves (CEAC) and scatter plots were generated. These visual tools enabled us to evaluate the likelihood of the treatment strategy being regarded as “cost-effective” across a range of thresholds. In addition, we conducted subgroup analyses stratified by PD-L1 combined positive score (CPS), claudin 18.2 (CLND18.2), and mismatch repair (MMR) status. For each subgroup, the ICUR was calculated by comparing the switch maintenance regimen with the continuation of first-line chemotherapy. This analysis was performed using the same PSM and cost-utility analysis framework as described in the main analysis. All the analyses were carried out using the R software (version 4.4.2, http://www.r-project.org).
Results
Curve fitting
Through the application of AIC, BIC, and visual inspection, we concluded that the log-normal distribution is the optimal function for extrapolating the OS of continuation of first-line group. Also, for the PFS of this group, the log-normal distribution is appropriate. For the switch maintenance group, the log-normal distribution is adopted for PFS, and the log-logistic distribution is used for OS. We generated replicated Kaplan–Meier survival curves and projected PFS and OS curves to compare the ramucirumab plus paclitaxel to the continuation of first-line regimen, as depicted in Figure 2. The scale and shape parameters of the projected curve of two treatment regimens can be found in Table 1. Furthermore, we created fit figures based on other alternative distributions, which are presented in Supplemental Figure 1. More detailed fitting curve parameters can be found in Supplemental Table 3.

Reconstructed Kaplan–Meier survival curve and the projected OS and PFS curve.
Cost-effectiveness analysis
Patients who received continuation of first-line regimen experienced a gain of 1.155 LYs, 0.754 QALYs, with an associated cost of ¥192,179. Meanwhile, patients who underwent the switch maintenance regimen achieved a gain of 1.421 LYs, 0.924 QALYs, at a cost of ¥378,760. In comparison to the continuation of first-line regimen, the switch maintenance regimen brought about an additional cost of ¥186,581. In terms of effectiveness, the switch maintenance regimen manifested an increase of 0.17 QALYs relative to the continuation of first-line regimen. The calculated ICUR for the switch maintenance regimen compared to the continuation of first-line regimen was ¥1,097,535/QALY. A summary of all the cost-effectiveness analysis results can be found in Table 3.
Results of the cost-effectiveness analysis.

ICER, incremental cost-effectiveness ratio; ICUR, incremental cost-utility ratio; LY, life-year; QALY, quality-adjusted life-year.
We also conducted a scenario analysis, where utility values in the model were derived from the RAINBOW trial. Specifically, a utility value of 0.75 was applied for the PFS state, and 0.61 for the PD state. Results of the scenario analysis showed an ICUR of ¥1,172,724 per QALY, which was slightly higher than the result of the base-case analysis.
Subgroup analyses based on PD-L1 CPS, CLND18.2, and MMR status were performed. The ICUR varied across different subgroups. For example, in the subgroup with PD-L1 CPS ⩾5, the ICUR was ¥698,140/QALY, while in the subgroup with PD-L1 CPS <5, it was ¥3,348,170/QALY. Similarly, differences were observed in subgroups stratified by CLND18.2 and MMR status. These results indicate that the cost-effectiveness of the switch maintenance regimen compared to the continuation of first-line chemotherapy may differ among various patient subgroups. Detailed data can be seen in Supplemental Figure 2.
In addition, we conducted a price-threshold analysis specifically for the cost of ramucirumab to identify the threshold at which the switch maintenance regimen becomes cost-effective. Figure 3 presented the price-threshold relationship for ramucirumab. The results indicated that when the unit cost of ramucirumab is reduced to ¥8443 (a 43.71% reduction from the original price), the resulting ICUR aligns exactly with the predefined WTP threshold. This figure also shows that when the WTP threshold is set at one times China’s per capita GDP, the unit cost of ramucirumab needs to be reduced to ¥6898, representing a 54.01% decrease from its original price.

Price-threshold analysis for ramucirumab plus paclitaxel regimen in advanced HER2-negative gastric or gastro-esophageal junction cancer.
Sensitivity analysis
The results of the one-way sensitivity analysis (OWSA) demonstrate the sensitivity of the analysis outcomes to various model variables. The diagram of tornado (Figure 4(a)) presented the top 10 sensitive variables sensitive to the results. As can be noticed from the figure, variables including the weight, the cost of ramucirumab, and the cost of subsequent treatment in the continuation of first-line regimen group exert a significant impact on the ICUR between the switch maintenance regimen and the continuation of first-line regimen. The ranges of variable alterations, based on the lower bound input and the higher bound input, are clearly visible, varying from ¥634,953 to ¥1,564,461 per QALY. A reduction in the cost of ramucirumab and the weight, as well as increasing the cost of subsequent treatment in the continuation of first-line regimen group, caused a decrease in the ICUR. The impact of other variables on the ICUR was not particularly prominent. To further explore the interaction between the two most influential factors identified in the OWSA, we conducted an additional two-way sensitivity analysis examining the combined impact of patient weight and the unit cost of ramucirumab on the ICUR. A 3 × 3 matrix was constructed, incorporating three levels for each variable: lower bound, base value, and upper bound. The results are presented in Table 4. Notably, the analysis reveals a clear synergistic effect: ICUR increases consistently with higher patient weights and higher ramucirumab costs, with the most favorable scenario (ICUR = ¥286,548/QALY) occurring at the lowest weight and lowest drug cost—approaching the predefined WTP threshold. This two-way analysis complements the one-way sensitivity findings and price-threshold results, providing a more nuanced understanding of how concurrent variations in key parameters affect the regimen’s cost-effectiveness.
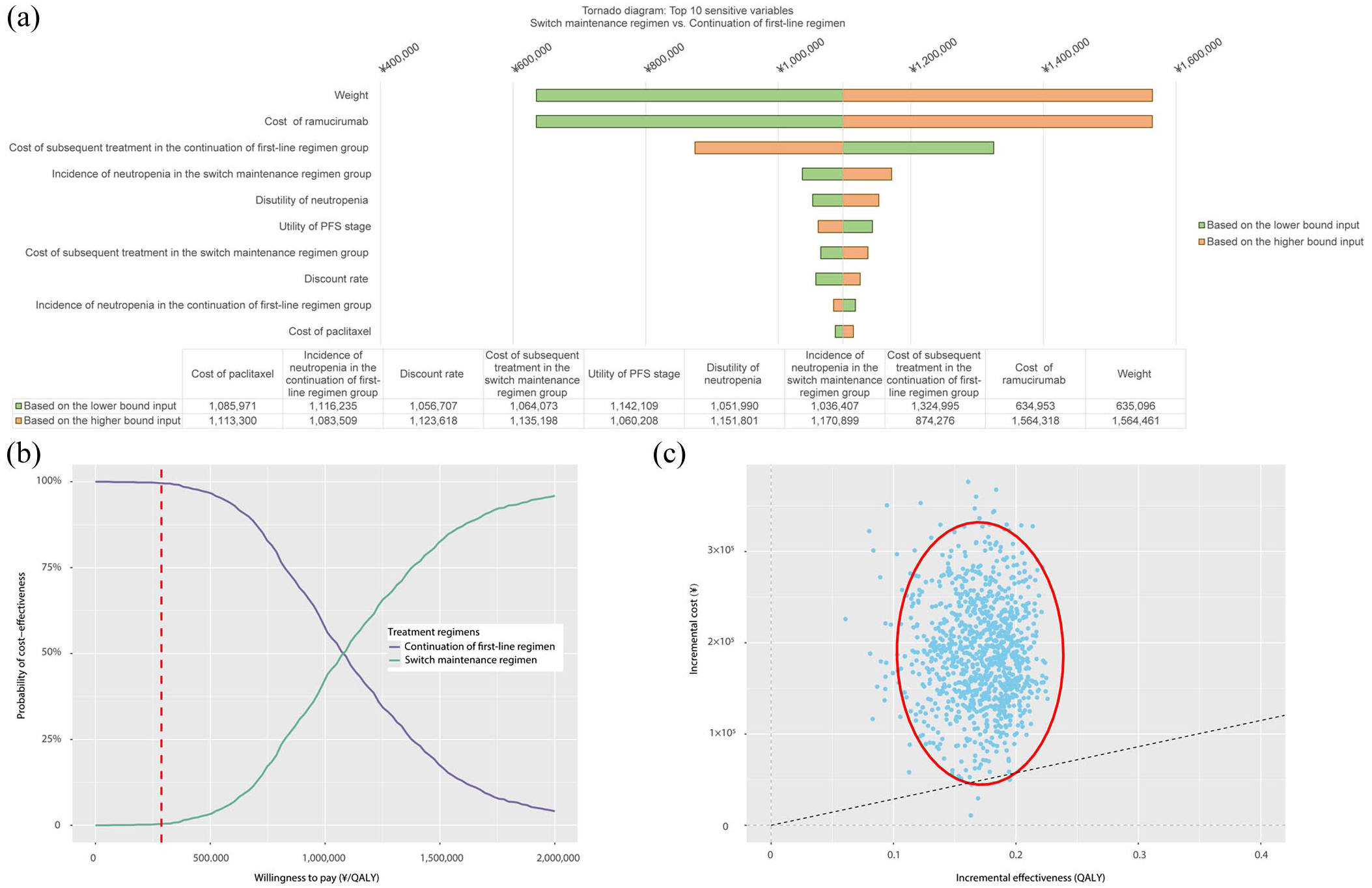
The plot output of the one-way and probabilistic sensitivity analysis. (a) The tornado diagram of the ICUR of switch maintenance regimen versus continuation of first-line regimen. (b) The cost-effectiveness acceptable curve of the switch maintenance regimen versus continuation of first-line regimen. (c) The incremental cost-effectiveness scatter plot of the switch maintenance regimen versus continuation of first-line regimen.
Two-way sensitivity analysis outcomes of two key inputs.

ICUR, incremental cost-utility ratio; QALY, quality-adjusted life-year.
The outcomes of the PSA are illustrated through CEAC (Figure 4(b)) and scatter plots (Figure 4(c)). The CEAC shows that, when the WTP threshold is set at ¥287,247 per QALY, the curve for the continuation of first-line therapy lies above that of the switch maintenance regimen. This indicates that the probability of cost-effectiveness is much higher for the continuation of first-line regimen (99.6%) compared to the switch maintenance regimen (0.4%). At this threshold, the continuation of first-line therapy is more likely to be cost-effective. Specifically, at lower WTP values, the continuation of first-line therapy may be more favorable, but as the WTP value increases, the probability of the switch maintenance regimen becoming cost-effective gradually rises. Once the WTP exceeds ¥1,097,535/QALY, the switch maintenance regimen becomes a more cost-effective option. The scatter plot depicts the distribution of incremental costs and incremental effects. In this plot, the blue dots represent the incremental cost-effectiveness in different simulations, and majority of these dots are clustered within the red oval area. The position and distribution of the red oval area suggest that in most simulations, the relationship between incremental costs and incremental effectiveness is relatively concentrated and above the WTP threshold line. This further supports the conclusion that the continuation of first-line regimen has advantages in terms of cost-effectiveness.
Discussion
Determining the optimal duration of first-line systemic chemotherapy for metastatic GC remains an unresolved clinical question. In some trials, chemotherapy is continued until disease progression or unacceptable toxicity, whereas others stop treatment after a predefined period. Prolonged administration of chemotherapy is associated with an increased risk of cumulative toxicities, which may adversely affect patients’ quality of life. Importantly, initiating a non-cross-resistant treatment regimen in the early stage, specifically prior to disease progression, has the potential to extend the therapeutic benefits of first-line therapy and delay clinical deterioration.12,26 Some trials investigated the feasibility of sequential therapy in advanced GC, and they showed the potential of sequential therapy in order to prolong therapeutic benefit of first-line treatment, but with the price of cumulative toxicities.27–29 Early use of a safer second-line regimen, such as ramucirumab combined with paclitaxel, may reduce toxicity, improve PFS, and ultimately enhance patients’ quality of life. From a clinical perspective, early implementation of such regimen appears to address the critical issue of rapid health deterioration in GC or GEJC patients, a population in which the majority are ineligible to receive second-line therapy due to disease progression. While this treatment strategy holds promise in improving patient outcomes, its economic implications must be thoroughly assessed to ensure its feasibility within healthcare systems, particularly in resource-constrained settings.
The current cost-effectiveness analysis indicates that the ICUR of ramucirumab plus paclitaxel as a switch maintenance regimen compared to the continuation of first-line oxaliplatin-based chemotherapy is ¥1,097,535 per QALY, significantly exceeding the WTP threshold defined in this study. Furthermore, PSA indicates that the probability of this regimen being cost-effective under the WTP threshold is almost zero. Similar findings have been reported in previous studies across different cancer types. For instance, a study based on the RAINBOW trial, conducted from the perspective of the Chinese healthcare system, concluded that ramucirumab plus paclitaxel is unlikely to be a cost-effective option compared to paclitaxel monotherapy in advanced gastric or gastro-esophageal cancer. 30 This outcome underscores the significant cost increase associated with ramucirumab, which offers only marginal additional clinical benefits. Beyond GC, similar conclusions have been drawn in non-small-cell lung cancer (NSCLC): a recent analysis evaluating ramucirumab combined with erlotinib for first-line treatment of EGFR-mutated metastatic NSCLC in China found that the regimen yielded an ICER of $128,302/QALY, far exceeding local WTP thresholds, and only became cost-effective after a more than 52.1% price reduction of ramucirumab. 31
Similar conclusions have also been drawn in international studies on GC. Kimura et al. 32 analyzed the cost-effectiveness of ramucirumab plus paclitaxel in Japan and found it to be less cost-effective when compared to the chemotherapy regimen. Likewise, Saito et al. 33 reported that ramucirumab plus paclitaxel regimen is not a cost-effective alternative to paclitaxel monotherapy in the Japanese healthcare setting.
Collectively, these findings suggest that despite its clinical efficacy in delaying disease progression across multiple indications, the high economic burden associated with ramucirumab-containing regimens restricts their adoption as cost-effective options. Our OWSA identified two key variables that significantly affect the cost-effectiveness of ramucirumab plus paclitaxel as switch maintenance regimen: patient weight and the cost of ramucirumab. Since weight-based dosing is a fixed clinical practice, adjusting the price of ramucirumab emerges as the most critical lever to improve cost-effectiveness. In China, where many innovative cancer treatments, including ramucirumab, are not yet included in the National Reimbursement Drug List, patients are often required to bear the full cost of such drugs. Policymakers and manufacturers should therefore consider revising the pricing structure and expediting its inclusion in the reimbursement list. A reasonable price adjustment and timely inclusion in the reimbursement list could alleviate the financial burden on Chinese patients with advanced HER2-negative gastric or GEJC, enabling broader access to this effective therapy.
Our study has several limitations. Firstly, utility values were derived from published literature rather than the ARMANI trial itself. Even though the utility values we selected in our study were sourced from research on a population similar to the target population of this study and measured using the EQ-5D questionnaire survey. Despite being highly relevant, they were still not direct utility data from the ARMANI trial. This inevitably brought in some degree of uncertainty. Secondly, in the process of extrapolating survival curves, two processes usually give rise to uncertainties. One is the conversion of “time-to-survival” data into “time-to-event” data, which may have some bias. However, Guyot’s algorithm seemed to be a relatively robust solution. This algorithm is frequently applied in survival analysis and has been proven to be superior compared to other methods. 34 Another source of uncertainty is the choice of survival model. It can be observed from the fit figures (Supplemental Figure 1) that the selection of survival model has a significant impact on the survival benefit. Thus, different choices of survival models may lead to substantial differences in the results. This is a currently unavoidable obstacle, and we can only attempt to choose a more reasonable survival distribution based on AIC and BIC values and visual inspection. Thirdly, subgroup analyses were limited to PD-L1 CPS, CLND18.2, and MMR status due to data constraints, and could not be extended to other patient characteristics. Although the ARMANI 3 trial provided HR values of PFS and OS stages in different subgroups, the method of survival simulation through curve extrapolation requires survival curves or IPD for different subgroups. The lack of such data restricted our ability to perform subgroup analysis on patients’ characteristics. However, the ARMANI 3 trial provided Kaplan-Meier curves of subgroups stratified by PD-L1 CPS, CLND18.2, and MMR status. We then performed the subgroup analyses based on PD-L1 CPS, CLND18.2, and MMR status. This can provide economic evaluation data for relevant researchers.
Conclusion
The cost-effectiveness analysis suggested that from the Chinese healthcare system perspective, the switch maintenance regimen at the current price is unlikely to be an advantageous regimen in terms of cost-effectiveness for patients with advanced HER2-negative gastric or GEJC at a WTP threshold of ¥287,247/QALY.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251375836 – Supplemental material for Cost-effectiveness analysis of ramucirumab plus paclitaxel as switch maintenance versus continuation of first-line oxaliplatin-based chemotherapy for advanced HER2-negative gastric or gastro-esophageal junction cancer
Supplemental material, sj-docx-1-tag-10.1177_17562848251375836 for Cost-effectiveness analysis of ramucirumab plus paclitaxel as switch maintenance versus continuation of first-line oxaliplatin-based chemotherapy for advanced HER2-negative gastric or gastro-esophageal junction cancer by Yitian Lang, Weican Cao, Yan Lin, Min Liu and Hui Zhao in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
