Abstract
Exosomes are lipid bilayer vesicles of endocytic origin ranging from 30 to 100 nm in size, and contain various nucleic acid molecules such as DNA, mRNA, miRNA, lncRNA and multiple proteins, which could be transferred into target cells. Recent study indicated that exosomes as information carriers between cells has introduced us to a new previously unknown biological communication system. Increasing evidences show that exosomes play a crucial role in gastric cancer because they are potential to influence normal cellular physiology and promote various states of the cancer. In this review, we focus on the latest findings on exosomes in the plasma of gastric cancer patients, mainly summarizing the functions of miRNAs, lncRNAs and multiple proteins in diagnosis, prognosis, and in establishing treatment regimens against gastric cancer. Furtherly, potential functions of exosomes as novel diagnostic biomarkers for gastric cancer are discussed extensively. Exosomes are believed to be a non-invasive disease biomarker with a dual capability to provide insights into the early diagnosis for gastric cancer.
Introduction
Gastric cancer (GC) is the fourth most common malignancy and the third leading cause of cancer-related mortality worldwide [1]. The incidence and mortality of gastric cancer are high in East Asian countries such as Korea, Japan, China, and Central South America such as Chile, Peru, Mexico [2, 3]. The poor prognosis of GC resulted from late detection, aggressive characteristics and poor response to available therapies. Early diagnosis of GC could significantly reduce the mortality of GC. A number of non-invasive detection methods for GC are available, including serum pepsinogen (PG), carcinoembryonic antigen (CEA) and carbohydrate antigen 19-9 (CA19-9). However, the sensitivity and specificity of these markers are not enough to improve the early detection of GC [4]. Currently, endoscopic screening for GC is the most reliable diagnostic tool, yet the use of endoscopic screening is limited due to its cost and invasive nature. Therefore, novel, non-invasive, cost-effective and high sensitive biomarkers are urgently needed to improve the efficiency of GC early detection [5].
Exosomes composition. Exosomes are small lipid bilayer vesicles sharing similar structure and contents with plasma membrane of origin cells. Exosomes contain typical transmembrane proteins and receptors, such as transmembrane proteins, membrane trafficking proteins as Annexins, Rabs proteins, adhesion molecules, the lipid raft associated protein. Furthermore, the exosomal membranes surface is assembled with immune regulator molecules, such as MHC-I/-II, CD80, CD86. Cytoskeleton proteins, HSPs, metabolic enzymes involving in MVB formation could be also found in exosomes. The contents of exosomes could be transferred from originals cell to target cells, consisting of DNA or RNA species, and a vast array of different proteins.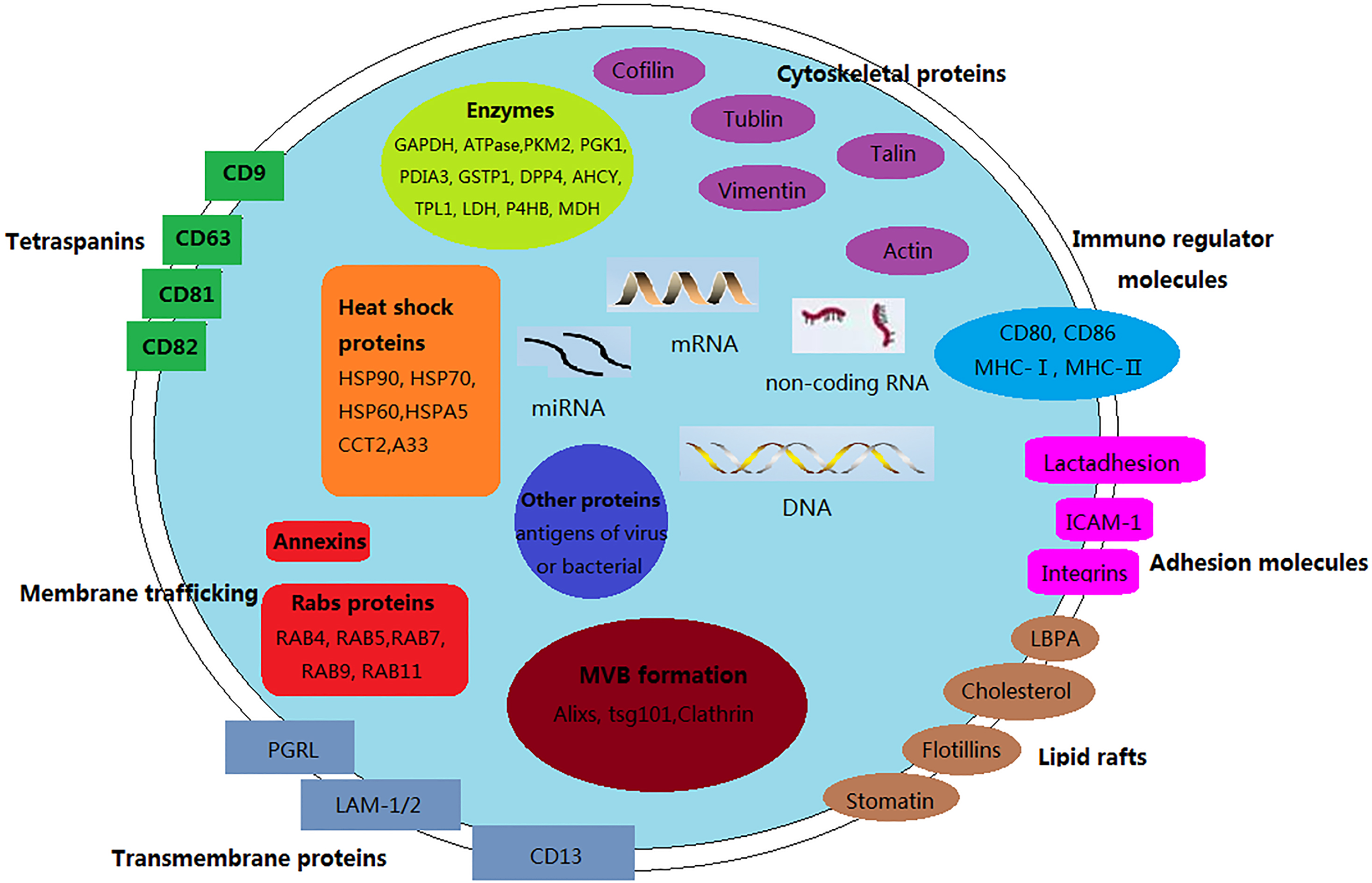
Exosomes are 30
Cancer-derived exosomes could be isolated from serum, pleural effusions, urine, and ascites fluids of cancer patients [12]. Alterations in expressions of certain exosomes suggest that exosomes could be an early marker for diagnosis and prognosis monitoring. Exosomes, present in multi-bodily fluids of patients stable, show promise as a tool for the evaluation of labile biomarkers. If exosomes can be perfected in cancer, manifestations of this cancer may be more effectively treated, thus offering improved survival [13]. Circulating exosomes-encapsulated miRNAs, lncRNAs presented as an early biomarker for cancer, and the expression pattern of exosomal miRNAs or lncRNAs is also correlated with tumor malignancy degree or prognosis such as breast cancer [14, 15]. MiRNAs and lncRNAs in plasma of gastric cancer patients could be mass-screened by RNA sequencing or RNA chip. Further, altered RNA expression in exosomes is detected by qRT-PCR method clinically. Moreover, – proteins could be mass screened by proteomics technology, and differential proteins in exosomes of gastric cancer could be detected in serum just like a tumor marker detection [16]. The biomarker potential of exosomes holds huge promise for diagnosing GC.
Exosomes have a number of advantages as a diagnosis target including: (a) stable conformational conditions of proteins; (b) effective molecular distribution due to the ability of microvesicles to re-circulate in body fluids and reach distal organs; (c) a more efficient association of antigen with target cells [17, 18]. In this review, we focus on the latest findings on the contents of exosomes derived from gastric cancer, the roles of miRNAs, lncRNAs and multiple proteins in exosomes in diagnosis, prognosis, and in establishing treatment regimens in gastric cancer are carried out a thorough discussion.
Major miRNAs involved in exosomes of gastric cancer patients
MicroRNAs (miRNAs) are non-coding (about 22 nucleotide) RNAs, which induce translational repression and/or degradation of their mRNA targets [19, 20]. MiRNAs could act as key regulators in a wide variety of biological processes, including cell development, differentiation, apoptosis, metabolism, and signal transduction [21]. To date, hundreds of miRNAs have been identified in human genomes. Accumulating evidences have illustrated the regulative role of miRNAs in the innate and adaptive immune response. Consequently, abnormal patterns of miRNAs also have been found in various human diseases, most notably in cancers [22]. Reports indicated that miRNAs are specific indifferent tumor types and miRNAs expression profiles could distinguish normal or tumor tissues, including gastric cancers and breast cancer [23]. Meanwhile, miRNAs are able to be detected stably in circulating plasma or serum. Thus, circulating miRNAs could be non-invasive biomarkers for the early diagnosis or the prediction prognosis of human cancers [24]. However, circulating miRNAs were considered to be passively leaked or actively transported from cells during tumorigenesis. These leaked miRNAs are shown to pack into small membranous vesicles or protected by the formation of protein-miRNA complexes [25, 26, 27]. At present, more and more circulating miRNA biomarkers with diagnostic and prognostic values for GC have been screened out [28, 29, 30].
MiRNAs could be wrapped in secretory microvesicles such as exosomes, apoptotic bodies, shedding microvesicles. These microvesicles could protect miRNAs from the degradation by RNase and serve as carriers for miRNAs to extracellular space or body fluids. Exosomes could also perform intercellular transfer of miRNAs, which participated in miRNA-based signalling mechanisms [31]. Indeed, exosomes contain substantial amounts of miRNAs involving in immune-independent regulatory mechanisms. Higher levels of miRNAs in exosomes have been detected in the plasma of gastric cancer patients [32]. These evidences suggest that exosomes-miRNA secretion is closely related to gastric cancer occurrence and development.
MiRNAs in exosomes could be consistently isolated from GC patients. Thus, miRNAs expression profiles could show the status of peritoneum in GC patients. Five miRNAs miR-320c, miR-1202, miR-1225-5p, miR-1207-5p, miR-4270 (1.91, 2.27, 2.23, 1.9, 2.25 fold changed respectively in exosomes from the peritoneum lavage fluid compared with normal individual) in exosomes from the peritoneum of GC patients were expressed diversely. Expressions of miR-21, miR-320c and miR1225-5p in the peritoneum lavage fluid of 18 CG patients were validated by qRT-PCR [33]. These results indicate that exosomal miR-21, miR-320c and miR-1225-5p (from peritoneum lavage fluid) may serve as biomarkers of peritoneal recurrence after curative GC resection. These results also provide a novel approach to the early diagnosis of peritoneal metastasis of GC.
Huang et al. reported that six serum miRNAs miR-10b-5p, miR-132-3p, miR-185-5p, miR-195-5p, miR-20a-3p and miR-296-5p (4.12, 1.55, 1.89, 6.12, 4.31, 6.71 fold increased respectively) were significantly expressed in GC compared with normal persons, and elevated significantly in exosomes from GC serum samples [34]. The diagnostic value of the six miRNAs was verified in GC tissues and serum exosomes, and the encouraging results increasingly demonstrated the important roles for the six miRNAs in tumorigenesis and progression. Let-7 miRNA acts as tumor suppressor by targeting oncogenes such as RAS and HMGA2. Let-7 miRNA is often down-regulated in many cancers [35]. Meanwhile, AZ-P7a cells release let-7 miRNA via exosomes into extracellular environment, resulting in decrease of let-7 within the cells to maintain their oncogenesis. Let-7 miRNA family in exosomes from AZ-P7a cells may reflect the oncogenic characteristics including tumorigenesis and metastasis of the cells [36]. Above abnormally miRNAs in exosomes associated with gastric cancer is briefly summarized in Table 1.
Recent studies show that miR-185, miR-20a, miR-210, miR-25 and miR-92b (4.32, 7.61, 4.66, 3.22, 7.14 fold enhanced respectively) were expressed differently in peripheral plasma of GC patients [37]. Yet, none of above five miRNAs from GC patients was demonstrated with a significantly different expression level in exosomes of peripheral plasma. The limited number of clinical specimens could explain the non-significant expression of five miRNAs in exosomes. More and more miRNAs in exosomes of GC are indentified, the clinical relevance and their biological functions are being systematically studied. These findings provide a novel serum miRNAs signature for GC diagnosis, and will serve as the basic application of circulating miRNAs in future clinical detection of GC.
lncRNAs as biomarkers in exosomes for GC
Long noncoding RNAs (lncRNAs) are non-protein-coding transcripts and longer than 200 nucleotides [38]. lncRNAs regulate the expression of associated genes at transcriptional, posttranscriptional, epigenetic levels [39]. Currently, numerous studies demonstrated that exosomes not only contain miRNAs, proteins, but also carry multiple lncRNAs [40]. In addition, some lncRNAs could function as tumor suppressor genes or oncogenes, which are often deregulated in GC [41, 42]. lncRNAs may be protected by exosomes from degradation in the circulation [43], lncRNAs could be therefore useful for diagnosing cancer at the early stage of GC [44]. Huang et al. found some lncRNAs in exosomes from patients’ plasma by RNA deep sequencing [45]. For instance, lincRNA-p21 in exosomes has been proved to be helpful to distinguish prostate cancer from benign disease [46].
LINC00152 could be isolated from the plasma di- rectly or exosomes in plasma of GC patients. Interestingly, LINC00152 mainly exists in exosomes from plasma of GC patients, but there is no significant difference of LINC00152 level between in plasma and in exosomes. LINC00152 was significantly increased in exosomes of GC patients, suggesting that LINC00152 in exosomes maybe a novel tumor biomarker for GC screening and diagnosis [47]. ZFAS1 is another newly-identified lncRNAs, which may promote the growth and metastasis of various tumors. ZFAS1 expression was elevated in GC cells, tumor tissues, serum and serum exosomes of GC patients. Moreover, ZFAS1 in exosomes could be transmitted to enhance the proliferation and migration of GC cells. These results suggest that ZFAS1 could be delivered by exosomes to promote GC progression. ZFAS1 mayserve as a potential diagnostic and prognostic biomarker for GC [48].
At present, only two major types of lncRNAs in exosomes have been identified in plasma of GC patients. Although there are various problems need to be solved, the detection of exosomal non-coding RNAs in body fluids is a booming research field. Numerous miRNAs, lncRNAs, and circular RNAs have been described, which could become potential early-diagnose and efficient treatment targets of GC. Certainly more lncRNAs will be discovered, and the exosomal non-coding RNAs will serve as superior biomarkers for early cancer diagnosis.
Diagnostic values of proteins in exosomes
Exosomes derived from tumor cells could exchange information between neighboring cancer cells or normal cells, and communicate with distant sites and various cells. They also could activate macrophages to increase the levels of pro-inflammatory factors. The pro-inflammatory factors in turn promote the proliferation and migration of tumor cells, and activate macrophages from human peripheral blood monocytes through the activation of NF-
Exosomes are enriched with multivesicular body (MVB) and related proteins, including Annexins, Rab, GTPases, flotillins; the proteins involved in MVB biogenesis containing ALIX, Tsg101; the membrane microdomain associated proteins include CD9, CD63, CD81, CD82 [51]. Moreover, exosomes are able to express Major Histocompatibility Complex-I/II (MHC-I/II) molecules and/or antigens. These specific proteins are important for the classification of exosomes and are attractive targets for the identification of novel biomarkers [52]. The most commonly identified exosomal proteins are heat shock protein-8 (HSP-8) and CD63 as well as Cytoskeletal proteins such as actins, cofillins, tubulins, and cellular signaling proteins (i.e.
CD97 proteins are tetraspanins of cell membrane, and also expressed on the surface of exosomes. Recently, studies reported that CD97 small isoform was associated with increasing invasiveness in vitro as well as elevated local growth and metastasis of gastric cancer in vivo [55]. In addition, exosomes isolated from cells which with high or low CD97 expression, promoting the proliferation and invasion of gastric cancer cells in vitro through the exosomes-mediated MAPK signaling pathway, and exosomal miRNAs are probably involved in the activation of the CD97-associated pathway [56]. Epidermal growth factor receptor (EGFR) located in the cytomembrane is well known to play a vital role in tumorigenesis and development. Studies showed that EGFR could be secreted from cells via the transport of vesicles and these EGFR contained in exosomes regulated the signalling pathways of endothelial cells and T cells [57, 58, 59]. Moreover, microvesicles containing EGFRvIII are found to merge with the plasma membranes of cancer cells to lack this type of receptor and the share of EGFR mutants between cancer cells promote tumor development [60]. EGFR in exosomes from gastric cancer cells could be delivered into the liver and integrated on the plasma membrane of liver stromal cells. The trans-located EGFR effectively activated hepatocyte growth factor (HGF) by suppressing miR-26a/b expression. Moreover, the up-regulated paracrine HGF, which binds to the c-MET receptor on the migrated cancer cells, providingfertile ‘soil’ for the ‘seed’, facilitating the landing and proliferation of metastatic cancer cells. All results indicated that EGFR in exosomes from GC cells could help the development of a liver-like microenvironment to promote liver-specific metastasis of gastric cancer [61]. Expression of adhesion related molecules, such as fibronectin-1 (FN-1) and laminin gamma-1 (LAMC-1), were increased in mesothelial cells after internalization of TEX (tumor-derived exosomes) from gastric cancer cell line and malignant pleural effusion. TEX internalization into gastric cancer cells significantly promoted migratory ability, while internalization of mesothelial cell-derived exosomes did not. Tumor-derived exosomes play a critical role in the development of peritoneal metastasis of gastric cancer, which may be partially due to induced increased expression of adhesion molecules in mesothelial cells [62].
Helicobacter pylori is a gram-negative bacterium that colony in human gastric epithelium, and is associated with the pathogenesis of gastric disorders, such as atrophic gastritis, peptic ulcers, and gastric cancer [63, 64, 65]. CagA as a virulence factor of H. pylori encoded by cytotoxin associated gene A (cagA) has been received increasing attention for its strong association with severe gastric lesions, particularly gastric cancer [66, 67]. Reports showed that CagA is present in serum-derived exosomes in patients with cagA-positive, and that exosomes containing CagA could be secreted from gastric epithelial cells inducibly expressing CagA. As purified CagA-containing exosomes into gastric epithelial cells formed an elongated cell shape, indicating that the exosomes deliver functional CagA into cells. These findings revealed that exosomes secreted from CagA-expressing gastric epithelial cells may enter into the circulation, delivering CagA to distant organs, tissues or cells. Thus, CagA-containing exosomes may be involved in the development of extra gastric disorders associated with cagA-positive H. pylori infection [68]. HSPs are groups of stress-response proteins which are either constitutively expressed or induced through the transcriptional action of heat shock factor (HSF), which have been proved to play a protective role in gastrointestinal tract [69, 70]. HSP-90, HSP-70, HSP-60, HSP-27 proteins in exosomes obtained from cell supernatant of gastric epithelial cells (RGM-1 cells) increased significantly, and these findings contribute to understand the functions of HSPs in H. pylori infected gastric epithelial cells, indicating that HSPs would be diagnostic markers for H. pylori infection [71].
There are many kinds of proteins in exosomes, and the protein components of exosomes secreted from different tumors are same approximately. But the amounts of their components vary greatly, and their functions are quite different. It is of great significance to analyze the expression difference of the protein components of exosomes in plasma of cancer patients by proteomics to find proteins closely related to cancer. Proteins in exosomes supplying a huge reservoir of potential markers for cancer, collectively, exosomes play a significant role in the study of cancer.
Conclusion
Exosomes play a critical role in signal transduction between cancer cells and their recipient cells, via releasing a wide variety of biological molecules, such as miRNAs, lncRNAs, proteins and their complexes, affecting the interaction of distant cancer cells in the tumor microenvironment and cancer progression. There is promising evidence supporting the use of exosomes as diagnostic tools for discovering biomarkers, exploiting them as drug delivery devices, and utilizing their inherent therapeutic potentials. In this review, we further elaborate the contribution of miRNAs, lncRNAs and proteins contained in exosomes derived from gastric cancers to regulate gastric cancer progression. With the development of novel diagnostic strategies by utilizing exosomes, it will lead to more effective diagnostic biomarkers in gastric cancer. However, it is critical to note that some crucial contents in exosomes are still not fully elucidated, and there is still a long road to fully understand the role of exosomes in gastric cancer progress, and exosomes serve as predictive and prognostic biomarkers still need to be firmly validated by further prospective clinical studies.
Footnotes
Acknowledgments
This work was supported by Science and Technology Project of Suzhou in China (NO: SYSD2015022) and by Medical Science and Technology Development Fund of Jiangsu University in China (JLY20140047).
Conflict of interest
None.
