Abstract
OBJECTIVE:
Cervical squamous cell carcinoma seriously threats to patient’s life and health. MiRNAs have role of regulating cell growth, proliferation, and death. MiRNAs can promote or inhibit cell growth and proliferation. This study discussed the role of miRNA373 in regulating cervical squamous cell carcinoma growth, proliferation, and apoptosis.
PATIENTS AND METHODS:
MiRNA373 and scramble miRNA were synthetized and transfected to cervical squamous cell carcinoma SiHa cells by lipofectamine. IAPs plasmid and miRNA373 were sequentially transfected to SiHa cells. MTT assay, caspase-3 activity, and flow cytometry were applied to test miRNA373 and IAPs impacts on cell growth, proliferation, and apoptosis. Western blot was adopted to determine IAPs expression.
RESULTS:
MiRNA373 transfection obviously reduced SiHa cell growth, induced phosphatidylserine eversion and caspase-3 activation, and declined IAPs expression. IAPs interference significantly enhanced miRNA373 induced SiHa cell apoptosis. IAPs overexpression markedly restrained miRNA373 induced SiHa cell apoptosis.
CONCLUSIONS:
MiRNA373 transfection suppressed SiHa cell growth and proliferation. MiRNA373 induced SiHa cell apoptosis possibly through downregulating IAPs, suggesting that IAPs might be a target for cervical squamous cell carcinoma treatment.
Introduction
Cervical squamous cell carcinoma is a type of malignant tumor in the female reproductive system, with the incidence at 3/100,000 [1, 2, 3, 4, 5]. Chemoradiotherapy and surgery are important methods for cancer treatment [6, 7, 8]. However, they also have various shortcomings and deficiencies. At present, multiple methods are combined and the curative effect is significant. How to improve the accuracy and success rate of the treatment of cervical squamous cell carcinoma becomes a key and difficult point. Molecular targeted therapy is the hot spot in the treatment of cervical squamous cell carcinoma [8, 9, 10]. Existing methods focused on survivn and apollon presented poor effect for the treatment of the cervical squamous cell carcinoma [11]. Therefore, more effective molecular target is needed in clinic [11, 12].
MiRNA218 inhibits cancer cell growth, whereas miRNA34a is associated with tumor metastasis [13, 14], suggesting that miRNAs may be involved in cervical squamous cell carcinoma occurrence and development [13, 14, 15]. Therefore, we tested miRNA expression in cervical squamous cell carcinoma tumor tissue and para-carcinoma tissue and found that miRNA373 was obviously upregulated in tumor tissue [16, 17]. This study intended to investigate the role of miRNA373 on SiHa cells growth and apoptosis.
Tumor treatment is mainly achieved by inducing tumor cell apoptosis. Cell apoptosis is regulated by antiapoptotic proteins and pro-apoptotic proteins [18, 19]. Ideal anti-cancer drugs can decline antiapoptotic protein level and upregulate pro-apoptotic proteins. Inhibitor of apoptosis proteins (IAPs) is a kind of widely studied antiapoptotic molecules [20, 21]. Currently, although there are numerous types of IAPs protein drugs, their effects are still unsatisfactory [22]. This study will also explore the potential molecules targeting IAPs.
Thus, this research applied SiHa cells as the model to explore the regulatory role of miRNA373 on SiHa cell growth and apoptosis.
Reagents and methods
Reagents and cell model
Cervical squamous cell carcinoma cell line, SiHa cells were purchased from AddexBio (San Diego, CA, USA). Protein extraction kit and BCA detection kit were from Beyotime. FBS and cell medium were from Hualan Biological Engineering.
MiRNA373, scramble miRNA, and siRNA IAPs were got from Genepharma. Primary antibodies were from Santa Cruz. Lipofectamine was from Invitrogen. MTT was from Beijing Dingguo Changsheng Biotechnology.
Cell culture
SiHa cells were resuscitated and cultured in DMEM high-glucose medium.
Transfection
MiRNA373 and control miRNA were transfected to SiHa cells using the lipofectamine method. Specially, the cells were seeded at the density of 65%. A total of 2
MTT assay
SiHa cell viability was detected by MTT according to the conventional method [10]. Specially, the cells were cultured for 8 h. After transfection for 24 h, the cells were inserted with MTT at 3 mg/ml for 3 h. At last, the reaction was stopped by DMSO for 7 min, and the plate was read on microplate reader at 520 nm [11].
Cell apoptosis assay
After miRNA373 transfection, SiHa cells were collected and added with 3
Western blot
Total proteins were extracted from the cells and separated by SDS-PAGE. After transferring and blocking, the membrane was incubated in primary antibody (1:600) at 4
Caspase-3 activity detection
After miRNA373 transfection, SiHa cells were collected and resuspended in DMEM. The cell protein liquid was added with chromophoric substrate at RT condition. Then the cells were put into the six-well plate and read on microplate reader [17]. Caspase-3 relative activity was calculated as the absorbance in miRNA373 group minus that in control.
IAPs overexpression or interference
SiHa cells were seeded in six-well plate at the density of 65%. A total of 3
Results
MiRNA373 transfection declined SiHa cell viability and inhibited its growth
MTT assay revealed that SiHa cell viability was significantly suppressed after 1
MiRNA373 transfection declined SiHa cell viability and inhibited its growth. 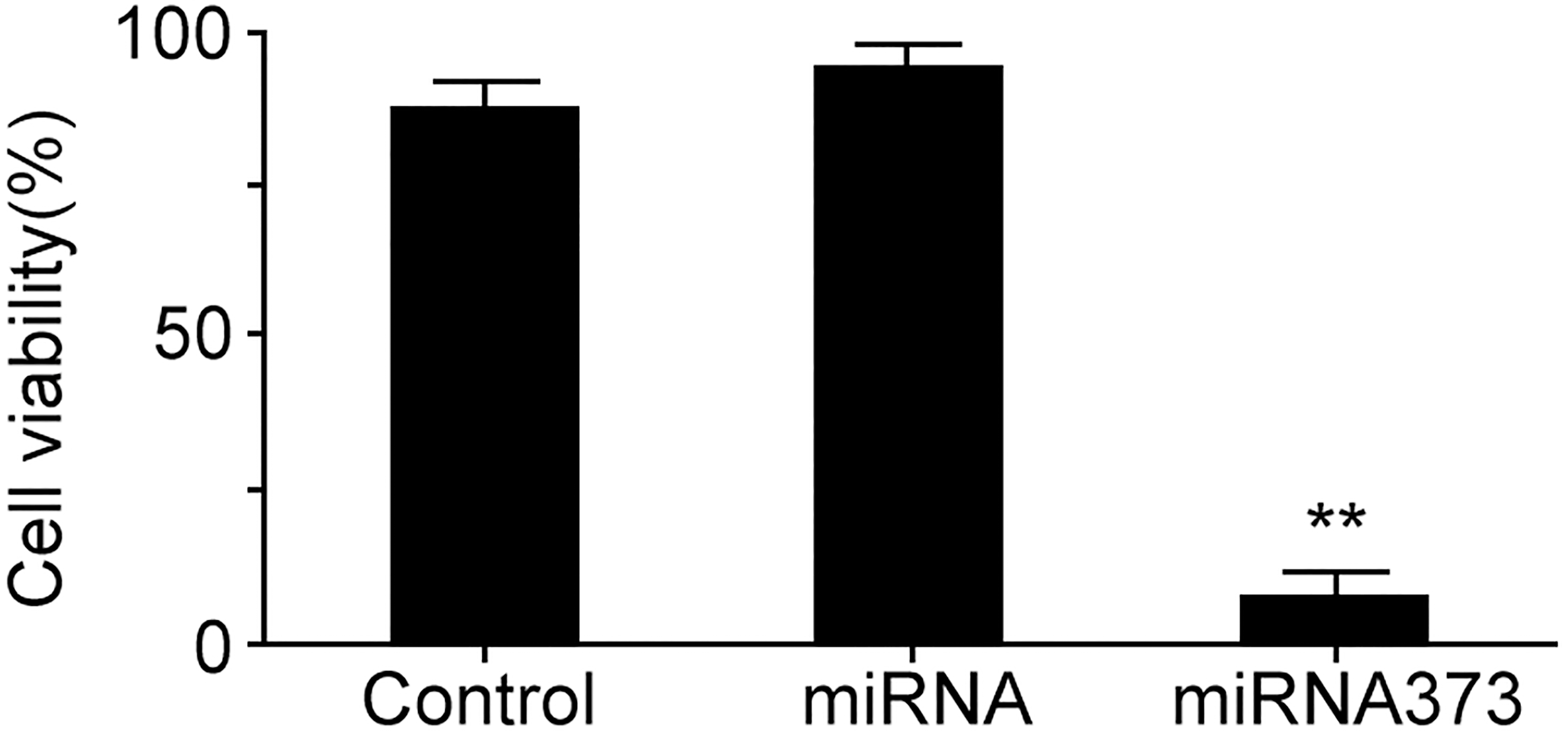
Annexin-V-FTIC was adopted to determine SiHa cell apoptosis. It was showed that phosphatidylserine eversion level was obviously higher after miRNA373 transfection (
MiRNA373 transfection induced SiHa cell apoptosis. 
It was demonstrated that Caspase-3 activity mark-edly increased in SiHa cells after miRNA373 transfection compared with miRNA group (
MiRNA373 transfection activated caspase-3 activity in SiHa cells. 
Western blot was performed to determine IAPs expression in SiHa cells. As shown in Fig. 4, IAPs level in SiHa cells from miRNA373 transfection group was obviously lower than that from miRNA transfection group (
MiRNA373 transfection reduced IAPs expression in SiHa cells.
To investigate the impact of IAPs on miRNA373 induced SiHa cell apoptosis, we applied siRNA to decline IAPs level and then transfected miRNA373 to SiHa cells.
IAPs knockdown enhanced miRNA373 induced SiHa cell apoptosis. 
As shown in Fig. 5, Western blot presented that siRNA IAPs markedly reduced IAPs expression compared with control. IAPs level exhibited statistical differences between miRNA373 group and siIAPs
Caspase-3 detection revealed that SiHa cell apoptosis was obviously enhanced in miRNA373 group after siRNA IAPs transfection (
To explore the effect of IAPs on miRNA373 induced SiHa cell apoptosis, we adopted overexpression of IAPs and detected cell apoptosis after miRNA373 transfection.
IAPs overexpression inhibited miRNA373 induced SiHa cell apoptosis. 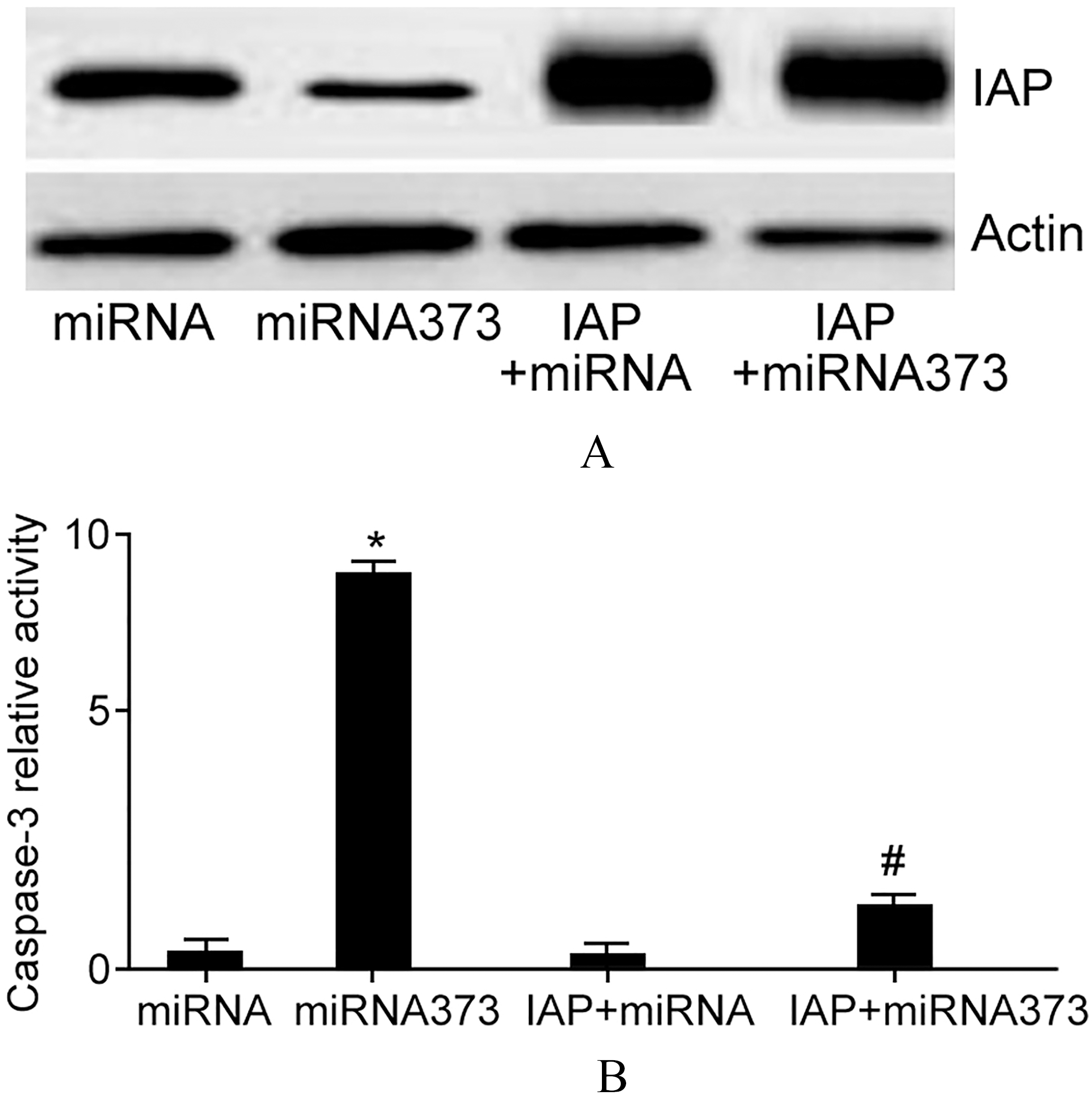
Figure 6 demonstrated that IAPs plasmid obviously upregulated its level in SiHa cells. IAPs level showed statistical differences between miRNA373 group and IAPs
Caspase-3 detection suggested that SiHa cell apoptosis was obviously suppressed in miRNA373 group after IAPs plasmid transfection (
This study adopted cervical squamous cell carcinoma SiHa cells as model to explore miRNA373 impact on SiHa cell apoptosis from molecular and protein levels. Our results showed that miRNA373 transfection reduced SiHa cells viability and growth, leading to SiHa cell apoptosis. This is in accordance with the previous research that miRNAs were involved in cell growth and survival [3]. There was still lack of reports about the role of miRNA in cervical squamous cell carcinoma [3]. MiRNA218 inhibits cancer cell growth, whereas miRNA34a is associated with tumor metastasis [13, 14], suggesting that miRNAs may be involved in cervical squamous cell carcinoma occurrence and development [13, 14, 15].
IAPs is a type of anti-apoptosis protein [23], while its role in SiHa cell growth and whether could be regulated by miRNA373 has not been elucidated [24, 25]. Our results suggested that miRNA373 transfection declined IAPs level in SiHa. MiRNA373 transfection combining with IAPs knockdown obviously elevated SiHa cell apoptosis, whereas IAPs transfection inhibited miRNA373 induced SiHa cell apoptosis.
Our study proved the role of IAPs protein in miRNA373 induced SiHa cell apoptosis. Firstly, miRNA373 transfection significantly decreased IAPs protein level in SiHa cells. Secondly, IAPs siRNA transfection obviously enhanced SiHa cell apoptosis induced by miRNA373 transfection. Lastly, IAPs plasmid markedly suppressed miRNA373 induced SiHa cell apoptosis. It suggested the function of IAPs protein in SiHa cell apoptosis induced by miRNA373. It indicated that IAPs protein might be treated as new strategy for the molecular target therapy of cervical squamous cell carcinoma [26]. Currently, IAPs also played an inhibitory role in other cancers [22, 25]. However, there is still lack of reports about the interaction between miRNA373 and IAPs in regulating cervical squamous cell carcinoma.
So far, no direct targeting effect of miR-373 on IAPs mRNA has been reported or validated by in silico analysis. RNA immune coprecipitation technique could be further included to detect the targeting relationship between IAPs and miR-373. Additionally, our data showed that the simple interference IAPs did not affect cell viability, suggesting that other molecules may be involved in miRNA373 induced apoptosis [25]. We proposed that indirect effect of miR-373 on IAPs may exist via targeting regulation of pro-apoptotic proteins during the induction of apoptosis, though the specific mechanism is still unclear. Therefore, further investigation will also focus to determine the direct target of miR-373 on the regulation of IAP-related apoptosis.
Our study also had some drawbacks. Firstly, we did not collect cervical squamous cell carcinoma tumor tissue and para-carcinoma tissue in clinic, thus cannot test IAPs protein levels in the clinical samples [27]. Secondly, we did not investigate IAPs levels in cervical squamous cell carcinoma after treatment, thus cannot confirm the relationship between IAPs and cervical squamous cell carcinoma. Lastly, this study was lack of animal model to evaluate the curative effect of miRNA373 on cervical squamous cell carcinoma
Footnotes
Acknowledgments
This project supported by the Jilin Provincial Health Special Project (No. 20160804), the Postdoctoral Science Foundation of China (No. 2013M540256), the Youth Foundation of Jilin Province (No. 20140520018 JH) and the Fundamental Research Project of Jilin University (No. 450060481216).
Conflict of interest
None.
Conclusion
To sum up, miRNA373 may suppress SiHa cell viability and induce cell apoptosis by downregulating IAPs, suggesting that IAPs may be treated as the therapeutic target for cervical squamous cell carcinoma.
