Abstract
OBJECTIVE:
This study aims to investigate the significance of combined detection of HER2 gene amplification and chemosensitivity in gastric cancer.
METHODS:
Immunohistochemistry (IHC), fluorescence in situ hybridization (FISH) and fluorescence reverse-transcription polymerase chain reaction (RT-PCR) were used to analyze the expression of HER2 protein, HER2 gene amplification and the mRNA expression of ERCC1, TUBB3 and TYMS genes in 135 cases of gastric carcinoma.
RESULTS:
The expression rate of HER2 protein was 39.3% (53/135). Among these positive cases, patients with HER2 protein (3+) accounted for 9.6% (13/135), patients with HER2 protein (2+) accounted for 13.3% (18/135), and patients with HER2 protein (1+) accounted for 16.3% (22/135). The amplification rate of the HER2 gene was 35.8% (19/53). In the detection of the mRNA expression of ERCC1, TUBB3 and TYMS, 45 patients had low and moderate single gene expression, 50 patients had low and moderate double gene expression, 22 patients had low and moderate mRNA expression for ERCC1, TUBB3 and TYMS, and 18 patients had no low and moderate expression. Among the 53 patients with HER2 protein expression and 22 patients with low and moderate mRNA expression of ERCC1, TUBB3 and TYMS, 12 patients received chemotherapy and trastuzumab. Follow-up results revealed that HER2 gene status was positively correlated with the therapeutic effect of the combined treatment in patients with low mRNA expression of ERCC1, TUBB3 and TYMS. Among these patients, five patients with extensive HER2 (3+), HER2 cluster-specific amplification, and low mRNA expression of ERCC1, TUBB3 and TYMS had a total survival of up to 19.1 months.
CONCLUSIONS:
The detection of HER2 in gastric cancer is highly heterogeneity, and the combined detection of HER2 protein expression, HER2 gene amplification and chemosensitivity can provide important reference markers for the benefit of antitumor drugs.
Introduction
“Precision medicine” has become a hot topic, which advocates treating the same disease with different treatments. Hence, a patient needs different treatment plans at different stages. With the development of molecular pathology, each patient can be accurately typed and treated [1, 2]. In recent years, focus has been directed at signal transduction, growth factors and their receptors, several novel drugs have been developed, and important results have been achieved. One of these protein products is human epidermal growth factor receptor 2 (HER2). Its high expression can activate the signal transduction system of cells, and induce cell transformation and proliferation [3, 4]. HER2 is a widely used therapeutic target in breast cancer [5, 6, 7]. Accumulating evidence [1, 2, 3, 4, 5] suggests that HER2 is an important marker of gastric cancer, and a high HER2 expression is most beneficial to tumor patients [8, 9]. Compared with breast cancer, gastric cancer exhibits unique immunostaining characteristics, and there is high tumor heterogeneity in HER2 positive patients. Hence, standardizing the determination of the expression of HER2 in gastric cancer is a guarantee of correct medication. In addition, chemotherapy is the main treatment option for patients with middle and late stage gastric cancer. In clinical practice, chemotherapeutic agents are blindly selected. As a result, some patients fail to benefit from chemotherapy. Instead, their quality of life is affected by many adverse reactions, and the best time of treatment is missed. The common chemotherapeutic agents for gastric cancer include fluorouracil, platinum-based drugs, and paclitaxel. In the present study, immunohistochemistry (IHC) was used to detect the expression of HER2 protein in gastric cancer, fluorescence in situ hybridization (FISH) was used to detect the status of the HER2 gene, and fluorescence reverse-transcription polymerase chain reaction (RT-PCR) was used to detect the expression of fluorouracil, platinum-based drugs and paclitaxel, in order to evaluate the diagnostic criteria of FISH, IHC and fluorescence RT-PCR for gastric cancer, provide a quantitative reference for the benefit of antitumor drugs, and guide the individualized selection of drugs.
Materials and methods
Materials
A total of 135 cases of radical resection of the stomach and esophagogastric junction were performed in the Pathology Department of the 152nd Central Hospital of Pingdingshan and the 159th Central Hospital of Zhumadian from January 2013 to October 2015, and samples were collected. The histological diagnostic criteria were based on the literature “Stomach Tumor Pathology” and the 2010 WHO Classification of Tumors of the Digestive System. The age of these patients ranged within 25–74 years, with an average age of 62.4 years old. Within 30 minutes after resection from the body, the samples were fixed with freshly prepared 10% neutral buffered formalin (NBF) for 8–48 hours. The fixation fluid-to-tissue volume ratio was 10:1. From the tumor tissues, 4–6 pieces was obtained (the proximal and distal margins were routinely cut, but the tumors, the adjacent gastric mucosa and other tissue blocks were not included), which included the deepest invasion point and the closest serosal layer. The samples were detected by hematoxylin and eosin (H&E), IHC, FISH and fluorescence RT-PCR, respectively.
Reagents
HER2 immunohistochemical staining, HER2 HercepTestTM kit and pharmDxTM kit approved by FDA were purchased from Dako Cytomation (Denmark).
Reagent and probe
Paraffin Pretreatment Kit II, which mainly contains the pretreatment solution and protease solution; PathVysionTM HER2 probe kit; Human ERCC1, TUBB3 and TYMS gene expression relative quantification kit (PCR, fluorescent probe method); ERCC1, TUBB3 and TYMS PCR reaction solution, primer/ probe mixture, positive controls, negative controls, internal PCR reaction solution, internal primer/probe mixture, internal positive control, and internal negative control. The same components between different batches could not be interchanged.
Methods
Immunohistochemical staining
Interpretation of the positive results of staining: when the cell membrane of tumor cells in all 4–6 slices of one case was unstained, the result was 0; when the membrane of tumor cells was slightly or faintly stained, the result was 1+; when the basal membrane, lateral membrane, or whole membrane of tumor cells were weakly or moderately stained, the result was 2+; when the basal membrane, lateral membrane, or the whole membrane of tumor cells had strongly positive staining, the result was 3+.
Interpretation of the positive area of staining: when the cell membrane of tumor cells in all 4–6 slices in one case was unstained, the result was negative; when the stained area was
FISH test
Reagents, probes and ISH procedures: The pretreatment procedures and process steps for the paraffin-embedded gastric cancer tissue sections were carried out according to a related literature [9] and the instructions of the kits.
Determination of FISH results
FISH results were determined mainly through the HER2 positive regions on the sections detected by IHC. First, the positive region of cancer cells was identified on H&E stained sections. Then, the same field of view of H&E staining was found on the FISH slice under a 10
Fluorescence RT-PCR
Paraffin sections should contain sufficient proportions of tumor cells, and have no protein or DNA contamination. Samples were processed using a commercially available RNA extraction kit, and the operation was carried out strictly in accordance with kit instructions. For ERCC1, TUBB3 and TYMS positive controls and internal positive controls, calibrated RNA controls did not need to be extracted. Instead, these were melted before use, subjected to vortex-induced oscillation for 10 seconds, centrifuged at 2,000 rpm for 15 seconds, and the number of reactions (the number of the first batch of samples to be inspected
Expression level criteria:
Sensitive chemotherapies: DDP (C) for low and moderate expression of ERCC1, docetaxel (D) for low and moderate expression of TUBB3, and 5-FU (F) for low and moderate expression of TYMS.
Statistics analysis
Data were statistically analyzed using statistical software SPSS 13.0. HER2 gene status, HER2 gene amplification, and the mRNA expression levels of ERCC1, TUBB3 and TYMS were compared between the two groups using t-test and chi-square test. P
Relationship between the expression of HER2 gene protein and histological type in 135 cases of gastric carcinoma
Relationship between the expression of HER2 gene protein and histological type in 135 cases of gastric carcinoma
The positive expression of HER2 protein in gastric adenocarcinoma, En Vision (
Histomorphology of gastric cancer
Among the 135 patients with gastric cancer, 25 patients had papillary adenocarcinoma (18.5%), 37 patients had tubular adenoma (27.4%), 23 patients had mucinous adenocarcinoma (17%), 11 patients had low adhesion adenocarcinoma (8.1%), and 39 patients had mixed adenocarcinoma (28.9%). Mixed adenocarcinoma refers to the mix of different histological structures of papillary adenocarcinoma, tubular adenocarcinoma, mucinous adenocarcinoma, and low adhesion adenocarcinoma. In the present study, all patients were at stage pT2-4.
Expression rate and expression characteristics of HER2 protein in gastric cancer
The relationship of the expression rate of HER2 protein and its histological types in gastric cancer is shown in Table 1. The positive expression of HER2 protein was located in the cell membrane, and the expression rate was 39.3% (53/135). Among these patients, three patients were HER2 (3+) extensive type (Fig. 1), seven patients were (3+) partial type, and three patients were (3+) focus type. These patients accounted for 9.6% (13/135). Furthermore, five patients were HER2 protein (2+) extensive type, seven patients were (2+) partial type, and six patients were (2+) focus type. These patients accounted for 13.3% (18/135). Moreover, seven patients were HER2 positive (1+) extensive type, eight patients were (1+) partial type, and seven patients were (1+) focus type. These patients accounted for 16.3% (22/135).
The positive intensity and area distribution of HER2 protein
The positive intensity and area distribution of HER2 protein
Comparison of HER2 protein positive and HER2 gene amplification results
Relationship between HER2 gene amplification and expression levels of ERCC1, TUBB3 and TYMS genes mRNA
Detection of the HER2 gene in gastric cancer using the FISH method. The red area is the probe signal and the green area is chromosome 17 (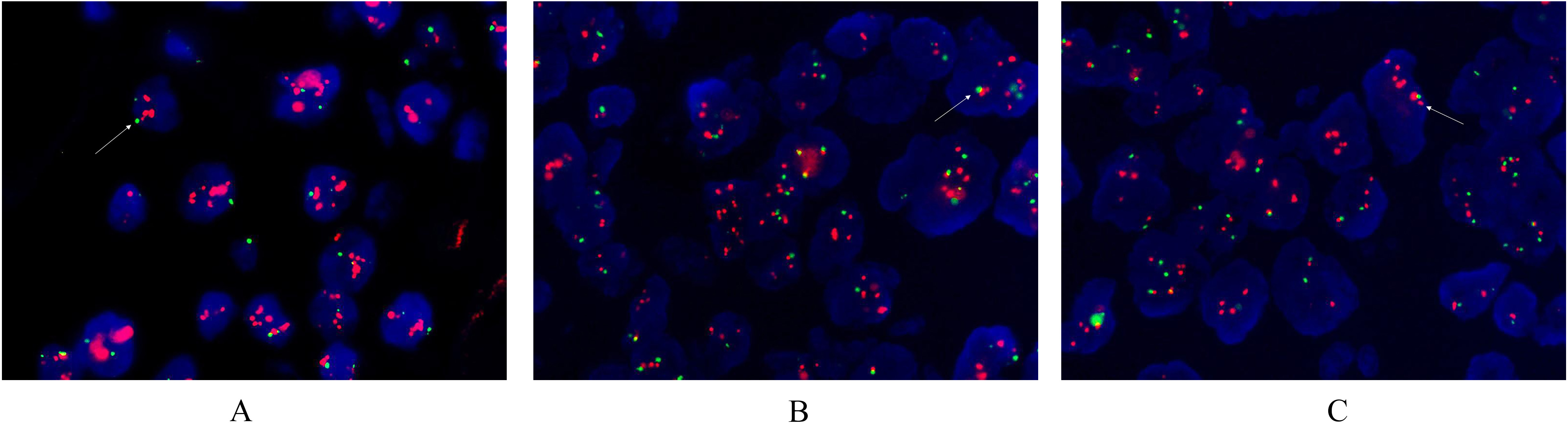
The significant feature of HER2 protein expression in gastric cancer is heterogeneity. If there was only either an extensive, partial, or focal type in 4–6 sections of one patient, it was defined as single-signal type; and single-signal type patients accounted for 62.3% (33/53). If there was two or more than two of HER2 (3+), HER2 (2+) and HER2 (1+) and the negative types in 4–6 sections of one patient, it was defined as multi-signal type; and multi-signal type patients accounted for 37.7% (20/53, Table 2).
Comparison of HER2 gene amplification with chemotherapy alone and trastuzumab in combination with chemotherapy in 11 patients
The mRNA expression of ERCC1, TUBB3 and TYMS. A: Low expression of a single-gene. The low expression of the ERCC1 gene, and the high expression of the TUBB3 and TYMS genes are shown. B: Low expression of a double-gene. The low expression of the ERCC1 and TYMS genes, and the high expression of the TUBB3 gene are shown. C: Low expression of a three-gene. The low expression of the ERCC1, TYMS and TUBB3 genes are shown.
The amplification rate of the HER2 gene was 35.8% (19/53, Table 3). Patients with HER2 protein (3+) accounted for 92.3% (12/13), patients with HER2 protein (2+) accounted for 33.3% (6/18), and patients with HER2 protein (1+) accounted for 4.5% (1/22). Morphological types of HER2 gene amplification in 19 patients: patients with HER2 gene cluster-specific amplifications accounted for 14.3% (7/19, Fig. 2), patients with large granular amplifications accounted for 10.2% (5/19), patients with spotty amplifications accounted for 6.1% (3/19), and patients with high multi-bodies accounted for 8.2% (4/19).
ERCC1, TUBB3 and TYMS gene mRNA expression and its relationship with HER2 gene amplification
For the mRNA expression of ERCC1, TUBB3 and TYMS, 45 patients had low and moderate single-gene expression (Fig. 3), 50 patients had low and moderate double-gene expression, 22 patients had low and moderate expression of all ERCC1, TUBB3 and TYMS mRNAs, and 18 patients had no low and moderate expression. The relationship between HER2 gene amplification and the mRNA expression of ERCC1, TUBB3 and TYMS genes is shown in Table 4.
Treatment based on the combined detection of HER2 gene amplification and ERCC1, TUBB3 and TYMS gene mRNA expression levels versus simple chemotherapy regimen
The treatment regimen based on the detection of HER2 gene amplification combined with ERCC1, TUBB3 and TYMS gene mRNA expression levels was called the combination regimen, while the treatment regimen that used chemotherapeutic drugs alone was called simple chemotherapy (chemotherapy regimens C, D and F). Among the 53 patients with HER2 protein expression and 22 patients with low and moderate mRNA expression of ERCC1, TUBB3 and TYMS, 12 patients received chemotherapy and trastuzumab (Table 5).
Discussion
In recent years, trastuzumab (trade name Herceptin), a monoclonal antibody targeting HER2 protein, has been verified to improve the survival rate of patients with primary and metastatic HER2 positive breast cancer [10]. In order to select the most suitable patient to receive this treatment, and avoid unnecessary medication for some patients, in 2007, the American Society of Clinical Oncology (ASCO)/American College of Pathologists (CAP) published an updated version of detection guidelines [11] based on the 2007 edition of the ASCO/CAP HER2 detection guideline [12]. This puts forward new requirements for the technical route, interpretation standard of results and quality control of HER2 detection in breast cancer, and is a reference with significance of evidence-based medicine, marking a major step towards the standardization of HER2 detection. For gastric cancer, related studies were subsequently carried out. A recent global multi-centre phase III clinical trial (ToGA) that involved 24 countries including China revealed the efficacy of trastuzumab for patients with HER2 positive advanced gastric cancer and gastroesophageal junction cancer [13, 14, 15]. Subsequently, the United States Food and Drug Administration (FDA) approved the drug for combination chemotherapy for the treatment of patients with HER2 positive metastatic gastric cancer and gastroesophageal junction cancer [11, 12]. At present, the IHC method is generally used to detect the HER2 protein expression status at home and abroad. However, in gastric cancer, the overexpression rates of HER2 protein in different studies were inconsistent, and most of these concentrated within 6.8–34.0% [16, 17, 18]. The detection of HER2 in gastric cancer is different from that in breast cancer, and these detection results are influenced by many factors, including the preoperative processing and fixation of samples, reagents and methods of detection, laboratory quality control, and the interpretation of staining results [19, 20]. In the present study, 135 patients with gastric cancer underwent combined detection using IHC and FISH techniques. The expression rate of HER2 protein was 39.3% (53/135). Among these positive cases, patients with HER2 protein (3+) accounted for 9.6% (13/135), patients with HER2 protein (2+) accounted for 13.3% (18/135), and patients with HER2 protein (1+) accounted for 16.3% (22/135). The amplification rate of the HER2 gene was 35.8% (19/53). Morphological types of HER2 gene amplification in 19 patients: patients with HER2 gene cluster-specific amplification accounted for 14.3% (7/19), patients with large granular amplification accounted for 10.2% (5/19), patients with spotty amplification accounted for 6.1% (3/19) and patients with high multi-body accounted for 8.2% (4/19). These results were consistent with those reported in related literatures [21, 22, 23, 24, 25]. The higher the expression level of HER2, the more likely the patient would benefit from the treatment of anti-tumor drugs targeting HER2. However, a literature stated that the interpretation standard for the expression of HER2 protein in gastric cancer is that patients with 3+ in
At present, chemotherapy for gastric cancer is usually based on combination regimens, and the common drugs of fluorouracil, platinum-based drugs and paclitaxel are commonly used. The combination of three drugs is a relatively intensive chemotherapy regimen, and the chemotherapy of a single-drug or a combination of two drugs is a non-intensive regimen. The combination of three drugs has significant advantages in survival, but the toxicity and side effects are greater. Since gastric cancer has a poorer prognosis in all solid tumors, heterogeneity is high, drug sensitivity is poor, and it has a higher requirement of individualized treatment. In clinical studies and clinical practice, attention must be given to these patients and tumor characteristics, in order to reasonably and individually select drugs. This is the only possible means that would really benefit patients and prolong their survival time. This would allow people focus more attention to the high heterogeneity of gastric cancer. Realizing an individualized treatment of cytotoxic drugs and targeted drugs in progressive gastric cancer has become a hotspot in this field [26, 27, 28]. In the present study, the combination of IHC, FISH and fluorescence RT-PCR was used to detect the targeted and chemotherapeutic agents for gastric cancer. Among the 53 patients with HER2 protein expression and 22 patients with low and moderate mRNA expression of ERCC1, TUBB3 and TYMS, 12 patients received regimen of cetuximab combined with oxaliplatin, leucovorin or fluorouracil; and these patients were followed up for 27 months. Follow-up results revealed that the HER2 gene status was positively correlated with the therapeutic effect of the combined treatment in patients with low mRNA expression of ERCC1, TUBB3 and TYMS. Among these patients, five patients with extensive HER2 (3+), HER2 cluster-specific amplification, and low mRNA expression of ERCC1, TUBB3 and TYMS had a total survival time of up to 19.1 months. Furthermore, the maintenance treatment of targeted drugs was used, and the progression free survival (PFS) was 9.7 months. In addition, both objective response rate (ORR) and disease control rate (DCR) significantly improved. The detection of HER2 in gastric cancer is highly heterogenous, and the combined detection of HER2 protein expression, HER2 gene amplification and the chemosensitivity marker can provide important reference markers for the benefit of antitumor drugs. The combined detection of targeted and chemotherapeutic drugs for gastric cancer is worth to be popularized in the selection of individualized regimens.
In summary, the standardization of the detection criteria of HER2 and drug sensitivity markers in gastric cancer is a guarantee of correct medication. The detection of HER2 in gastric cancer is highly heterogeneity, and the specification and refinement of HER2 detection is helpful to guide the medication. The classification of extensive partial, and focal types, as well as the range and intensity of HER2 detection, can guide and help to evaluate the benefit of targeted drugs. In the targeted therapy of gastric cancer, it is necessary to simultaneously detect the chemosensitivity markers. It is an inevitable trend to select chemotherapy regimens for the individualized treatment of gastric cancer by detecting these chemosensitivity markers.
Footnotes
Acknowledgments
Key scientific and technological projects in Henan Province funded plan, No. 132102310008.
