Abstract
Background:
Studies on the prognostic significance of site-specific distant metastasis, multiple-site metastases, and the impact of surgery of the primary tumor and metastatic lesion on survival outcomes of patients with metastatic gastric cancer (GC) remain elusive. Therefore, this study aimed to investigate the prognostic significance of the site of distant metastasis among patients with metastatic GC. Furthermore, the effect of surgery of the primary tumor and metastatic lesion on the prognosis of metastatic GC was also analyzed.
Methods:
The data of 4,221 eligible patients, who were diagnosed with metastatic GC between 2010 and 2015, were identified from the Surveillance Epidemiology and End Results (SEER) database. Multivariate logistic regression analysis was performed to assess the association between potential prognostic factors, including the site of metastasis and surgery, and survival of patients with metastatic GC. Overall survival (OS) and cause-specific survival (CSS) were determined using the Kaplan-Meier survival curves and differences were assessed using the Log-rank test.
Results:
Out of the total 4,221 GC patients with definite organ metastases, 3312 patients had single-site metastasis while 909 patients had multiple-site metastases. GC patients with single-site metastasis of liver or lung exhibited better CSS and OS compared to those with bone metastasis. Furthermore, GC patients with liver metastasis benefited from surgery of both the primary and metastatic lesions, while those with lung metastasis benefited from surgery of metastasis resection only. Multivariate Cox regression analysis revealed that GC patients with single-site metastasis, well-differentiated tumors, GC patients who underwent surgery of the primary tumor and those who received chemotherapy exhibited favorable prognosis.
Conclusions:
The site of metastasis was an independent prognostic factor for metastatic GC. Surgery had survival benefits in certain cases of metastatic GC; however, further studies are warranted to clarify these benefits in carefully selected patients.
Background
Gastric cancer (GC) ranks the fifth most frequently diagnosed cancer worldwide. Due to its increased tendency to metastasize to vital organs including the liver, lungs, bone, and brain, GC is the third leading cause of cancer death. 1 According to the estimation of American Cancer Society in 2019, there would be approximately 27,510 new cases and 11,140 deaths in the USA in 2019. 2 Notably, prognosis of GC depends largely on the clinical stage at presentation. 3 However, due to a lack of early alarming symptoms and limited availability of early screening programs, GC remains unnoticed until advanced stages and approximately 40% of patients will present with distant metastases at the time of initial diagnosis, where treatment options are limited. 4 Consequently, the prognosis of patients with metastatic GC remains poor with median survival being less than 1 year and the 5-year survival rate being less than 10% without surgery. Nevertheless, surgery as the standard curative therapy has markedly improved the survival of patients with early GC. Besides, a multimodality treatment strategy including surgery combined with neoadjuvant and adjuvant therapies has been recommended to improve the survival of locally advanced stages. 5,6 Thus, surgery represents a curative opportunity for patients with GC and is considered the foundation of multimodal management of GC. 7 In this context, several retrospective studies have provided evidence of a potential benefit of gastrectomy in metastatic GC as well. 8 -14
Although chemotherapy or chemotherapy combined with targeted therapy remains the cornerstone of the palliative treatment of advanced or metastatic GC, surgery has demonstrated survival benefit in patients with metastatic GC. Accumulating studies have revealed that liver resection in GC patients with liver metastasis is considered favorable with marked 5-year survival and could be offered to selected patients. 8 While improved surgical interventions and adjuvant therapies remain the upfront research topic, studies on careful patient selection remain scarce. Moreover, due to the paucity of epidemiological studies, predicting the prognosis of the patients with metastatic GC remains elusive. 15 Besides, the decision to perform surgery of primary tumors and/or metastases remains clinically challenging. Therefore, it becomes imperative to identify patterns of metastases in order to articulate the best treatment decision and optimize the follow-up strategies. Considering these pieces of evidence, this study aimed to determine the epidemiology of preferential sites of distant metastasis in patients with metastatic GC. Furthermore, the effects of surgical resection of the primary tumor and metastatic lesion on the prognosis of metastatic GC were also analyzed.
Methods
Data Collection
For this study, the data of metastatic GC patients were retrieved from the Surveillance Epidemiology and End Results (SEER) database of the USA National Cancer Institute, which is the largest publicly available cancer database and consists of a consortium of 18 population-based regional cancer registries with accurate and consistent data and includes representative cancer statistics from an estimated 28% of the American population, The SEER registries routinely collect data on patients’ demographic information and clinicopathological characteristics including the primary tumor site, morphology, stage at diagnosis, first course of treatment, and follow-up for vital status. We used the SEER*Stat software version 8.3.6 with the approval from the SEER program (Username: 10053-Nov2018) to assess the data. The SEER database provided patients data up to 2016 based on the November 2018 submission. This study was approved by the Institutional Review Board of the Affiliated Hospital of Southwest Medical University. As it was based on the public data of the SEER database, the requirement for informed patient consent was waived.
Patient Selection
A total of 40,728 patients with GC were identified within the SEER database between 2010 and 2015. Data on the peritoneum and other uncommon metastatic organs such as adrenal glands and spleen was unavailable. Based on the inclusion and exclusion criteria, a total of 4,221 gastric adenocarcinomas with defined liver, lung, bone, and brain metastases were included in this study. Patients’ age at diagnosis, gender, ethnicity, primary tumor site, grade, histological type, TNM stages based on the 7th edition staging manual updated by the American Joint Committee on Cancer (AJCC), marital status, surgery of the primary tumor, surgery of the metastases, chemotherapy, radiotherapy, survival months, and cause of death for patients with GC were collected from the SEER database. The inclusion criteria were as follows: The primary site code represented “stomach” (C16.0-16.9); histology codes denoted “adenocarcinoma” (ICD-03, 8140/3, 8142/3, 8143/3, 8144/3, 8145/3, 8210/3, 8211/3, 8255/3, 8260/3, 8261/3, 8263/3, 8310/3, 8323/3), “mucinous” (ICD-03, 8480/3, 8481/3) and “signet ring cell carcinoma” (ICD-03, 8490/3); patients with identified bone, brain, lung, or liver metastasis. The exclusion criteria were as follows: patients who survived less than 1 month; patients with incomplete data about distant metastatic sites; patients with incomplete data about primary and/or metastatic surgery; patients with missing or incomplete information about survival, follow-up months, cause of death or other characteristics. The unknown clinical data were presented as an unknown category. The data extraction process is illustrated in Figure. 1.

Schematic representation of the data extraction procedure applied in the study.
Statistical Analysis
Cause-specific survival (CSS) and overall survival (OS) were defined as two primary endpoints of the present study. CSS was defined as the survival time from the time of diagnosis to the date of the death from GC. The patients who died from causes unrelated to GC or who survived after the follow-up deadline were defined as censored. OS was calculated from the date of diagnosis to the date of death from any cause or the last follow-up. In this study, CSS estimated the probability of surviving GC. Survival curves were estimated using the Kaplan-Meier method, and the significant differences between the survival curves were assessed by the Log-rank test.
Independent variables were first analyzed using univariate analysis. Variables that were significantly associated with surgery of primary and metastatic lesions in GC patients identified by univariate analysis were then entered into a Cox proportional hazards regression model for multivariate analysis, yielding hazard ratios (HR). The model analyzed nine significant variables, including age, gender, primary tumor site, marital status, race, histological type, TNM staging, metastatic site, and chemotherapy. All statistical analyses were two-sided. P-values of < 0.05 were considered statistically significant. All statistical analyses were performed with R software (version 3.6.0).
Results
Patient Characteristics
A total of 4221 eligible GC patients with definite organ metastasis (liver, lung, bone and brain) were included in this study, of whom 3312 had single-site metastasis while 909 had multiple-site metastases. In the overall patient cohort, there were 3133, 1041, 956, and 156 patients with liver, lung, bone, and brain metastases, respectively. Notably, the most common single-site of metastasis was liver (70.5%), followed by bone (15.8%), lung (11.9%), and brain (1.8%). The median age at diagnosis was 65 years. The clinicopathological characteristics of patients with metastatic GC are summarized in Table 1. A total of 457 patients underwent surgical resection, of whom 303 had surgical resection of primary GC and 218 had the resection of metastasis. In the subgroup of patients with primary tumor resection, the percentage of patients with bone metastasis was the lowest, approximately 4.0%, while in the subgroup with metastatic tumor resection, the percentage with liver metastasis was found to be the lowest, approximately 3.7%. Furthermore, a total of 2796 patients received chemotherapy and 972 patients were administered with radiotherapy.
Clinicopathological Characteristics of Patients With Metastatic Gastric Cancer, n (%).

*: Non-cardia included fundus, body, gastric antrum, pylorus, greater curvature and lesser curvature of stomach.
**: Differentiated included well and moderately differentiated; undifferentiated included poorly differentiated and undifferentiated.
***: Unmarried included single, divorced, separated, widowed, unmarried or domestic partner.
****: Others include American Indian/AK Native, Asian/Pacific Islander.
Survival Outcomes
OS and CSS in patients with advanced GC were evaluated based on single-site or multiple-site metastases and surgeries performed on the primary or metastatic tumors. The results indicated that advanced GC patients with single-site bone metastasis (p = 0.03 for CSS), brain metastasis (p = 0.03 for CSS), or liver metastasis (p < 0.001 for CSS) benefited the most from primary surgery (p < 0.05) (Figure 2). Furthermore, for GC patients with single-site metastasis undergoing resection for metastatic lesions, those with lung metastasis (p = 0.007 for CSS) and liver metastasis (p = 0.02 for CSS) benefited significantly from surgery than their counterparts with bone metastasis and brain metastasis (Figure 3). However, in terms of GC with multiple-site metastases, there was no significant difference between patients who underwent resection for primary tumor and those who did not have the surgery in their prognosis, nor was there significant difference between those who had resection for metastatic lesions and those who did not (Supplementary Fig. 1).
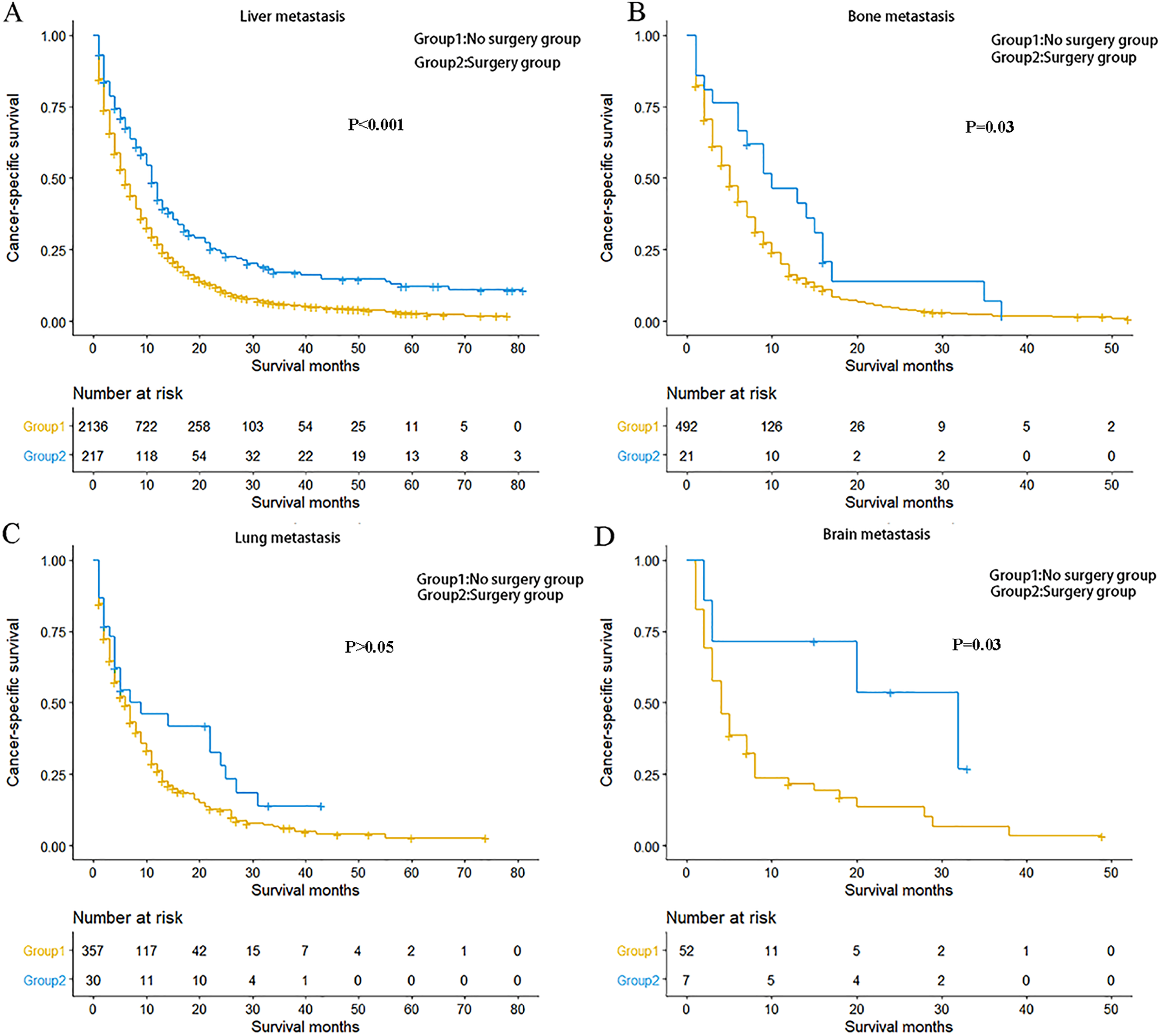
Kaplan-Meier curve of cancer-specific survival based on whether or not surgery of the primary tumor was performed. A, Patients with single-site liver metastasis. B, Patients with single-site bone metastasis. C, Patients with single-site lung metastasis. D, Patients with single-site brain metastasis.
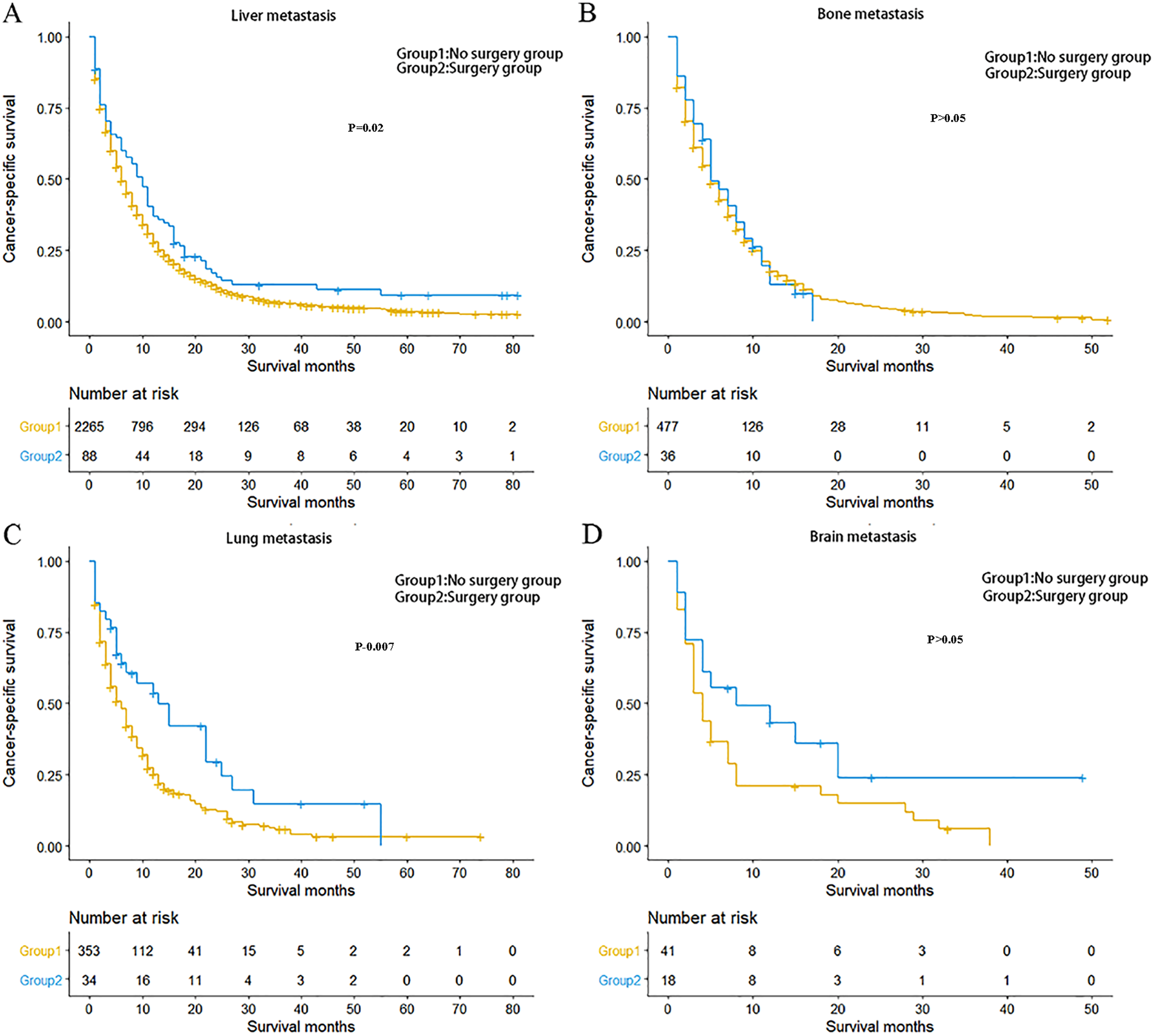
Kaplan-Meier curve of cancer-specific survival based on whether or not surgery of the metastatic lesion was performed. A, Patients with single-site liver metastasis. B, Patients with single-site bone metastasis. C, Patients with single-site lung metastasis. D Patients with single-site isolated brain metastasis.
In the cohort of patients who received chemotherapy, advanced GC patients with the brain (p = 0.02 for CSS) or liver (p < 0.001 for CSS) metastasis benefited significantly from surgery of the primary tumor (Supplementary Fig. 2). Meanwhile, patients with lung (p = 0.01 for CSS) or liver (p = 0.02 for CSS) metastasis benefited significantly from surgery of the metastases (Supplementary Fig. 3).
The median OS for advanced GC patients with single-site bone, brain, lung, and liver metastases were 5, 4, 6, and 6 months, respectively. Advanced GC patients with isolated lung or liver metastasis exhibited better CSS and OS compared with those with bone metastasis (p = 0.001 for CSS and p = 0.002 for OS in lung metastasis; p = 0.002 for both CSS and OS in liver metastasis). However, no significant difference was observed between the prognosis of patients with single-site lung metastasis and those with single-site liver metastasis (p > 0.05 for both CSS and OS). Also, there was no significant difference between the prognosis of patients with single-site bone metastasis and those with single-site brain metastasis (p > 0.05 for both CSS and OS) (Figure 4). Furthermore, the prognostic value of the pattern of metastases was also evaluated for stage IV GC patients. The median OS for patients with one, two, three, and four metastatic sites was 6, 4, 4, and 3 months, respectively. Patients with single-site metastasis exhibited survival benefits over those with multiple-site metastases (p < 0.001 for both CSS and OS). However, no significant difference in the prognosis was observed among patients with two, three, and four metastatic sites (p > 0.05 for both CCS and OS) (Supplementary Fig. 4).
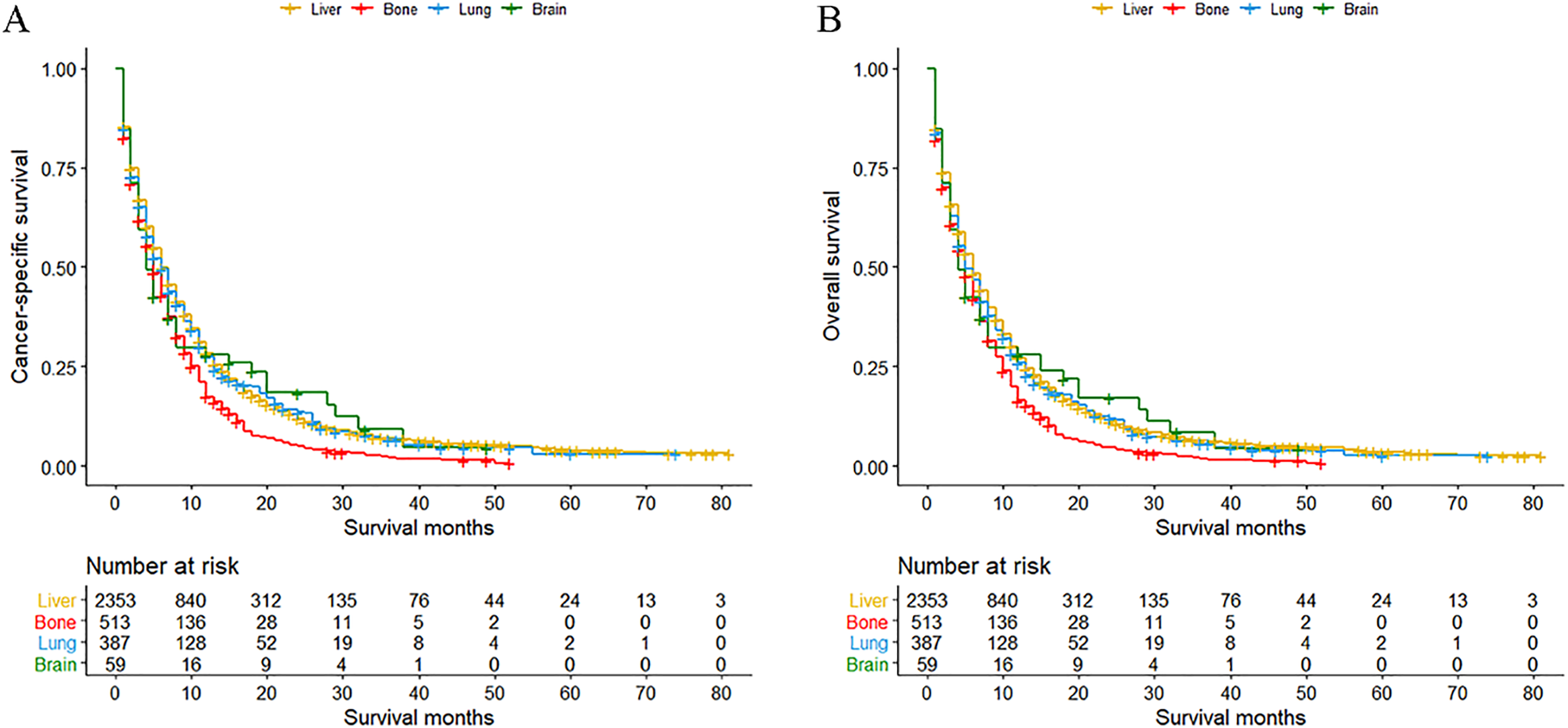
Kaplan-Meier curve of (A) cancer-specific survival and (B) overall survival based on the single-site organ metastases.
Analysis of Prognostic Factors Using Multivariable Cox Regression
Multivariate Cox regression analysis was performed to evaluate the association of significant variables with the prognosis of metastatic GC. The results indicated that both CSS and OS were significantly improved in GC patients who underwent primary resection (HR, 0.48; 95% CI, 0.41-0.56; p < 0.001) and those who received chemotherapy (HR, 0.37; 95% CI, 0.34-0.40; p < 0.001). Lung metastasis (HR, 0.77; 95% CI, 0.67-0.89; p < 0.001) and liver metastasis (HR, 0.82; 95% CI, 0.74-0.92; p = 0.001) were significantly associated with improved CSS and OS compared with bone metastasis (Table 2). In the overall patient cohort, age > 65, singleness, undifferentiated cancer, signet ring cancer, or multiple-site metastases were found to be statistically significant independent prognostic factors for poor survival. However, in the single-site metastasis cohort and the overall patient cohort, there was no significant association between the GC patients who underwent metastatic surgery and CSS/OS (Supplementary Table 1).
Multivariate Cox Regression Analysis for CSS and OS in GC Patient With Single-Site Metastasis.
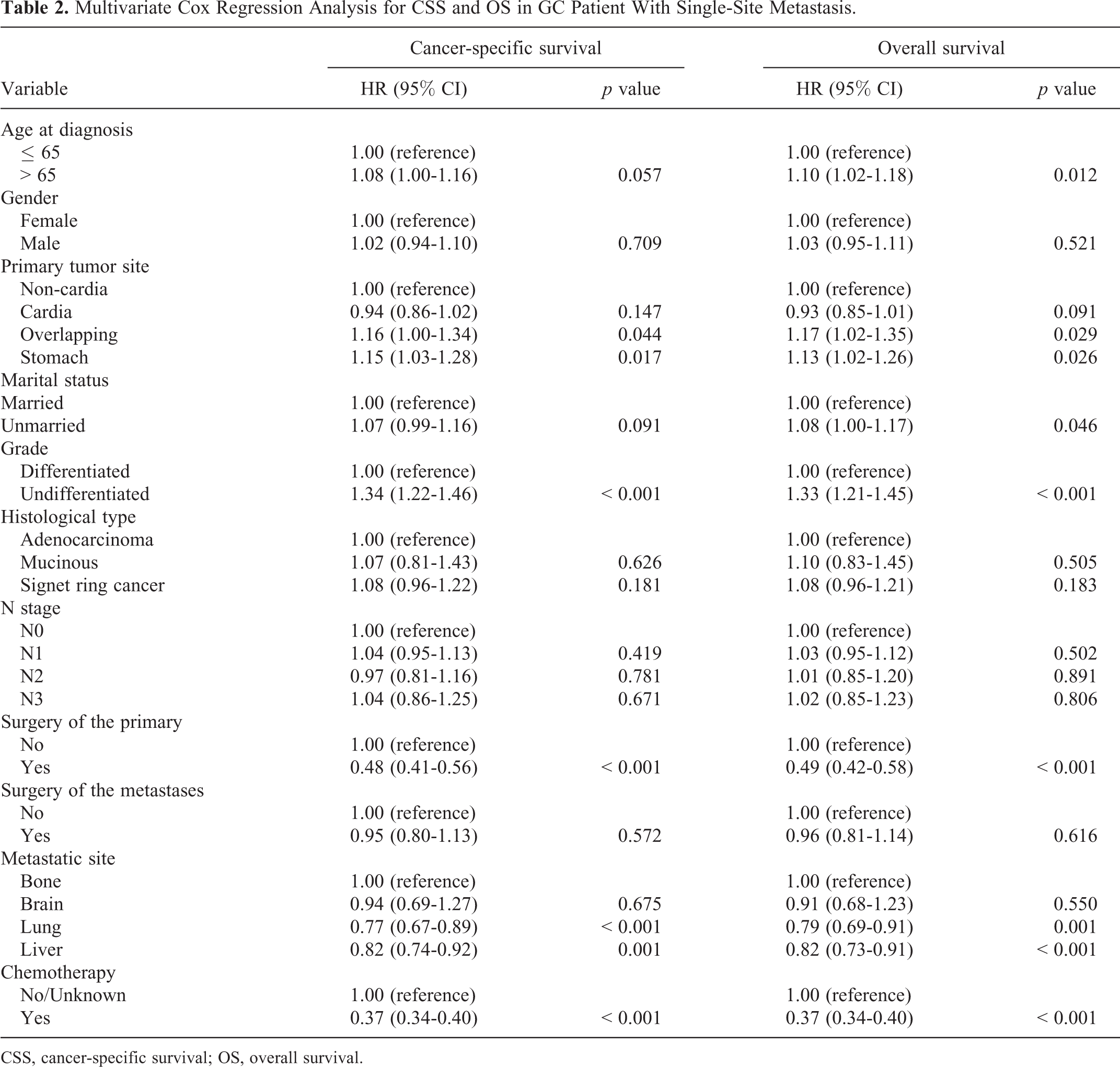
CSS, cancer-specific survival; OS, overall survival.
Prognostic Factors Associated With Surgery
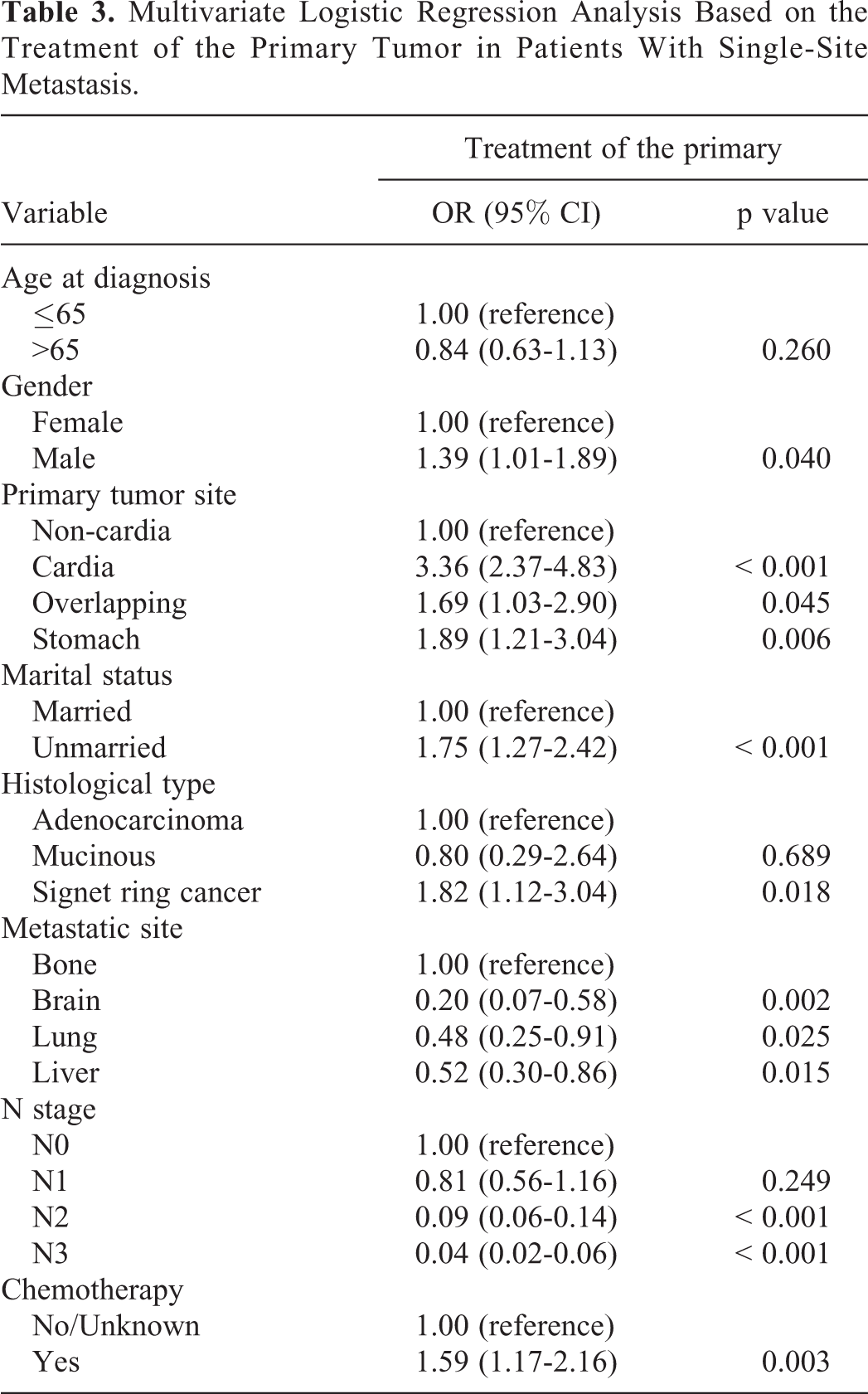
In a multivariable logistic regression model, patients with gastric cardia cancer (OR, 3.36; 95% CI, 2.37-4.83; p < 0.001), who were unmarried (OR, 1.75; 95% CI, 1.27-2. 42; p < 0.001), had signet ring cell cancer (OR, 1.82; 95% CI, 1.12-3.04; p = 0.018), or who underwent chemotherapy (OR, 1.59; 95% CI, 1.17-2.16; p = 0.003) were less likely to undergo primary surgery. Compared with bone metastasis, GC patients with brain (OR, 0.20; 95% CI, 0.07-0.58; p = 0.002), lung (OR, 0.48; 95% CI, 0.25-0.91; p = 0.025) or liver (OR, 0.52; 95% CI, 0.30-0.86; p = 0.015) metastases were more likely to have a more favorable prognosis (Table 3). In the metastatic tumor resection cohort, GC patients who were older than 65 years (OR, 1.44; 95% CI, 1.03-2.02; p = 0.036) or who were male (OR, 1.61; 95% CI, 1.35-2.27; p = 0.007) were less likely to undergo metastatic tumor resection. In addition, patients with mucinous adenocarcinoma of the stomach (OR, 0.27; 95% CI, 0.13-0.61; p < 0.001) and those who underwent primary resection (OR, 0.13; 95% CI, 0.08-0.19; p < 0.001) were more likely to have metastasis resection. Meanwhile, compared with those with liver metastasis, GC patients with brain metastasis (OR, 0.07; 95% CI, 0.04-0.14; p < 0.001), lung metastasis (OR, 0.37; 95% CI, 0.24-0.58; p < 0.001) or bone metastasis (OR, 0.44; 95% CI, 0.28-0.69; p < 0.001) were more likely to undergo metastatic surgery (Supplementary Table 2).
Multivariate Logistic Regression Analysis Based on the Treatment of the Primary Tumor in Patients With Single-Site Metastasis.
Discussion
Although metastatic GC is recognized as the terminal stage, carefully selected patients may exhibit an improved prognosis following primary tumor resection surgery or metastasectomy. 11 Generally, advanced GC has a tendency to metastasize to important organs, including the liver, lung, brain and bone, hence a poor prognosis. On the other hand, different sites of metastases may have different impacts on the prognosis. Therefore, a better understanding of the epidemiology of metastatic GC remains highly desirable to actively investigate and articulate new treatment strategies. 16 In our study, we found the prognosis of patients with liver or lung metastasis was significantly better than that of patients with bone metastasis. Meanwhile, GC patients with liver, brain or bone metastasis will significantly benefit from primary resection, while metastatic resection might prove to be highly beneficial to those with lung metastasis.
Metastatic Pattern of Advanced GC
In our study, the metastatic rate of GC to liver, lung, bone and brain was 16.0%, 5.3%, 4.9%, and 0.8%, respectively, which is consistent with previous reports. There are two reasons to explain the higher rate of liver metastasis. Firstly, the common routes for metastasis are direct invasion and hematogenous metastasis, thus making liver the first site of distant metastasis 17 -22 . Secondly, thanks to increased availability of abdominal CT and ultrasound, there is a noticeable increase in the detection rate of liver metastasis in GC patients. Our study also indicated that the proportion of patients with isolated bone metastasis was significantly higher than that of patients with isolated lung metastasis, which is different from Qiu et al. suggesting GC patients with bone metastases and those with lung metastases are comparable in number. 23 The smaller proportion of liver metastasis in Qiu et al. could be explained by the fact that early liver metastases are usually asymptomatic and thus often go unnoticed. Luckily, with the boom of economy and popularity of bone scans, it is likely that an increasing cases of liver metastasis will be diagnosed in the early asymptomatic phase. 24
Factors Affecting Patient Surgery Compliance
In the multivariate logistic regression analysis, age, marital status, histological type, metastatic sites, and chemotherapy are all independent factors affecting patient surgery compliance. As the median age of our study population was 65 years, we selected 65 years as the cutoff point. We also found unmarried patients were less likely to undergo primary surgery, which is consistent with Liu et al. indicating being single or living alone was associated with lower surgical rates. 25 We speculate that such a low resection rate in single patients might be attributed to the lack of financial and emotional support from their families. Concerning the histopathological type, signet ring cell carcinoma is more aggressive and usually has an even worse prognosis, so patients usually exhibit poorer surgical compliance. 26 In terms of the site of metastasis, due to the low resection rate of the primary tumor in the GC patients with bone metastasis in our study, we used bone metastasis as a reference to analyze the effects of other metastatic sites on primary tumor resection. Similarly, the lower rate of liver metastasis resection helped determine our analysis of impact of other metastatic sites on the metastatic lesion resection. Lastly, we found GC patients who had received chemotherapy were less likely to opt for surgery. By contrast, Song et al. Suggested that chemotherapy can markedly support patients’ choice of resection of primary or metastatic tumors and thus improving the prognosis. 27 We assumed that the positive effect of chemotherapy on patients’ choice of surgery in Song et al. might be related to the transient tumor-shrinking effect of chemotherapy.
Resection Is Beneficial to Survival of GC Patients
Notably, we found that only 10% of Patients with metastatic GC received surgical treatment. This might result from the surgical intolerance or a lack of awareness in a majority of patients. Meanwhile, our study revealed the prognosis of GC patients with isolated liver metastasis would be improved as a result of resection of the primary and metastatic lesions. This corroborates Cheon et al. 28 which included 1013 GC patients with liver metastasis. They found 41 of the 58 patients undergoing gastrectomy also received resection of liver metastasis in the process of gastrectomy, and that their 1-year, 3-year, and 5-year survival rate were 75.3%, 31.7% and 20.8% respectively, suggesting the resection of liver metastasis significantly prolonged the survival time of patients. Besides, our study also indicated there was no significant difference in the prognosis between patients with isolated liver metastasis and those with isolated lung metastasis. However, Guner et al. indicated that the prognosis of isolated lung metastasis is more favorable than that of isolated liver metastasis. For now, it is hard to explain the disagreement between our study and Guner et al., yet it may be on account of the different sizes of patients in these two studies. Moreover, our study demonstrated the prognosis of GC patients undergoing resection of isolated lung metastasis is favorable, which is in line with previous studies. The retrospective analysis by Clinton et al. on 21 studies showed the 5-year survival rate of GC patients undergoing resection of their lung metastasis was around 30%. Similarly, Sun et al. included 1014 lung metastatic GC patients and found that there was a positive correlation between resection of primary tumor and a better prognosis. As the lung function of GC patients with lung metastases is poor, the quality of life may be improved with the removal of the diseased lung. Considering these pieces of evidence, we believe that resection of lung metastasis is a reasonable decision. 29 -33
Conversion Therapy Is Beneficial to Survival of Metastatic GC Patients
According to the current guidelines, palliative chemotherapy is the main treatment strategy for stage IV GC. 34 With the upgrading of chemotherapy drugs, the survival benefit of these patients is greater than before, yet it is not satisfactory. Then a prospective clinical trial (REGATTA) revealed that postoperative chemotherapy together with surgery of the primary tumor was not beneficial to patients and therefore was terminated, indicating the combination of postoperative chemotherapy with primary tumor resection is not necessarily beneficial. 35 On the other hand, conversion therapy, an extension of conversion chemotherapy, helps perform R0 resection of a tumor that was initially considered technologically unresectable or oncologically incurable, including a locally advanced tumor, or one with positive margin, through chemotherapy, radiotherapy, or target therapy. Compared with palliative treatment, conversion therapy enables patients with longer survival time and higher quality of life. 36,37 Recently, an Italian Research Group enrolled 282 patients with stage IV GC by adopting a variety of treatments including conversion therapy, chemotherapy, and a combination of surgery and chemotherapy. They found conversion therapy is significantly advantageous to improve prognosis of patients. 38 Similarly, Cascinu et al. included 82 patients with stage IV GC and found the median survival time of patients undergoing conversion surgery was significantly longer than that of patients receiving chemotherapy alone (17 months vs 12 months). Also, Yamaguchi et al. recruited 259 stage IV GC patients to receive chemotherapy, among which 84 cases had resection. The results showed there was significant difference between the median survival time of patients who underwent and those who did not undergo surgery, and also the median survival time of patients receiving R0 resection was longer than those with R1-2 resection (56.2 months vs 16.3 months). 39 -42 Overall, these findings will allow for improved surgical planning strategies for metastatic GC patients. Our study indicated that chemotherapy combined with resection of the primary tumor could improve the survival rate of GC patients with brain, bone or liver metastasis, and that chemotherapy combined with resection of the lung metastasis is beneficial to GC patients with lung metastasis. However, due to the limitation of the database, we could not distinguish the sequence of chemotherapy and resection, and could only roughly suggest combination combined with resection might be beneficial to the survival of metastatic GC patients. Therefore, future clinical trials are needed to investigate the accurate treatment for advanced GC patients. Meanwhile, different treatments should be targeted to different sites of metastases, so that optimal survival rate could be achieved and to improve quality of patients.
Prognosis Varies With Metastatic Sites
Our study also revealed that the survival outcomes of GC patients with a single-site distant metastasis varied with metastatic sites. Compared to patients with brain and bone metastases, GC patients with lung and liver metastases exhibited better CSS and OS. Besides, GC patients with single-site metastasis exhibited better CSS and OS than GC patients with multi-organ metastases. Also, surgery of the primary tumor largely improved the prognosis of these patients, and likewise, the sites and number of metastases also affect the prognosis of metastatic GC patients. It is worth mentioning that both lung and liver metastases are easy to detect at the early phase owing to the advancement in imaging technologies, thus the prognosis of these GC patients is relatively better than for those with brain metastasis or bone metastasis. These trends were similar to colorectal cancer. However, in pancreatic cancer, no survival difference was observed between patients with single-site metastasis and those with multiple site metastases. 43,44
Limitations
Although this study was carefully conducted, several limitations do exist. First, this is a retrospective study based on the SEER database; therefore, information relevant to the specific therapy and personal history of patients that could affect prognosis was not available. Second, we only included four metastatic sites as information about metastases to other sites was unavailable, especially about peritoneal metastasis which plays a crucial role in the diagnosis and treatment of advanced patients with GC, thus selection bias might exist. Third, as only less than one third of the patients underwent both primary and metastatic lesion resection at the same time, these patients could not be picked up for single analysis. Fourth, the database of SEER began to release information on GC metastases only in 2010, which means the follow-up time of the sample in our study is not adequately long enough. Finally, there are no surgical details, such as the number of distal / total gastrectomy, nor descriptions about whether resection of metastasis was performed for the sake of R0 or relief of disease. Therefore, it is essential to assess the application value of conversion therapy in IV GC through prospective trials. At present, the Asian Association of Clinical Oncology (FACO) is conducting large-scale retrospective and prospective cohort studies in China, Japan and South Korea, which are hopefully to fulfill the gap. However, despite the above limitations, to the best of our knowledge, our study for the first time comprehensively analyze the effect of primary and metastatic lesion resection on the prognosis of metastatic GC.
Conclusions
The findings of our study indicated that the site of metastasis was an independent prognostic factor for metastatic GC. The prognosis of GC patients with liver and lung metastases was better than that of those with bone or brain metastases. Furthermore, compliance to primary or metastatic tumors were associated considerably with age, site of tumor, histological type, site of metastasis, and chemotherapy. Overall, the curative effect of primary tumor resection on GC patients with liver and lung metastases was favorable for their survival. Also, surgery had some survival benefits to patients with metastatic GC; however, further studies are warranted on carefully selected patients to validate these findings.
Supplemental Material
Supplemental Material, Supplementary_Fig._1 - Prognostic Value of the Site of Distant Metastasis and Surgical Interventions in Metastatic Gastric Cancer: A Population-Based Study
Supplemental Material, Supplementary_Fig._1 for Prognostic Value of the Site of Distant Metastasis and Surgical Interventions in Metastatic Gastric Cancer: A Population-Based Study by Yinghua Li, Danna Xie, Xiaojing Chen, Teng Hu, Simin Lu and Yunwei Han in Technology in Cancer Research & Treatment
Supplemental Material
Supplemental Material, Supplementary_Fig._2 - Prognostic Value of the Site of Distant Metastasis and Surgical Interventions in Metastatic Gastric Cancer: A Population-Based Study
Supplemental Material, Supplementary_Fig._2 for Prognostic Value of the Site of Distant Metastasis and Surgical Interventions in Metastatic Gastric Cancer: A Population-Based Study by Yinghua Li, Danna Xie, Xiaojing Chen, Teng Hu, Simin Lu and Yunwei Han in Technology in Cancer Research & Treatment
Supplemental Material
Supplemental Material, Supplementary_Fig._3 - Prognostic Value of the Site of Distant Metastasis and Surgical Interventions in Metastatic Gastric Cancer: A Population-Based Study
Supplemental Material, Supplementary_Fig._3 for Prognostic Value of the Site of Distant Metastasis and Surgical Interventions in Metastatic Gastric Cancer: A Population-Based Study by Yinghua Li, Danna Xie, Xiaojing Chen, Teng Hu, Simin Lu and Yunwei Han in Technology in Cancer Research & Treatment
Supplemental Material
Supplemental Material, Supplementary_Fig._4 - Prognostic Value of the Site of Distant Metastasis and Surgical Interventions in Metastatic Gastric Cancer: A Population-Based Study
Supplemental Material, Supplementary_Fig._4 for Prognostic Value of the Site of Distant Metastasis and Surgical Interventions in Metastatic Gastric Cancer: A Population-Based Study by Yinghua Li, Danna Xie, Xiaojing Chen, Teng Hu, Simin Lu and Yunwei Han in Technology in Cancer Research & Treatment
Supplemental Material
Supplemental Material, Supplementary_Table.1 - Prognostic Value of the Site of Distant Metastasis and Surgical Interventions in Metastatic Gastric Cancer: A Population-Based Study
Supplemental Material, Supplementary_Table.1 for Prognostic Value of the Site of Distant Metastasis and Surgical Interventions in Metastatic Gastric Cancer: A Population-Based Study by Yinghua Li, Danna Xie, Xiaojing Chen, Teng Hu, Simin Lu and Yunwei Han in Technology in Cancer Research & Treatment
Supplemental Material
Supplemental Material, Supplementary_Table.2 - Prognostic Value of the Site of Distant Metastasis and Surgical Interventions in Metastatic Gastric Cancer: A Population-Based Study
Supplemental Material, Supplementary_Table.2 for Prognostic Value of the Site of Distant Metastasis and Surgical Interventions in Metastatic Gastric Cancer: A Population-Based Study by Yinghua Li, Danna Xie, Xiaojing Chen, Teng Hu, Simin Lu and Yunwei Han in Technology in Cancer Research & Treatment
Supplemental Material
Supplemental Material, Table_and_Figure_legends - Prognostic Value of the Site of Distant Metastasis and Surgical Interventions in Metastatic Gastric Cancer: A Population-Based Study
Supplemental Material, Table_and_Figure_legends for Prognostic Value of the Site of Distant Metastasis and Surgical Interventions in Metastatic Gastric Cancer: A Population-Based Study by Yinghua Li, Danna Xie, Xiaojing Chen, Teng Hu, Simin Lu and Yunwei Han in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Authors’ Note
This study was approved by the Ethics Committee of the Affiliated Hospital of Southwest Medical University, Luzhou City, China (KY2020035). Yinghua Li contributed to the manuscript preparation and data analysis. Yunwei Han contributed to the study design. Danna Xie contributed to the manuscript editing. Xiaojing Chen, Teng Hu, and Simin Lu contributed to the image collection. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by the Department of Science & Technology of Luzhou City (No.2018LZXNYD-GP01), and by the Department of Science & Technology of Liangshan Prefecture (No.18YYJS0104)
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
